Abstract
MULTIMEDIA
Six video recordings of feline SCI cases, both pre- and postoperative, are included in the online version of this article doi:10.1016/j.jfms.2011.09.006
TRAUMA AND ITS CONSEQUENCES
Spinal cord injury (SCI) is a common and devastating condition, both in humans and animals. Approximately 30,000 cases of SCI occur each year in the United States in humans. While there are no comparable figures in the veterinary field, in a retrospective post-mortem study exogenous SCI represented the third most common feline disorder; however, the actual figure is likely to be higher than even this suggests. 1,2
Primary SCI is the direct effect of exogenous or endogenous trauma, which results in concussion, contusion and/or physical laceration of the spinal cord. The primary injury triggers a cascade of secondary events (vascular changes, ion disturbances and excitotoxicity, free radical production and inflammation) with subsequent exacerbation of the original trauma. 3 Vascular changes caused by the secondary injury impair the autoregulatory mechanisms of the spinal cord. When coupled with systemic hypotension, this results in significant changes to spinal cord perfusion. 4
Most of the effort in the medical field is concentrated on reducing the cascade of secondary events. Unfortunately, no medical therapy has yet been definitively proven to be effective, either in humans or in animals. 3,5 In humans, appropriate use of methylprednisolone sodium succinate (MPSS) has been shown to produce some degree of improvement in motor function at 6 weeks and 6 months. 6 This benefit may not be evident in our patients because the level of improvement attributed to the use of MPSS may not be enough to produce a clinical improvement. Surgical decompression and stabilisation, with the aim of preventing further neural damage and relieving the compression from the spinal cord, still remains the main treatment protocol for the primary injury. 3,7,8 The timing of surgery is controversial but there is increasing evidence that early decompression and stabilisation is associated with shorter hospitalisation times and can help to reduce the cascade of secondary events. 5,9
EXOGENOUS SPINAL CORD INJURY
Vertebral trauma: fracture, luxation, subluxation
Exogenous trauma causing vertebral fracture, luxation and subluxation is an important cause of acute SCI in cats, representing 7% of spinal cord diseases according to one study. 2 However, that study involved a referral, post-mortem population, and so the actual proportion is likely to be higher. Most cats, in fact, are not referred because of the perceived poor outcome or are not treated due to financial constraints.
In most cases the trauma is caused by either a road traffic accident (RTA) or a fall from a height, but other causes such as gun shot wounds and bite wounds are also described. 10 –13 It is important to thoroughly evaluate any cat with suspected spinal trauma because of possible concomitant injuries and prioritise them accordingly. Between 30–83% of spine and spinal cord trauma cases have concomitant injuries (pulmonary contusions, pneumothorax, rib fractures, long bone fractures, pelvic fractures, abdominal organ injuries, haemoperitoneum and head trauma). 10,13,14
It is also very important to recognise the possibility of vertebral fracture or luxation at an early stage to avoid any risk of further neural damage during examination of the cat. 8 Even though SCI is rarely fatal, time is a key factor in terms of maximising the chances of a positive outcome. If practical, the cat can be secured to a flat board or a stretcher. Approximately 50% of all vertebral fractures and luxations/ subluxations involve the thoracolumbar junction (T10—L2); the lumbar region (31–67%) and sacrococcygeal junction (14–36%) are other commonly affected areas. 10,13,14 Fractures involving the cervical vertebrae are comparatively uncommon in cats (2%). 10 Approximately 20% of cats with traumatic spinal injuries also have acute intervertebral disc herniation secondary to trauma. 11
The examination and management of a cat with suspected spinal trauma should focus first on systemic stabilisation (see ABC box). Hypovolaemia and hypoxaemia must be recognised and treated immediately, as they can exacerbate secondary SCI. 3 It is paramount to assess ventilation, partial pressure of oxygen in arterial blood and systemic blood pressure in cats with SCI and to correct any deviation from normal. 3 Packed cell volume, total protein level, urea, creatinine and electrolytes should be evaluated as a minimum as soon as possible.
Spinal trauma ABC

Once the immediately life-threatening problems have been addressed, neurological examination should take place to localise and assess the severity of the lesion (Tables 1 and 2). Non-ambulatory cats should be handled very carefully until an unstable spinal lesion has been ruled out. The absence of nociception associated with vertebral fracture/luxation is a poor prognostic indicator (see box above). 11,12 In dogs with absent nociception following spinal fracture/luxation, the reported prognosis for functional recovery is <5% for a patient with displaced vertebrae and <25% if there is no displacement. 15 No similar data are available in cats.
Assessment of nociception

