Abstract
A 7-month-old male neutered cat was referred for paraparesis and painful sensation at the level of T13 vertebra where a dermal cyst was observed. Spine radiographs and magnetic resonance imaging (MRI) showed a well-encapsulated cyst communicating with the meninges and spinal cord, suggestive of hydromyelia and myelodysplasia. Dorsal laminectomy was performed and the cyst was completely removed. The day after surgery, the cat was ambulatory paraparetic. Involuntary defecation was observed for only a few days. The surgical specimen was cystic and covered by skin. Microscopic examination revealed a hollow hemispheric mass of glial fibrillary acidic protein (GFAP)-positive neural tissue lined by ependyma and formed of glia and vascular structures consistent with myelomeningocele (MMC). Only anecdotal descriptions of MMC have been published in the veterinary literature, mainly in the lumbosacral spinal cord. To the authors’ knowledge, this is the first report of a MMC with tethered spinal cord syndrome in a cat successfully treated surgically.
A 7-month-old male neutered cat was referred for neurological evaluation. The owner indicated that the cat has never been able to jump normally and, occasionally, attempts to move could induce the cat to cry out, which was interpreted as a response to an acutely painful stimulus. On clinical examination, a palpable dermal cyst at the level of T13 vertebra was detected (Fig 1) and the neurological examination revealed pelvic limb ataxia and ambulatory paraparesis. The postural reactions were decreased and the segmental spinal reflexes were increased in the pelvic limbs. Thoracolumbar hyperaesthesia and allodynia were present. The neuroanatomical localisation was suggestive of T3–L3 transverse myelopathy.
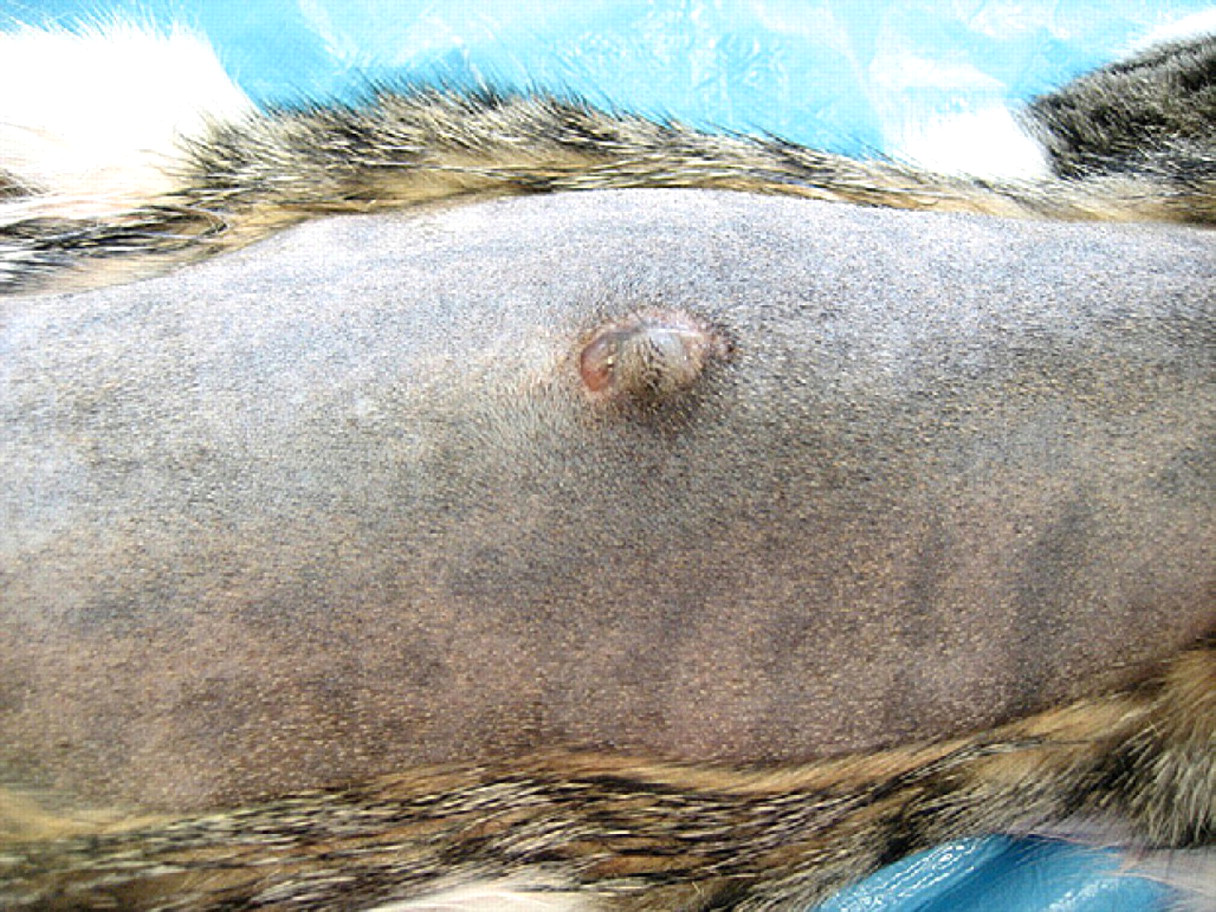
Skin clipped at surgical preparation revealing the dermal cyst.
