Abstract
Clinical report
A stray male domestic shorthair kitten was admitted to Albano Small Animal Hospital, Danderyd, Sweden, at approximately 12 weeks of age with clinical signs of paraplegia and severe extensor rigidity of the pelvic limbs (Fig 1). Physical and neurological examinations were performed upon initial presentation and repeatedly during the 3 weeks of hospitalisation. Neurological examination included assessment of consciousness, behaviour, posture, cranial nerves, postural reactions, spinal reflexes, muscle tone and pain perception, as described by Lorenz and Kornegay. 1

(a) Pelvic limbs of the affected kitten showing deformity consistent with arthrogryposis. Note the involvement of the tail. (b) Arthrogryposis in the pelvic limbs causing severe hip flexion
General physical examination was unremarkable, aside from bilateral pelvic limb rigidity, which had resulted in persistent angular deformity of the stifle and hock joints consistent with arthrogryposis. It was impossible to flex the pelvic limbs even with force under general anaesthesia. The proximal portion of the tail also showed rigidity. On neurological examination, the severe stiffness precluded evaluation of the pelvic limb postural reactions and spinal reflexes. Pain perception was intact in both pelvic limbs and in the distal part of the tail. Discomfort was not noted except when manually trying to flex the pelvic limbs. Urinary and faecal function was intact. The remainder of the neurological examination, including assessment of the thoracic limbs, was unremarkable.
Based on these examinations and the age of the kitten, an inherited or acquired neuromuscular or spinal cord disorder was suspected. Differentials included fetal hypotonia resulting in arthrogryposis, an inherited motor neuron degeneration, a form of muscular dystrophy, an in utero infection, or possibly traumatic injury to the lumbar spine.
A minimum database was performed consisting of a complete blood count and serum biochemistry profile. Creatine kinase, alanine aminotransferase and aspartate aminotransferase activities were within the respective reference ranges. Serum antibody titres for retroviruses (feline leukaemia virus and feline immunodeficiency virus) and protozoal infections (Toxoplasma gondii and Neospora caninum) were negative. No abnormalities, except the angular limb deformities, were detected on radiographs of the spine, hips and pelvic limbs. On two occasions, lumbar cerebrospinal fluid collected under general anaesthesia showed normal cell counts and protein concentration.
Electromyography (EMG) was performed using a concentric needle electrode and a subcutaneous electrode for grounding. Spontaneous activity, including fibrillation potentials, was identified in several pelvic limb muscles (quadriceps, semimembranosus, biceps femoris, gastrocnemius), with the most severe changes found in the gastrocnemius muscle. EMG of the thoracic limbs (triceps, biceps brachii) was unremarkable.
Biopsies from the triceps, biceps brachii, gastrocnemius, quadriceps and semimembranosus muscles, and from the ulnar, radial and peroneal nerves, were collected under anaesthesia. Specimens were either immersion fixed in 10% buffered formalin and evaluated in paraffin sections or frozen in isopentane pre-cooled in liquid nitrogen and evaluated in cryostat sections. Biopsies from the pelvic limb muscles showed moderate generalised atrophy, with atrophic fibres of both fibre types, ranging in size from 25–50 μm. Fibrosis, internal nuclei and muscle fibre loss were limited to the gastrocnemius muscle. No organisms were identified.
Peripheral nerve specimens were resin embedded and evaluated in semi-thin (1 μm thick) sections, as previously described. 2 Nerve biopsies showed regional nerve fibre loss, endoneurial fibrosis and several foamy macrophages containing lipid debris. These findings were consistent with chronic axonal damage resulting in the neurogenic pattern of muscle fibre atrophy observed in the pelvic limb muscle biopsies.
Due to the poor prognosis, the cat was euthanased and a post-mortem examination of the central and peripheral nervous system was immediately conducted at the National Veterinary Institute, Uppsala, Sweden. The brain and spinal cord were fixed in 10% buffered formalin. After fixation, multiple specimens from the cerebral cortex, nucleus caudatus, hippocampus and thalamus, rostral colliculi, medulla oblongata, cerebellum and segments from the cervical, thoracic and lumbar spinal cord were paraffin embedded and processed for routine histopathology. All sections (5 μm thick) were stained with haematoxylin and eosin (H&E) and examined by light microscopy. On gross examination, the quadriceps and sartorius muscles were bilaterally severely contracted, with extension deformities in both stifle joints. No macroscopic lesions were present in the vertebral column or spinal cord.