Differentiation between upper (UMN) and lower motor neuron (LMN) signs

Neuroanatomical localisation by neurological examination
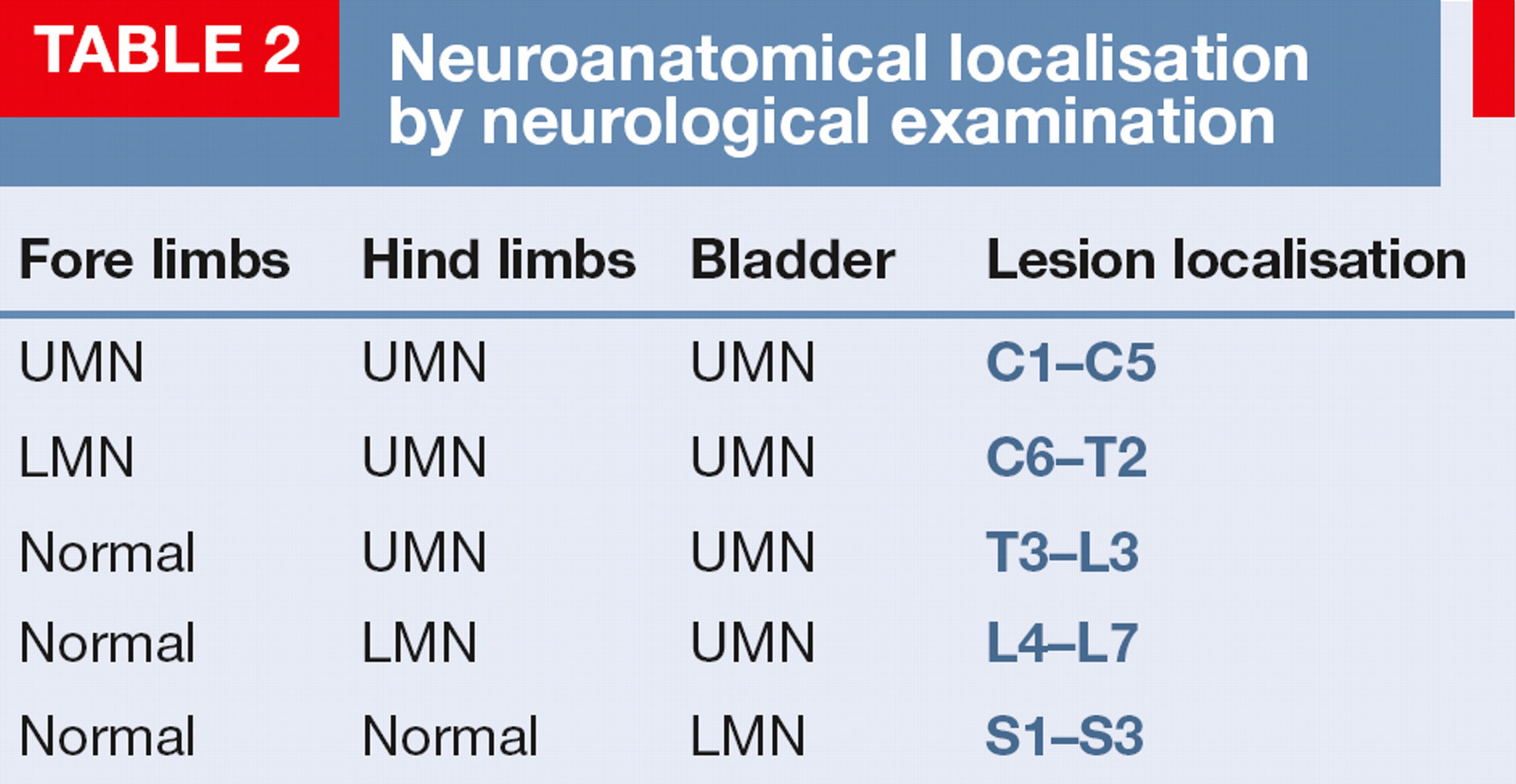
It is important to be aware of the possibility of multiple spinal cord lesions and /or spinal shock (see box below) while interpreting the neurological examination. Indeed, approximately 5–20% of cats with thoracolumbar fractures have a second spinal column fracture/luxation. 11,13 As a general rule, lower motor neuron (LMN) signs mask upper motor neuron (UMN) signs. For example, if there is a lesion between the L4 and S3 segments and a second lesion between T3 and L3, hindlimb segmental spinal cord reflexes will be reduced. This is because the LMN is the last in the chain of neurons that produces muscular contraction and directly controls the spinal cord segmental reflexes. Similarly lesions affecting the brainstem can mask the clinical signs caused by C1-C5 lesions. 8
Lateral survey radiographs of the whole spine should be obtained in any cat with suspected spinal trauma, 8 together with survey chest radiographs and abdominal radiographs or ultrasound. Anaesthesia is necessary for a diagnostic radiographic study, and the utmost care must be taken to prevent any further neural damage in the case of an unstable fracture. Thus, where possible, it is preferable to take ventrodorsal views with the cat in lateral recumbency (using a horizontal beam). Radiographs are sufficient to diagnose the majority of vertebral fractures and luxations, but cannot be reliably used to rule out an unstable vertebral lesion. 16 It should be borne in mind that radiography does not give any information about the amount of displacement that occurred at the time of injury. Strong paraspinal muscles could reduce the fracture/luxation to a less displaced or even a normal position by the time of radiography. While myelography could provide information over and above that provided by plain radiography about any compressive lesions of the spinal cord, the required positioning and manipulation to perform myelography could potentially worsen the neurological status of the patient.
Advanced imaging such as computed tomography (CT) or, preferably, magnetic resonance imaging (MRI) would be necessary for definitive evaluation of the integrity of or degree of damage to the spinal cord. CT has almost 100% sensitivity in detecting acute traumatic spinal lesions (Figs 1 and 2), is less financially restrictive and is quicker to perform than MRI; however, MRI provides more detailed and definitive information about the spinal cord parenchyma (Fig 3). 17
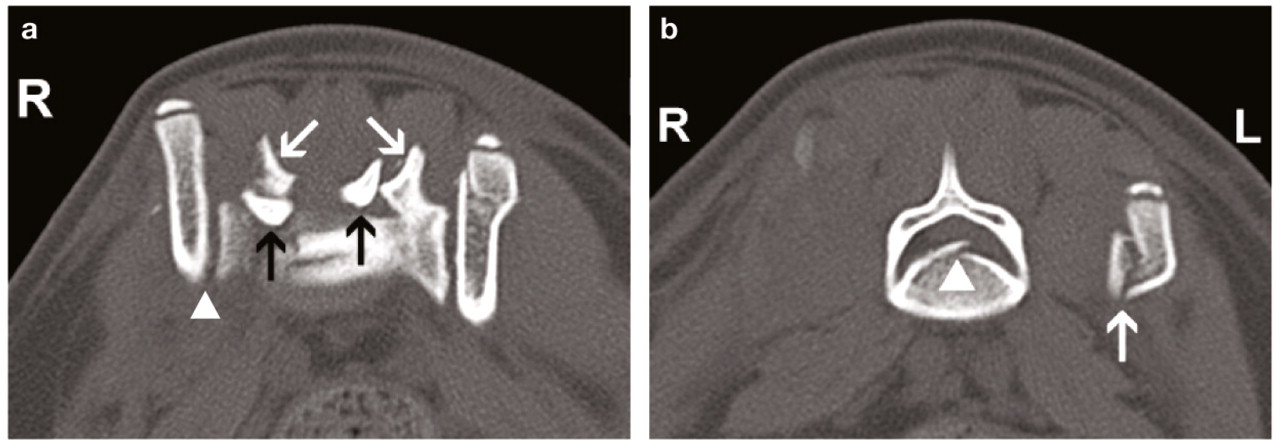
Transverse CT image (bone window) of an L7-S1 fracture and bilateral subluxation of the dorsal articular facets in a domestic short hair (DSH) cat. (a) Arrow-head indicates the right sacroiliac subluxation; white arrows indicate the dorsal articular facets of S1; black arrows indicate the dorsal articular facets of L7. (b) Fracture of the L7 body (arrow-head) with left sacroiliac fracture (arrow). Courtesy of Dr L Benigni, The Royal Veterinary College

