Abstract
Objective
High altitude environments present unique medical treatment challenges. Medical providers often use small portable pulse oximetry devices to help guide their clinical decision making. A significant body of high altitude research is based on the use of these devices to monitor hypoxia, yet there is a paucity of evidence that these devices are accurate in these environments. We studied whether these devices perform accurately and reliably under true mountain conditions.
Methods
Healthy unacclimatized active-duty military volunteers participating in mountain warfare training at 2100 m (6900 feet) above sea level were evaluated with several different pulse oximetry devices while in a cold weather, high altitude field environment and then had arterial blood gases (ABG) drawn using an i-STAT for comparison. The pulse oximeter readings were compared with the gold standard ABG readings.
Results
A total of 49 individuals completed the study. There was no statistically significant difference between any of the devices and the gold standard of ABG. The best performing device was the PalmSAT (PS) 8000SM finger probe with a mean difference of 2.17% and SD of 2.56 (95% CI, 1.42% to 2.92%). In decreasing order of performance were the PS 8000AA finger probe (mean ± SD, 2.54% ± 2.68%; 95% CI, 1.76% to 3.32%), the PS 8000Q ear probe (2.47% ± 4.36%; 95% CI, 1.21% to 3.75%), the Nonin Onyx 9500 (3.29% ± 3.12%; 95% CI, 2.39% to 4.20%), and finally the PS 8000R forehead reflectance sensor (5.15% ± 2.97%; 95% CI, 4.28% to 6.01%).
Conclusions
Based on the results of this study, results of the newer portable pulse oximeters appear to be closely correlated to that of the ABG measurements when tested in true mountain conditions.
Introduction
Pulse oximetry has widespread clinical acceptance in medicine for the noninvasive monitoring of arterial oxygen saturation (Sa
Because of technological advances, the newer generation of pulse oximeters are smaller, lighter, and more reliable. According to the device manufacturers, the accuracy of current handheld pulse oximeters is reported to range from ±2% to ±4% depending on the manufacturer and attachment probe used. Such performance is consistent with the independently tested accuracy of the previous generation of larger pulse oximeters, and is accepted to be representative of performance in the controlled clinical setting.
Portable pulse oximeters appear to be gaining acceptance for field applications. These smaller pulse oximeters are seeing increased use in the field setting by mountaineers, special operations units, and the aviation community as a means of monitoring for hypoxia. Several models of portable pulse oximeters have received aeromedical approval for clinical use in medevac and other helicopter-based operations in the US military. 3
However, a review of the scientific literature reveals that published data on the performance of such devices under field conditions are lacking. Conditions present in the field may impact the accuracy of pulse oximeters. For example, individuals who are cold have lower peripheral perfusion, and they may be shivering; both factors make signal processing more difficult and may affect the accuracy of the pulse oximeter readings. Two manufacturers (Nonin and BCI) claim that current digital processors make their devices suitable for use in demanding environments by using reflectance sensors to address the problem of poorly perfused or shivering extremities. We studied whether these devices perform accurately and reliably under true mountain conditions when compared with standard arterial blood gases (ABG). In our study, pulse oximeters that have incorporated this technology were tested using sensor sites on the finger, earlobe, and forehead. 1 ,2,4–6
Methods
This comparative study of 49 subjects evaluated different pulse oximetry devices measured in a cold, high altitude, field environment compared with the gold standard of ABG measurements. Testing was performed on unacclimatized US Navy and Marine Corps volunteers at an elevation of 2100 m (6900 feet) in an outdoor environment.
Study Setting and Subjects
This study was conducted from February 13, 2011, to March 13, 2011, at the US Marine Corps Mountain Warfare Training Center (MWTC) in Bridgeport, California. Participants were 18 years or older and present for active-duty military training at this facility. Our goal was to assess individuals who are still in the initial stages of respiratory adaptation undergoing the hypoxic ventilatory response (HVR), the normal physiologic adaptation to altitude-related hypoxia, and thus may have increased risk of developing altitude-related illness symptoms. We therefore excluded acclimatized individuals (defined as being at an altitude of greater than or equal to our study altitude of 2100 m [6900 feet] for more than 48 hours). Pregnancy was also an exclusion criteria.
