Abstract
Ultramarathon races are fairly demanding and impose substantial physiological stress on healthy athletes. These competitions may thus be considerably more challenging for individuals with diabetes. This case study aims to describe glycemic control, muscle damage, inflammation, and renal function in 3 athletes with type 1 diabetes during a successful performance in a relay ultramarathon. The team completed the race in 29 hours and 28 minutes, earning third place. The total distance covered by each athlete was 68.7, 84.5, and 65.1 km. Most blood glucose levels showed that athletes were in a zone where it was safe to exercise (90–250 mg/dL or 5.0–13.9 mmol/L). Creatine kinase, lactate dehydrogenase, and aspartate aminotransferase serum levels increased 1.2- to 50.7-fold prerace to postrace, and were higher than the reference ranges for all the athletes postrace. Blood leukocytes, neutrophils, and serum C-reactive protein (CRP) increased 1.6- to 52-fold prerace to postrace and were higher than the reference ranges for 2 athletes after the race. Serum creatinine increased 1.2-fold prerace to postrace for all the athletes but did not meet the risk criteria for acute kidney injury. In conclusion, our main findings show evidence of satisfactory glycemic control in athletes with type 1 diabetes during a relay ultramarathon. Moreover, elevation of muscle damage and inflammatory biomarkers occurred without affecting renal function and challenging the maintenance of blood glucose among athletes. These findings are novel and provide an initial understanding of the physiological responses in athletes with type 1 diabetes during ultramarathon races.
Introduction
Ultramarathons are foot race competitions that vary from 50 to 1600 km and are performed on various terrains. Tolerating the physiological impact is fairly demanding for ultrarunners, 1 –5 and could be more challenging for chronic disease patients. Athletes with diabetes have participated in ultramarathons.6,7 However, to our knowledge, glycemic control and muscle damage resulting from these foot races were not assessed among these athletes. Here we address these issues and focus on athletes with type 1 diabetes mellitus (T1DM).
According to the American Diabetes Association, persons with T1DM who do not have complications and have good glycemic control can perform all types of exercise.8,9 During exercise, however, hypoglycemia is the most frequent and dangerous consequence for T1DM patients. 10 This may be due to the inability of the pancreas to promote a reduction in plasma insulin levels during exercise, combined with an exercise-induced glucose uptake via an insulin-independent mechanism. 11 T1DM patients experience hypoglycemia during exercise because of increased insulin sensitivity and augmented exogenous insulin absorption rates.12,13 Some people with diabetes may present a blunted counterregulatory hormone response to exercise and an impairment in hepatic glucose production. Conversely, high-intensity exercise may lead to hyperglycemia, given an increase in plasma catecholamine concentrations and subsequent liver glycogenolysis elevation. 10 –14 Therefore, glycemic control constitutes a challenge for T1DM patients during exercise, and glycemic management strategies are essential. 12
Ultramarathons cause marked muscle damage in healthy individuals 2 ,3,15,16 because of mechanical, metabolic, and inflammatory processes. 17 Muscle protein blood level and muscle soreness are common measures of muscle damage.18,19 Generally, muscle damage is unrelated to health complications. However, the release of excessive amounts of intramuscular proteins into the blood stream can lead to precipitation in kidney tubules, negatively affecting the renal function. 18 This can become clinically relevant to ultrarunners with T1DM who have an increased risk of kidney complications related to the pathophysiology of diabetes. 20 Exercise-induced muscle damage can lead to transient insulin resistance in healthy subjects. 21 Muscle damage initiates a series of immune reactions, which involves the production of cytokines and leukocyte infiltration into the damaged muscle, systemic release of leukocytes and cytokines, 22 and the production of a large number of acute-phase proteins. 23 Transient insulin resistance is caused by inflammatory factors generated by muscle damage. 24 Therefore, profound muscle damage and inflammatory responses observed in ultramarathons 1 ,2,16 may influence glycemic control in athletes with T1DM.
Based on the above statements, this study aims to describe glycemic control and muscle damage in a team comprising 3 athletes with T1DM during their participation in a relay ultramarathon. We also describe the athletes’ inflammatory response and renal function during the competition.