CT images from a DSH cat. (a) Sagittal image showing a T12-T13 fracture (arrow) and traumatic intervertebral disc extrusion (IVDE) (arrow-head). (b) Transverse image showing a T12-T13 fracture of the right articular facets and traumatic IVDE in the vertebral canal (arrow-head). Courtesy of Dr L Benigni, The Royal Veterinary College

(a) Sagittal T2-weighted (T2W) MRI scan of a 3-year-old Oriental Shorthair cat with a T12 compression fracture (arrow) sustained during an RTA. (b) Transverse STIR MRI scan of the same cat; there is signal change within the spinal cord (arrow) and soft tissue swelling within the paraspinal muscles (arrow-head). (c) Scuffed nail and foot pad excoriation secondary to the trauma. A video clip of the cat, which was showing paraplegia (video 1), is available at doi:10.1016/j.jfms.2011.09.006
Spinal shock
Occurs immediately after severe SCI
Causes loss of spinal cord segmental reflexes caudal to the lesion, regardless of the localisation
Cutaneous trunci reflex is helpful with localisation if the lesion is between segments T3-L3
Signs of spinal shock disappear with 24–48 h
Lack of nociception associated with luxated/fractured vertebrae on radiographs suggests a hopeless prognosis.
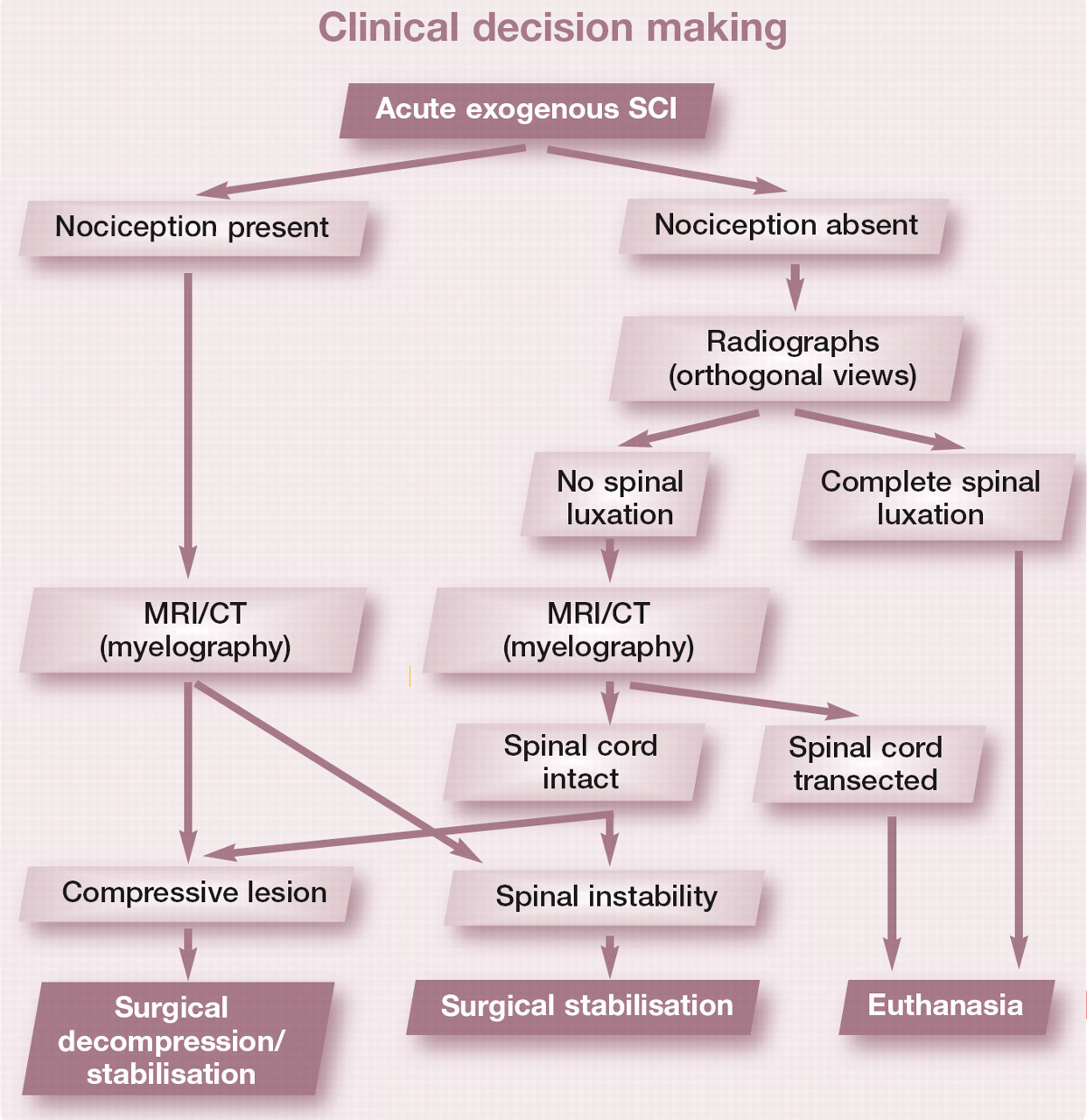
The benefits of advanced imaging aside, a fundamental point is that lack of nociception associated with luxated/fractured vertebrae on survey spinal radiographs suggests a hopeless prognosis due to the functional transection of the spinal cord implied by complete sensory deficits (see clinical decision-making box and Fig 4).