At the light microscopic level, a striking, bilaterally symmetrical loss of approximately 70% of large motor neurons was found in the ventral grey matter horns of the entire lumbar spinal cord (Fig 2a). Vascular prominence was also of note. The residual population of unaffected lower motor neurons and overall neuronal loss were illustrated using an antibody against non-phosphorylated neurofilament medium (M) and heavy (H) (Fig 2b,c; SMI 33, Covance Labs). In ventral columns and lateral funiculi, severe axonal necrosis, myelin degeneration, astrocytosis and macrophage infiltration were observed (Fig 2d,e). The proliferation of gemistocytes and astrocytes, and vascular prominence were interpreted as part of the regenerative process and may be found where there is severe loss of neurons or damage to the astrocytes. The marked gemistocytic- astrocytic response to neuronal loss was demonstrated using an antibody against glial fibrillary acidic protein (GFAP) (Fig 2f; #20334 Dakocytomation, Carpinteria, CA, USA). Macro phages were highlighted using an antibody against feline CD18 (not shown, FE39F2, gift of PF Moore, Leucocyte Antigen Biology Laboratory, University of California-Davis, USA).

Transverse section of a lumbar segment of the spinal cord, showing bilateral and almost symmetrical loss of most of the lower motor neuron population in the ventral motor horns and some loss of myelin staining in the ventral and lateral funiculi. H&E/Luxol fast blue stain. Bar = 1 mm. (b) Transverse section of a lumbar segment of the spinal cord showing the small residual population of unaffected lower motor neurons in the ventral horns. Phosphorylated neurofilament M and H immunostain. Bar = 0.9 mm. (c) Higher magnification of the lumbar spinal cord segment shown in Fig 2b with some intact lower motor neurons in the ventral horn. Phosphorylated neurofilament M and H immunostain. Bar = 45 μm
(d) Transverse section of the lumbar spinal cord with neuronal loss, gemistocytic astrocytosis and focal and diffuse microgliosis. H&E stain. Bar = 35 μm. (e) Lumbar spinal cord segment showing ventral funiculi with severe axonal necrosis, demyelination, astrocytosis and macrophage infiltration. H&E stain. Bar = 35 μm. (f) Transverse section of the lumbar spinal cord showing a marked gemistocytic-astrocytic response to the neuronal cell loss. GFAP. Bar = 35 μm
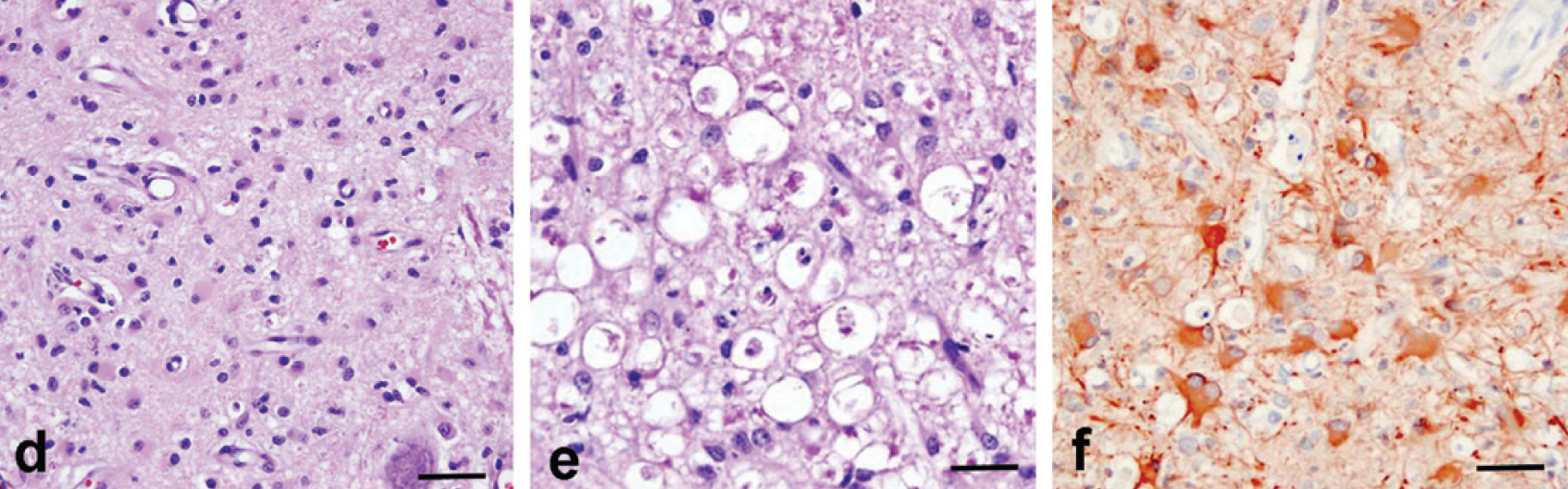
Axonal degeneration was also found in the dorsal tracts of the cervical and thoracic segments of the spinal cord (not shown).
