Abstract
A 36-year-old man with a history of migraine headache attempted to hike from Lukla, Nepal, to Mount Everest Base Camp. On the sixth day of hiking, he had a migraine headache. After achieving resolution with typical therapies and rest, he ascended higher. Another headache developed that was interpreted to be a migraine. The headache was treated, and he ascended higher, after which severe symptoms of acute mountain sickness developed, necessitating his evacuation by helicopter. Persons with headaches in daily life may present challenges to diagnosis when traveling to high altitude. Careful evaluation and decision making are needed to achieve proper diagnosis and treatment of acute mountain sickness.
Introduction
For the active trekker at low altitude, headache is usually induced by exercise or dehydration, but for the person at high altitude, headache may be an important harbinger of serious high altitude illness. The cause of headache at high altitude can be complex in persons with previous headache history. In addition to common sea level causes, acute mountain sickness (AMS) and high altitude cerebral edema (HACE) must be considered. It is critical to differentiate various types of headache at high altitude to treat it appropriately. We discuss a specific case of migraine headache in a trekker that contributed to a delay in diagnosing AMS sufficiently severe to warrant evacuation by helicopter.
Case Presentation
A 36-year-old man made his first trek in the Khumbu Region of Nepal in April 2012, on a planned walk to Mount Everest Base Camp (5364 m [17,598 feet]). The trek plan was a 2-day stay in Kathmandu (1400 m [4593 feet]), to be followed by a flight to Lukla, Nepal (2860 m [9383 feet), followed by a 9-day walk to Everest Base Camp.
His medical history included migraine headaches, which he had had since his late teenage years. Past migraines were characterized by unilateral throbbing or pulsating pain accompanied by nausea, photophobia, and phonophobia. His worst headaches had been preceded by a visual scotoma described as multicolored lights, moving right to left in a counterclockwise fashion in the field of view, followed by a rapid transition in which his vision “went black.” Nausea and vomiting accompanied these headaches. He had headaches once or twice a year, usually associated with poor sleep and increased stress. In more recent years, he had noted headaches with fewer scotomas. He had never used any prescription medication to treat his headaches, and noted that treatment with ibuprofen 600 mg to 800 mg, acetaminophen 650 mg, or over-the-counter acetaminophen 250 mg/aspirin 250 mg/caffeine 65 mg combined with rest would eliminate his symptoms. Rest was most efficacious if completed in a cool, dark, and quiet place. He had never had a migraine headache lasting more than 48 hours, with most resolving in 12 to 24 hours.
The morning of the flight to Lukla was uneventful. The subject was participating in a clinical study to determine an efficacious dose of acetazolamide to prevent AMS. He was randomly assigned to ingest either 62.5 mg or 125 mg of acetazolamide by mouth twice a day in a blinded fashion. At 0300 hours he took his assigned dose of acetazolamide. He felt well during the flight from Kathmandu to Lukla and while hiking that same day from Lukla (2860 m [9383 feet]) to Phakding (2610 m [8562 feet]). The first night after trekking he ate well and slept without incident, with acetazolamide taken at 2000 hours.
He awoke without incident on the morning of the second day. Acetazolamide was taken at 0800 hours. The trek to Namche Bazaar (3440 m [11,285 feet]) began after breakfast. After ascending above the Larja Bridge (2800 m [9186 feet]), he had a headache characterized as dull, global, and worsened by exertion. After arrival in Namche, the headache resolved within an hour with rest and hydration and without pain medication. Pulse oximetry the first night in Namche was 80%. The subject remained in Namche Bazaar for 2 days to allow acclimatization, and his headache did not return during the stay. The stay included an acclimatization hike of a few hours to Syanboche (3720 m [12,204 feet]) on the second day. He took his dose of study assigned acetazolamide before bedtime. Pulse oximetry the second evening showed 88% oxygen saturation.
