Abstract
A 13-year-old male neutered domestic shorthair cat presented with an acute onset of dyspnoea. Thoracic radiographs revealed marked, bilateral, caudal lung lobe consolidation. A diagnosis of anatomically mixed T-cell lymphoma with pulmonary, renal and alimentary involvement was confirmed on histopathology. Pulmonary involvement in cases of feline lymphoma is uncommon and the radiographic appearance of pulmonary lymphoma is highly variable. Lung lobe consolidation has been described with primary lung tumours in cats, but not previously in association with pulmonary lymphoma. This unusual presentation serves to alert practitioners to the possibility of lymphoma as a cause of severe bronchopulmonary disease in the cat.
Lymphoma is the most frequently diagnosed malignancy in cats accounting for up to 40% of all feline neoplasms.1–3 Lymphoma can be classified according to anatomic location, histological criteria (mitotic rate and appearance of the neoplastic lymphocytes) and immunophenotype.4,5 Anatomical categories of feline lymphoma include abdominal, nodal, mediastinal, atypical and mixed. Atypical lymphoma refers to the involvement of any non-lymphoid tissues including the central nervous system, skin, eyes, mammary glands, lungs, larynx or nasal cavity. The mixed form of lymphoma includes any combination of two or more of the other forms.4,6 Pulmonary involvement in feline lymphoma is rare.4,7 This report describes an unusual presentation of anatomically mixed T-cell lymphoma with renal, alimentary and pulmonary involvement resulting in marked, bilateral, caudal lung lobe consolidation.
A 13-year-old male neutered domestic shorthair cat presented with a 2-week history of lethargy and a 5-day history of inappetence, intermittent vomiting and dyspnoea. At presentation, the cat was in severe respiratory distress with open-mouth breathing and tachypnoea (respiratory rate 60 breaths per min). The cat had a restrictive pattern of respiration with a marked increase in inspiratory effort and paradoxical abdominal wall movements. Thoracic auscultation revealed harsh lung sounds bilaterally that were loudest over the left caudodorsal hemithorax. A mass was also palpable in the mid-cranial abdominal region, caudal to the liver. The remainder of the physical examination was unremarkable. The cat's respiratory distress stabilised after supplemental oxygen by mask and the cat was sedated with butorphanol (Torbugesic; Fort Dodge) (0.2 mg/kg intravenously) for thoracic radiography. There was complete loss of aeration of the left caudal lung lobe with no evidence of gas within the bronchi of this lobe and decreased aeration of the right caudal lung. There was also evidence of a low-volume pleural effusion and mild cardiomegaly (vertebral heart score 9) (Fig 1). On thoracic ultrasonography, both caudal lung lobes were non-aerated (hepatised). The bronchi within these lobes had markedly thickened walls and contained anechoic intraluminal material. A low-volume pleural effusion was confirmed within the dependent pleural space. A limited echocardiographic examination was performed. Cardiac chamber size and ventricular wall thickness were within reference intervals. 8 Ultrasound-guided, fine needle aspirate biopsies of the consolidated lung lobes and a sample of the pleural fluid were collected for analysis.

Right lateral and dorsoventral thoracic radiographs. There is marked bilateral caudal lung lobe consolidation and a low-volume pleural effusion on the right side at the level of the fifth and sixth intercostal spaces and ninth and tenth intercostal spaces, and on the left side at the level of the seventh and eighth intercostal spaces (white arrowheads). There is free gas separating the caudal lung lobes from the diaphragm due to iatrogenic introduction of gas or pulmonary perforation during collection of ultrasound-guided fine needle aspirate samples (black arrowheads).
