Abstract
Localised saprophytic fungal infections in cats usually result from the traumatic introduction of infectious propagules, either spores or hyphae, through a breach in the normal integument. Although this can occur via a variety of penetrating traumatic injuries, a very common inciting event is the cat scratch or cat fight wound in which the subcutis is contaminated with vegetative material from soil; similar material may be also present on the claws of the perpetrator.1,2
Normally, these infections are investigated by obtaining appropriately collected specimens for cytology, histopathology (including use of special stains) and microbiological culture. Determining the aetiological agent and the in vitro susceptibility provides very useful information to help guide therapy. However, in some instances the causal organism grows slowly, or fails to produce characteristic spore-forming morphological features on culture (ie, it will not sporulate), despite using different specialised media (eg, V8 juice agar, potato dextrose agar, cornmeal agar, malt agar) and growth conditions (temperature, light, ionic composition, etc). In such instances, use of the polymerase chain reaction (PCR) and sequence analysis of certain conserved regions of the fungal genome (eg, the internal transcribed spacer regions [ITS1 and ITS2 regions] of the ribosomal DNA [rDNA] gene cluster), in a so-called panfungal PCR, 3 can be very helpful in permitting accurate species identification. While this does not circumvent the problem of determining antifungal susceptibilities for fungi that grow only very slowly in vitro, determination of the specific aetiological agent via PCR and sequence analysis does allow literature review of reported susceptibilities or response to therapy for that or closely related species.
While waiting for laboratory data, it is generally critical to start empiric therapy immediately after excisional biopsy in an attempt to stop disease progression. In this setting, use of a safe drug with a broad spectrum of antifungal activity is of great benefit. Based on preliminary data from veterinary medicine, and more information from the human field, posaconazole would appear to be a very appropriate choice in the cat, 4 although it is currently very expensive. Itraconazole is a good second choice, and much more affordable, while terbinafine is another option, depending on the particular fungal pathogen involved.
Case history
Presenting findings and empiric treatment
An 11-year-old spayed female domestic short-hair cat (2.8 kg) was presented on 7 July 2004 with two lesions affecting the skin. Abnormal physical findings consisted of swelling of the subcutis over the bridge of the nose and a focal swelling of the dewclaw pad of the left thoracic limb. Tartar and moderate periodontal disease were also detected. The cat had been in a fight 3 weeks earlier and it was thought that the skin lesions developed subsequent to penetrating injuries resulting from the fight, either bites or scratches.
The cat was treated empirically using amoxicillin clavulanate (50 mg twice daily).
Lesional biopsy
Eleven days later, the two lesions had improved slightly but had not resolved. This suggested that the primary problem remained, with improvement due to resolution of a secondary bacterial infection. It was considered most likely that a saprophytic pathogen was present in the lesions. Such pathogens can include fungi, Nocardia species, 5 Mycobacterium species 6 or conceptually similar bacterial, fungal or algal saprobes. 7 Accordingly, the cat was given a general anaesthetic and the lesions were biopsied by excision. Due to financial constraints, haematology, serum biochemical testing, and determination of FIV and FeLV status were not undertaken.
The lesions were examined carefully under anaesthesia, prior to marginal resection. The lesion on the bridge of the nose comprised a firm subcutaneous swelling, extending laterally in the direction of the medial canthus of the left eye. Although the lesion proximal to the left dewclaw had epithelialised, a prominent and fluctuant swelling was present within the subcutis. Representative material from both sites was collected aseptically using sharp dissection, placed in buffered formal saline and submitted for laboratory examination. Histopathological examination revealed pyogranulomatous inflammation in both lesions; each contained identical yeast-like elements which could be discerned in haematoxylin and eosin (H&E) stained sections. The fungi present were well demonstrated by periodic acid-Schiff (PAS) and Gomori silver staining techniques.

Appearance of the pad lesion after initial surgical debridement and biopsy. A crust is visible over the lesion

Location of the lesion affecting the subcutis of the nasal bridge. The photograph was taken about 10 days after incisional biopsy, and skin sutures (arrow) are visible along the line of the surgical incision
The location of the lesions is evident from Figs 1 and 2, which show the appearance of the biopsy sites some days after the procedure.