(a, b) Lateral and ventrodorsal radiographs showing a T8 fracture with displacement and spinal cord transection. Note the subtle change on the lateral projection compared with the gross spinal displacement visible on the ventrodorsal projection. (c) Lateral radiograph of a 2.5-year-old DSH cat showing an L6—L7 fracture with complete over-riding of L6 and L7 vertebrae; surgical exploration revealed spinal cord transection
Acute exogenous SCI

Treatment should be aimed at controlling the pain (Table 3); preventing further neural damage by physically securing the patient to a flat board or a stretcher, thereby ensuring full body stabilisation; limiting the extent of secondary SCI; decompressing the spinal cord; and stabilising the fracture/luxation when indicated.
Over the years a number of studies have investigated how to prevent and limit secondary injury. Many different therapeutic agents, including calcium channel antagonists, sodium channel antagonists, potassium channel blockers, polyethylene glycol and MPSS, have been investigated and some are under further investigation. 3 However, the basics such as maintaining adequate blood pressure and tissue oxygenation and decompression of the spinal cord still constitute the most effective therapy for acute SCI. 18,19 Use of glucocorticosteroids such as MPSS remains controversial and the beneficial effects are not fully proven in dogs and cats.
Analgesics commonly used in cats by the authors

NB Where appropriate, if licensed products are available they should be used first or non-licensed drugs can be administered according to the cascade.
Therapeutic plasma concentration of fentanyl is reached within 12 h in cats and stays in the analgesic range for up to 72 h after application of the patch.
NSAIDs should be used with care in hypovolaemic patients or those receiving glucocorticosteroids.
IM = intramuscularly, IV= intravenously, SC = subcutaneously, PO = by mouth, ICP = intracranial pressure
No clinical study has evaluated the efficacy and side effects of MPSS in traumatic spinal cord injury in cats.
MPSS – the conflicting evidence
MPSS — the conflicting evidence MPSS has been shown to have some efficacy in human patients, but only when treatment was initiated within 3 h of trauma at 30 mg/kg followed by 5.4 mg/kg/h for 24 h; or, alternatively, initiated within 3–8 h of trauma at 30 mg/kg followed by 5.4 mg/kg/h for 48 h. 6 However, as mentioned, the small degree of motor improvement attributed to this protocol in humans may not be significant in our patients. Thus, MPSS is contraindicated if more than 8 h have elapsed since the traumatic event, and in patients that have voluntary motor function. 18 Moreover, one retrospective study reported a high incidence of adverse effects after dexamethasone administration in dogs with thoracolumbar disc herniation, and no difference in outcome was found compared with dogs treated with surgery alone. 20
The treatment of choice in any cat with SCI and associated spinal instability and/or spinal cord compression is surgical (Fig 5). When compared with patients managed conservatively, early decompression and stabilisation is associated with a significant improvement in neurological status. 7,14 In carefully selected rare cases with no spinal instability, conservative treatment has been reported to allow good outcomes. 10,11,14 However, a consideration during the decision-making process is that it is often easier and less painful to care for animals with vertebral fracture/luxation soon after surgical stabilisation than it is to provide optimal nursing care for animals that must be rested or retain external coaptation devices for prolonged periods. 8

