Abstract
Objective
To study the effectiveness of ibuprofen versus placebo in preventing acute mountain sickness (AMS) and high altitude headache (HAH).
Methods
Double-blind, randomized, placebo-controlled trial.
Results
Two hundred ninety-four healthy Western trekkers were recruited on the Everest approach at 4280 m or 4358 m and randomly assigned to receive either 600 mg of ibuprofen or placebo 3 times daily before and during ascent to 4928 m. One hundred eighty-three of 294 participants completed the trial. Of the participants who did not complete the trial, 62 were lost to follow-up and another 49 broke trial protocol. In an intent-to-treat analysis (232 participants), ibuprofen was found to be more effective than placebo in reducing the incidence of AMS (24.4% vs 40.4%; P = .01) and the incidence of HAH (42.3% vs 60.5%; P < .01). Ibuprofen was also superior to placebo in reducing the severity of HAH (4.9% vs 14.7%; P = .01). The end point of oxygen saturation was also higher in the ibuprofen group (80.8 % vs 82.4%; P = .035). For the 183 participants who completed the trial and conformed to the protocol, the incidence of AMS between placebo and treatment groups was not significant (32.9% vs 22.7%; P = .129 for AMS incidence, 9.6% vs 8.2%; P = .74 for AMS severity, 54.8% vs 42.7%; P = .11 for HAH incidence, and 8.2% vs 3.6%; P = .18 for HAH severity).
Conclusions
Ibuprofen was found to be effective in preventing AMS in the intent-to-treat analysis group but not in those who completed the trial. This loss of significance in the subjects who completed the trial may be explained by persons in the placebo group having a higher burden of illness and associated decreased compliance with the protocol. An important limitation of this study may be the possibility that ibuprofen can mask headache, which is a compulsory criterion for the diagnosis of AMS.
Keywords
Introduction
Acute mountain sickness (AMS) is a disease process associated with hypobaric hypoxemia and defined by a group of symptoms occurring at altitudes greater than 2500 m as defined by the 1991 Lake Louise Consensus Group. 1 These symptoms include gastrointestinal upset, sleep disturbances, weakness/fatigue, dizziness/lightheadedness, and most importantly high altitude headache (HAH)—the requisite symptom for the diagnosis of AMS. Both AMS and HAH commonly occur within 6 to 24 hours of ascent to altitudes greater than 2500 m with substantial morbidity with increasing altitude (42% at 3000 m for AMS and 47%–68% at 4928 m for HAH). 1 –5 Although AMS is often a self-limiting and benign process, if untreated it can progress to high altitude cerebral edema (HACE), a condition characterized by ataxia, altered level of consciousness, vasogenic cerebral edema, coma, and death.1,6 The potential lethality of HACE underscores the importance of effective prevention of AMS and HAH.
Although the mainstay of AMS prevention is a gradual ascent, acetazolamide has been used as the standard prophylactic medicine for decades. 7 In addition to acetazolamide, nonsteroidal anti-inflammatory drugs (NSAIDs) such as aspirin and more recently ibuprofen have been shown to be effective in the prevention of HAH. 8 –11 In a previous study, 11 our research group found ibuprofen to be similar to acetazolamide in preventing HAH and AMS. These results, taken together with other data suggestive of an inflammatory component to hypobaric hypoxemic pathology, suggested that an NSAID such as ibuprofen could counter AMS. For this reason, we designed a field-based trial to verify and extend our previous clinical findings in the Himalaya.
Methods
This study was designed as a prospective, double-blind, randomized, placebo-controlled, field-based clinical trial. Enrollment took place between October and November 2009 along the approach trail to Mount Everest in the Nepal Himalaya. This study was conducted in accordance with the Declaration of Helsinki; the Oxford Tropical Research Ethics Committee and the Nepal Health Research Council provided institutional review. The trial was duly registered (ISRCTN 91790322).
