Abstract
A 12-year-old female neutered cat presented with acute onset unilateral vestibular syndrome, a spontaneous cutaneous wound, polyuria, polydipsia, and diabetes mellitus. Hyperadrenocorticism was demonstrated by means of hyper-responsiveness to adrenocorticotropic hormone stimulation, elevated urine cortisol-to-creatinine ratio, bilaterally enlarged adrenal glands on abdominal ultrasound, and pituitary enlargement on computed tomography imaging. The cat was euthanased and post-mortem histological examination revealed feline skin fragility syndrome; confirmed a pituitary cromophobe macroadenoma; and generalised toxoplasmosis with tachyzoites in the pancreas, bowel and brain. This report is the first to describe the concurrence of macroadenoma pituitary-dependent hyperadrenocorticism and generalised toxoplasmosis in a cat with central vestibular syndrome.
Feline spontaneous hyperadrenocorticism (HAC) is rare and in approximately 80% of cases caused by a functional pituitary adenoma (pituitary-dependent hyperadrenocorticism; PDH). About 50% of pituitary tumours are microscopic in size; the remaining tumours are large (usually greater than 3–4 mm in greatest diameter). 1 In rare cases pituitary macroadenomas have been associated with specific central nervous system (CNS) neurological signs, 2,3 but in the majority of reports lethargy was the only potential neurological sign. 4–7
Seroprevalence studies suggest that
Feline spontaneous HAC has never been described in association with generalised toxoplasmosis, although it has been associated with
A 12-year-old spayed female domestic shorthair cat weighing 3.8 kg, which lived indoors and was fed a commercial diet for the last year (previously was an outdoor cat with possibility to hunt) was referred to Milan University Medical Teaching Hospital with an acute onset of ataxia, a spontaneous skin wound and polyuria and polydipsia (PU/PD). Physical examination revealed an enlarged abdomen, dehydration (∼10%), diffuse muscle wasting, the presence of a 5 cm×5 cm skin tear on the right hind limb (Fig 1) and severe, generalised thinning of the skin with prominent dermal blood vessels (Fig 2). Neurological examination revealed abnormal mentation with heavy obtunded mental status, unilateral head tilt, imbalance, ataxia, dysmetria, circling to right side, and ipsilateral proprioceptive deficits and weakness. Head tilt, imbalance, ataxia and circling indicated a vestibular system dysfunction. Obtunded mental status, dysmetria and ipsilateral proprioceptive deficits and weakness could be explained by a CNS central diffuse or multifocal lesion with a major component involving the cerebellum, pons and medulla.12

12-year-old neutered female domestic shorthair cat with PDH. Note the spontaneous skin tear on the left caudal limb.

12-year-old neutered female domestic shorthair cat with PDH. Note the thin skin with prominent dermal blood vessels.
Blood examinations revealed neutrophilia (15.0; range 2.5–12.5×109/l), lymphopenia (0.3; range, 1.5–7.0×109/l), and hyperglycaemia (20.1 nmol/l; range, 4.4–6.1 nmol/l). The urine had a specific gravity of 1.020 with glucose presence of 3+. Urine was negative for bacterial and fungal growth. A combined enzyme-linked immunosorbent assay (ELISA) for feline leukaemia virus (FeLV) antigen and feline immunodeficiency virus (FIV) antibody were negative (Snap Combo Plus FeLV Ag/FIV Ab, Idexx Laboratories). Indirect fluorescent antibody (IFA) technique for feline coronavirus (FCoV) antibody was negative and indirect haemagglutination test (IHA) for
The pituitary-adrenal axis was assessed using an adrenocorticotropin (ACTH) stimulation test with synthetic ACTH (0.125 mg intramuscularly; Synacthen; Novartis Farma). This revealed hyper-responsiveness with baseline serum cortisol of 726 nmol/l (range 20–270 nmol/l), 30 min post-stimulation cortisol of 985 nmol/l (range<400 nmol/l), and 90 min post-stimulation cortisol of 226 nmol/l (range<400 nmol/l). The urinary cortisol-to-creatinine ratio (UC:CR) measured on an home-collected urine sample was 11 (range 2–3.6×10−5). 15 No further blood tests were undertaken. Abdominal ultrasonography revealed the liver was enlarged with normal echogenicity and an homogeneous parenchyma. The adrenal glands were hypoechoic, homogeneous, and enlarged (width of left adrenal 0.66 cm, right adrenal 0.46 cm; range<0.39 cm). 16 These data were consistent with a diagnosis of PDH. To correct the dehydration, hyperglycaemia and to prevent a systemic infection from the skin tear the cat was initially treated with intravenous fluids, porcine lente insulin (0.5 U/kg subcutaneously q 12 h; Caninsulin; Intervet), and cefazolin (20 mg/kg intravenously q 12 h; Dorom; Teva Pharma), respectively.
A computed tomography (CT) scan of the head was performed 3 days after the admission of the cat. Pre-contrast images showed a large mass (10 mm× 10 mm× 6 mm) with well-defined margins within the sella turcica. The mass appeared slightly hyper-dense compared to the surrounding parenchyma, and showed high homogeneous contrast enhancement in post-contrast images (Fig 3). There was no evidence of mass effect or compression of the ventricular system. The tomographic findings were consistent with a pituitary macroadenoma associated with a thin surrounding hypo-dense lesion consistent with a mild perilesional oedema. There was also a hypo-dense area in the caudal portion of the cerebellum.