(a) Ventrodorsal radiograph of an L5—L6 fracture/subluxation in a 2-year-old DSH cat. (b) Ventrodorsal postoperative radiograph of the same cat
Sacrococcygeal and intercoccygeal vertebral luxations
Sacrococcygeal and intercoccygeal vertebral luxations are well recognised and common injuries in cats that have suffered an RTA and are thought to result from traction on the tail (Fig 6). 8,21 Typically there is devastating central nervous system (CNS) or peripheral nervous system (PNS) injury, which relates to the fact that, in cats, the spinal cord extends to the first sacral vertebra and the cauda equina to the first coccygeal vertebra. 22 The clinical signs and prognosis depend on the extent of the injury to the spinal cord and PNS (nerve rootlets, cauda equina and peripheral nerve) and could range from a paralysed tail, and urinary and/or faecal incontinence to partial loss of sciatic nerve function or even complete pelvic plexus dysfunction (Fig 7). 21 Fractures/luxation of sacrococcygeal vertebrae should be stabilised as soon as possible to prevent further traction injury to the cauda equina. 21 Because of the size of the coccygeal vertebrae, amputating the tail immediately would provide a simpler and more viable option.
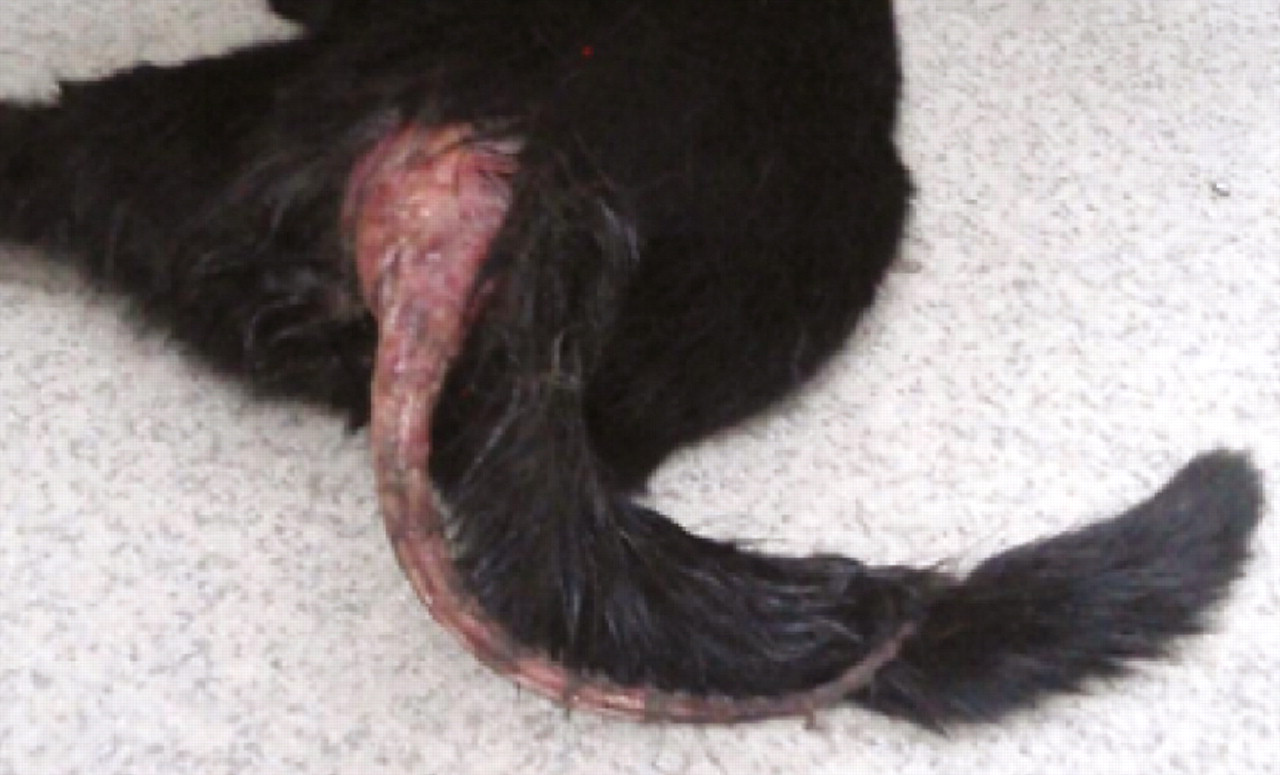
Tail traction injury with extensive soft tissue involvement in a 4-year-old DSH cat

Intercoccygeal luxation in a 4-year-old DSH cat. This cat subsequently made a full recovery; his perineal reflex and tail base sensation were intact on presentation. A video clip of a cat with sacrococcygeal luxation (video 2) is available at doi:10.1016/j.jfms.2011.09.006
Many affected cats present with difficulty in urination as a result of temporary or permanent damage to the pudendal and/or pelvic nerves (Fig 8). Although anatomically the lesion is affecting the LMN, the bladder may be difficult to express due to increased sympathetic tone secondary either to pain or to trauma at the level of the sympathetic sacral spinal chain. 23 In these cases, and in cats that have developed reflex dyssynergia (detrusor contraction combined with inadequate urethral relaxation), it is mandatory to provide the necessary pharmacological aids during the recovery period. Various drug combinations can be used for this purpose (see box below). In animals in which there has been complete severance of the nerves (neurotmesis) that supply the bladder sphincter mechanism, the prognosis for recovery of bladder function is hopeless (Fig 9). 24 Cats with tail base sensation on presentation are expected to regain urinary continence; reportedly, 60% of cats without tail base sensation on presentation could also regain their urinary continence. 24 The prognosis for return of urinary continence is good if anal tone and perineal sensation are present on initial examination. Cats that do not become continent within 1 month usually fail to regain urinary function. 25

Neuroanatomy of the bladder. The hypogastric nerve originates from the L2—L5 spinal cord segments in cats and controls the relaxation of the detrusor muscle and contraction of the internal urethral sphincter during the urine storage phase. The pudendal nerve originates from the S1—S3 spinal cord segments and provides sensory and motor innervation to the external urethral sphincter. The pelvic nerve controls contraction of the detrusor muscle during voiding. The possible area of damage to the spinal cord in the case of traction injury secondary to sacrococcygeal luxation is shown in red. Intact spinal cord is shown in white