Methods
Subjects
Three male athletes were studied during their participation in the Brazil 135 Ultramarathon relay race. Demographics of the athletes are presented in Table 1. They all had satisfactory glycemic control over the preceding year, as glycated hemoglobin (HbA1c) values ranged from 6.9 to 8.7% (52–72 mmol/mol). They received multiple daily injections via insulin pen (Athlete A: intermediate and short-acting human insulin and Athlete B: long and rapid-acting analog insulin) or continuous subcutaneous infusion via insulin pump (Athlete C: rapid-acting analog insulin). This study was approved by the Ethics Research Committee of the São Paulo State University (UNESP) (no. 037/2008), and informed written consent was given by each athlete.
Characteristics of athletes with Type 1 diabetes mellitus
CHO, carbohydrate ingestion.
Research Design and Race Details
This descriptive field study occurred at the Brazil 135 Ultramarathon. This 217-km international foot race is a single-stage mountain ultramarathon performed on dirt roads in the most difficult segment of a Brazilian pilgrimage route called “The Walk of Faith” (Mantiqueira Mountains, Brazil), with a total positive and negative elevation change of 12,200 m (Figure 1). Runners may participate in the individual (solo) or relay race (teams comprising 2, 3, or 4 athletes) and must arrive at the finish line within 60 hours to be considered an official finisher. Seven teams comprising 3 athletes, including the team comprising our athletes, participated in the race in 2009. Six of these teams completed the competition, and their race time ranged from 21 hours and 35 minutes to 43 hours and 30 minutes. Volunteers could eat and drink ad libitum during the race. Ambient temperatures were 8 to 30°C.
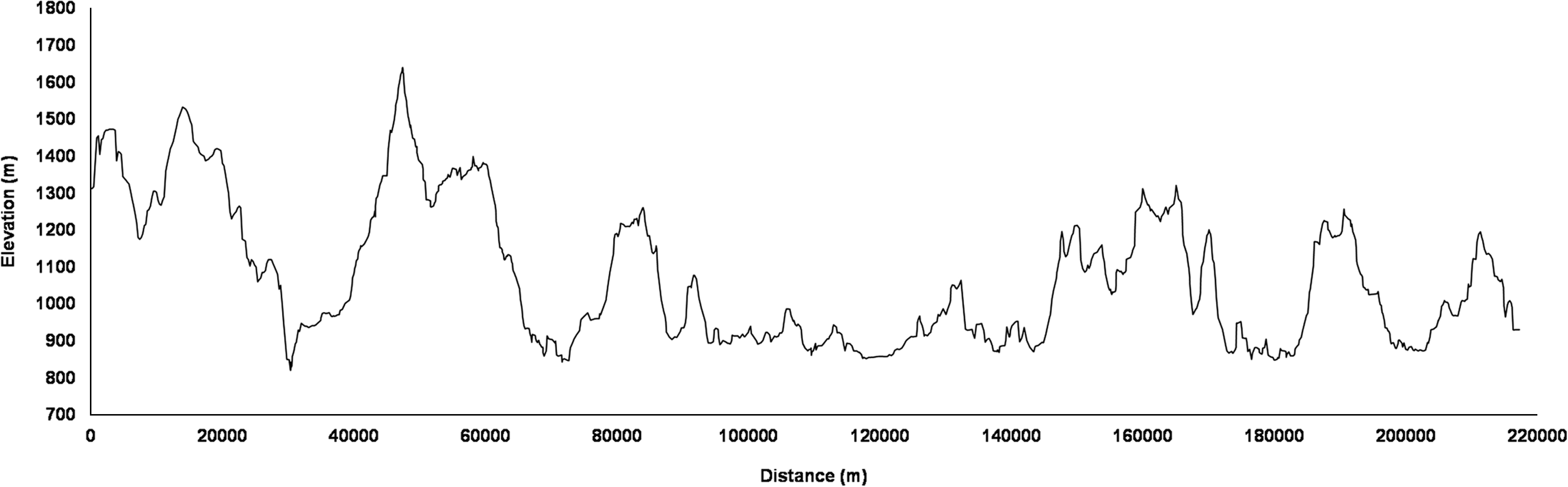
Elevation profile throughout the Brazil 135 Ultramarathon route.
Aerobic Performance Test
Critical velocity was individually determined 1 week before the competition from 4 maximal runs of 800, 1200, 1600, and 2000 m performed randomly once daily. Critical velocity was determined by the angular coefficient of the linear fit of distance versus running time. 25 Critical velocity intensity was found to occur at approximately midway between anaerobic threshold and maximal oxygen uptake. 26 Critical velocity denoted the highest sustainable rate of the athletes’ aerobic metabolism.
