Abstract
An 11-year-old castrated male domestic medium hair cat was presented with neurological signs consistent with a right thalamocortical lesion. Computed tomography (CT) images revealed a heterogeneously, hyperattenuating, poorly contrast enhancing intra-axial mass within the right lateral ventricle. The histological diagnosis at post-mortem examination was vascular hamartoma with hemorrhage and necrosis. This is the first report of a vascular hamartoma affecting the thalamocortex in a geriatric cat. Also, this is the first time that CT images of a feline cerebral vascular hamartoma have been reported.
An 11-year-old castrated male domestic medium hair cat was presented to the Ohio State University Veterinary Medical Center for evaluation of a 1-week history of lethargy, changes in sleep habits, and disorientation, and a 2-day history of vision loss. The cat was current on vaccines and was negative for feline leukemia and feline immunodeficiency viruses.
On presentation, the cat showed dull mentation. Physical examination was unremarkable. Neurological examination revealed inappropriate mentation consisting of disorientation, absent menace in the left eye and decreased menace in the right eye, with bilateral normal pupillary light reflexes (PLRs). The remainder of the cranial nerve examination was unremarkable. Postural reactions (lateral tactile placing and hopping) were mildly delayed in the right thoracic and pelvic limbs. Spinal reflexes and cutaneous trunci reflex were normal. Spinal palpation revealed mild discomfort at the level of the thoracolumbar junction and diffusely throughout the lumbar spine. Based on the history and the neurological findings, a right thalamocortical lesion with possible extension across midline, or significantly increased intracranial pressure was suspected. Involvement of the optic chiasm was also considered as a possible explanation for the bilateral visual deficits. Differential diagnoses included neoplasia (meningioma, glioma, ependymoma, lymphoma) and inflammatory diseases, either infectious (toxoplasmosis, cryptococcosis, feline infectious peritonitis (FIP), non-FIP viruses, bacterial) or non-infectious (immune-mediated). Ophthalmological examination revealed a normal fundus and confirmed the menace deficits and normal PLRs consistent with central blindness.
A complete blood count showed mild neutrophilia (11×109/l, reference interval 3–9.2×109/l) and mild monocytosis (0.7×109/l, reference interval 0–0.5×109/l) consistent with a mild inflammatory leukogram. A biochemical profile revealed mildly increased blood urea nitrogen (34 mg/dl, reference interval 13–30 mg/dl) and hypercalcemia (11.3 mg/dl, reference interval 8.4–10.1 mg/dl). Ionized calcium was normal. A non-invasive Doppler blood pressure was normal. Thoracic radiographs were unremarkable.
The cat was anesthetized for computed tomography (CT) (GE Lightspeed Ultra) of the brain and cerebrospinal fluid (CSF) collection. Anesthesia consisted of premedication with intravenous midazolam (0.2 mg/kg) and hydromorphone (0.05 mg/kg), induction with intravenous propofol (3 mg/kg) and maintenance with inhalatory isofluorane using mechanical ventilation. The CT study consisted of 1.3 mm contiguous transverse acquisitions, pre- and post-contrast administration (iohexol, Omnipaque 240 mg/ml, dose: 2 ml/kg IV). A multilobulated, irregularly marginated, heterogeneously hyperattenuating mass was evident located within the right lateral ventricle, extending into the left lateral ventricle (Fig 1A, B). Both lateral ventricles (right more than left) and the left olfactory bulb recess appeared dilated (Fig 1C). There was a marked midline shift with left-sided deviation of the falx cerebri (Fig 1C). The mass showed poor contrast enhancement after intravenous contrast administration (Fig 1D, E). CSF collection was not attempted due to concerns about increased intracranial pressure and risk for herniation. Differential diagnoses for the mass included neoplasia and less likely granuloma (such as fungal granuloma). An intra-axial origin of the mass was considered more likely, with choroid plexus tumor and ependymoma strongly considered due to the ventricular involvement of the mass; however, a glial cell neoplasm with erosion into the ventricle could not be excluded. A meningioma arising from arachnoid cells in the choroid plexus, though rare, was also considered. The cat recovered uneventfully from anesthesia. Surgical excision, radiation therapy and palliative medical treatment options were discussed with the owners, and palliative corticosteroid therapy was elected. The cat was discharged with oral prednisolone (0.5 mg/kgPOq12 h) and famotidine (0.5 mg/kgPOq12 h). The cat improved temporarily with corticosteroid therapy. Approximately 8 weeks after diagnosis, the patient's neurological signs worsened and he became anorectic, at which time the owners elected humane euthanasia. A complete post-mortem examination was performed. At necropsy, there was a 6×6×4 mm, discrete, dark red, cavitated lesion which filled the rostral portion of the right lateral ventricle and extended across midline into the left lateral ventricle. Both lateral ventricles were mildly dilated. No other gross abnormalities were noted. Microscopically, the lesion consisted of a well-demarcated expansile mass composed of variably sized blood filled spaces within the choroid plexus of the right lateral ventricle which extended into the left lateral ventricle and adjacent cerebral parenchyma (Fig 2A). These spaces were lined by flattened spindle cells that were morphologically similar but of two distinct immunophenotypes (Fig 2B). The predominant population was immunohistochemically positive for Factor VIIIa (Von Willebrand factor), interpreted as endothelial cells (Fig 2C). The second population was positive for smooth muscle actin (SMA), interpreted as pericytes (Fig 2D). The lining cells were either supported by a collagenous stroma (Masson's Trichrome positive), or by closely spaced intervening astrocytes (fibrous astrocytosis, Glial fibrillary acid protein (GFAP) positive) (Fig 2E). Multifocally, the tissue surrounding the blood filled spaces contained many clusters of hemosiderin laden gitter cells and deeply basophilic fragments of mineral, indicating chronic hemorrhage and necrosis. A diagnosis of focal vascular hamartoma with chronic hemorrhage and mild secondary obstructive hydrocephalus was made.