Serological tests to detect feline immunodeficiency virus antibody and feline leukaemia virus p27 antigen were negative (Snap FIV/FeLV Combo Test; Idexx laboratories). Cytological examination of the lung aspirates revealed erythrocytes, scattered, low numbers of macrophages and moderate numbers of monomorphic medium-to-large round cells (20 μm diameter), with occasional mitoses, large round or oval, often nucleolated, nuclei and a modest rim of deeply basophilic cytoplasm containing a juxtanuclear pale zone. Pleural fluid analysis was consistent with a modified transudate (total protein 35 g/l, nucleated cells 700 × 106/l, erythrocytes 90,000 × 106/l). Cytological examination of the pleural fluid revealed 38% large vacuolated macrophages, 50% neutrophils, 7% small lymphocytes, 1% eosinophils and 4% medium-to-large round cells similar to those seen in the lung aspirates. Cytological findings were highly suggestive of round cell neoplasia, specifically lymphoma. Further diagnostic and therapeutic procedures including abdominal ultrasonography and aspiration cytology of the abdominal mass were declined and the owner elected euthanasia.
At post-mortem examination the caudal lung lobes had a pallid solid appearance and failed to collapse bilaterally (Fig 2). The airways within the caudal lung lobes were markedly thickened and a mucosal pseudomembrane was present. The tracheobronchial and sternal lymph nodes were enlarged. A low-volume blood-tinged effusion was present in both the pleural and peritoneal cavities and the heart was grossly normal. The kidneys were normal in size but several firm, white, nodular lesions (10–15 mm diameter) were detected in the right and left renal cortices. Enlarged mesenteric lymph nodes, with the largest measuring 5 cm × 3 cm, were identified as the palpable abdominal mass. On histopathology, a heavy infiltrate of large round cells was identified in the caudal lung lobes, mesenteric lymph nodes, small intestine and kidneys. The infiltrative round cells were large with large round, irregular or cleaved nuclei containing large prominent central nucleoli and an open to vesiculate