(a) Sagittal T2W MRI scan of an 8-year-old cat showing signal change, loss of integrity of the spinal cord at L7 level (arrow-head) and suspected severe spinal cord damage at the level of the origin of the sacral nerves (arrow). (b) Sagittal STIR image of the same cat showing transection at the level of the origin of the pelvic and pudendal nerves (arrow-head) and soft tissue damage (arrow) ventral to the tail
UMN bladder
Pharmacological intervention
Smooth muscle relaxants
Phenoxybenzamine (0.25–0.5 mg/kg PO q12h)
Prazosin (0.25–0.5 mg/cat PO q8–12h)
Striated muscle relaxants
Dantrolene (0.5–2 mg/kg PO q8h)
Diazepam (0.2–0.5 mg/kg PO q4–8h)
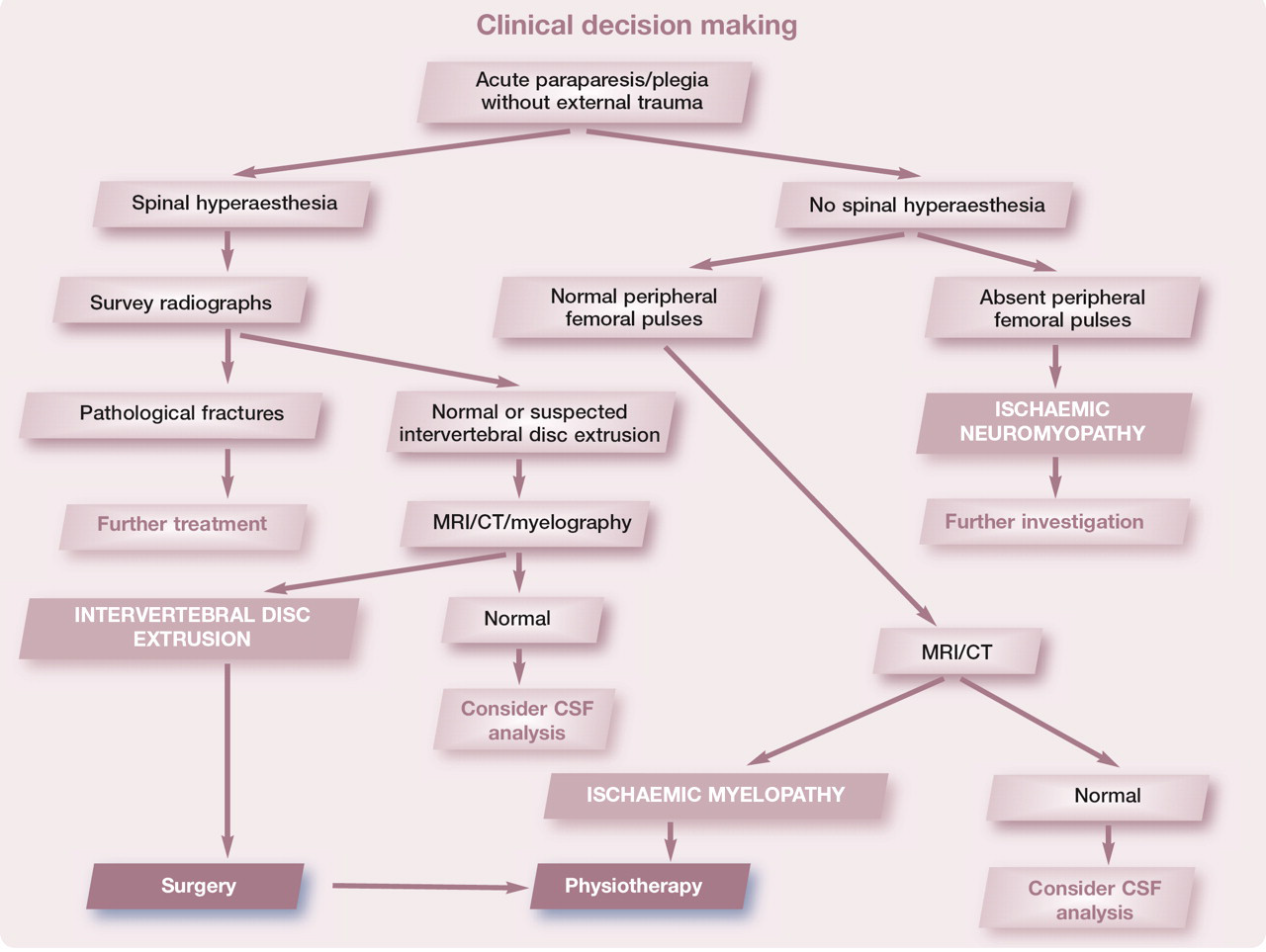
ENDOGENOUS SPINAL CORD INJURY
Intervertebral disc extrusion
Intervertebral disc disorder is not a common presentation in cats. Both acute intervertebral disc extrusion (IVDE) and chronic intervertebral disc protrusion (Hansen type I and II, respectively) have long been reported to occur in cats. 26 Because the incidence of feline IVDE has been reported to be as low as 0.09%, 27 many veterinarians are still not aware of the occurrence of IVDE in cats. It is certainly less common than it is in the canine population, in which the incidence has been reported to be as high as 2%. 28 The age of cats with IVDE is usually about 8 years (range 1.5–17 years). 29 There is no gender predilection and the majority of feline IVDE cases described in the literature have been domestic shorthair cats. 27,28,30 The onset of neurological signs is variable, but usually acute-progressive. 27,29 The most common intervertebral spaces involved are T13—L1 and L4—L5, but L7—S1 involvement and cervical IVDE have also been reported. 29 –33
As always the neurological signs depend on the neuroanatomical localisation. Signs include spinal hyperaesthesia, reluctance to jump, urinary and faecal incontinence, ataxia, and limb and tail paresis (see video 3, doi:10.1016/j.jfms.2011.09.006). 27,30 –38 A practical five-point system has been proposed for grading IVDE in the thoracolumbar area in dogs (Table 4), and is particularly useful in trying to elicit information about the expected outcome. 36 Grades 1–4 have a very good prognosis after decompressive surgery, with more than 90% of dogs making a successful recovery; in comparison only about 60% of dogs with grade 5 IVDE will recover successfully. 36 This system has not yet been evaluated in cats; however, it can be used as a rough guide for the clinician dealing with a suspected case of feline IVDE (Table 4).