Discussion
There are very few descriptions of feline motor neuron degeneration in the literature.3–5 An inherited form of juvenile onset motor neuron degeneration, with a simple autosomal mode of inheritance, has been described in a family of Maine Coon cats. 5 Affected kittens exhibited tremor, proximal muscle weakness and muscle atrophy beginning at about 4 months of age. Loss of function was initially rapid but progressed slowly after 7–8 months of age. Variably disabled cats lived for up to 8 years. Additionally, a 10-week-old kitten has been described with rapid development of tetraplegia resulting from motor neuron degeneration and accumulation of neurofilaments. 3 Acquired motor neuron degeneration has been described in three unrelated adult cats between 6–12 years of age. 4 Clinical signs were slowly progressive and included generalised weakness, a crouched gait and ventroflexion of the neck. Clinically, cats developed both upper and lower motor neuron signs consisting of tremor, fasciculations (including the tongue), muscle atrophy and dysphagia.
A specific cause for motor neuron degeneration and loss in our cat was not found; however, a vascular, metabolic or toxic aetiology was suggested. As the cat was a stray, no prior history was available. While a congenital disorder cannot be ruled out, an acquired motor neuron loss (arising either in utero or postnatally), rather than an inherited genetic mutation, is suspected based on the apparent recent cellular response of microgliosis, reactive gemistocytic astrocytosis and, most notably, the prominent vascularity. Prominent vascularity has not been described in inherited motor neuron diseases. No evidence of a traumatic insult was observed on physical examination at the time of presentation at approximately 12 weeks of age. While protozoal radiculitis in young dogs may lead to contractures and pelvic limb fixation, 6 there was no laboratory evidence of protozoal nfection in our case; nor were infectious organisms identified at necropsy. Iliac arterial occlusion from emboli resulting in decreased perfusion was not noted on necropsy and even though there was axonal necrosis the lesion was not frankly malacic (Fig 2e).
Arthrogryposis is defined as a persistent flexure or contracture of a joint, and in most cases is congenital in nature. Most commonly, impaired fetal movement causes malformations and is triggered by environmental and genetic factors. 7 Recently, mutations in the acetylcholine receptor and related genes7,8 or maternally transferred acetylcholine receptor antibodies 9 have been implicated in human fetal akinesia syndrome. Arthrogryposis has been described as a complication of motor neuron degeneration in growing animals; 10 however, none of the previous cases, even very young cats, showed clinical signs of limb rigidity.
Limb rigidity has also been described in cats with tetanus,11,12 laminin α2 (merosin)-deficient muscular dystrophy,13,14 and as a complication of femoral fracture management. 15 None of these conditions were identified in the present cat. In this case, joint flexion is likely to be a result of skeletal muscle denervation and contraction, with continued growth of the long bones, resulting from motor neuron loss and contraction. The descending projections to coccygeal spinal segments influencing tail movements are partly located in the lumbar spinal cord. 16 Involvement of these projections may account for the tail rigidity seen in this cat.
Obtaining an ante-mortem diagnosis of either acquired or inherited motor neuron degeneration and loss is difficult. Evaluation of muscle biopsies by histological and histochemical methods can usually differentiate myopathic from neuropathic disorders, the main differential diagnoses. Further differentiation of the causes of neurogenic atrophy relies on consideration of clues from the signalment, neurological examination and electrodiagnostic evaluations. Although generalised weakness, muscle atrophy and fasciculations can occur with both peripheral neuropathy and motor neuron disease, spinal reflexes are usually preserved in cases of motor neuron degeneration until the disease is advanced. 4 However, reflexes could not be evaluated in our cat due to the pelvic limb rigidity. Similarly, until late in the course of the disease, motor nerve conduction velocities are within (or only marginally lower than) the reference range in motor neuron degeneration, while the motor nerve conduction velocity is usually decreased in peripheral nerve disease. 4
Definitive pathological diagnosis of motor neuron loss and degeneration relies, as in this case, on post-mortem examination of the ventral horn cells of the spinal cord.
Footnotes
Acknowledgements
The authors are very grateful to Dr R J Higgins for his invaluable help with providing and reading the histopathological images.