Participants and Outcome Measures
Trekkers completed questionnaires after giving signed informed consent. Inclusion criteria specified healthy non-Nepali males and females 18 to 65 years of age traveling directly between the baseline villages of Pheriche or Dingboche (4280 m and 4358 m, respectively) and the end point in Lobuje (4928 m). Potential participants were excluded if they had any more than 2 mild symptoms (≤1) on the Lake Louise AMS Questionnaire (LLQ), signs or symptoms of a substantial acute infection, had slept above 4500 m, or had taken any NSAIDs or acetazolamide within 1 day or 3 days before enrollment, respectively.
The predetermined primary outcome measure was AMS incidence at the study end point as calculated on the LLQ, a validated field standard for the diagnosis of AMS.
12
Predetermined secondary end points included evaluation of AMS severity, as well as HAH incidence and severity on the LLQ and visual analog scale (VAS). Other secondary measures included pulse oximetry (Sp
Study Design
Commercial pharmaceutical-grade ibuprofen and placebo were packed in visually identical capsules by Deurali-Janta Pharmaceuticals (Kathmandu, Nepal). Study medications were randomized via computer-generated code. Participants were sought out on a daily basis in all baseline village hotels and sequentially enrolled to minimize selection bias.
Newly arrived trekkers were recruited daily to participate, and volunteers meeting inclusion criteria were interviewed to obtain data. Interviewers gathered demographics, ascent profile data, LLQ, VAS, and Sp
On their ascent from baseline, a minority of participants stopped overnight at a lodge at 4595 m, but all were expected to arrive at the end point altitude for data collection. The VAS and LLQ scores were self-reported the night of arrival and the morning after arrival, at which point the study was complete. End point data collection represents morning-after-arrival data (unless missing, then replaced with night-before data) to emphasize specificity in the diagnosis of HAH and AMS.
Study administrators at baseline and end point sites were trained to be vigilant for severe illness that might be attributable to altitude or a reaction to medications, equipped with appropriate rescue medications and Gamow bag (portable hyperbaric chamber), and always ready to help with descent.
Statistical Analysis
Power calculations suggested a minimum total of 164 participants (82 per arm) was needed, a total calculated to be sufficient to identify a statistically significant difference (at 80% power) between treatment and placebo based on published HAH incidence of 50% and AMS incidence of 32% at the study end point. 11 Intent-to-treat analyses, univariate comparisons (Pearson χ2, Fisher exact tests, t tests), and logistic regression were used to examine multivariate risk factors for binary outcomes. Probability values less than 0.05 were considered significant. All analyses were conducted using R 2.6.0 software (R Foundation for Statistical Computing, Vienna, Austria).
Results
Two hundred ninety-four total participants were enrolled in the study, with 183 completing the entirety of the protocol (Figure). Forty-nine participants broke protocol for various reasons, but did present at the completion of the study to allow for data acquisition. The remaining 62 participants (26.7%) were lost to follow-up; an expected finding consistent with previous studies.