Ascent from Namche Bazaar to Tengboche (3860 m [12,663 feet]) took place on the fourth day on the trail. Day 5 on the trail was spent trekking from Tengboche to Pheriche (4240 m [13,910 feet]). The subject began the day headache free. Acetazolamide was taken at 0830 hours, and the group was on the trail at 0925 hours. After stopping in Pangboche, the group had lunch in Shomare (4010 m [13,156 feet]). The subject felt fatigued. He ate lightly and drank more than a liter each of sports drink and water. Upon arrival at Pheriche, the patient was tired but without headache. He ingested acetazolamide at 2100 hours, just before going to bed.
He awakened the next morning at 0600 hours with a right-sided, throbbing headache. He noted in his journal that this was bad timing. The headache was accompanied by photophobia and nausea. He notified one of the trip leaders of the headache and that he was of the opinion that this was a typical migraine for him. Fortunately, it was a scheduled rest day for acclimatization. Ondansetron 4 mg oral dissolving tablet and ibuprofen 600 mg were taken. Garlic soup and sports drink were ingested. After an hour, a short hike was attempted, but when the pain increased, he stopped ascending and returned to Pheriche. The afternoon was spent at rest, but the headache and photophobia persisted. Before dinner, another 600 mg ibuprofen and 5 mg hydrocodone/500 mg acetaminophen were taken. By 2100 hours, his symptoms were gone. Acetazolamide was taken before bed per study protocol.
The next morning the subject awakened without headache or other symptoms. He took acetazolamide at 0830 hours, and the group was on the trail by 0925 hours. The subject noted having a great day of hiking through the Thokla pass (4830 m [15,846 feet]) and then to Lobuche (4910 m [16,108 feet]). Dinner and socializing were uneventful, and bedtime was 2030 hours, after a dose of acetazolamide. That night, he arose once to urinate. He noticed that a headache was developing and at first thought it was yet another migraine, so did not alert trip leaders to its presence. Upon return to his sleeping bag, he observed that his breathing was shallower than normal.
At 0530 hours, the subject noted a persistent headache that was intense and diffuse. There was no associated photophobia or nausea. He did not join the group for breakfast until 0800 hours. After a light breakfast, he informed one of the trip leaders about the headache and poor rest the prior evening. The trip leader questioned whether this was another migraine; in the subject’s opinion, the headache was different from his previous migraines. He was advised to withdraw from the acetazolamide study and began a dose of 250 mg orally twice daily, beginning immediately. The subject was closely observed and given the choice to remain behind or to accompany the group. He elected to remain with the group and was advised to keep the trip leaders informed of his condition with further ascent. As the hike progressed, he felt more fatigued, but he continued to be able to drink without difficulty. The group arrived at Gorak Shep (5140 m [16,863 feet]) at 1500 hours. The subject reported fatigue as his main complaint (Figure 1). He excused himself from lunch to rest and nap, intentionally missing a planned acclimatization hike to the summit of Kala Patthar (5550 m [18,208 feet]).

Fatigue at Gorak Shep. (Photograph by Paul Auerbach, MD.)
He slept for a short time, and upon awakening took ibuprofen 600 mg and hydrocodone 5 mg/acetaminophen 500 mg, then proceeded to eat a full dinner of chicken soup, steamed rice, and fried potatoes. He reported to trip leaders that he felt much better. Just before leaving the hall at 2100 hours, his pulse oximetry was checked and showed his oxygen saturation to be 71%. After returning to his room, he ingested 250 mg acetazolamide with approximately half a liter of water. After a short period of sleep, he awoke at 2330 hours with moderate headache and urinary urgency. After voiding, he returned to his room and drank more fluids. Awakening to void occurred 3 more times during the night.