Plain ventrodorsal radiographs of the thoracolumbar spine were characterised by a radiolucent oval bone defect of the caudal aspect of the spinous process of T13 (spina bifida) and dorsal lamina (Fig 2). The lateral projection of the thoracolumbar spine showed a focal lucency of the caudal aspect of the dorsal lamina of T13. Magnetic resonance imaging (MRI: Hitachi Aperto, 0.4 Tesla) was performed. T1- and T2-weighted sequences demonstrated an encapsulated cutaneous cyst at the level of T13 vertebra, communicating with the subarachnoid space through a defect of the dorsal lamina of T13. The thoracolumbar spinal cord presented features suggestive of hydromyelia and myelodysplasia. The dorsal subarachnoid and epidural spaces were mildly dilated. T1-weighted images after intravenous injection of 1 ml of gadoterate meglumine (Dotarem; Guerbet SA) showed signal enhancement of the cystic wall and tissues leading to the spinal cord (Figs 3 and 4).

Ventrodorsal radiographic projection of the thoracolumbar spine. The caudal aspect of the spinous process of T13 appears to bifurcate caudally (arrows), outlining the cranial margin of a radiolucent oval defect of the dorsal lamina.

Sagittal pre-(A) and post-contrast transverse (B) T1-weighted images. There is continuity of tissue between the spinal cord and the cutaneous mass. Markers on the skin indicate the cranio-caudal extent of the cutaneous mass. A focal hypointense signal originates from the cutaneous mass, suggestive of the presence of fluid.

Markers on the skin indicate the cranio-caudal extent of the cutaneous mass. Sagittal (A) and transverse (B) T2-weighted images. There is a communication between the subarachnoid space and the cutaneous mass through a defect of the dorsal lamina. The cutaneous mass is hyperintense, suggesting that it contains fluid. The dorsal epidural and subarachnoid spaces are enlarged likely due to tethering to the skin.
The surgical treatment consisted of a T12–T13 dorsal laminectomy, durotomy and myelotomy, and the cyst was completely removed. Dura regeneration matrix (Durepair; Medtronic) was placed over the dural defect prior to standard closure. Analgesia was provided throughout the procedure with a multimodal approach, it included: medetomidine (0.004 mg/kg) and methadone (0.2 mg/kg) administered intramuscularly as preanaesthetic medication, preoperative lumbosacral extradural injection of morphine (0.1 mg/kg, total volume 0.6 ml), intraoperative intravenous constant rate infusion of ketamine (0.3 mg/kg loading dose and 0.01 mg/kg/min) and fentanyl (0.003 mg/kg loading dose and 0.005 mg/kg/h), meloxicam (0.2 mg/kg) was administered intravenously at the end of the procedure. General anaesthesia was uneventful, monitored variables (including heart rate, arterial blood pressure, end tidal carbon dioxide, haemoglobin saturation and rectal temperature) were stable within normal physiological ranges. Good control of nociception was confirmed by absence of sympathetic surges during surgical stimulation. Ketamine infusion was continued postoperatively (0.005 mg/kg/min) for 12 h and an intravenous constant rate infusion of medetomidine (0.001 mg/kg/h) was started just prior to the recovery and carried on for 24 h postoperatively. The day after surgery, the cat was ambulatory paraparetic and with good urinary control; however, involuntary defecation was observed for few days. Ketamine and medetomidine were both stopped postoperatively and neurogenic pain was controlled with methadone, meloxicam and gabapentin. Cardiorespiratory parameters, temperature and analgesic requirements were regularly evaluated and eventually the cat was discharged after 1 week of hospitalisation.