chromatin pattern. There was a small to moderate amount of basophilic to eosinophilic cytoplasm, and moderate anisokaryosis and cellular pleomorphism. Mitoses were common (>10/high power field) with numerous abnormal mitoses noted. In the caudal lung lobes, the neoplastic infiltrate was strikingly restricted to the vicinity (including the epithelium) of the moderate-to-large airways and blood vessels (Fig 3). There was widespread congestion and oedema with large numbers of alveolar macrophages present and scattered free red cells in alveolar spaces. In the cranial lung lobes, the round cell infiltrate was absent while the congestion, oedema and alveolar macrophages were similar to the caudal lobes.

Gross post-mortem appearance of the lungs. Note the pallid caudal lung lobes which failed to collapse (arrows) (a) and the thickened bronchial walls (arrow) (b).
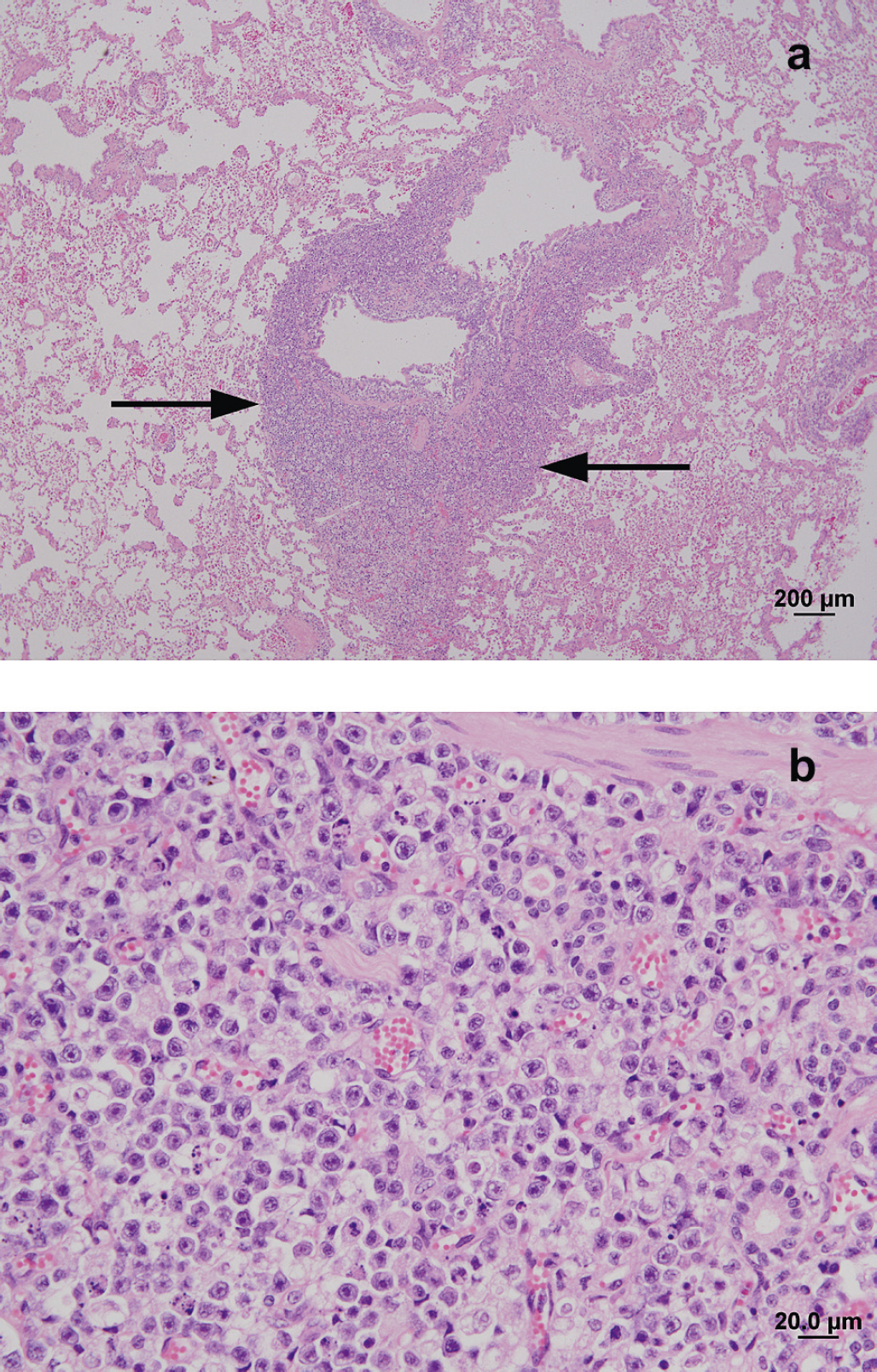
(a) Photomicrograph of a haematoxylin-eosin stained section of the left caudal lung lobe. Note the extensive cellular infiltrate of neoplastic lymphocytes (arrows) surrounding the segmental bronchus. (b) Higher magnification of the peribronchial neoplastic infiltrate, comprising large round cells with large round, irregular or cleaved nuclei, large prominent central nucleoli and an open to vesiculate chromatin pattern.
In the kidneys, there were multifocal interstitial neoplastic round cell infiltrates. In the small intestine, while there was no evidence of gross thickening or nodular mass lesions at necropsy, a similar neoplastic round cell infiltrate extended from the lamina propria into the submucosa, tunica muscularis and serosal tunic. In the mesenteric lymph nodes the normal lymph node architecture was completely effaced by the neoplastic large round cells which extended through the capsule and into the mesentery. There was no evidence of neoplastic infiltration in the liver, spleen, bone-marrow or heart. On immunohistochemical staining (CD3 and CD79a) 90–95% of the neoplastic lymphocyte population stained positive for CD3. A diagnosis of anatomically mixed, high-grade T-cell lymphoma was made.