Mycological culture and identification
Several days later, subsequent to the detection of fungal elements in the biopsies, material from both lesions was collected for mycological culture (Fig 3); cotton-tipped swabs were used without any chemical restraint. Cytology of exudate expressed from the pad lesions revealed fungal elements with a morphology consistent with what had been observed in the tissue sections. A non-sporulating dematiaceous (pigmented) hyphomycete was isolated in pure culture on Sabouraud dextrose agar from both anatomical sites. The isolate (strain 2190/04) was sent to a fungal reference laboratory (Australian Reference Laboratory in Medical Mycology) for morphological identification, but species identification was not possible as the organism could not be induced to sporulate and classical identification relies on the morphology of reproductive structures.

The same lesion as shown in Fig 1, but after removal of the crust. A moistened swab was inserted into the lesion to obtain material for further cytology and mycological culture
The isolate was referred to a third laboratory (Mycology Laboratory, Westmead Hospital, Westmead, NSW) for molecular identification. DNA was extracted, 8 and universal fungal primers were used to amplify the ITS1, 5.8S and ITS2 regions of the rDNA gene cluster, 9 and the D1/D2 variable region of the 28S (large subunit [LSU]) ribosomal RNA. 10 PCR products were purified and sequenced. 8 ITS and LSU sequences were edited and merged using Chromas (version 2.23) software (Technelysium) and entered into a BLASTN sequence analysis search 11 provided by ANGIS/Biomanager (http://biomanager.info/) for species identification. The isolate was identified as most closely resembling Paraphaeosphaeria species, the teleomorph of Microsphaeropsis species, but could not be identified more reliably as the organism had not yet been listed in the GenBank database.
The sequence was aligned, using ClustalW, 12 with similar sequences from four isolates previously identified morphologically as Microsphaeropsis arundinis13,14 and a fifth organism, strain 04–359–2614, isolated from a patient at Concord Hospital (kindly provided by Dr Tom Gottlieb). These five isolates had also been assigned a molecular identification most closely resembling Paraphaeosphaeria species. Sequence alignments showed greater than 99% identity between the isolates, indicating our isolate and 04–359–2614 were M arundinis. Subcultures of the newly identified isolates (2190/04 and 04–359–2614) were submitted to the Orange Agricultural Institute where their identification was confirmed morphologically to be M arundinis. The GenBank accession numbers for the merged ITS and LSU sequences of all six M arundinis isolates are EF094551, EF094552, EF094553, EF094554, EF094555 and EF094556.
The slow growth habit of the organism precluded the determination of meaningful susceptibility data, even at the reference laboratory. The limited available literature on this organism suggested ketoconazole, itraconazole and terbinafine as being potentially useful antifungal agents that could be given orally, while amphotericin B could be given intravenously, subcutaneously or intralesionally.13–16
Our assessment was that the cat had been inoculated by a large amount of a saprophytic mould as a result of a fight with another cat; presumably the nasal bridge had been scratched deeply and there was also a penetrating injury in the distal left thoracic limb. Most likely, the heavy inoculum of fungal elements was present either on the claws of the perpetrator, or in the ground on which the cats fought, or both.
Treatment and outcome
Treatment was commenced on 16 August 2004 using itraconazole (50 mg with food once daily) as empiric therapy. Itraconazole was well tolerated, with no reduction in appetite to suggest drug-induced hepatotoxicity. The lesions resolved slowly but steadily during therapy, which extended approximately until January 2005, at which point the owner elected to discontinue treatment. The cat was outwardly completely normal on veterinary re-examination in March 2005.
Unfortunately, the nasal lesion recurred on 6 May 2006 (first recurrence), albeit it was situated somewhat more proximally (‘higher up’) on the bridge of the nose (Fig 4). The lesion was marginally excised on 15 May 2006 using an 8 mm skin biopsy punch inserted to the depth of the underlying periosteum and submitted for full investigation. At the laboratory, the biopsy specimen was partially bisected. Purulent material removed from a central cavity was used to make a smear and provide a specimen for culture. The biopsy was then placed in formal saline and submitted for histopathological examination.

Appearance of the nasal bridge at the time of re-biopsy on 15 May 2006, before (a) and after (b) excisional biopsy. The arrow in (a) highlights the lesion in the subcutis; the arrow in (b) shows the skin sutures along the incision line
Diff Quik-stained smears demonstrated numerous inflammatory cells (80% neutrophils, 20% large macrophages, with the occasional giant cell and scattered lymphocytes) and large numbers of spherical fungal elements within neutrophils and macrophages (Fig 5). The spherical fungal elements contained one to three compartments and occasionally elongated into septate hyphae (Fig 5). Histologically, there was a relatively well circumscribed, deep dermal granuloma comprised of sheets of large histiocytic cells with large amounts of vacuolated eosinophilic cytoplasm, often containing PAS-positive spherical bodies that sometimes formed short septate hyphae (Fig 6). There was a scattered infiltrate of neutrophils throughout the granuloma and a central space. The granuloma approached the deep surgical margin, with only a thin margin of loose connective tissue. At one location, there was a smaller granuloma adjacent to the main mass, closely approaching the lateral margin. While the granuloma may have been completely removed grossly, the surgical margin was not considered sufficiently ‘clear’ to be definitive. Indeed, the pathologist's impression was that excision may not have been complete.