CT scan of the head of a 12-year-old neutered female domestic shorthair cat with PDH: post-contrast transverse slice at the level of the diencephalon. A well-defined space-occupying lesion (10 mm high, 10 mm wide, and 6 mm long) is evident in the hypothalamic-pituitary region (arrows).
Due to the cat's poor clinical condition the owners requested she be euthanased when the cat was still under anaesthesia for CT, and gave their permission for an autopsy. The main gross autopsy findings were bilateral adrenal enlargement, a multi-nodular firm pancreas and a severely enlarged (7 mm in greatest diameter) pituitary gland. A large, poorly defined malacic area in the cerebellar white matter was also detected.
Examination of coronal sections of the brain, performed a few days thereafter on formalin fixed tissue, showed a large (1×0.9 cm diameter), poorly defined malacic area within the cerebellum and brainstem. Specifically, the lesion comprised the area of right medial, rostral and lateral vestibular nuclei, and caudal cerebellar peduncle, extending cranially and dorsally to the flocculus into the cerebellar folia.
Histologically, the skin was characterised by diffuse, severe, epidermal orthokeratotic hyperkeratosis, dermal–epidermal atrophy, and large areas of epidermal detachment, consistent with feline skin fragility syndrome (FSFS). 17 Diffuse adrenal cortical hyperplasia and severe, multifocal to coalescing, necrotising, neutrophilic and macrophagic, subacute pancreatitis with intense fibrosis were also present. Innumerable intralesional protozoal tachyzoites, 3–4 μm, with small basophilic nuclei and clear cytoplasmic halo were recognisable within pancreatic necrotic areas, both free and within macrophage or pancreatic acinar cell cytoplasm (Fig 4). A small area of intramural necrosis with neutrophilic infiltration was detected within duodenal wall muscle layer, in close proximity to pancreatic duct. Pituitary mass was consistent with a chromophobe adenoma, composed of well differentiated small chromophobe cells, supported by a thin fibrovascular stroma (Fig 5). The malacic area grossly detected within the cerebellum was consistent with necrotising and purulent encephalitis with intralesional free tachyzoites, occasional protozoal cysts and peripheral lymphoplasmacytic perivascular cuffing. Similar, smaller lesions were multifocally detected scattered within brain parenchyma.
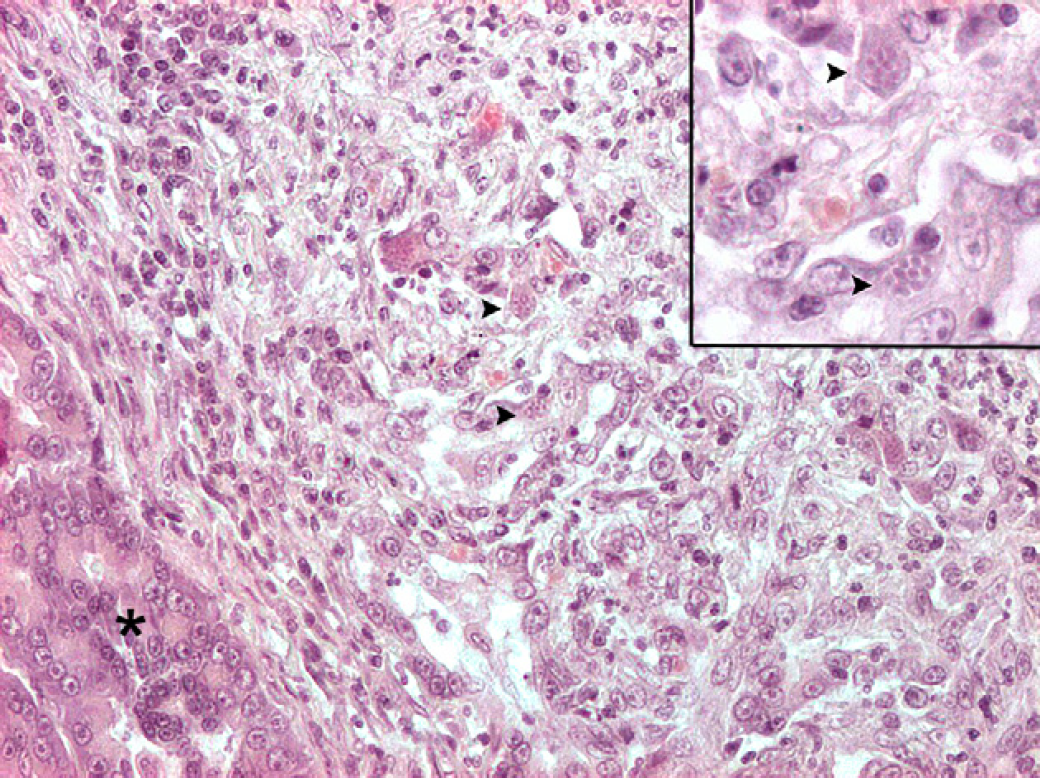
Exocrine pancreas (asterisk): a large area of necrosis, neutrophilic and lymphoplasmacytic infiltration and fibrosis effaces normal architecture. Numerous tachyzoites and cysts are scattered throughout the lesions (arrowheads). Haematoxylin and eosin, 200×. The small insert at the upper right shows higher magnification of the same protozoal cysts (haematoxylin and eosin 400×).