On neurological follow-up at 1 and 3 months the cat was showing continuous improvement, with no more signs of hyperaesthesia and allodynia. However, the cat was still ambulatory paraparetic. At the 12 and 24 months re-examination the owner reported that the cat was able to jump and run freely as never before and neurologically, the cat displayed only pelvic limb ataxia.
A follow-up MRI examination was performed 5 weeks after surgery. T1- and T2-weighted sequences were obtained and revealed that the dorsal subarachnoid and epidural spaces remained enlarged and the spinal cord looked smaller and more hyperintense than before surgery (Fig 5). On post-contrast T1-weighted images, enhancement was present within the subcutaneous tissue in the surgical area.
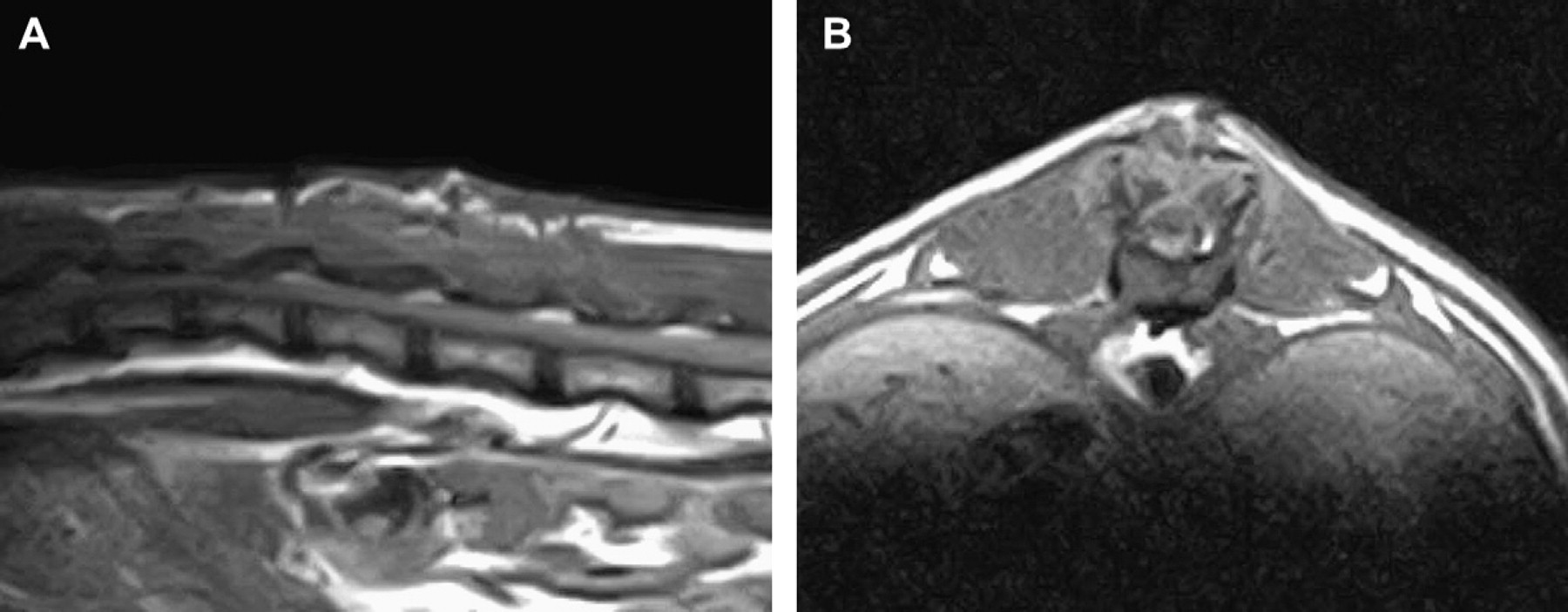
Sagittal (A) and transverse (B) T1-weighted images obtained after surgery to remove the cutaneous mass and its tract to the spinal cord. The dorsal subarachnoid and epidural spaces are still enlarged, and the spinal cord looks smaller and more hyperintense than before surgery. Tissue scarring at the surgical site is manifested by irregular skin outline and subcutaneous heterogeneous signal.
