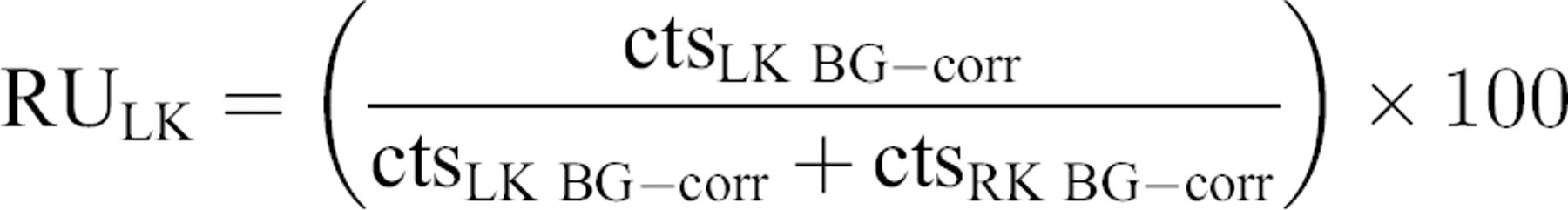Abstract
In this study we investigated the influence of technical factors (positioning, background (BG) correction and attenuation correction) on qualitative and quantitative (absolute (AU) and relative (RU) uptake) assessment of feline kidneys with 99mtechnetium labelled dimercaptosuccinic acid (99mTc-DMSA). Eleven healthy adult cats were included. Influence of BG and depth correction on quantitative assessment was evaluated. Depth correction was based on the geometric mean method (using dorsal and ventral images) and the use of two standards placed over each individual kidney. Visual evaluation showed superiority of dorsal and ventral over lateral positioning due to increased separation of the kidneys permitting region of interest (ROI) placement without overlap. No apparent influence of BG correction was found for RU. However, AU was systematically overestimated without BG correction. Depth correction did not seem to affect RU in most cases, however, in some cats the differences were not negligible. The values for AU without depth correction were lower compared to depth corrected values.
Acquiring information on the morphology and functionality of the kidneys in feline medicine is commonly based on radiographical and ultrasonographical examinations, combined with the results of blood tests and urinalysis. Techniques that combine both morphological investigation and functional assessment can be found in nuclear medicine. Determination of the glomerular filtration rate (GFR) using plasma sampling methods is considered the best representation of renal function. The need for optimal accuracy requires a cumbersome method based on multiple blood samples, although several simplified methods have been developed for practical use. 1–3 However, these methods do not give information on the individual kidney function, which may be needed for some pathological conditions. In this regard, several nuclear medicine tracers have been reported in dogs and humans, each representing different renal parameters. The effective renal plasma flow (ERPF) can be calculated with the aid of 99mtechnetium labelled mercaptoacetyltriglycine (99mTc-MAG3) 4 or 123I- or 131I-labelled orthoiodohippuric acid (123I- or 131I-OIH). 5 GFR can be estimated using 99mTc-diethylenetriaminepentacetic acid (99mTc-DTPA). 6–8 These tracers are used with a gamma-camera method, allowing the calculation of the individual kidney function. However, each tracer has some inherent disadvantages for relative renal function calculation. Labelling with 123I makes OIH a rather expensive tracer to use, whereas labelling with 131I does not yield highly qualitative images. 5 99mTc is very suitable for gamma-camera imaging, but specifically in cats, MAG3 has a high percentage of excretion by the liver, and is not an optimal tracer for renal function determination in cats. 9 99mTc-DTPA is suitable for imaging and affordable. However, because GFR is a dynamic process, its measurement is susceptible to physiological changes. 10
99mTechnetium labelled dimercaptosuccinic acid (99mTc-DMSA) is a well-known nuclear tracer in human medicine. 11–15 where it is frequently used, particularly in paediatrics. 16–19 99mTc-DMSA gradually accumulates in the proximal tubular cells, 20,21 allowing assessment of size, shape, homogeneity of tracer uptake and position of the kidney in the abdomen. Its main indications in human medicine are visual evaluation of regional kidney impairment (eg, in case of pyelonephritis), follow-up after instalment of treatment and the evaluation of relative and absolute individual kidney function (eg, prior to renal surgery). 16,21 Calculation of the relative renal function gives an indication of the contribution of each kidney to the total kidney function, while the amount of functional renal mass of the individual kidney is reflected by the absolute uptake (AU) calculation. Because of the gradual accumulation and delayed imaging, its presence in the kidneys suffers less from haemodynamic or physiological changes at the moment of image acquisition.