He awoke at 0515 hours for the fifth time and arose to urinate. The diffuse headache he now had was noted to be far worse than the most severe migraine he had ever had. After sitting for a minute or two, he began to vomit. His roommate awakened and administered the subject ondansetron, a 4 mg oral dissolving tablet. Within 5 minutes, the patient vomited again. At 0630 hours, the roommate summoned trip leaders, who after examining the subject administered dexamethasone 8 mg orally. That was vomited soon thereafter, so another 4 mg was given. He was examined for ataxia and none was identified. Lungs were clear on auscultation. He was then placed supine on a bed and supplemental oxygen was administered at 1 to 2 L per minute by facemask.
Considering the recent ascent and the character of the subject’s headache, the trip leaders made a preliminary diagnosis of severe AMS. It was decided that, with impending bad weather, the subject was in no condition to attempt an assisted descent to Pheriche. The patient was lucid but in worsening condition with severe headache, persistent nausea, and difficulty walking secondary to fatigue. The decision was made to arrange helicopter evacuation, first to Lukla and then to Kathmandu. Because of bad weather, it was not possible for the helicopter to land at Gorak Shep until 1300 hours, at which time a hot load was undertaken, and the subject was flown down to Lukla within 15 minutes.
Upon landing in Lukla, a medic established intravenous access and began fluid administration. The headache was subsiding but the subject’s fatigue persisted. After a short layover, he was flown by helicopter to the Kathmandu airport (1400 m [4592 feet]), and transferred by ground transportation to Norvic International Hospital for further evaluation. His headache had improved more with descent from Lukla to Kathmandu. By 2100 hours, the headache and nausea had resolved, and the subject was resting comfortably as an inpatient at the hospital, continuing to receive IV fluids. The next day he was informed that electrocardiogram and chest roentgenogram were normal. Complete blood count showed an increase in the neutrophil count on his white cell count differential, and it was advised the patient take azithromycin for 5 days for upper respiratory infection characterized by shortness of breath and harsh cough productive of dark sputum and blood. He was discharged late that afternoon.
Discussion
This case highlights the diagnostic confusion that can arise in persons who ascend to altitude and carry a diagnosis of previous migraine headache.
Migraine Headache And Migraine Attacks At Altitude
Migraine headache is a moderate to severe, recurrent, unilateral, and throbbing headache lasting hours to days, and generally accompanied by nausea, photophobia, and phonophobia, and worsened by routine physical exertion. 1 Traditionally, the headache has been thought to result from dilation of blood vessels, whereas the aura results from vasoconstriction. This interpretation is no longer considered viable. 2 Studies have shown a link between the phenomenon known as cortical spreading depression (CSD) and migraine headache.1,2 The CSD is a self-propagating wave of neuronal and glial depolarization thought to be responsible for the propagation of aura and migraine headache. 3 –5
Another theorized cause of migraine is activation of the trigeminocervical pain system. 1 ,2,6–9 This system of neurons may be triggered by events such as menstruation or ovulation, or by exercise. 1 Activation may follow CSD. 1 ,2,10 Headaches caused by an identifiable secondary source may involve this pathway, but for migraineurs, recurrent triggering with no ongoing identified cause is seen. 1 The essential difference between migraineurs and the rest of the population is their vulnerability to recurrent activations of the trigeminovascular and upper cervical pain systems. This causes headache that cannot be traced to an easily identifiable pathological process (infection, inflammation, tumor, and so forth). 1 Other triggers for headache at altitude include various causes of vasodilation, stimulation by noxious agents, and perhaps lower pain threshold.8,9
Altitude can trigger migraine headaches in persons who already have them. 6 ,11,12 Studies by Watson et al, 6 Murdoch, 13 and Jaillard et al 14 indicated that persons who travel at altitude may have migraine-type headaches as a result of factors such as travel, hypoxia, low pressure exposure, hypercapnia, stress, poor fluid or food intake, and sleep deprivation. Persons living at altitude with a diagnosis of migraine headache show similar prevalence of migraine headache as persons at sea level. 14 However, persons who have a diagnosis of migraine headache have been shown to have more painful and prolonged, but not more frequent, attacks when at altitude, sometimes presenting with focal neurologic deficits.12,13 All agree that study populations are small, and further investigations are needed.