Surgical specimens, consisting of a cyst wall and the overlying skin, were fixed in 4% buffered formalin, longitudinally split and normally processed for histology. Serial 4 μm tissue sections were stained with haematoxylin and eosin for standard morphological evaluation. Selected sections were chosen for Goldner trichrome and Luxol fast blue stains. For immunohistochemistry (IHC), tissue sections were placed on positively-charged slides (SuperFrost Plus; Menzel-Gläser, Braunschweig, Germany). Endogenous peroxidase activity was quenched through immersion in 3% H2O2 methanol solution and unspecific binding was reduced with a 25% normal goat serum solution in phosphate buffered saline Tween 20 (PBST). Rabbit anti-glial fibrillary acidic protein (GFAP) (1:1000, Dako, Carpinteria, CA), vimentin (1:200, Novocastra Laboratories, Newcastle, UK), desmin (prediluted, Ventana Medical Systems, Tucson, AZ, USA), and mouse anti-NeuN (1:1500, Millipore, Billerica, MA, USA) and phosphorylated neurofilaments (2F11, Dako, Carpinteria, CA) were used as primary antibodies. The reaction was detected after incubation with a secondary antibody conjugated with multiple peroxidases (Universal ImmPress, Vector Labs, Burlingame, UK) and addition of a chromogen (DAB, brown, ImmPact Dab; Vector Labs, Burlingame, UK). Negative controls were obtained by omission and substitution of the primary antibodies and appropriate tissues were used as positive controls.
Histologically, the epithelium was poor of dermal adnexa and raised by a cyst. Abundant, disorganised and vascularised tissue expanded the subcutis. The cystic lumen was surrounded by a continuous layer of cuboidal cells and a thick band of variably intermixed tissues (Fig 6). The inner layer of the cystic wall, including the cyst epithelium, was characterised by a strong GFAP immunoreactivity (Fig 7). Dorsally, islands and streams of GFAP-positive tissues were intermingled with immature connective tissue, abnormal vessels, and groups of myocytes, melanocytes and thick cords of meningeal tissue (Fig 8). Within the inner layer of the cystic wall, anti-2F11 and anti-NeuN antibodies allowed detection of neuronal axons and ganglional cell bodies, respectively.
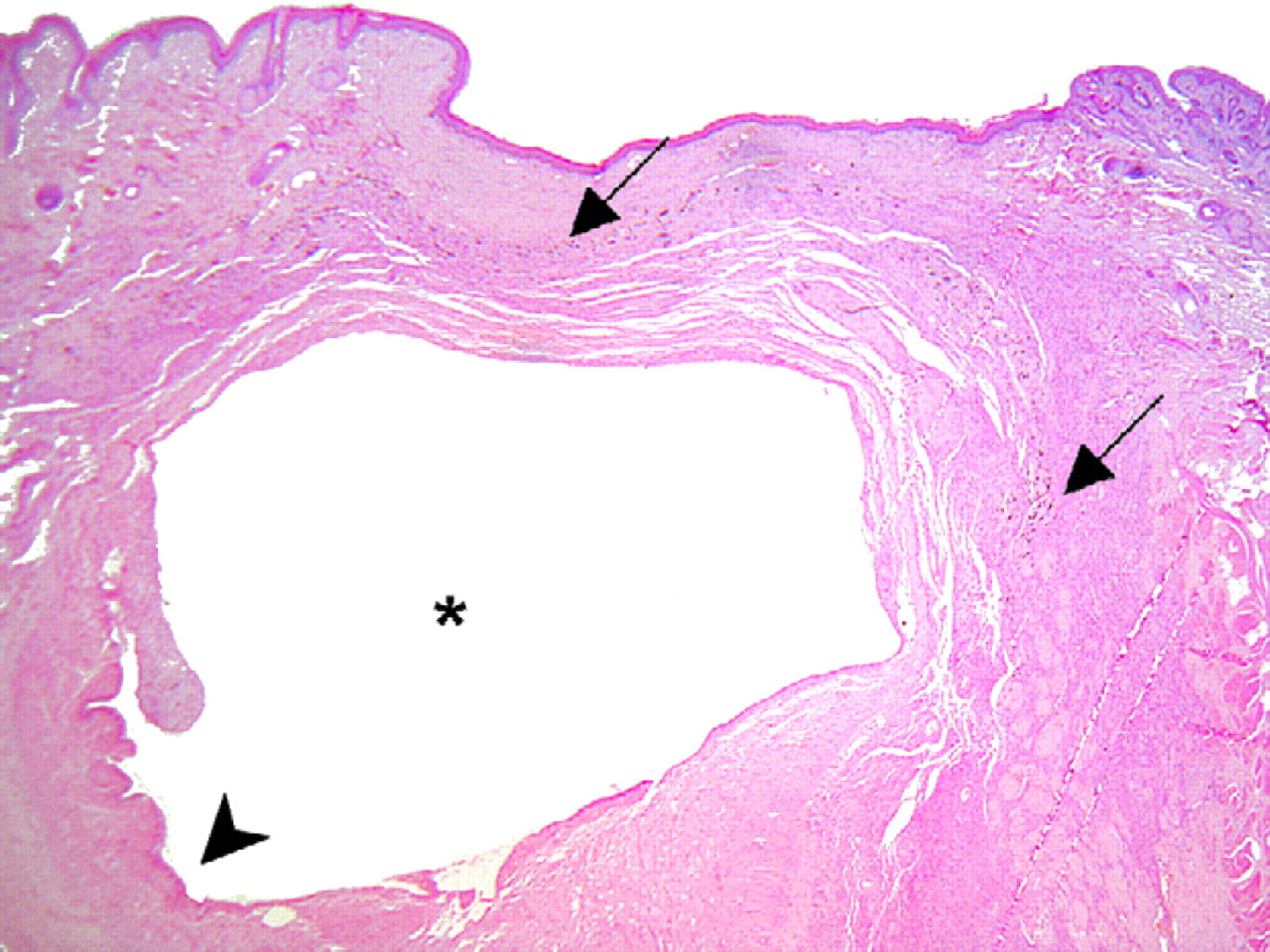
Beneath the skin there is a cystic cavity (asterisk) lined by neuroglial tissue (arrowhead) and surrounded by loosely textured collagen tissue. Scattered melanocytes (arrows) are visible within the deep dermis. Haematoxylin and eosin, 25×.

Anti-GFAP immunohistochemistry. The lumen of the cystic cavity is lined by a positive cylindrical epithelium (arrowheads), consistent with ependyma. Within the dense GFAP-positive stroma, whorls of negatively stained fibrovascular tissue are evident (asterisks). ImmPress universal method, DAB-horseradish peroxidase and haematoxylin counterstain, 125×.

Islands of dark blue stained neuroglial tissue (arrows) are intermingled with bundles of light green stained mature collagen. Goldner's trichrome stain, 320×.
The histological evaluation of the surgical specimen demonstrated that the dorsal aspect of the cystic lesion, covered by epidermis, was constituted by a mixture of different tissues, consistent with the ‘area medullovasculosa’. 1,2 Only the inner portion of the cystic wall and the overlying skin showed structural continuity. The extruded spinal cord parenchyma, presumably consistent with the dorsal half of the spinal cord, was reduced to a thin band of compact glial tissue with rare neurons and neurofilaments, ventrally lined by ependyma-like cells. The sub-epidermal arrangement of different tissues in islands and whorls reflected the incomplete penetration of the histological elements derived from the somitic mesenchyma between the surface ectoderm and neural tube. 3 Moreover, the presence of numerous melanocytes within the ‘area medullovasculosa’ is consistent with a blocked centrifugal migration of melanocyte-precursors from the neural folds. On the basis of the clinical, imaging, surgical and histopathological data, a diagnosis of myelomeningocele (MMC) was achieved.