Race Measurements and Blood Analyses
Records of distance covered and duration of exercise bouts (Garmin Forerunner 305), blood glucose, and insulin requirements (Accu-Chek glucose meter, Roche Diagnostics) were obtained from our athletes during the relay race. The athletes conducted glycemic measurements for both prebouts and postbouts of exercise during the relay race. They conducted additional blood glucose analyses during longer bouts of exercise. Race time was obtained from official race reports.
The athletes were also evaluated prerace and postrace. Prerace measurements were performed in the afternoon before the race at 5 hours postprandial, whereas postrace evaluation occurred immediately after the race. Athletes completed a questionnaire on basic demographics and prerace training and underwent anthropometric evaluations. Twelve mL blood samples were collected from each athlete by a certified nurse and distributed across a number of tests. Automated equipment (Autolab 18, Boehringer Mannheim and KX-21N Sysmex) and typical laboratory procedures were used to generate the following blood constituents: creatine kinase (CK), lactate dehydrogenase (LDH), aspartate aminotransferase (AST), creatinine, urea, total protein, albumin, urate (Wiener kits), C-reactive protein (CRP) (Biotécnica kit), total and differential white cells counts, red blood cells, and platelets parameters. 27 All blood serum analysis results were corrected for changes in plasma volume estimated using total protein. 28
Athletes rated their level of muscle soreness prerace and postrace. Runners selected a number that best described any general feeling of pain, soreness, and muscles ache using a 10-point Likert scale: 1 (no soreness), 2.5 (dull, vague ache), 4 (slight soreness), 5.5 (more than slight soreness), 7 (sore), 8.5 (very sore), and 10 (unbearably sore). 2
Results
The team completed the race in 29 hours and 28 minutes, earning third place. The athletes completed the competition in 21 bouts of exercise: 7 bouts alternately performed by each athlete. The total distance covered by Athletes A, B, and C was 68.7, 84.5, and 65.1 km, respectively.