Bolay and Rapoport 10 studied hypoxia versus hypobaria as a trigger for migraine at high altitude. They looked specifically at low barometric pressure triggering migraine, noting that atmospheric weather changes are commonly listed as migraine triggers. They also considered studies by Messingler et al 7 to determine which specific weather components had such effects. Their findings were not conclusive. Atmospheric pressure changes drive air movement, such as Chinook winds. Winds, along with warm weather, clouds, and dust, are associated with low pressure systems. They found that such alterations of barometric pressure do not trigger trigeminovascular neurons and may not serve to trigger migraines. This was restated by Pun 15 in a letter to the editor, in which he argued that hypobaric hypoxia rather than low atmospheric pressure is a cause for altitude-triggered migraine.
Other Types Of Headache
The medical provider must carefully consider several headache syndromes when making a determination of the etiology of cephalgia (Table 1). 16 This may make the diagnosis of AMS/HACE more difficult when symptoms occur at high altitude, especially if the symptoms are subtle or overlapping. Table 2 presents a comparison of the other major headache types pertaining to this case. 7 ,17,18
International Headache Society headache classification and descriptions 16
Other headache types and characteristics
There is an argument that benign exertional headache accompanies heavy straining activity only, whereas effort/exertional migraine headache may accompany any exertional activity.
High Altitude Headache And Acute Mountain Sickness/High Altitude Cerebral Edema
High altitude headache (HAH), AMS, and HACE may represent a spectrum of a single disease process. It is generally accepted that headache is the most common and frequently seen symptom in this spectrum. 8 ,12,17–19
The diagnosis of AMS is based on the setting, symptoms, physical findings, and exclusion of other illnesses. Rapid ascent of unacclimatized persons above 2500 m (8,202 feet) is a common setting for AMS, but AMS can be also seen in partially acclimatized persons abruptly ascending to higher altitude, with overexertion, accompanying use of respiratory depressants, and perhaps during onset of infection. The cardinal symptom is headache, followed by fatigue, dizziness, and anorexia. Sleep disturbance is common. Periodic breathing produces a feeling of suffocation, and frequent awakening may fragment sleep. Sleep disturbances occur almost universally for persons traveling at altitude; those with AMS have marked exaggerations in this disturbance. Facial pallor and a sensation of deep inner chill are commonly noted. Vomiting, dyspnea on exertion, and irritability are also seen. The victim may have disabling lassitude.8,20
Severe AMS may progress to HACE, which is heralded by encephalopathy. Hallmarks include ataxic gait, severe lassitude, and altered consciousness, including confusion, impaired mentation, drowsiness, stupor, and coma. Headache, nausea, and vomiting are frequent but not always present. Hallucinations, cranial nerve palsy, hemiparesis, hemiplegia, seizures, and focal neurologic signs have been reported. It is important to note that mild AMS can progress to HACE within 12 hours.8,20
The International Headache Society (IHS) defines HAH as a headache occurring after ascent to an altitude greater than 2500 m (8202 feet); it must develop within 24 hours after ascent and have at least 2 of the following characteristics: 1) bilateral; 2) frontal or frontotemporal; 3) dull or pressing quality; 4) mild to moderate intensity; and 5) aggravated by exertion, movement, coughing, straining, or bending. To meet the definition, this headache must also resolve within 8 hours of descent.8,21 A similar definition by Silber et al 12 includes the headache being associated with at least 1 other symptom typical of high altitude illness, including: 1) Cheyne-Stokes respiration at night; 2) desire to overbreathe; and 3) exertional dyspnea.
