Abstract
A 34-year-old man presented to a clinic at a ski resort in the Rocky Mountains at 9000 feet (2743 m) with shortness of breath and fatigue, a few days after arriving to altitude from sea level. He was found to be hypoxic with radiographic findings consistent with high altitude pulmonary edema (HAPE). He was treated with high flow oxygen, steroids, and calcium channel blockers and transferred to a lower altitude tertiary care hospital for intensive care unit monitoring and further treatment. During his diagnostic evaluation, he was found to have both a patent foramen ovale and influenza B infection. While patent foramen ovale is a known risk factor for HAPE, there is also some evidence that upper respiratory tract infections in general and influenza in particular may also be risk factors for HAPE. The 2 diseases may share an underlying pulmonary pathophysiology, as both cause noncardiogenic pulmonary edema and alveolar hemorrhage. We report an unusual case of influenza B virus compounded by previously undiagnosed patent foramen ovale, travel to high altitude, and subsequent development of HAPE.
Keywords
Introduction
High altitude pulmonary edema (HAPE) is a potential hazard to mountaineers, trekkers, skiers, tourists, and military personnel ascending to high altitudes. The incidence of HAPE depends upon several variables, including the altitude attained, speed of ascent, physical activity after arrival, gender, age, and individual susceptibility. According to a study from Peru, adults ascending to 12,408 feet (3782 m) experience a 3% incidence of HAPE. 1 Among Swiss climbers at 14,957 feet (4559 m), the incidence was 5.2%. 2 Subclinical pulmonary edema occurs in many more persons. For example, Hackett and Rennie 3 observed rales in 23% of trekkers in Nepal, of whom only 4.5% had clinical HAPE. Despite educational efforts directed toward prevention, early recognition, and treatment, approximately 20 reported deaths occur from HAPE annually throughout the world. 4
To better counsel patients and travelers about high altitude travel, it is important to know what physiologic and environmental characteristics may predispose certain persons to HAPE. One of these comorbidities is patent foramen ovale (PFO), a disease entity that is present in more than 25% of the general population. 5 Patent foramen ovale has been shown to be 4 times as common in subjects with HAPE, 6 but the exact pathophysiology of the association is still uncertain. 7 There are anecdotal reports, animal models, and small previously published series of infectious processes as a risk factor for HAPE. 8 -11 Influenza, specifically, has been associated with noncardiogenic pulmonary edema and pulmonary hemorrhage in particularly severe and fatal cases; this correlation may indicate an underlying common mechanism with HAPE, or may place persons with this particular infection at higher risk of complications if traveling to altitude. We report a case of influenza B virus compounded by previously undiagnosed PFO, travel to high altitude, and subsequent development of HAPE.
Case Presentation
A 34-year-old man presented to a clinic at a ski resort in the Rocky Mountains (base elevation 9000 feet [2762 m]) complaining of 2 to 3 days of progressive shortness of breath, malaise, fever, cough, and headache. Three days earlier he had arrived at this elevation from his sea level hometown in Maryland. One week before arrival, he had an upper respiratory infection with sinus congestion and nonproductive cough. His upper respiratory infection symptoms were present but improving on arrival to altitude; however, they began to worsen with exertion after several days. He skied on the first 2 days of his visit without difficulty, and had tried to ski the day before, but on the day of presentation he felt too short of breath to get out of bed. A friend became concerned that he looked “blue” and seemed confused and so brought him to the clinic for evaluation. He had no known medical problems, and denied a history of altitude illness on multiple prior ski visits to a similar altitude. He was not a smoker, did not use illicit drugs, and had no relevant family medical history. He had been taking the over-the-counter medications DayQuil (acetaminophen/dextromethorphan/phenylephrine) and Robitussin (guaifenesin) as directed.
On physical examination, the patient was a Caucasian male of average height and weight. His initial vital signs at the ski clinic were as follows: blood pressure, 128/70 mm Hg; heart rate, 76 beats/min; respiratory rate, 40 breaths/min; temperature, 101.1°F; oxygen saturation, 14% on room air. The marked hypoxia is documented in multiple places as having a reliable tracing on the monitor, and the recorded printout of vital signs from the clinic show an oxygen saturation of 22%. The patient appeared fatigued and in moderate respiratory distress, but was alert and oriented. His lung examination was remarkable for tachypnea, 2-word dyspnea, rhonchi in bilateral lower lung fields, and supraclavicular retractions. His skin was dusky, and his lips and fingers were cyanotic. His Glasgow coma scale score was 15, but he was slow to respond. The remainder of his physical examination including head, neck, heart, and abdomen was unremarkable. His chest radiograph (Figure) was interpreted as bilateral pulmonary edema or infiltrates—right greater than left—and a small right pleural effusion.