MMC is a congenital spinal cord malformation included in a large group of congenital neural tube defects that result from a combination of genetic and environmental predisposing factors. 3 The term MMC refers to the protrusion of spinal cord parenchyma and meninges through a dorsal fissure of the vertebral arch, the consequence of incomplete fusion of neural folds during the embryonic neural tube formation. 4 The extruded tissues are variably intermixed in a highly vascularised area (‘area medullovasculosa’) and are arranged in a cystic cavity filled by cerebrospinal fluid. 1,2 The incomplete development of neural parenchyma at the site of extrusion, the compression atrophy by the cystic content, and the progressive traction on cranial spinal parenchyma during physiological postnatal vertebral elongation (‘tethered cord syndrome’) are responsible for the development of neurological deficits. 5
In human beings, the prevalence of MMC is around 3.3% of 10,000 live births. Thus, MMC is consistently associated with abortion and stillbirth and it is responsible for a poor prognosis due to progressively worsening neurological deficits. 3
Although MMC has been experimentally induced in animal models, 6,7 naturally occurring MMC has only been rarely diagnosed in veterinary medicine and the lesion was mainly localised at the lumbosacral spinal cord segments. 5,8–11 The aetiology of MMC includes a complex interaction between genomic and environmental predisposing factors. In cats, the experimental administration of griseofulvin during pregnancy and of toxic compounds, such as methylmercury and ethylenethiourea, were capable of inducing MMC in kittens. 12 The high prevalence of neural tube defects within Manx cats, characterised by a breed-specific sacrocaudal dysgenesia, supports the hypothesis of a highly favourable genetic background. 13 During neurulation, the meeting and fusion of the neural folds is associated with the closure and differentiation of the overlying tissues. The term ‘spina bifida’ indicates a single or multiple defect of the vertebral arch, characteristically located along the lumbar tract and associated with neurological dysfunctions. 13
Commonly, at the site of the MMC defect, a palpable mass is clinically appreciable as a dome-shaped protrusion of meninges (meningocele) and spinal cord parenchyma (MMC) beneath the dermis. Frequently, the attachment of the meninges to the dermis exerts traction on the spinal cord (tethered spinal cord), especially during postnatal axial skeleton elongation. Subsequent cranial ischaemic lesions can cause worsening of the neurological condition due to massive and progressive upper motor neuron system involvement. 5
Imaging a patient with spinal cord anomalies can be performed by various methods, including ultrasound, radiography, computerised tomography (CT) and MRI. Ultrasound is used in humans to diagnose fetal neural tube defects and associated vertebral abnormalities 14 but to the authors’ knowledge, there are no reports of its use in veterinary medicine in the same context. In the present case, ultrasound was not performed, and although it might have been possible to detect the presence of fluid in the subcutaneous cyst, it was judged unlikely that the information gained would have led to sufficient characterisation of the lesion, such as that obtainable with MRI. Plain radiography is known to have low sensitivity for the diagnosis of soft tissue anomalies of the spinal cord, but can be useful in detecting bony abnormalities of the vertebrae when the disease is sufficiently advanced. 