Discussion
The restrictive respiratory pattern, increased inspiratory effort and harsh lung sounds in this case were most suggestive of bronchopulmonary disease. In a cat of this age, bronchopulmonary neoplasia, infection or congestive heart failure were considered the major differentials. 9 The ultrasonographic and radiographic findings of caudal lobar pulmonary consolidation were unusual and an inflammatory or neoplastic aetiology was suspected.
The radiographic appearance of primary pulmonary neoplasia in cats is variable. The most commonly diagnosed primary lung tumours in cats are adenocarcinomas and carcinomas of bronchial or bronchoalveolar origin. The radiographic findings most frequently described for these tumours include solitary or multiple pulmonary nodules/masses and/or pleural effusion. 10 Lobar consolidation involving one or more lung lobes is less common.11,12 Radiographic evidence of pulmonary consolidation has been reported in 15–30% of feline primary lung tumours, including adenocarcinomas, bronchoalveolar carcinomas, anaplastic carcinomas, squamous cell carcinomas and also in lymphomatoid granulomatosis in cats.11–14 The latter is a slowly progressive diffuse ornodular angiocentric lymphoproliferative disorder with a propensity to involve pulmonary parenchyma. 14 The lobar pulmonary consolidation in our case was a striking radiographic finding and demonstrates that lymphoma should be included amongst the differential diagnoses for this sign.
Although lymphoma is well recognised as a cause of dyspnoea in cats, this is typically because of mediastinal rather than bronchopulmonary involvement. In a report of 15 dyspnoeic cats with severe primary pulmonary parenchymal disease, lymphoma was the cause of dyspnoea in only two cats. Both cats had mediastinal involvement as well as masses and/or nodules present in all lung lobes. 15 Well-demarcated nodules and/or masses within the pulmonary parenchyma are the most commonly described radiographic feature of feline pulmonary lymphoma.4,15–17 Pulmonary masses and/or nodules (3/7 cats), a bronchial pattern (3/7 cats) and an unstructured interstitial pattern (1/7 cats) were all described in a recent study. 16 An alveolar pattern was also reported in one cat with concurrent pulmonary lymphoma and a lung lobe torsion of the cranial portion of the left cranial lung lobe. 16 This contrasts the situation in dogs where pulmonary lymphoma was most commonly associated with an unstructured, interstitial pattern (10/16 dogs) followed by an alveolar pattern (5/16 dogs), masses and/or nodules (5/16 dogs) and a bronchial pattern (1/16 dogs). 16 Pulmonary involvement is reported in more than 60% of dogs with lymphoma, while pulmonary involvement in cats with lymphomais unusual.16,18 Pulmonary involvement was reported in less than 3% of cases of feline lymphoma in one study and accounted for less than 3% of all the atypical lymphoma cases in another.4,7
In humans, primary pulmonary lymphoma arising from the mucosa-associated lymphoid tissue is rare, accounting for less than 1% of all cases of lymphoma. The criteria for diagnosing primary pulmonary lymphoma in humans are that the lung and/or bronchus are involved without evidence of mediastinal or extrathoracic lymphoma prior to, at, or for 3 months following diagnosis.16,19 Primary pulmonary lymphoma has not been described in the cat or dog. Secondary pulmonary lymphoma may occur by direct extension, via haematogenous or lymphatic dissemination from distant sites or from foci of lymphoid tissue within the lung parenchyma itself.16,19 Histologically, primary pulmonary lymphoma in humans has a peribronchovascular distribution while the histological appearance of secondary pulmonary lymphoma is more variable. 16 The histological appearance of pulmonary lymphoma in cats is not well characterised. In the case reported here, the dense infiltrate of neoplastic lymphocytes was strikingly restricted to the vicinity of the medium-to-large airways and moderate-sized blood vessels. A similar histological pattern has been reported in a case of large granular lymphocyte lymphoma in a cat. 20
This case represents an unusual clinical and radiographic presentation of lymphoma due to extensive bronchopulmonary neoplastic infiltrates. Lymphoma should be considered in the differential diagnosis of cats with severe bronchopulmonary disease.