Most glycemic values ranged from 90 to 250 mg/dL (5.0–13.9 mmol/L) during the race (Figure 2), which is a safe range for diabetic individuals who want to exercise. 8 Athletes took their usual insulin dose a day before the race. Insulin dosage adjustments throughout the competition were individually conducted and were fundamentally based on the interaction among glycemic levels, carbohydrate counts, and exercise bout characteristics. Athlete A took 75% of the basal dose plus 50% of the bolus insulin dose, Athlete B took 85% of the basal dose plus 10% of the bolus insulin dose, and Athlete C took 50% of the basal dose plus 50% of the bolus insulin dose.
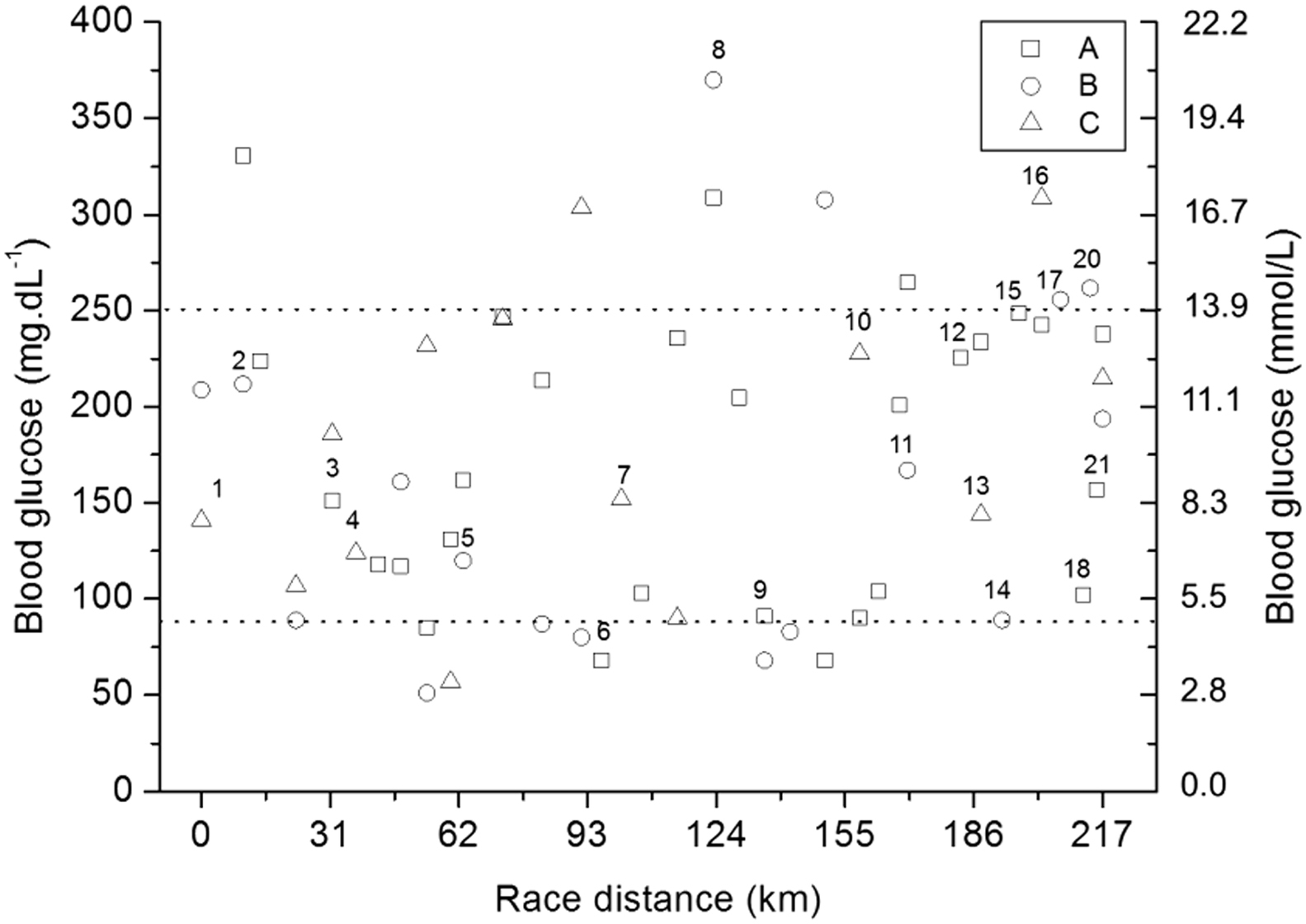
Blood glucose responses of 3 athletes (A, B, and C) during the race. Dot lines delimit the range from 90 to 250 mg/dL (5.0–13.9 mmol/L) of blood glucose. Numerical order above the markers (squares, circles, and triangles) represents which athlete (A, B, and C) was running during the competition. Segment 19 was run by athlete C. This is omitted from the figure because there was no blood glucose measurement.
Muscle protein serum levels were higher than the reference ranges for all athletes postrace (Table 2), with marked percentage changes (prerace to postrace) of serum CK (A = 844%, B = 4978%, and C = 891%), LDH (A = 123%, B = 161%, and C = 82%), and AST (A = 23%, B = 934%, and C = 106%). Moreover, muscle soreness increased from 1 (no soreness) to 2.5 (dull, vague ache) for Athlete A and from 1 (no soreness) to 5.5 (more than slight soreness) for Athletes B and C prerace to postrace. Considering all the data, we observed that athletes had high muscle damage after the competition.
Biochemical and hematological measures of the athletes prerace and postrace
MCV, mean corpuscular volume; MCH, mean corpuscular hemoglobin; MCHC, mean corpuscular hemoglobin concentration; RDW, red cell distribution width.
Blood leukocytes, neutrophils, and serum CRP were higher than the reference ranges for Athletes A and B after the race. Blood monocytes were higher than the reference ranges for all athletes after the race (Table 2). Increases in white blood cells count and acute-phase protein (ie, CRP) indicated that the athletes showed an inflammatory response prerace to postrace.
Urea levels were higher than the reference ranges for all athletes after the race. However, serum creatinine increased 1.2-fold prerace to postrace in all athletes (Table 2) and did not meet the risk criteria for acute kidney injury (ie, increased serum creatinine x 1.5). 29 Finally, slight changes were observed for total protein, albumin, urate, erythrocytes, and platelets prerace versus postrace (Table 2).