The common standard operating procedure in human medicine is to perform the measurements on dorsal views only, with the patient in dorsal recumbency above the gamma camera. 17,23 Oblique views are obtained for inspection of renal morphology and homogeneity of tracer uptake. 17,23 In some indications, eg, the suspected presence of an abdominal mass or ectopic kidney and for depth correction using the geometric mean (GM) method, ventral views are also obtained. 17,23–25 Relative and absolute kidney function is estimated from the background (BG-corrected) and depth-corrected count rate in each kidney on dorsal and ventral views.
In veterinary medicine, 99mTc-DTPA is regularly used to calculate GFR and relative renal function. 6–8 The animals are often placed in lateral position and dorsal images are acquired. 6,7,26 However, especially in cats, the kidneys are loosely fixated in the abdomen and will move when the position of the cats changes. On the scintigram, the activity of both kidneys thus may overlap when positioned laterally, making the unequivocal placement of regions of interest (ROIs) over the individual kidneys difficult.
The aim of this study was to investigate the applicability of 99mTc-DMSA in feline medicine. For this purpose, the images firstly were assessed to determine which position is best for visual analysis of the kidneys. Further, technical factors that may influence the quantification of relative and absolute function, more particularly the influence of BG correction and depth correction using the GM method, were investigated. Secondly, this study aimed to obtain normal absolute and relative renal uptake values of 99mTc-DMSA uptake in cats.
Material and methods
Animals
All procedures were carried out after approval of the Ethical Committee of the Faculty of Veterinary Medicine, Ghent University. Eleven cats were included, two males and nine females. The average age was 5.1 years (range 2–12 years); average bodyweight was 5.01 kg (standard deviation (SD)±0.96 kg, range 3.3–6.1 kg). Serum chemistry (blood urea nitrogen and creatinine) and urinalysis were performed, to ensure no clinicopathological evidence of renal dysfunction was present.
The cats were kept fasted at least 12 h prior to the scans, but access to water was allowed at all times.
Tracer
An average activity of 99.1 MBq (±5.3 MBq; average of 19.8 MBq/kg bodyweight) of 99mTc-DMSA (TechneScan DMSA, Covidien Belgium, Mechelen, Belgium) was injected intravenously through an indwelling catheter placed in the cephalic vein. The exact amount of injected activity was calculated by measuring the activity in the syringes in a dose calibrator before and after administration.
Further, two syringes containing on average 5.6 MBq (±1.0 MBq) 99mTc-DMSA were prepared for use as calibrated sources (one per kidney), and scanned with the corresponding cat. Decay correction between the measurements was taken into account.
Immediately after administration of the tracer a 0.9% saline solution was used to rinse the catheter.
Scans
All scans were performed with the cats under general anaesthesia. The cats were induced using propofol (4 mg/kg, PropoVet; Abbott Lab Laboratories, Kent, UK), and were kept under anaesthesia with a constant rate infusion of propofol for the duration of the scan (0.3 mg/kg/min).
For the scans, a dual-head gamma camera (Toshiba GCA7200 DI) was used, equipped with low energy, high-resolution (LEHR) collimators. Then, 240 s static ventral and dorsal images were obtained at 4 h after intravenous tracer injection using a zoom factor ×2 and a matrix size of 128×128 (pixels size=2 mm) with the cats positioned in dorsal, ventral and lateral recumbency. Additionally, in dorsal position, dorsal and ventral images were obtained with the two calibrated sources centred on the kidneys to calculate AU values of 99mTc-DMSA in normal cats.
Visual evaluation
Each planar acquisition was visually assessed to determine which acquisition portrays the kidneys the most distinctly. The distance between the kidneys was measured for the cats positioned in the dorsal and ventral recumbency. This distance was calculated on a profile through the kidneys and 20% of the maximum activity was taken as internal kidney limit.
Quantitative evaluation
A ROI was drawn manually over the left (LK) and right kidney (RK) separately. BG activity was measured with C-shaped ROIs manually drawn surrounding the ROIs of the kidneys, close to but avoiding inclusion of activity originating from the kidney tissue (Fig 1).

Ventral view of a cat in dorsal position with the calibrated sources (indicated by ∗) placed at the level of the kidneys, obtained 4 h after injection of 99mTc-DMSA. ROIs 1 and 3 are placed around the kidneys including the calibrated sources; ROIs 2 and 4 are used for BG correction. The urinary bladder (B) is visible caudal from the kidneys. The liver is marked with ‘L’.