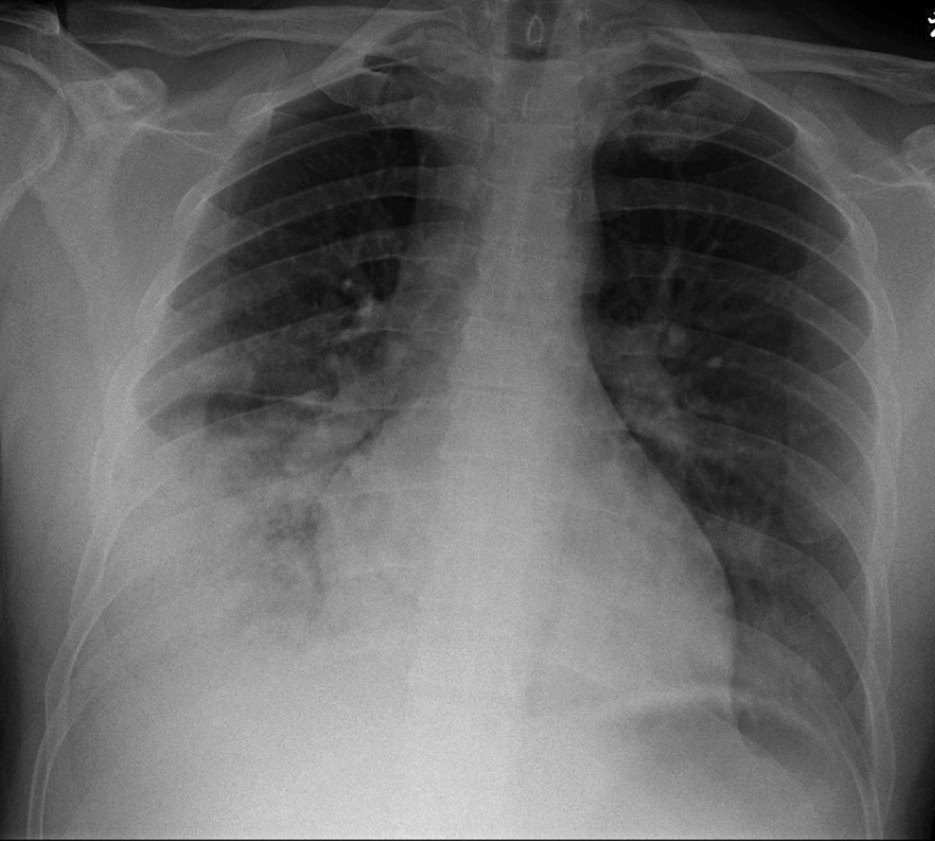
Chest radiograph showing bilateral pulmonary edema or infiltrates, right greater than left, and a small right pleural effusion.
The patient was placed on 15 L oxygen through a non-rebreather mask, which brought his oxygen saturation up to 85%. He received ketorolac 30 mg intravenously, dexamethasone 5 mg orally, acetaminophen 1000 mg orally, nifedipine extended release 30 mg orally, and ondansetron 4 mg intravenously. A transfer was arranged to the closest tertiary care facility both for descent and for further intensive treatment. En route, his oxygen saturation was maintained at approximately 90% on a 15 L facemask. When he arrived in the emergency department at Denver Health Medical Center (elevation 5280 feet [1609 m]), he was placed on continuous positive airway pressure with 100% fraction of inspired oxygen (Fi
During the inpatient diagnostic workup, given the patient's history of upper respiratory infection, he had an influenza swab obtained that was positive for influenza B, confirmed by polymerase chain reaction, at which point he was started on oseltamivir antiviral therapy. The remainder of his laboratory data was unremarkable. He also had a transthoracic echocardiogram, which showed normal left ventricular size and function, no valvular disease, and normal pulmonary artery pressures, but revealed a patent foramen ovale (PFO). On hospital day 2, he was transferred out of the medical intensive care unit to a floor bed on 3 L oxygen through nasal cannula, no longer requiring a facemask or positive pressure ventilation. His repeat chest radiograph at that time showed a residual small, right-sided pleural effusion without focal infiltrates or signs of edema. On hospital day 3, he was discharged to fly back home to Maryland, with instructions to complete a course of levofloxacin and oseltamivir.