Silber et al 12 looked specifically at the clinical aspects of headache at high altitude in attempt to more clearly define HAH. Their study investigated members of the 1998 MEDEX expedition to Kanchenjunga base camp in eastern Nepal. Of the 69 persons evaluated, 138 headaches were reported. Twenty-six percent of these headaches woke subjects at night or were present upon waking in the morning. Most were described as mild, with grading of greater than 5 of 10 on 35% of occasions. Most were bilateral and commonly were frontal, diffuse, or temporal. One third had a pulsatile quality, and 16% of sufferers reported nausea. Almost half of the headaches were worsened by movement or exertion. Headaches were severe enough to require the patient to lie down and rest in one third of cases.
Twenty-two subjects (37%) reported a medical history of headaches, and of these, 7 (30%) had migraine with aura and 4 (17%) had migraine without aura. Comparing headaches experienced by those with migraine history (n = 45) and those without (n = 93), Silber et al 12 reported that there were no differences in the proportion of headaches that were unilateral, pulsatile in nature, or worsened by movement. Migraineurs were more likely to report nausea and photophobia. Conclusions were that even with gradual ascent, headache is possible and very common, and that headache is even more likely and severe with rapid ascent. The presence of headache in the medical history did not influence the risk of HAH or frequency of headaches. Persons with medical history of headache tended to have headaches of greater severity. Migraineurs tended to experience typical migraine at high altitude, precipitated in susceptible persons by exertion and degree of altitude. Silber et al 12 proposed new criteria for the diagnosis of HAH (Table 3), noting that the IHS and Lake Louise diagnostic criteria differ, that neither the IHS nor Lake Louise definitions were able to define headaches for a large number of this group, and that not all headaches that develop at altitude are sufficient per criteria to be classified as HAH or AMS.
Diagnostic criteria for high-altitude headache, proposed by Silber et al 12
Serrano-Duenas 19 took a different approach. He felt that the nomenclature was best termed “hypoxic headache.” He offered that exposure to high altitude and consequent hypobarism entail a complex series of adaptive mechanisms that depend on the rate of ascent and altitude reached. He believed that although HAH is currently considered to be an essential symptom of AMS, it is too often considered only as a component of AMS and not as an independent process. He pointed out that the IHS 1988 and 2004 classifications considered the presence of hypoxic headache a different entity from HAH, occurring only after ascent above 2500 m (8202 feet). Serrano-Duenas 19 further noted that these definitions make no reference to HAH for people living above 2500 m (8202 feet) or to experienced, well-acclimatized mountaineers, who may have headache without evidence of AMS. Serrano-Duenas 19 went on to conclude that there are 3 possibilities of headache when considering high altitude and hypobarism: 1) headache that per criteria is consistent with AMS clinical manifestations; 2) headache that occurs independently of AMS and is probably due exclusively to hypoxia; and 3) altitude-triggered migraine or migrainelike episodes.
Although the pathophysiology for HAH is not well understood, low pressure and hypoxia are at the center of many discussions regarding the causes of HAH and AMS. 8 ,21–24 Queiroz and Rapoport 9 discuss how the carotid body chemoreceptors, which are sensitive to changes in arterial partial pressure of oxygen during ascent at altitude, cause compensatory changes. Neurohumoral and hemodynamic responses result in overperfusion of microvascular beds, increased hydrostatic capillary pressure, capillary leakage, and edema. In this interpretation of events, hypoxia-induced cerebral vasodilation most likely produces the headache, and hypobaric hypoxia is the primary but not sole cause of AMS. Several other mechanisms are thought to play a role. These include alteration of the blood-brain barrier by endothelial growth factor, nitric oxide, and bradykinin, and activation of the trigeminovascular system by both chemical and mechanical stimulation. We note that the hypothesis of Sanches del Rio and Moskowitz 25 is similar to that for the production of migraine headache discussed earlier.