Discussion
We aimed to describe glycemic control, muscle damage, inflammation, and renal function in a team comprising 3 athletes with T1DM during their participation in a 217-km relay ultramarathon. Previous studies reported these responses based on 226- 30 and 452-km 31 triathlon competitions in these athletes. Recent investigations documented that they were able to complete 161-km solo ultramarathon races.6,7 However, to our knowledge, no study investigated these physiological responses as a result of an ultramarathon among these athletes. Our main findings indicate that they could maintain good glycemic control through appropriate management strategies during the race. Moreover, elevation of muscle damage and inflammatory biomarkers occurred without affecting the athletes’ renal function and glycemic control.
The results presented herein expand the limited number of findings about glycemic control in the study athletes during endurance32,33 and ultraendurance30,31 competitions. Additionally, 73% of the blood glucose analyses showed that these athletes were in a zone where it was safe to exercise (90–250 mg/dL or 5.0–13.9 mmol/L). 8 Among the 60 blood glucose measurements performed by the team, only 3 hypoglycemia records (<70 mg/dL or 3.9 mmol/L) 20 were seen in the athletes during the race.
Insulin dose adjustment conducted on an individual basis was essential for the athletes to maintain a satisfactory glycemic level during the race. Athletes had a solid expertise about insulin pharmacokinetics, glycemic monitoring and interpretation, balanced diet, and exercise physiology. Consistent with these findings, Toni et al. 13 and Lumb and Gallen 14 reported that the exact adjustments of insulin dose during exercise must be made from each athlete’s own experience.
Our results showed that the athletes in this study, running ∼65 to 85 km over episodic periods, had similar increases in muscle damage and inflammatory biomarkers, as reported previously where 1 athlete was evaluated after a 452-km triathlon competition. 31 Different from triathlon competitions, mountain ultramarathons are performed on off-road trails, coastal mountains, and paths with considerable cumulative downhill courses,5,34 resulting in substantial muscle damage and subsequent inflammatory responses. 22
High serum CK values are common after ultramarathons, but rarely result in adverse consequences among healthy ultrarunners. 15 Athlete B presented the highest serum CK levels (6930 U/L) among our athletes, which may be attributed to him running a longer distance (84 km) at a higher average exercise intensity (∼62% of critical velocity) than Athletes A (68.7 km, 57% of critical velocity) and C (65.1 km and 61% critical velocity). Exercise duration and intensity are reportedly associated with higher muscle damage. 18 Athlete B showed the highest postrace blood urea level (15.98 mmol/L). However, leakage of muscle cell contents into the circulation was insufficient to elicit renal function impairments. We observed that increases in serum creatinine prerace to postrace did not meet the risk criteria for acute kidney injury 29 for all athletes.
Increases in muscle damage biomarkers observed herein did not challenge blood glucose maintenance among the study athletes. Although exercise commonly improves insulin sensitivity, it may paradoxically promote transient insulin resistance if it causes muscle damage. 21 The mechanism underlying exercise-induced insulin resistance may be associated with the acute-phase response, impairment in the insulin signaling pathway, 24 and decreased noninsulin-mediated glucose uptake. 35 This effect is observed in people unaccustomed to exercise and attenuated with repeated exposures. Multiple bouts of eccentric exercise-induced muscle damage have failed to elicit disruptions in glucose homeostasis in healthy subjects. 36 –38 Extending this prior knowledge, we found evidence that repeated bouts of exercise increased muscle damage biomarkers and did not appear to influence the glycemic control among our athletes.
Conclusions
We observed satisfactory glycemic control in 3 athletes with T1DM who successfully completed a relay ultramarathon. Elevation of muscle damage and inflammatory biomarkers occurred without affecting renal functions and challenging blood glucose maintenance among athletes. These findings are novel and provide an initial understanding of the physiological responses in the study athletes during ultramarathon races. Future investigations in a large sample, even during an individual (solo) ultramarathon, can better demonstrate these case report findings. Late hypoglycemia, 10 –14,32 pronounced muscle damage, and inflammatory conditions1,16 may occur hours after the exercise has finished, which could constitute a possible limitation of the present investigation. Future studies on the participation of these athletes in ultramarathon events should also focus on these issues.
Acknowledgments: The authors would like to thank the volunteers, organizers, and staff of the Brazil 135 Ultramarathon.
Author Contributions: Study design
Financial/Material Support: Fundação de Amparo à Pesquisa do Estado de São Paulo (FAPESP) (Proc. 2009/08535-5), Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES) and Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) (Proc. 305650/2009-2).
Disclosures: None.
Footnotes
Submitted for publication October 2016.
Accepted for publication April 2017.