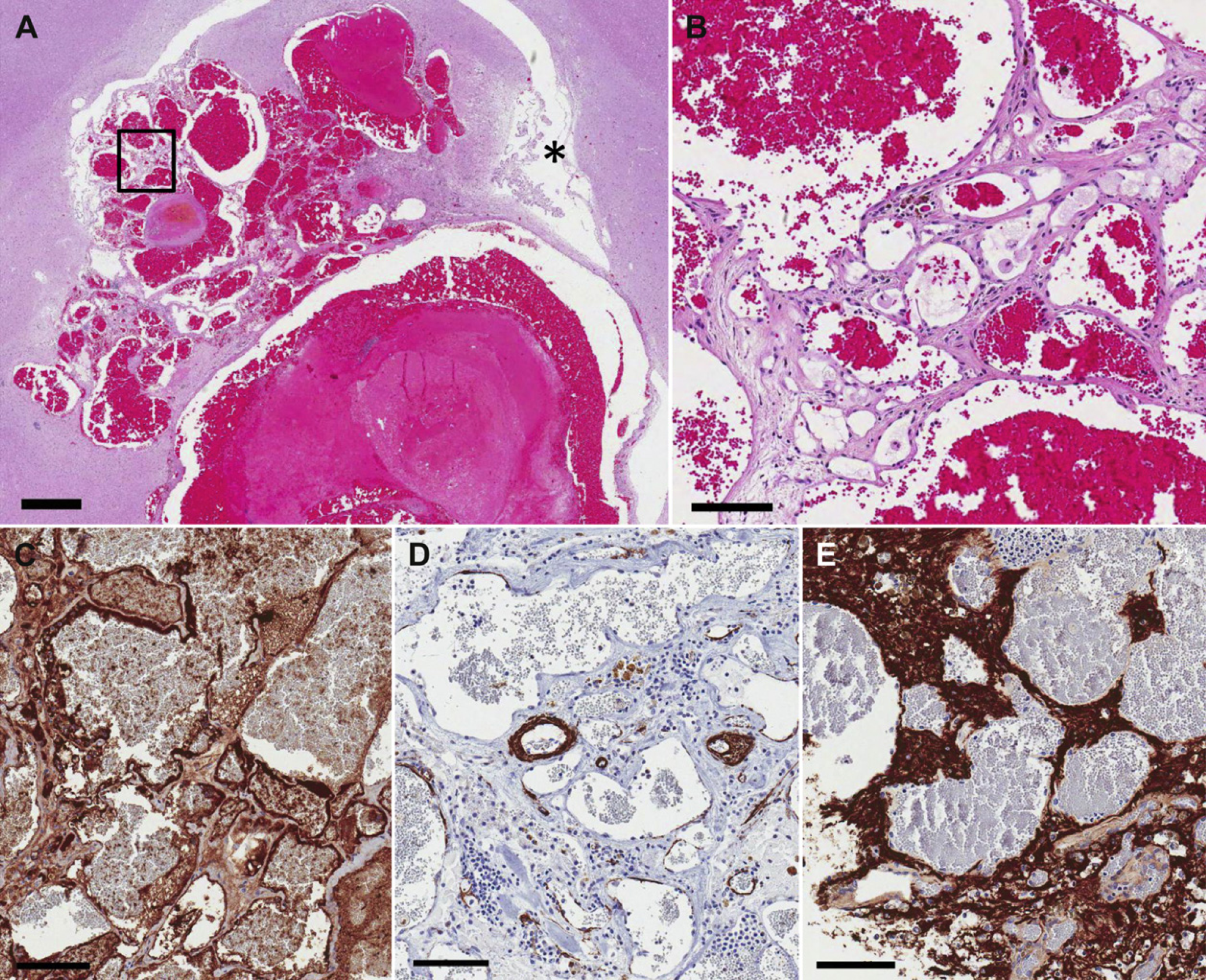
(A) The lesion is located within the choroid plexus, extending into the lateral ventricle (asterisk) and the adjacent cerebral parenchyma (hematoxylin and eosin, bar=1 mm). (B) At higher magnification, it is clear that the lesion consists of variably sized blood filled spaces lined by spindle cells, separated by eosinophilic matrix (hematoxylin and eosin). (C) These spindle cells are identified as endothelial cells via Von Willebrand factor immunohistochemistry. (D) The presence of pericytes within the walls of the vascular spaces is variable as demonstrated via SMA immunohistochemistry. (E) Astrocytic fibers are labeled dark brown via GFAP immunohistochemistry, highlighting the presence of intervening neural tissue exhibiting fibrous astrocytosis (B–E, bar=100 μm).