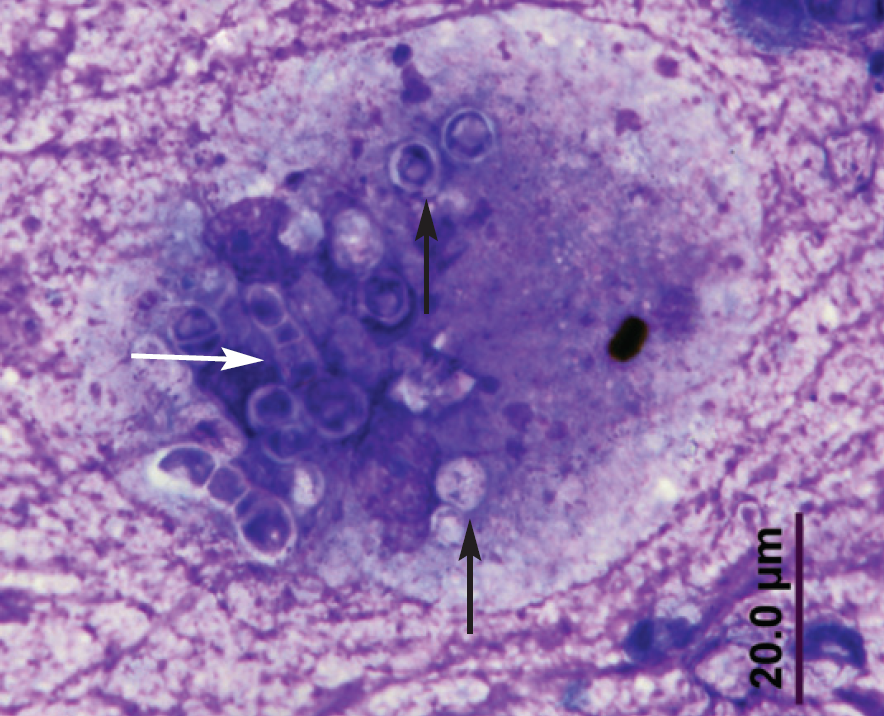
Diff Quik-stained smear from a swab collected from the lesion in Fig 4. Fungal elements were commonly yeast-like (black arrows) although some hyphal forms were also evident (white arrow). Fungal elements were often within macrophages

Photomicrographs of H&E-stained sections from a cat with a Microsphaeropsis arundinis infection at increasing powers of magnification. There is a marked pyogranulomatous cellular infiltrate, and organisms (arrows) are visible in the lesions at high magnification
Accordingly, the cat was again given itraconazole (50 mg once daily) for several months following surgery. This led to an apparent resolution of the problem, according to the owner, although the lesion subsequently recurred at exactly the same site in October 2007 (second recurrence). In the absence of reliable susceptibility data, the cat was treated empirically with fluconazole (33 mg twice daily), even though this agent is normally not very effective against dematiaceous fungi. This therapy seemed to have little impact on the appearance of the lesion, and fluconazole was considered inferior to itraconazole in terms of its in vivo efficacy.
At this time, a bottle of posaconazole syrup was donated by a human infectious diseases clinician, and this new triazole agent was tried at a dose (5 mg/kg once daily) suggested by McLellan et al, 17 and used subsequently by Wray et al. 4 The formulation (40 mg/ml oral suspension) proved to be palatable and allowed very precise dosing. Treatment resulted in a rapid decrease in the size of the lesion. The full bottle was administered to the cat over a 5-month period, and the cat remained apparently asymptomatic until February 2009 when the lesion again recurred (third recurrence). As no more donated posaconazole was available, the cat was given itraconazole again and it was anticipated that the cat would receive this agent indefinitely.