BG activity
In order to compensate for activity from non-renal tissue included in the kidney ROI, a correction for BG activity per kidney was made in the calculation of tracer uptake (expressed in counts):
Corrected and non-corrected kidney counts were compared to evaluate the influence of BG activity.
Relative uptake (RU)
RU was based on the BG corrected renal counts obtained for the left and right individual kidney. It is expressed in percentage (sum of both values=100%). RU was measured using values without or with depth correction. Calculations without depth correction were based on the dorsal views only. Calculations with depth correction used the values obtained from the GM method, as it places the kidneys at virtually the same depth in the cat's body.
The GM of the kidneys was calculated using the counts in the LK and RK obtained from ventral and dorsal views. The GM value is equal to the square root of the counts from the dorsal projection multiplied by those from the ventral projection:
RU of the LK (RULK) was then calculated using following formulae:
without depth correction:
with depth correction:
AU
The AU is expressed as the percentage of the injected dose of activity that is accumulated in the individual kidneys, and is calculated with the aid of the calibrated sources.
To attain the counts in the calibrated sources, one kidney ROI was used and the counts of this ROI without the calibrated sources were deducted from the counts of the same ROI with the calibrated sources (Fig 1), for the dorsal and ventral images.
The GM of the calibrated source (GMCS) is then calculated as follows:
Based on the calculations mentioned above and the measured activity in the calibrated sources, the AU of tracer can be calculated for each kidney separately, based on the following formula (depth corrected):
With:
ActivityCS: the activity in the calibrated source, expressed
in MBq GMCS: GM of the calibrated source GMKidney BG—corr: GM of the counts in one kidney Activity full syringe/activity empty syringe: both are expressed in MBq
measured before and after injection.
The AU was also calculated based on dorsal views only. The counts in the calibrated source then were derived from the view with the non-attenuated syringes. Normal AU and RU values were calculated using the above formulae and the GM method, based on 240 s static scans (ventral and dorsal views) at 4 h after tracer injection, with the cats in dorsal position. A BG correction was applied.
Results
Visual interpretation
Figure 2 shows a normal dorsal view of the kidneys with the cat in dorsal and ventral position. A high tracer uptake in the kidneys compared to BG is visible. Visual inspection shows that images with the animals in lateral recumbency were mostly not suitable for proper kidney evaluation due to overlap of the kidneys on each other (Fig 3).