Two papers discuss HAH as the most common symptom associated with acute and chronic exposure to high altitude. Arregui et al 26 looked at a South American population exposed chronically to hypobaric hypoxia and found an increase in migraine and episodic tension-type headaches that increased with age. They tried to associate this with erythrocytosis, which increases with age at altitude. Appenzeller 27 discussed these findings, but noted that chronic mountain sickness is a disease of a high altitude Andean population and may be a maladaptation to high altitude living for some in this region alone, because it is not found among indigenous populations living at high altitude in the Himalaya. However, in a more recent article, Clarke 22 discusses briefly that Asian populations are now reporting symptoms of chronic mountain sickness.
Case Analysis
In this case, the patient made an ascent to 3440 m (11,285 feet) before the onset of any headache. On the sixth day, a migraine attack occurred, was treated, and subsequently resolved. Upon further ascent, another headache developed. That was initially thought to be a repeat migraine attack, even though the quality and pain distribution (unilateral to global) changed. The second headache occurred closer in timing to the development of AMS symptoms. Whether the first 2 headaches were a function of AMS is unclear, but what is clear is that 2 distinct headache patterns occurred for the patient.
The headache experienced in Pheriche was clearly a migraine, as it presented with typical symptoms and responded to therapies used successfully for resolution at sea level. The patient recognized his symptoms and was not overwhelmed by them, having been subject to previous migraine attacks. That probably was his first mistake, namely, being at high altitude and not considering AMS as a possibility.
After resolution of the initial headache, the patient was completely asymptomatic and had an uneventful ascent to the next destination. However, that soon changed. The next headache was different in quality and distribution. The patient was aware of his symptoms, but downplayed them because he was somewhat stoic and his headache was not close to what he would have previously considered to be a bad headache. Therefore, although this headache was taken seriously by trip leaders, it was considered to perhaps be another migraine, and therefore not treated as aggressively as would have been if suspected or diagnosed as AMS. That is consistent with the consideration that the headache experienced in Lobuche was a migraine headache but of greater intensity brought on by the exposure to altitude, which would fall into agreement with points made by Murdoch 13 and Silber et al, 12 and hinted at by Pun 15 and Bolay and Rapoport, 10 referring to increased frequency and intensity of migraine attacks with associated focal neurologic deficits noted with further ascent. No other symptoms of AMS were present at the time, and it was not diagnosed. As a precautionary measure, the patient was removed from the acetazolamide prophylaxis study that regulated a possibly disadvantageous dose of acetazolamide for treatment of AMS, and moved to a treatment dose. In hindsight, the discussion about whether to allow the patient to ascend to Gorak Shep should have led to a decision by the trip leaders to have the patient descend or at least remain at a constant altitude to achieve acclimatization and observe resolution of any AMS symptoms.
The patient could have better communicated his symptoms. In Lobuche, he should have been denied the opportunity to ascend until his AMS symptoms resolved, or even advised to descend. Descent from this location to a location where symptoms were resolved and rest and acclimatization could occur would have been guideline recommendations 27 and optimal. Dexamethasone therapy could have been initiated sooner.
Any headache occurring at high altitude must be taken seriously as a precursor to AMS, even if the patient has a medical history of headache at sea level. Any change in what would be considered a typical headache should be a red flag for that traveler and trip leaders/physicians. It should be thoroughly investigated for a high altitude related cause. Figure 2 shows an algorithm that may aid in identifying and treating headache at altitude. The intent is that with careful notation of history and symptoms, appropriate treatment decisions can be made, including the decision to descend or administer medications, or both. This patient was fortunate in that a helicopter rescue could be arranged. Had that not been the case, he would have spent hours in a pressurized Gamow bag while being administered dexamethasone and supplemental oxygen, hopefully improving sufficiently to tolerate ambulatory or assisted descent.