15 Myelography could be useful in demonstrating anomalies that are communicating with the subarachnoid space, but cross-sectional imaging methods such as CT and MRI are, on the other hand, non-invasive and able to distinguish tissue types. CT has an excellent spatial resolution, and is, therefore, indicated when small areas of mineralisation, gas, or early haemorrhage are present. MRI provides excellent contrast resolution, which makes it the preferred imaging method for most neurological diseases. 15 Interestingly, in humans, it has been shown that prone MRI can provide additional information, such as anteroposterior movement of the medullary cone in patients suspected of having a tethered cord and in whom supine MRI were unremarkable. 16
Over the last 20 years, the prevalence of MMC and related neural tube defects among human beings has been reduced by supplementation of high doses of folic acid before conception. 4 However, the prognosis for MMC is poor and the life-expectancy for an affected infant is reduced, due to persistent and irreversible neurological deficits. 3,17
Early diagnosis, associated with fetal and neonatal surgery, is a key factor in the reduction of the severity and progression of neurological signs in human patients. 17 Surgical repair of MMC, in veterinary medicine, was previously reported only in an English bulldog, 5 a Yorkshire Terrier, 8 and a Manx cat with a related sodium deficiency syndrome. 18 In those reports and in our cat, the neurological condition improved significantly after surgery. It is likely that the use of multimodal approach to control perioperative nociception has contributed to fasten the recovery. Administration of analgesic drugs acting with different mechanisms and at different anatomical sites in the pain pathway permits lowering of the doses of each single agent, thereby reducing the chances of unwanted effects. It also provides an overall better control of nociception due to a synergistic action of the different molecules. 19
The Manx cat represents one of the most flagrant examples of breeding induced malformation. Most of the kittens are culled soon after birth or early in the life due to the presence of severe malformation – sacrocaudal meningocele, meningomyelocele, myelodysplasia – leading to severe sensory motor and excretory neurological deficits incompatible with the life. The perpetuation of a tailless Manx cat is unethical and inhuman breeding practice considering that selecting for a Manx cat with normally developed tail could readily prevent all these deficits.
In conclusion, when a slowly progressive postnatal ambulatory deficit in the hindlimbs and tail is observed, in association with urinary/faecal incontinence and a dorsal axial abnormality, a suspicion of neural tube defect could be sustained. The surgical approach, as we reported in a feline case of MMC, allowed a significant neurological improvement leading to a normal physical activity and good quality of life. To the authors’ knowledge, this is the first report of thoracolumbar MMC with tethered spinal cord syndrome occurring in a cat successfully treated surgically. However, the surgical outcome and prognosis remain difficult to predict, due to the limited ability of central nervous system to compensate severe lower and upper motor neuron damages.
Footnotes
Acknowledgements
We acknowledge the contributions of Susan Bravo Rodriguez for the MRI examination, and Lisa Baroncini for the histological preparations.