2
,4,11,13 An intent-to-treat analysis per protocol was used to analyze all accumulated data. Analysis of demographic data revealed no statistically significant differences between the ibuprofen and placebo groups with respect to gender, age, nights spent at altitude, mode of arrival to the Himalayas, LLQ score at baseline, previous use of medication for altitude illness, Sp
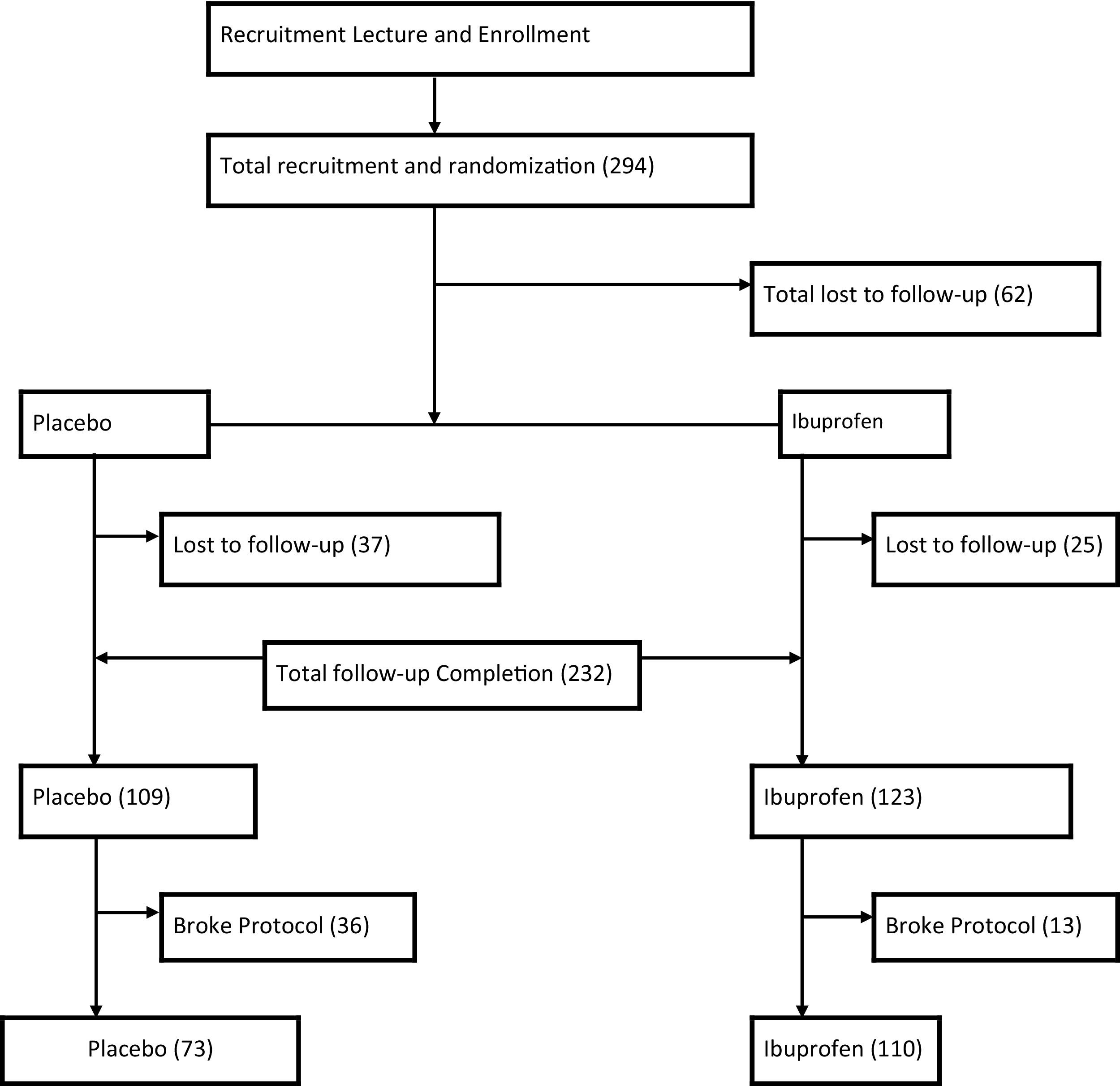
Participant flow diagram.
With regard to adverse events, one participant among the 232 reported black stool and was advised to stop the study medication (the code was broken and he was found to be on ibuprofen). He was followed closely with timely physical examinations with no untoward event. Ten other persons reported stomach pain, 3 in the ibuprofen group and 7 in the placebo group. For all, the pain subsided within a day with no additional medication. There were no patients with HACE or HAPE.