The investigation of a cat with suspected IVDE starts with the signalment and the collection of a thorough history. Particular attention should be paid to any suspected trauma, and other causes of spinal cord lesions should also be considered during the investigation (ie, neoplastic, inflammatory, infectious or vascular diseases). Thorough neurological examination will help localise the lesion to specific spinal cord segments and also helps in constructing working diagnoses and further diagnostic plans (see box, page 857). The quality of the femoral pulses, pad temperature and colour of the nail beds should be assessed in a cat with acute-onset paraparesis or paraplegia in order to rule out aortoiliac thromboembolism as a cause of the ischaemic neuromyopathy. Haematology and biochemistry should be performed in order to assess the safety of general anaesthesia and surgery, if required. Jugular sampling should be performed with especial care in tetraparetic cats.
Cerebrospinal fluid (CSF) analysis in cats with IVDE may be normal, or may reveal albuminocytological dissociation or an elevated nucleated cell count, and therefore using this test without further imaging might lead to an incorrect diagnosis and is not helpful in diagnosing IVDE in the cat. 30
After appropriate neuroanatomical localisation, survey spinal radiographs of the area of interest may be useful in cases of suspected trauma and acute onset of neurological signs. 13 Mineralised radiodense material inside the vertebral canal, with associated narrowing of the intervertebral disc space, can be seen in some cases (Fig 10). 27,34,35 Myelography has greater sensitivity in identifying the compressive extradural lesion of the spinal cord in dogs; 37 however, feline data are lacking. Myelography is not always diagnostic, however, 27,30 and further advanced imaging modalities should be considered in cats with suspected IVDE. In dogs, CT reportedly shows similar sensitivity to myelography in diagnosing IVDE, 38 but no large-scale studies have been published regarding CT sensitivity in cats. CT might reveal a hyperattenuating compressive lesion, especially in cases where the intervertebral disc is mineralised. 27,33
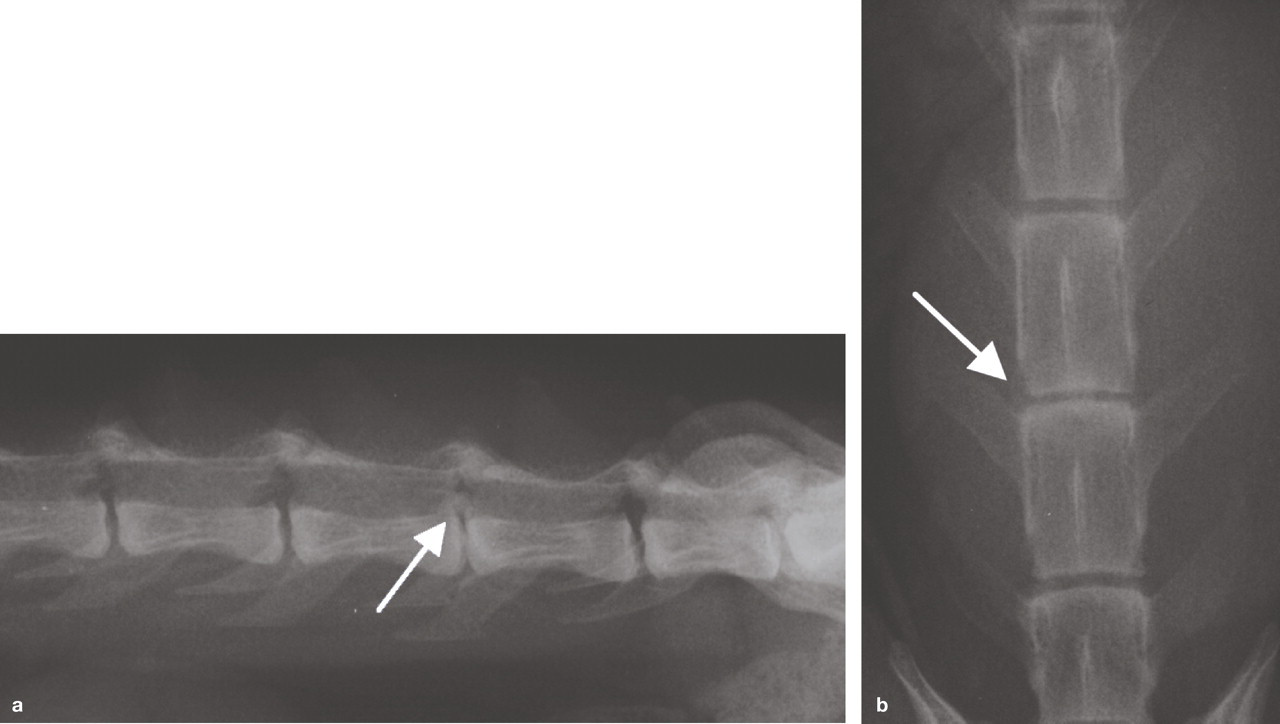
Survey radiographs of the lumbar vertebral column of a DSH cat; (a) lateral projection and (b) ventrodorsal projection. There is narrowing of the L5—L6 intervertebral space with calcified material in the L5—L6 intervertebral foramen (arrow). MRI scans from this cat are shown in Fig 11