Procedures
After institutional review board approval at Naval Medical Center San Diego, consent was obtained from subjects who were within 48 hours of arrival to altitude. Subjects were taken out into the cold field-training environment without adequate or appropriate cold weather garments (average temperature, 40°F) for field training exercises. After their scheduled training event, subjects were evaluated at the outdoor study site situated at 2100 m (6900 feet) with 2 different pulse oximetry devices (Nonin PalmSAT 2500, Nonin Onyx 9500; Nonin Medical, Inc, Plymouth, MN) using 5 different pulse oximetry probes. The following probes were used with the Nonin PalmSAT 2500 device: Nonin PalmSAT 8000AA and 8000SM finger probes, Nonin PalmSAT 8000Q ear probe, and Nonin PalmSAT 8000R forehead probe. The Nonin Onyx 9500 finger pulse oximeter is a standalone device. All probes were used on each patient, and all measurements were recorded. Subjects were then asked to complete a Lake Louise Altitude Illness questionnaire. After completion of the measurements and questionnaire, they were moved into a room temperature-controlled space for ABG draw. All ABGs were drawn by certified nurses and emergency physicians. The ABG samples were analyzed using the i-STAT Portable Handheld System (I-STAT Corp, Princeton, NJ) and using the CG8+ABG cartridges (part number: 03M86-01). The room temperature environment was used to ensure higher ABG sampling success and to ensure proper functioning of the i-STAT equipment. Any abnormal laboratory values or concerning symptoms were addressed and evaluated by local medical providers. All female participants completed a urine pregnancy test before participation.
Statistical Analysis
Using an α of .05 and β of .80, we determined that we would need 35 participants to adequately power the study based on similar validation studies, but planned for 50 given the likelihood of complications of data collection and equipment problems in the environment we were working in. 7 Data were compared using an analysis of variance with Scheffé comparison of differences. Bland-Altman comparison was also used for comparison of differences among diagnostic tests. Acute mountain sickness (AMS) data were compared using linear regression looking for correlation between AMS score and measured ABGs. All statistics were calculated using the STATA Statistical software package (StataCorp LP, College Station, TX).
Results
We compared the data for 49 subjects as we were unable to reach our goal of 50 owing to small class numbers and lack of full participation during the data collection period. There was no statistically significant difference between any of the devices and the gold standard of ABG. The best performing device was the PalmSAT 8000SM finger probe with a mean difference of 2.17% and SD of 2.56% (95% CI, 1.42% to 2.92%; Figure 1), then the PalmSAT 8000AA finger probe (mean difference ± SD, 2.54% ± 2.68%; 95% CI, 1.76% to 3.32%), next the PalmSAT 8000Q ear probe (2.47% ± 4.36%; 95% CI, 1.21% to 3.75%), then the Nonin Onyx 9500 (3.29% ± 3.12%; 95% CI, 2.39% to 4.20%), and the poorest performing device the PalmSAT 8000R forehead reflectance sensor (5.15% ± 2.97%; 95% CI, 4.28% to 6.01%). The PalmSAT 8000R forehead probe was the only device showing a statistically significant difference from the other devices (P < .005; Figures 1 and 2).