Discussion
Members of the class Coelomycetes are emerging as an important group causing soft tissue infections, especially in immunosuppressed human patients. Infections are usually localised and follow traumatic implantation, but may progress to invasive subcutaneous disease, as in the cat of the present report and a previous feline patient. 13 Airborne transmission of these fungi is unlikely to occur as they typically produce their conidia within enclosed structures (so-called conidiomata) that are not likely to be aerosolised. 18
Species of the genus Microsphaeropsis are coelomycetous anamorphic fungi that have probable affinities to the ascomycete genus Paraphaeosphaeria O Eriksson. 19 Microsphaeropsis species are typically found as saprobes and parasites of terrestrial plants. They inhabit branches and leaves of various plant hosts and are ubiquitous in soil and freshwater environments. 14 M arundinis is a plant inhabitant that was first described from the grass Arundo donax in Pakistan. In Australia, A donax is a ‘garden escape weed’ known as giant reed or elephant grass (Fig 7). It is a robust perennial that grows to a height of 6 m, 14 and is typically found growing along watercourses, roadsides and wetlands.

M arundinis was first described as an inhabitant of Arundo donax, a garden escape weed known as giant reed or elephant grass
M arundinis has been shown to be the cause of soft tissue infections in two human patients, both of whom had diabetes and were receiving immunosuppressive medications for intercurrent illness. 14 Antimicrobial susceptibility of these two isolates was determined using broth microdilution; both were susceptible to itraconazole (minimum inhibitory concentration [MIC] 0.25 mg/l) and amphotericin B (MIC 0.25 mg/l; and 0.125–1.0 mg/l). Treatment with terbinafine appeared to be curative in the first patient, while the clinical course in the second case was more complicated and hard to interpret.
A third isolate of M arundinis was obtained from the infected forelimb of a cat; the site of the lesion (the fourth digit) was amputated, and the cat was treated with ketoconazole and topical terbinafine without subsequent relapse. 13
A fourth isolate was obtained in 1981 in Sydney, Australia, from an ankle nodule of a patient being treated for acute myeloid leukaemia. Histopathology of the nodule showed both a leukaemic infiltrate and the presence of fungal elements. The fungus was grown on three separate occasions from specimens obtained from this site. 14
Other Microsphaeropsis species have been implicated in soft tissue infections. Microsphaeropsis olivacea was reported as causing a skin lesion in an otherwise healthy woman, 20 and was also isolated from the eye of a man with keratitis following ocular trauma. 21 Coniothyrium-Microsphaeropsis complex was isolated from the ulcerated knee of a diabetic woman who had received a renal transplant. 22
It is interesting that the lesion on the distal limb of the cat of this report responded quite promptly to therapy, with an apparently permanent cure of the infection at this site. A favourable response was also seen in the cat described by Kluger et al, 13 although the amputation of the toe may have facilitated the cure. In contrast, the nasal bridge lesion, although seemingly focal and of limited invasiveness, tended to recur, despite ‘debulking’ and appropriate follow-up antifungal therapy. Quite possibly the cytoreductive surgery was less aggressive than optimal; or the cat might have benefited from adjunctive topical therapy such as peri-incisional amphotericin B.15,16 Alternatively, perhaps the cool temperature of this superficial site may have favoured growth or persistence of this saprophytic organism.
Posaconazole is a very promising azole agent for treating fungal infections in cats. It has a very broad spectrum of activity, including Aspergillus species, Neosartorya species, pigmented fungi, Mucor species, 4 oomycetes (Pythium species) and yeasts. The formulation is a liquid suspension, which is palatable for most cats and permits very precise dosing on a mg/kg basis. Once daily administration seems to be effective. More pharmacokinetic data is required concerning its use in cats, and higher doses may prove to be even more effective! In our opinion, it is justified for empiric therapy of filamentous fungal infections, despite its high cost, until susceptibility data confirms that less expensive agents such as itraconazole or terbinafine are likely to be effective in vivo.
Addendum
In January 2010, a further two bottles of posaconazole were donated and given to the owner of the cat. There was a prompt improvement after re-instituting this agent. Although it is hoped that several months of continuous therapy with posaconazole will cure the infection, our advice will be to continue azole therapy indefinitely with either itraconazole or posaconazole, subject to availability.
Footnotes
Acknowledgements
The authors wish to thank Dr Tom Gottlieb for sharing information concerning his patient and Dr Michael Priest from the Orange Agricultural Institute for assigning a morphological identification. Richard Malik is supported by the Valentine Charlton Bequest of the Centre for Veterinary Education of The University of Sydney.