Pituitary gland chromophobe adenoma: well differentiated small chromophobe cells, organised in small packets supported by a delicate fibrovascular stroma. Haematoxylin and eosin, 100×.
Immunohistochemistry was performed on deparaffinised sections of all tissues sampled at necropsy. The standard avidin–biotin–peroxidase complex (ABC) technique
18
was used. Primary antibody, a rabbit antiserum directed against

Cerebellum, numerous

Exocrine pancreas: numerous
In feline HAC the most common clinical signs include PU/PD, polyphagia, weight loss, abdominal enlargement, thin skin, skin tears, and hair loss. 1 In the case described here the predominant signs were PU/PD, cutaneous lesions due to FSFS, and neurological signs of central vestibular disease.
PU/PD are often the first overt signs of HAC and are usually associated with marked hyperglycaemia because 80–100% of cats with HAC are diabetic. 19 FSFS is a rare disorder characterised by markedly fragile, thin skin which is easily damaged by minor trauma. Moreover, FSFS has been associated with several underlying disease processes, most commonly spontaneous or iatrogenic HAC, and the use of progestagens.20
In feline medicine, disorders that may cause a central vestibular syndrome are meningoencephalitis due to infectious-inflammatory disorders of CNS (eg, toxoplasmosis, feline infectious peritonitis (FIP), FIV), aberrant parasitic migration, neoplasm (eg, meningioma, lymphoma), or thiamine deficiency.
10,12
In the cat described in this report, neurological examination revealed the presence of head tilt, imbalance, ataxia and circling to right side indicated a vestibular system dysfunction, and obtunded mental status, dysmetria and ipsilateral proprioceptive deficits and weakness which could have been explained by a CNS central diffuse or multifocal lesion with a major component involving the cerebellum, pons and medulla.
12
Even if the serology for
In cats immunosuppression caused by high doses of drugs such as cyclosporine and prednisolone 21 or a lymphotropic virus such as FIV 22 may reactivate a latent infection result in clinical toxoplasmosis.
In this cat, lack of recent exposure history to
Many cats with pituitary gland adenomas die from complications relating to endocrine disease before neurological signs manifest as the tumour size increases. Pituitary macroadenomas are responsible for CNS signs when the enlarged hypophysis compresses the surrounding structures in the brain. 25 Neurological deficits commonly involve diencephalic signs such as mental changes, circling, visual and pupillary light reflex deficits (mydriasis), and seizures.25
In this case of macroadenoma PDH in association with generalised toxoplasmosis and neurological signs, it is difficult to establish if the neurological signs were caused by the pituitary macroadenoma and/or the CNS toxoplasmosis, because both diseases may cause similar clinical signs. The majority of the reports of macroadenoma PDH in cats in the veterinary literature showed no obvious central neurological signs.
4–7
One case has been described as macroadenoma with central neurological signs, but the pituitary gland was larger than the case reported here.
3
In our case there was no evidence of mass effect or compression by the pituitary adenoma detectable at CT scan, but there were encephalic lesions (cerebellar malacia, multiple necrosis foci in the brain) due to