Two acquisitions of the same cat in ventral (right) and dorsal (left) position. The distance between the kidneys is clearly greater when the cat is in dorsal position, facilitating ROI placement (R=right kidney, L=left kidney, B=urinary bladder, Cr=cranial direction).
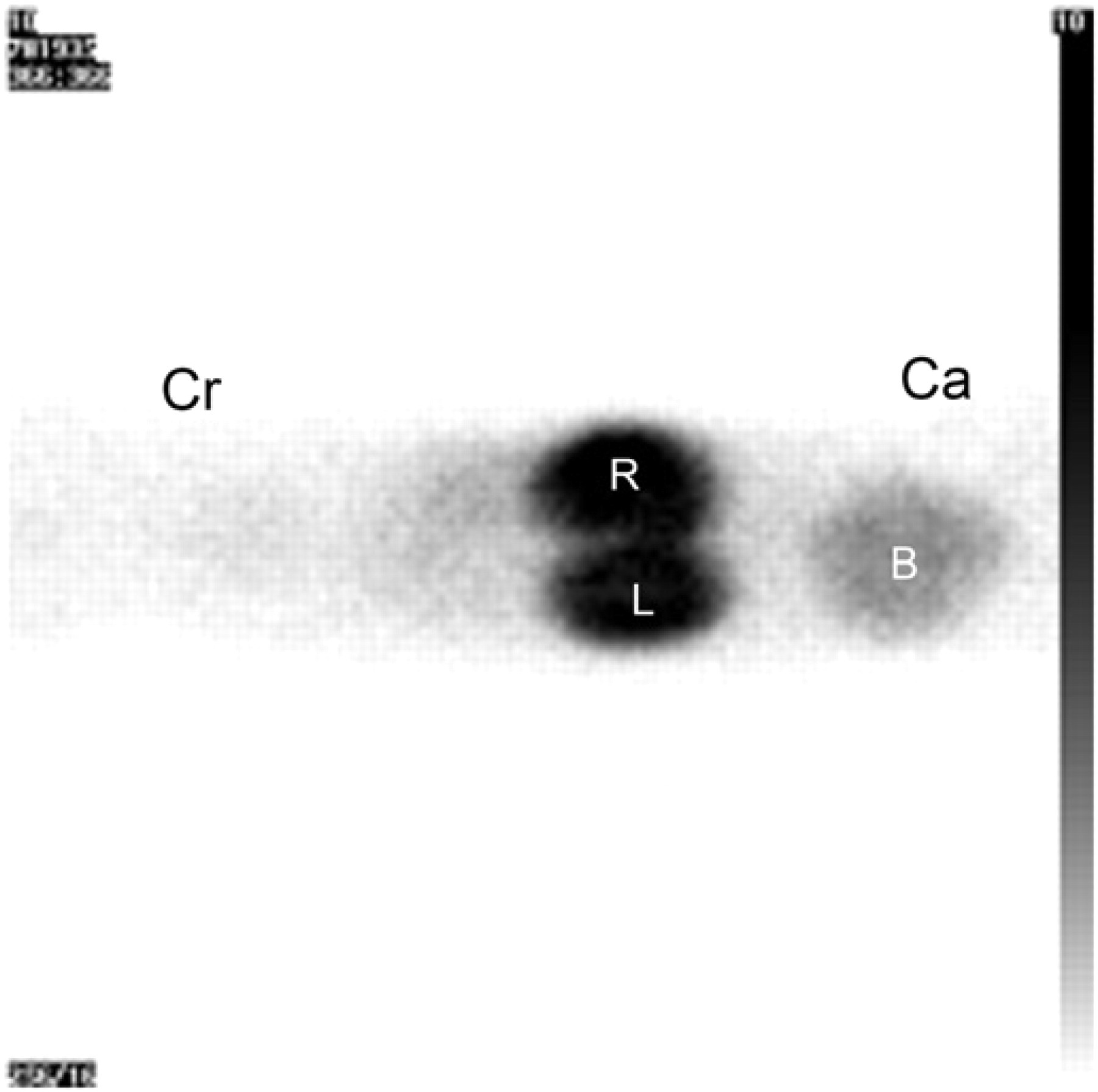
Dorsal acquisition of a cat placed in lateral recumbency. Due to the flexibility of the kidneys in the abdomen, correct ROI placement is not possible (R=right kidney, L=left kidney, B=urinary bladder, Cr/Ca=cranial/caudal direction).
On the images acquired with the cat in ventral or dorsal recumbency, the kidneys were well separated from each other although the distance between them is more prominent when the cat is positioned dorsally. This may be favourable for ROI placement over the kidneys. The mean distance on the dorsal views between the kidneys with the cat in dorsal position was 7.3 cm (SD±1.4 cm, range 5.4–8.8 cm); as opposed to 4.6 cm on average in ventral position (SD±1.8 cm, range 2.4–8.4 cm).
Quantitative analysis
BG correction
After correction for surrounding non-renal activity, the calculated kidney activity was systematically lower. On kidney-to-kidney basis, the contribution of BG activity was variable.
The mean of the differences (±SD) between kidney counts with and without BG correction (expressed in %) for the depth corrected values was 13.7% (±1.6%; range 12.1–16.2%) for the LK and 14.8% (±2.0%; range 11.6–17.9%) for the RK. When using dorsal images only (no depth correction), these values were 13.7% (±2.1%; range 11.2–17.5%) and 15.5% (±2.1%; range 11.2–18.0%), respectively.
RU
The RU values of the LK and RK were calculated without and with depth correction, and then compared. Mostly, the differences seemed to be rather small. The mean % difference between both methods was 0.6% for the LK (±5.2%; range 0.1–9.4%), and −1.1% (±5.2%; range 0.1–9.0%) for the RK. The average normal RU of the LK and RK, respectively, was 49.5% (±2.2%; range 48.4–52.7%) and 50.5% (±2.2%; range 47.3–51.6%) (4 h after injection, dorsal position, BG corrected). When only dorsal images were used, these values were 49.9% (±3.2; range 45.7–55.1%) for the LK and 50.1% (±3.2%; 44.9–54.3%) for the RK (4 h after injection, dorsal position, BG corrected).
AU
The average normal AU of the LK and RK was 21.3% (±3.4%; range 16.1–25.4%) and 21.7% (±3.8%; range 15.7–27.0%) (4 h after injection, dorsal position, BG corrected). This value for both kidneys combined was 43.0% (±7.0%; range 32.6–53.3%). The mean individual AU was 25.1% (±4.0%) without BG correction, and 21.5% (±3.6%) with BG correction. When calculated on dorsal images only, a mean value of 11.5% instead of 21.5% was obtained.