Intent-to-treat Analysis
Table 1 reveals the baseline characteristics of the intent-to-treat group, and the main outcome profile of that group is presented in Table 2. (The intent-to-treat group comprises the group based on initial treatment intent, not on treatment eventually administered). Several statistically significant results were observed for the ibuprofen group as compared with placebo, including a decreased incidence of HAH (42.3% vs 60.5%; P < .01) and AMS (24.4% vs 40.4%; P < .01), as well as a decrease in severity of HAH on the LLQ and VAS (HAH > 1 on LLQ, 4.9% vs 14.7%; P = .01; VAS, 0.7 ± 1.5 vs 1.1 ± 1.7; P = .04). Additionally, end point Sp
Baseline characteristics of the intent-to-treat group
AMS, acute mountain sickness; LLQ, Lake Louise AMS Questionnaire; N/A, not applicable; Sp
End point cohort consists of participants enrolled at baseline and followed up at end point (n = 294).
The Lukla airport is at ∼2800 m, and trekkers starting from Jiri (2000 m) pass through Lukla.
Enrollment occurred in the villages of Dingboche (4358 m) and Pheriche (4280 m).
Participants were allowed to have one mild (LLQ = 1) nonheadache symptom on the Lake Louise questionnaire to prevent self-selection bias.
Main outcome profile (intent-to-treat)
AMS, acute mountain sickness; N/A, not applicable; Sp
Probability value for comparison between placebo and ibuprofen groups.
Severe headache incidence as determined by a value of 2 or greater (of 0–3) on the Lake Louise AMS Questionnaire, “do you have a headache?”
AMS defined as Lake Louise AMS Questionnaire score of 3 or greater, requiring headache plus one or more symptoms.
Severe AMS incidence as determined by a Lake Louise AMS Questionnaire score of 5 or greater.
Participants who broke protocol were defined as those taking a medication conflicting with their assigned treatment group (crossovers) or those who missed 2 or more doses.
Analysis of Fully Compliant Participant Data
Table 3 shows the main outcome profile of the fully compliant participants. Analysis among fully compliant participants (those who hiked from the baseline to end point, missed fewer than 2 doses of assigned medication, and abstained from using nonstudy medications) found no statistically significant differences in any outcomes measured. Although not statistically significant, the data did demonstrate a difference between ibuprofen and placebo groups with respect to HAH and AMS incidence and severity, consistent with results of the intent-to-treat analysis (42.7% vs 54.8% for HAH incidence; 3.64% vs 8.2% for HAH score > 1; 22.7% vs 32.9% for AMS incidence; 8.2% vs 9.6% for AMS score > 5 on LLQ).
Main outcome profile excluding those who broke protocol a
AMS, acute mountain sickness; N/A, not applicable; Sp
Participants who broke protocol were defined as those taking a medication conflicting with their treatment group (crossovers) or those who missed 2 or more doses.
Probability value for comparison between placebo and ibuprofen groups.
Severe headache incidence as determined by a value of 2 or greater (of 0–3) on the Lake Louise AMS Questionnaire; “do you have a headache?”
AMS defined as Lake Louise AMS Questionnaire score of 3 or greater, requiring headache plus one or more symptoms.
Severe AMS incidence as determined by a Lake Louise AMS Questionnaire score of 5 or greater.
Multivariate Analysis
Multivariate models were developed for 6 different outcomes: headache incidence; severe headache incidence; AMS incidence; severe AMS incidence; end point VAS; and Sp
Statistically significant differences in outcome between treatment groups were found for headache incidence (P = .01), and indications of differences were observed for AMS incidence (P = .07).
Analysis of Participants Who Broke Protocol
Analysis of the participants breaking protocol compared with fully compliant participants revealed a higher incidence of AMS (51% vs 26.8%; P = .001) and incidence of more severe HAH scores based on both LLQ and VAS (HAH score > 1 on LLQ 24.5% vs 5.5%; P < .01; VAS 1.75 ± 2.33 vs 0.62 ± 1.26; P < .01). Although not quite a statistically significant difference, participants who broke protocol had an increased incidence of HAH (63.3% vs 47.5%; P = .051) compared with those who were fully compliant. No statistically significant difference was observed in Sp
Lost To Follow-up Analysis
Analysis of those who completed the study versus those lost to follow-up revealed 2 measured variables for which statistically significant differences between the 2 groups were observed; nights spent at altitude (3.42 ± 0.778 vs 4.37 ± 1.42; P < .01) and baseline Sp
Exclusion Analysis
Analysis of persons enrolled in the study vs persons excluded revealed only 1 variable with a statistically significant difference between the groups: age (36.5 ± 11.1 vs 31.6 ± 10.6; P = .011). There were no significant differences between groups with respect to gender, mode of arrival, nights spent at altitude, or oxygen saturation at baseline.