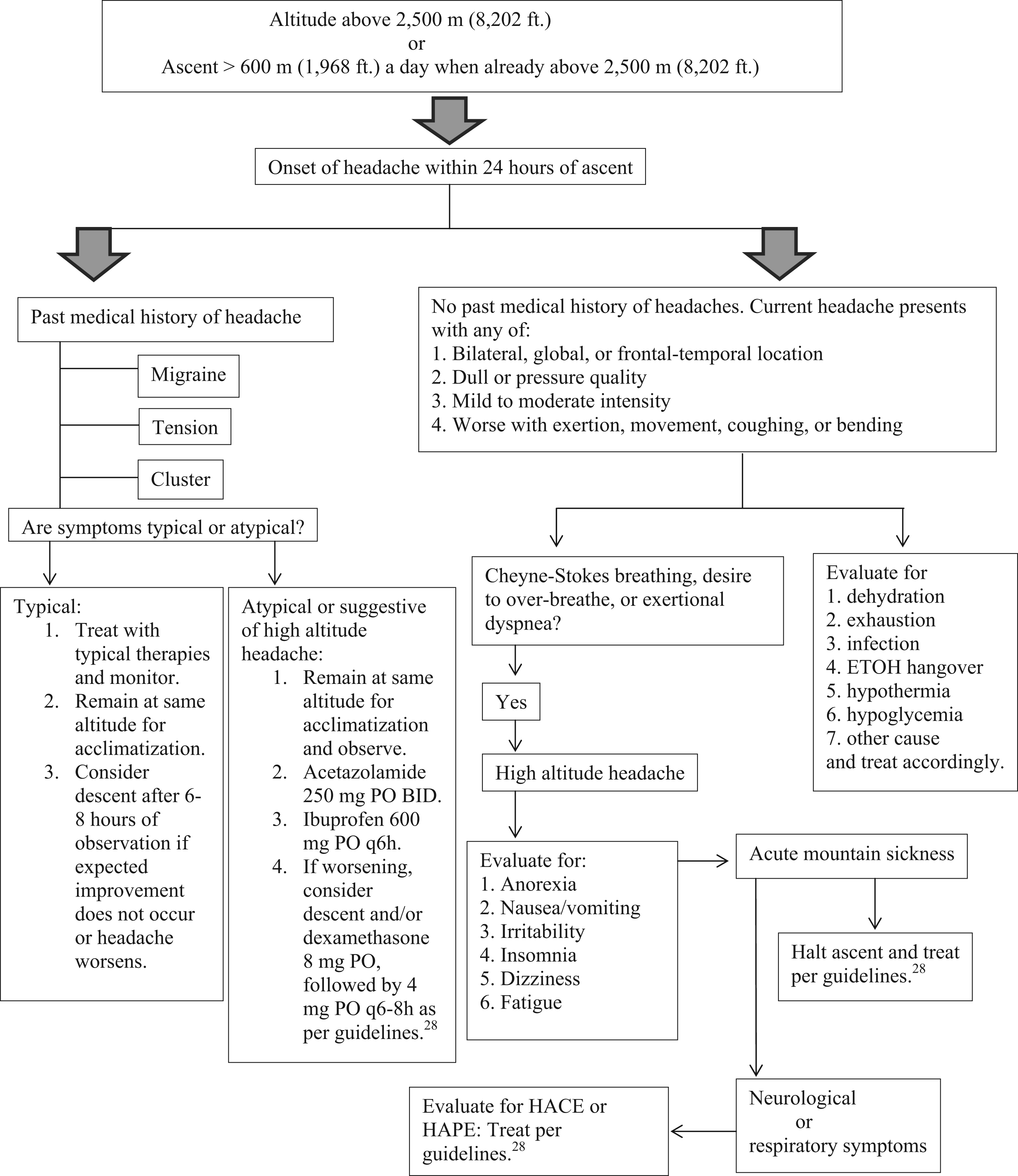
Algorithm for identifying and treating headache at altitude. ETOH, ethanol; HACE, high altitude cerebral edema; HAPE, high altitude pulmonary edema. 28
Footnotes
Acknowledgments
The authors give special thanks to Bob Veno of Global Rescue for braving the weather and providing expert clinical care.