Discussion
Historically, DMSA was used as a renal protective agent in cases of heavy metal poisoning in humans. 11 The exact mechanisms of uptake in the kidney cells (direct tubular uptake or reabsorption after glomerular filtration) remain debated in human medicine. The result is accumulation of the tracer in the renal tubules, primarily in the proximal tubular cells. 99mTc-labelled DMSA can be used for scintigraphic examination of the renal cortex. Indications in human medicine are the diagnosis of acute pyelonephritis, the evaluation of renal scarring after resolution of the primary disease, and the assessment of renal masses. 16,22
In this study, firstly, the applicability of 99mTc-DMSA as a tracer for the assessment of kidney function in cats was evaluated. Comparable to the optimal scan time in human medicine, 18,27 scans at 4 h after administration give a good target/BG ratio and thus seem adequate to estimate kidney function in cats. 26 This time interval allows a good renal uptake of the tracer, yet it does not necessitate the cats to be hospitalised overnight.
Visual inspection of the scans reveals the importance of careful positioning of the cat, due to the mobility of the kidneys in the cat's body. Scintigraphic renal studies in veterinary medicine often describe a lateral recumbency, 6,7,26 but an unambiguous interpretation of the individual kidney function can be hampered by this position. In this study also the kidney activity showed overlap when the cats were placed laterally, not providing the space needed to draw ROIs around each kidney separately without interference of activity originating from the other kidney.
The kidneys are clearly separated from each other when the cat was placed ventrally or dorsally. However, when placed ventrally, the kidneys seemed to move closer towards each other. Still, both positions are suitable for image processing as they allow good distinction between the kidneys. We elected to use the dorsal position for RU and AU quantification. Average normal RU and AU values were calculated, based on acquisitions at 4 h after tracer injection, with and without BG and depth correction.
The question arises whether, as in humans, correction for tracer uptake in the surrounding non-renal tissue is necessary (BG correction). 28 Our results indicate that the contribution of BG activity is not negligible for 99mTc-DMSA in cats as the AU will systematically be overestimated without BG correction and as a consequence this is mandatory for absolute kidney uptake quantification. Without BG correction a mean AU value for both kidneys of 25.1% (±4.0%) was obtained, compared to 21.5% (±3.6%) with BG correction. For normal functioning kidneys, BG correction generally does not change the assessment of the RU. 19,29 However, as BG counts are not relative to the kidneys uptake but rather an absolute value of the surrounding tissue, the relative contribution of a poorly functioning kidney would be overestimated when BG correction is not applied. 16
For further uptake quantification, a choice must be made between a method based on only the dorsal image (without depth correction) and that using the GM (with depth correction). Whether or not a correction for the possible difference in depth of the kidneys within the body should be applied for calculation of RU still is a matter of debate in human medicine. 30,31 Some studies report the necessity of such corrections for adult patients, where it is not needed for paediatric patients. 24,26,32 For the latter population, the additional ventral acquisitions needed for GM calculation are considered to be too distressing. 17 In veterinary medicine it has been reported that in cats depth correction for GFR measurements using 99mTc-DTPA is not necessary due to their small body volume. 8 The differences between RU calculations without and with depth correction were mostly rather small in this study (mean % difference for the LK was 0.6% (±5.2%), and −1.1% (±5.2%) for the RK). In some cases however, the differences were not negligible. On a theoretical basis, there are less causes of error when using the GM, 24 but this must be performed carefully. GM correction based on one standard for depth correction (placed between the kidneys) implies that the body thickness at the site of the LK is equal to that of the RK. 24 The flexibility of the body and the mobility of the kidneys in cats may introduce differences in thickness of attenuating tissue, which may compromise the use of depth correction based on only one standard. Therefore, we placed two standards, one over each kidney. From a practical point of view, the use of the dorsal acquisition only is acceptable for determination of the relative kidney function, provided that the cat is positioned dorsally and symmetrically above the camera.
The average normal AU of, respectively, the left and right kidneys with BG and depth correction was 21.3% (±3.4%) and 21.7% (±3.8%), and 43.0% (±7.0%) for both kidneys together. The values obtained for AU without depth correction were clearly lower than the AU values with depth correction. Therefore, it may be concluded that dorsal images alone do not permit accurate calculation of AU values in cats.
Conclusions
99mTc-DMSA can be a useful tool in cats for kidney investigation when individual kidney information is required. Normal values of absolute and relative 99mTc-DMSA uptake are presented. In future studies, the possibility for split renal function calculations based on 99mTc-DMSA imaging combined with plasma sampling methods for GFR estimation will be explored as is applied in human medicine. 33,34