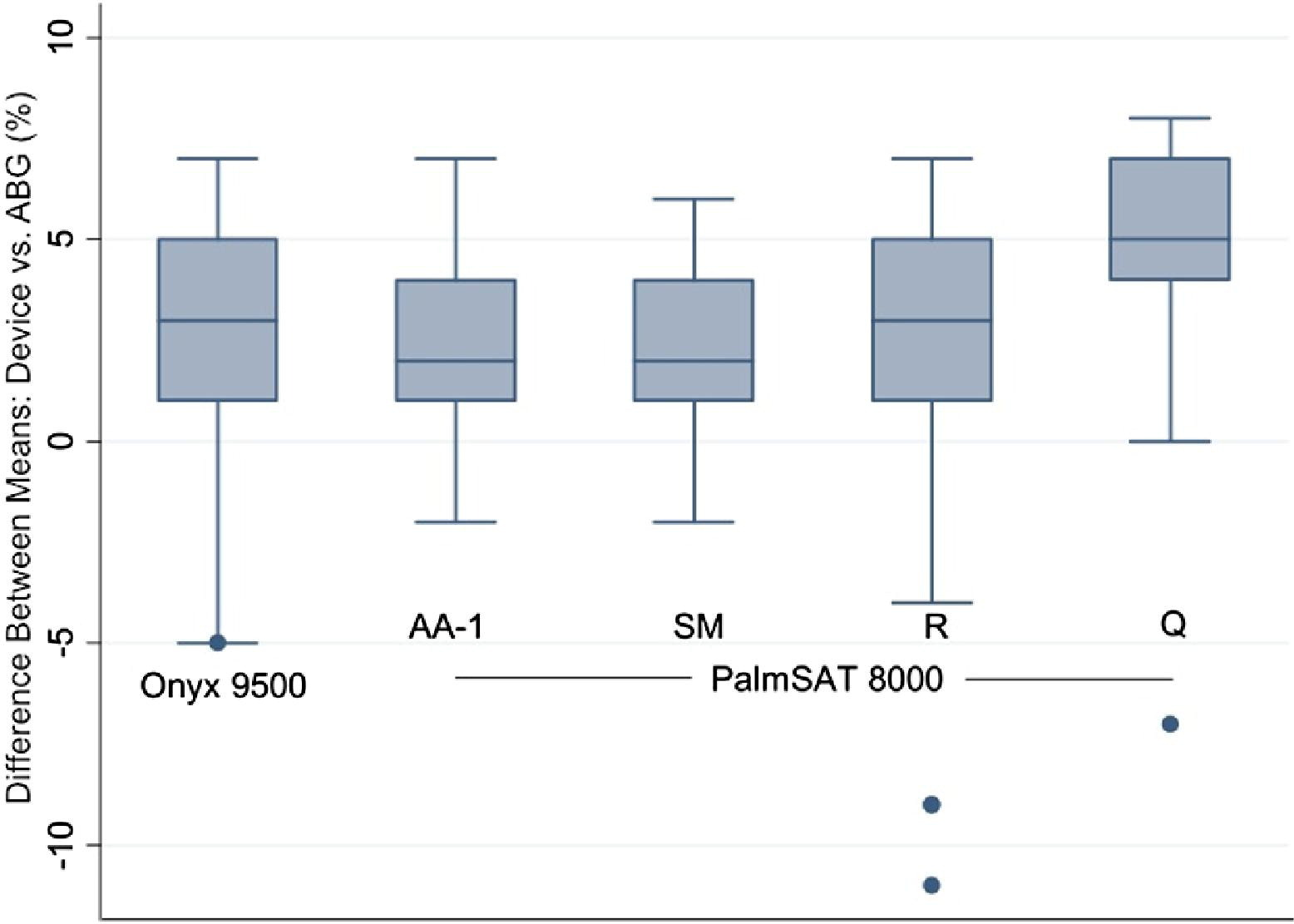
Difference between means of pulse oximetry device vs arterial blood gas (ABG).
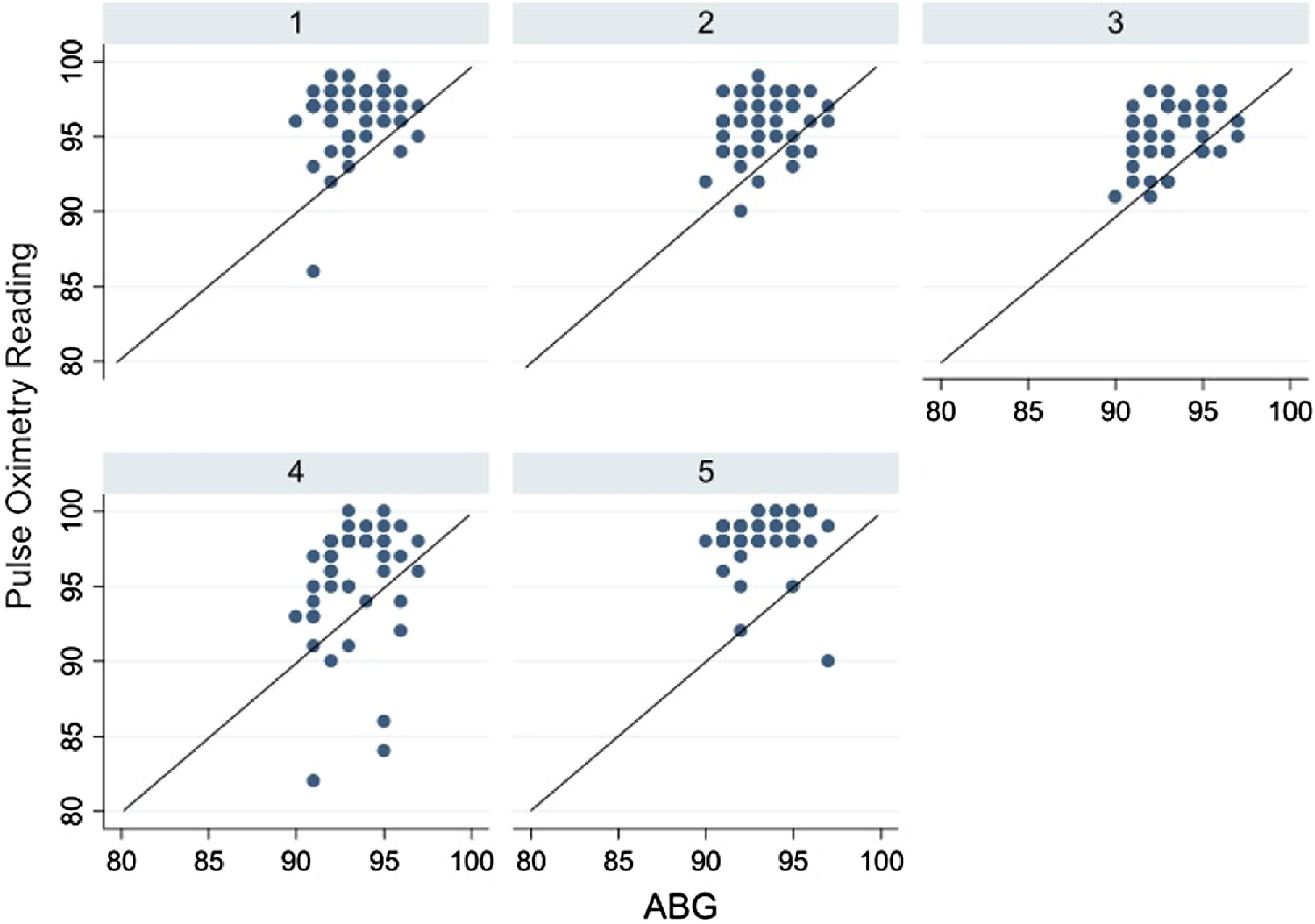
Pulse oximetry device readings vs arterial blood gas (ABG) readings. Line demonstrates perfect agreement between the two. Key for devices: 1, Onyx 9500; 2, PalmSAT 8000AA-1; 3, PalmSAT 8000SM; 4, PalmSAT 8000R; 5, PalmSAT 8000Q.
The average oxygen saturation for the various pulse oximeters and the ABGs is listed in the Table. The overall average oximeter saturation was 96.18% and the average ABG saturation was 93.32%. The range for oxygen saturation measured by pulse oximetry device varied by device, with the greatest range being 82% to 100%, and saturation measured by ABG ranged from 91% to 97% (Table).
Performance of individual pulse oximetry devices and arterial blood gas measurements
ABG, arterial blood gas.
When comparing ABG oxygen saturation with scores on the Lake Louise AMS questionnaire there was no significant correlation (P = .9373).
Discussion
These results indicate that at the altitude of 2100 m (6900 feet) above sea level, all studied pulse oximetry devices perform within the standard deviation reported by the manufacturer. No statistically significant difference exists among the majority of the devices (except for PalmSAT 8000R). Our evaluation indicates the performance of these devices can be ranked, with the most accurate device being the Nonin PalmSAT 2500 using the 8000SM finger probe. We believe that the PalmSAT 8000R forehead probe may have had the worst performance because of the difficulty with proper placement owing to poor adhesive duration. This required additional manual pressure, which may have reduced the accuracy of the reading.
Both the Nonin PalmSAT 2500 device and Nonin Onyx 9500 have been tested and approved by the US Army and US Air Force for use in flight operations, and the Nonin Onyx 9500 has been certified by the Navy Experimental Dive Unit for safety in hyperbaric chamber use. Although these devices have been proven to be highly versatile, there previously have been limited data on the accuracy of these devices in cold mountainous environments. This study demonstrates that at 2100 m (6900 feet) in a cold-weather environment, the devices perform well and can be recommended for use.