Discussion
Although HAPE is a relatively rare diagnosis, it can be rapidly fatal if not recognized and treated appropriately. It is caused by the abnormal accumulation of fluid in the alveolar airspaces due to a breakdown in the blood-gas barrier within the lungs, triggered by hypobaric hypoxia, resulting in a patchy accumulation of extravascular fluid in the alveoli. The underlying mechanism is elevated pulmonary artery pressures (>40 mm Hg), which disrupt the alveolar-capillary barrier and cause leakage of fluid into the alveolar space. Factors known to be associated with an increased risk of HAPE development include preexisting conditions or anatomic abnormalities that lead to increased pulmonary blood flow or pulmonary vasoconstriction, such as primary pulmonary hypertension, congenital absence of 1 pulmonary artery, and left-to-right intracardiac shunts such as atrial and ventricular septal defects. A PFO in the setting of rising pulmonary vascular resistance during hypoxic pulmonary vasoconstriction may reverse the direction of blood flow, shunting blood from right to left and further exacerbating hypoxemia. 12 Patent foramen ovale has been found to be 4 times more common among HAPE-susceptible persons. 6 For this reason, patients with a known PFO should be counseled about their increased risk before ascending to high altitude. Our patient was unaware of having a PFO; while this is clearly a risk factor for the development of HAPE, his numerous prior visits to altitude without altitude illness or HAPE imply that his PFO alone is not enough to account for the development of HAPE in this instance.
Influenza is an important cause of mortality in the United States and globally. The Centers for Disease Control estimates that from the 1976–1977 season to the 2006–2007 flu season, annual flu-associated deaths in the United States ranged from a low of approximately 3000 to a high of approximately 49,000 people. 13 Prior studies on the effect of infection as a risk factor for HAPE have relied on small case series and animal models; animal models have shown that “priming” of the pulmonary endothelium with endotoxin and elevated levels of vascular endothelial growth factor by infection can lead to increased vascular permeability, leakage, and pulmonary edema,10,11 which may in turn cause the resulting greater mortality seen in humans with pulmonary infections at altitude.8,9 Influenza infection in particular has not been associated with HAPE; however, a higher mortality has been observed among people with pneumonia and influenza living at altitude when compared to their sea-level counterparts. 9 In particularly severe cases of the recent influenza A H1N1 outbreaks, there have been case reports of respiratory failure due to alveolar hemorrhage and noncardiogenic pulmonary edema (although these have not been reported with influenza B).14,15 The clinical manifestation is quite comparable to HAPE: marked hypoxia, audible rales, and radiographic findings of alveolar consolidation. This similarity may indicate an underlying common pathophysiology between the 2 processes, but further research into the role of pulmonary infection as a predisposing factor for HAPE remains to be done.
Although concomitant HAPE, PFO, and influenza is likely to be a rare combination, it seems our patient had all 3: an undiagnosed and previously asymptomatic PFO, a viral upper respiratory infection (which ultimately proved to be influenza B), and in the setting of a sojourn to high altitude, respiratory status and hypoxemia that worsened acutely and significantly, indicating a component of HAPE. Because the patient reported improving before his arrival at altitude, with subsequent worsening, we suspect that the combination of prior infection plus exposure to hypobaric hypoxia ultimately led to his clinical picture. The presence of a viral upper respiratory infection in the right season and the right setting should indicate caution for potential moderate to high altitude sojourners. The presence of an underlying infection might require very little exacerbation by hypobaric hypoxia to result in severe respiratory distress and pulmonary edema. In addition, patients with suspected HAPE should be queried as to recent infections and associated respiratory symptoms; if given early in the course of influenza, antiviral therapy may be beneficial. 16 Above all, patients should likely not ascend to moderate altitude until respiratory symptoms associated with influenza have resolved.