Discussion
The present study represents an attempt to verify and extend observations from our previous trial. 11 Many of the findings were reconfirmed here in an intent-to-treat analysis (Table 2); ibuprofen again showed a significant clinical reduction in the incidence (but not severity) of AMS and HAH. However, for the participants who completed the trial without breaking protocol (Table 3), estimated differences in the incidence and severity of both HAH and AMS in the ibuprofen group vs the placebo group dropped below the threshold necessary for statistical significance. We hypothesize that the loss of statistical significance observed in some outcome measures when persons breaking protocol were excluded stems from the exclusion of data from the sickest participants. The data in Table 2 demonstrate that participants in the placebo group bore a greater burden of illness, and thus would be expected to have a higher rate of noncompliance resulting in exclusion from the data set in Table 3 (placebo [n = 36] vs ibuprofen [n = 13]; P = .01; odds ratio, 4.2). Thus, we recommend that the loss of statistical significance shown in Table 3 should be interpreted with caution. Indeed, both AMS incidence and HAH severity were statistically significantly higher in the cohort that broke protocol. Those who broke protocol also had a higher HAH incidence than those continuing on in the study (P = .051).
We were surprised to find that many outcomes that were significantly different between treatment groups in univariate analyses were no longer significant in our multivariate analyses. Although some loss of precision in estimation is to be expected with the loss of degrees of freedom accompanying increased terms in a model, we posit that the inclusion of a covariate specifying “broke protocol” in our multivariate models also contributed to the “washing out” of significant differences. The colinearity of treatment status and “broke protocol” status results in larger standard errors for each of these coefficient estimates. Indeed, when the “broke protocol” covariate was removed from the multivariate models, outcomes of AMS and HAH incidence were again statistically significant (data not shown).
Finally, although not statistically significant (P = .10), persons taking placebo were estimated to be somewhat more likely to be lost to follow-up than were persons in the ibuprofen group, as was the case noted above with breaking protocol. We suggest our observed results in the fully compliant analyses may be partially biased, whereby treatment appears less effective than it actually is. The less compliant participants who broke protocol were likely more affected by the altitude. This group was also more likely to be those individuals taking placebo than those taking ibuprofen. Thus, analyses of fully compliant participants, including adjustments for breaking protocol, contribute to a diminished observed treatment effect among the fully compliant.
Previous HAH prevention studies with aspirin demonstrated a significant protective effect.8,9 This is consistent with the present study, which demonstrated a reduction in the incidence and severity of HAH in the intent-to-treat analysis. Similarly, Meehan et al 14 studied naproxen for prevention of AMS; however, they found no statistically significant differences to suggest a protective effect. These results should be viewed with caution, as the study likely lacked sufficient power to come to any strong conclusion with a sample size of just 11 participants. As we were submitting this article, another study 15 carried out at a lower altitude (3810 m) with a smaller sample size (86 participants) than this present study was published. The study, which was conducted in the White Mountains of California, showed ibuprofen to be efficacious in the prevention of AMS.