When humans ascend to altitude the partial pressure of oxygen decreases in the air they inspire. The partial pressure of oxygen in the atmosphere decreases at a logarithmic rate owing to decreasing barometric pressure; at 5800 m the atmosphere contains one half the partial pressure of oxygen as at sea level. 8 Complex physiologic modifications must be made to compensate for the decreased amount of inspired oxygen and to prevent hypoxia. Collectively these changes are known as acclimatization, and individuals acclimatize at varying rates. Diverse interactions between genetic factors and the environment determine the ability of an individual to acclimatize, making it difficult to determine who will acclimatize well and who will have difficulty. 9 Improved physical fitness does not appear to aid in acclimatization. 8 Those who acclimatize poorly are more susceptible to experiencing high altitude illnesses.
Currently, the only diagnostic tools available to field medical personnel for the detection of altitude-related illnesses are medical history and physical examination, but the physical findings of altitude illnesses are not specific and can be misleading. The physical findings most often associated with high altitude pulmonary edema (HAPE) are tachypnea, tachycardia, fever, and rales on pulmonary examination; these physical findings are consistent with many other common illnesses. 9 –12 The use of pulse oximetry under field conditions may be a vital tool for discriminating non-altitude-related illnesses from the more serious condition of HAPE and aid in the determination of who is in more dire need of treatment. 8 –13
Studies performed at the Frisco Medical Center in Colorado found the mean oxygen saturation measured by pulse oximetry (Sp
It is important to note that pulse oximetry has known limitations at the extremes of altitude. Pulse oximetry accuracy declines when the Sp
In our study there was no clear correlation between scores on the Lake Louise AMS questionnaire and the Sp
Study Limitations
I-STAT machines have well-defined operating parameters for temperature but no have calibration adjustments recommended for altitude. The devices are regularly used in hospital settings with elevations greater than 2134 m (7000 feet), although without reported inaccurate data. 9 ,19,20 Subjects were also moved into a room temperature environment before taking ABG samples, and this warmer environment may have resulted in changes in perfusion and blood flow to the extremities before ABG sampling. We did not compare the measured pH and temperatures or make attempts to correct for these variables in our data.21,22
Only unacclimatized individuals were used for this study to maximize the rate of detecting individuals with symptomatic AMS to correlate with oxygen saturation. We were unable to determine an accurate ascent profile as each participant was from a different coastal military command. Among our study population, the range of oxygen saturation was fairly narrow. It may be interesting to see whether the range would differ if acclimatized individuals were included.
Only one manufacturer of pulse oximetry devices was used, and it is likely these devices use very similar or identical algorithms to calculate the oxygen saturation. Algorithms used in other devices may be more sensitive to changes in temperature and altitude than those tested. Future investigations should compare different devices and different calculation algorithms.
Conclusions
Portable pulse oximetry devices can be a valuable medical tool used to provide greater insight into the development of high altitude sickness and lay the foundation for early identification and treatment interventions. Data from this study indicate that at an operating altitude of up to 2100 m (6900 feet), medical personnel may recommend the new generation of pulse oximeters for use in the field during high altitude operations to assist in clinical decisions. Furthermore, we have shown that the new generation of Nonin pulse oximeters give reliable and accurate Sa
Funding and Corporate Sponsorship
This study was funded by intramural sources and no financial support was provided by the Nonin Corporation for this study, although the tested devices were loaned by Nonin for this study. The pulse oximetry devices were chosen based on the prevalence of use in the current operational environment to ensure the greatest relevance.
Department of Defense Disclaimer
The views expressed in this article are those of the authors and do not reflect the official policy or position of the Department of the US Navy, Department of Defense, or the US government.
Footnotes
Acknowledgments
We would like to thank LT Darren Thomas for his help with the coordination of our study site and the US Marine Corps Mountain Warfare Training Center for their cooperation with our study. We would also like to thank Anna Dasher, RN, for all her hard work and assistance with the completion of this study. We would like to thank Robert Riffenburgh, PhD, for his assistance with the statistics and graphics for this study. We have no conflicts of interest to declare, devices were obtained through a cooperative research and development agreement, and we received no financial contributions from the makers of the devices tested. This article was completed in memory of LCDR Fred Trayers, a friend, a teacher, an outdoor enthusiast, and an inspiration to all those who knew him.