(A) Transverse pre-contrast image at the level of the thalamus showing a multilobulated, irregularly marginated, heterogeneously hyperattenuating mass located in the mid aspect of the falx cerebri, extending into the cerebral hemispheres (right more than left) and lateral ventricles. Dilation of the ventral aspect of the lateral ventricles is also noted. (B) Transverse pre-contrast image at the level of the midbrain, showing a more caudal section of the mass. (C) Dorsal reconstruction after iohexol administration. Marked displacement of the falx cerebri and dilation of both lateral ventricles and the left olfactory bulb recess (white arrow) are noted. (D) Transverse post-contrast image, obtained at the same level as image (A). Note the poor contrast enhancement of the mass. (E) Transverse post-contrast image, obtained at the same level as image (B). Note the poor contrast enhancement of the mass.
Cerebral vascular malformations are generally categorized as hamartomas, which are non-neoplastic, developmental anomalies of the vasculature. 1–3 They are further classified into four categories: arteriovenous malformations, venous malformations, cavernous malformations, and capillary telangiectasias. 1,3,4 Various vascular malformations of the central nervous system (CNS) have been described in domestic animals, 5–13 with the majority of these cases diagnosed early in life. 8,9,12,13 However, a delayed onset of signs has been reported in a few canine patients. 7,10,11 In this case, the cat was 11 years old at the onset of signs. This is in contrast with the two previously published cases of CNS vascular hamartomas in cats, where a 16-month-old and a 15-month-old cat developed neurological signs due to a cerebellar and a cervical spinal cord vascular hamartoma, respectively. In humans, clinical signs due to cerebral vascular malformations have a peak incidence in the third decade of life. 14,15 In humans, late onset of signs has been explained by continued hemodynamic stress and consequent attenuation of the abnormal vessels which eventually leads to hemorrhage. 15,16 In addition, there is strong evidence that some CNS vascular malformations in people may be acquired, as a result of trauma, radiation, or other injury to the CNS. 1
Based on the location of the mass in this case, the main differential diagnoses were choroid plexus tumor, glioma (oligodendroglioma, astrocytoma), ependymoma, or a meningioma arising from the choroid plexus. Choroid plexus tumors can be hyperdense to isodense on pre-contrast CT images, but tend to enhance strongly after iohexol administration, 17 which was not a feature of this mass. Gliomas have variable density on CT and can range from none to strong or ring enhancement. 17 Ependymomas tend to also enhance strongly. Meningiomas arising from the choroid plexus are rare, but have been reported in the cat. 17 They, too, most often strongly enhance after iohexol administration. 17
The hyperdense appearance of intracranial masses on CT images may be attributed to hypercellularity, calcification, or hemorrhage in certain stages. 18 CT appearance of hemorrhage has been described extensively. 19 The unique imaging characteristics of this mass were likely due to extensive hemorrhage and mineral deposition noted at necropsy.
The hamartoma reported here was structurally similar to the capillary telangiectasias described in people. This is based on two features: the composition of the vessel walls within the hamartoma and the presence of intervening normal brain parenchyma between the hamartoma vessels. 1,3,10,20 Capillary telangiectasias are generally small, measuring less than 1 cm in diameter. 1 Microscopically, they appear as a cluster of small, dilated, capillary-type vessels. The diameter of the vessels may be larger than expected for a normal capillary, but morphologically the vessel walls resemble those of capillaries, devoid of smooth muscle or elastic fibers. 1,3,20 A key feature of capillary telangiectasias is that the component vessels are separated from each other by normal neural parenchyma, as noted in the case reported here. 1,3,20 Presence of factor VIII-specific staining confirmed the endothelial nature of the vessel-lining cells. Also, some of these lining cells were actin-positive. This feature has been previously reported in canine CNS vascular hamartomas. 10,11 These cells likely represented pericytes, which are modified smooth muscle cells that surround capillaries and venules. 10 The presence of pericytes as well as intervening neural parenchyma within the mass suggest a malformative pathogenesis rather than neoplastic, differentiating the diagnosis of vascular hamartoma from the morphologically similar diagnosis of cavernous hemangioma. 3,10,12,20
In this case, the development of clinical signs is attributed to the presence of the hemorrhage observed within and surrounding the lesion as well as compression of the surrounding cerebral parenchyma, and secondary obstructive hydrocephalus. 13
To the authors’ knowledge, this is the first report of a cerebral vascular hamartoma in a cat and the first time that CT images of an intracranial hamartoma in a feline patient are reported. Also, it is the first time that a cerebrovascular malformation is reported in a geriatric cat. Although this diagnosis is expected to be rare, it is important to include vascular malformations as possible differential diagnoses for intracranial masses in cats, especially when the CT imaging features are consistent with a hemorrhagic mass.