Our study was inconsistent with the results of Kayser et al 16 looking at the effects of calcium carbasalate for prevention of AMS and HAH. They found no evidence of calcium carbasalate having a protective effect for AMS or HAH; however, the study also suffered from significant methodologic problems with blinding and an inadequate sample size. 17
Roach et al 18 demonstrated elevated levels of anti-inflammatory markers in individuals less susceptible to AMS. The protective effects of anti-inflammatory markers point toward inflammation as important in the pathophysiology of AMS. There has been recent interest in the concept that inflammation may contribute to the development of cerebral forms of altitude illness. Vasogenic cerebral edema is known to occur secondary to exposure to hypobaric hypoxemia and increased cerebral blood flow.19,20 It is also widely recognized that the generation of this vasogenic cerebral edema requires some disruption of the blood-brain barrier. 21 In addition to such edema, hypobaric hypoxemia has been shown to result in elevated levels of inflammatory markers and anti-inflammatory markers in those susceptible and resistant to altitude illness, respectively. 18 ,22,23 Although the causal relationship of these findings remains unclear, their association has led to the development of the theory that hypobaric hypoxemia-induced inflammation may play a role in blood-brain barrier breakdown, which is one important step in the development of AMS and subsequent HACE. Ibuprofen may inhibit this blood-brain barrier breakdown.
The etiology of the pain of HAH also continues to be hypothetical. What is known is that irritation of the meningovascular system by various mechanisms is important in the development of various forms of headaches. 6 ,7,22,24–26 Traction on the meningovascular system as a consequence of brain swelling and increased cerebral blood flow has been proposed as a possible mechanism for the development of the characteristic HAH of AMS.19,20 Chemical irritation or sensitization of this system is also thought to take place. For example, inflammatory mediators such as prostaglandins are known to potentiate nociception and thus magnify the mechanical irritation described above. 23 The possible role of inflammation in the development of HAH is another pathway by which anti-inflammatory drugs such as NSAIDs may prevent HAH. 27 –30
Limitations of the Study
There are several limitations to the application of these data in clinical practice. This cohort we studied at high altitude is partially acclimatized even though the end point data were collected at about 700 m higher than the study enrollment altitude. We have used this site for a number of randomized, controlled trials in the past, 2 ,3,11 whose results have been verified by other studies from other venues. 31 It is well known that acetazolamide can be taken episodically to hasten acclimatization even while trekkers are ascending. We reasoned that another drug that performed likewise would be a useful alternative. Importantly, adequately powered high altitude studies are scarce, and this site gives us an opportunity to study large numbers of participants. Second, because just over a fifth of enrollees were lost to follow-up, our results may be biased, particularly when being lost to follow-up may be directly related to AMS status. Analysis of those lost to follow-up suggests participants in the placebo group were more likely to drop out of the study. Attrition of this magnitude is expected as in our previous studies in the past in this region,2,4 given the realities of conducting a study in a wilderness setting. Third, we observed a statistically significant difference between the age of participants and the age of persons excluded from the trial (who on average were about 5 years younger). More advanced age is known to have a protective effect on brain volume, and this difference could conceivably result in a lower AMS incidence and severity bias. Fourth, the mean baseline oxygen saturation of the group lost to follow-up was significantly lower than that of persons who completed the study. This difference too may have led to lower AMS incidence bias in the cohort, 32 although it is difficult to know whether a median difference of 1% between the 2 groups in baseline oxygen saturation is clinically significant, albeit statistically significant. Finally, an important limitation of the study is that ibuprofen is effective in prevention and treatment of headache, which is not only the most prevalent symptom of AMS but a compulsory criterion for the diagnosis of AMS.33,34 Hence, it may be problematic to distinguish the pure analgesic effect from a treatment effect of AMS.
Conclusions
Until basic research and more studies confirm the mechanism behind AMS using ibuprofen, we urge caution when using ibuprofen and other NSAIDs for the prevention of HAH and AMS. The clinical results we observed suggest ibuprofen is an inexpensive, over-the-counter prophylactic agent. However, an alternative, arguable conclusion is that ibuprofen merely masks a useful marker (headache) of AMS. Further study is warranted.
Footnotes
Acknowledgments
Funding for this study was provided by Wellcome Trust, UK. We would like to thank all the staff of the Himalayan Rescue Association in Nepal and members of Mountain Medicine Society of Nepal (MMSN) who helped us tremendously to carry out this study. We are especially indebted to the trekker participants who were very kind to enroll in our study.