MRI scans of the DSH cat radiographed in Fig 10. (a) Sagittal T2W image showing the hypointense extradural material associated with the L5—L6 intervertebral space and compressing the spinal cord ventrally. (b) Transverse T2W image of the L5—L6 intervertebral space showing the hypointense extradural material displacing the spinal cord to the left side. Both images are characteristic of IVDE, which was also confirmed during surgery (Fig 12)

Clinical grading system for SCI

From Sharp and Wheeler 36
Acute endogenous SCI
MRI provides better contrast resolution than any of the above-mentioned imaging modalities and is therefore the ‘gold standard’ for imaging intervertebral discs. 17 MRI usually reveals extradural material compressing the spinal cord above the affected intervertebral space (Fig 11). 39 Intramedullary migration of the intervertebral disc and focal intramedullary oedema associated with extrusion of a small volume of intervertebral material have also been reported in cats. 31,35 The only disadvantages of MRI and CT are accessibility and cost. However, the cost—benefits of these advanced imaging modalities outweigh other less expensive diagnostics by aiding assessment of the spinal cord and allowing more accurate planning of further therapy. The morbidity that often accompanies myelography is also avoided with CT/MRI. 17
Conservative and surgical therapy are both recognised treatment options, as in dogs. 36 The scattered information from published cases of feline IVDE seems to suggest that surgical treatment has better success, 29 and it is also the authors' treatment of choice for any cat with spinal cord compression and associated neurological deficits and/or persistent pain. Cats are more difficult to restrain for conservative treatment than dogs and thus early decompression might prevent further deterioration of neurological signs and improve the prognosis. However, in selected cases conservative management may also be an option to consider from the outset. 31 The standard surgical techniques of hemilaminectomy (Fig 12), dorsal laminectomy and ventral slot are used, depending on the site of the lesion. 27,30,32
Post-surgical management is tailored to the patient's needs and includes management of the urinary bladder, postoperative analgesia, nursing and physiotherapy. The prognosis is generally good for a full return to normal routine (see video 4, doi:10.1016/j.jfms.2011.09.006).
Feline ischaemic myelopathy
Feline ischaemic myelopathy (FIM) has generally been considered to be a rare condition in cats. However, the reported occurrence has increased in recent years, probably due to increased accessibility to MRI and awareness of this condition among veterinary surgeons. 29 As in dogs, FIM is suspected to be secondary to fibrocartilaginous embolism (FCE), but can only be definitively diagnosed on postmortem histological examination. 40 There are several reports in the veterinary literature of cats and other non-domestic felids with suspected or confirmed FCE; 40 –47 however, the exact incidence of FCE has not yet been investigated in feline patients.
Neurological signs occur due to obstruction of the spinal vessels by a small amount of nucleus pulposus from the intervertebral disc, causing an ischaemic necrotising myelopathy. 41 Several mechanisms have been suggested to explain how the degenerated intervertebral disc material enters the bloodstream in humans 48 and dogs. 49
Affected cats are usually middle aged and older, with no breed or gender predisposition. 29 The most commonly affected site in cats seems to be the C6—T2 spinal cord segments; however, other sites can be affected. 50 An explanation for this neuroanatomical localisation is currently lacking; however, an association with protruded intervertebral discs frequently found in that area of the spinal cord has been proposed. 51 The neurological signs vary according to the localisation, 29 and are usually lateralised. This might be due to the particular vascularisation of the feline spinal cord: approximately half of the vertical arteries branch either to the left or to the right side, whereas the other half run centrally (Fig 13). Furthermore, it has been found that there are more unilateral arteries in the cervical area compared with the rest of the spinal cord. 52

(a) Diagram of the spinal cord blood supply. The outer part of the spinal cord is supplied by the radial arteries and the inner part is supplied by the vertical artery. The middle part is supplied by both the radial and vertical arteries. Modified from King. 57 (b) Transverse STIR image showing a hyperintense intramedullary lesion lateralised to the right side, suggesting infarction of the portion of the spinal cord supplied by the radial arteries on the right side (arrow)
Nursing and supportive care for a cat with SCI
Fluid therapy for hypovolaemic cats with trauma and SCI

NB All doses are recommended for use in cats without cardiopulmonary disease. Patient parameters and trends should be continuously monitored: heart rate, pulse quality, arterial blood pressure, oxygen saturation, temperature (core and extremities), ECG, capillary refill time, chest auscultation and urine production. IV = intravenous

Nonambulatory cat with indwelling urinary catheter and closed urinary drainage system
FIM is very acute in onset and not progressive, which can differ from IVDE where owners usually report an acute onset, but often with progressive deterioration. Moreover, ischaemic myelopathy usually presents as a hyperacute, non-painful myelopathy and is the main differential in those cats without spinal hyperaesthesia (see video 6, doi:10.1016/j.jfms.2011.09.006). However, in some cases, cats, similar to dogs, can show discomfort or pain during the initial phase. 45,49 In the absence of external trauma, ischaemic neuromyopathy caused by aortic or iliac thromboembolism is another mimic of acute-onset myelopathy that needs to be ruled out; paraparesis/paraplegia has been reported with this common feline condition and the neurological signs may deteriorate within the first 24 h, usually with gradual improvement thereafter. 49 Reported clinical signs of thromboembolism include a weak or absent femoral pulse, cold limb extremities, cyanotic nail beds and painful muscles. 53,54
MRI is the diagnostic imaging modality of choice when assessing a cat with non-painful myelopathy. 17 Typically on MRI FIM presents as a focal well-defined T2-weighted and short-tau inversion recovery (STIR) hyperintense intramedullary lesion affecting either grey or white matter and often with obvious lateralisation (Figs 13 and 15). 44,46 Contrast enhancement on T1-weighted images might be observed after 5–6 days. 44

MRI scans of the cervical spinal cord of a cat. (a) Sagittal STIR image showing a hyperintense intramedullary lesion at the level of C5 (arrow). (b) Sagittal T2W image showing a hyperintense intramedullary lesion at the level of C2 (arrow). Both images are suggestive of ischaemic myelopathy in the light of a peracute presentation
Treatment consists of supportive therapy, urinary bladder management, nursing and physiotherapy. In dogs, the prognosis depends on the severity of the neurological signs at the time of presentation and the extent of the lesion on MRI. The involvement of intumescences, presence of symmetrical clinical signs and decreased nociception have also been found to be associated with a poor prognosis. 55,56 An equivalent study is, however, lacking for feline patients.
KEY POINT
Always perform a thorough physical examination to assess concurrent injuries in a cat with spinal cord injury (SCI).
ABC stabilisation is the first priority and duty.
Neurological examination and localisation is mandatory prior to imaging.
Securing the patient to a stretcher or flat board is necessary to avoid further neural damage in the case of an unstable fracture/luxation.
Advanced imaging is irreplaceable for the appropriate treatment of SCI.
Early decompression and stabilisation is paramount for spinal fracture/luxation.
Analgesia is an important and ethical part of the management of SCI.
Consider euthanasia only in the case of absent nociception in association with marked spinal displacement.
Intervertebral disc extrusion and fibrocartilaginous embolism should be included as differential diagnoses in cats with SCI.
Physiotherapy forms a significant part of rehabilitation after SCI.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
