Abstract
Clinical report
History
A 14-year-old female neutered domestic short-hair cat was presented to the referring veterinarian with a 2-week history of progressive hair loss and hind limb weakness. Diabetes mellitus (DM) had been diagnosed 8 months earlier and good glycaemic control achieved using 2 units of insulin glargine (Lantus; Sanofi-Aventis) q12h subcutaneously. Despite this, polyphagia, polyuria and polydipsia persisted.
Examination by the referring veterinarian revealed a sparse coat with hair that epilated easily, dry skin and a gallop rhythm. Serum biochemistry performed 5 months and 3 weeks before referral showed persistent hypokalaemia (3.2 and 3.0 mmol/l, respectively; reference range [RR] 3.7–5.4 mmol/l), hypochloridaemia (98 and 103 mmol/l; RR 106–123 mmol/l), metabolic alkalosis (serum bicarbonate 32 and 29 mmol/l; RR 12–24 mmol/l), and elevations in creatine kinase (CK) (1466 and 2165 U/l; RR < 361 U/l) and urea (12.3 and 11.6 mmol/l; RR 3.0–10.0 mmol/l). Serum total T4 (TT4) and free T4 (fT4) were both subnormal (TT4 < 5 nmol/l; RR 10–48 nmol/l; fT4 8.5 pmol/l; RR 15–48 pmol/l). Potassium supplementation was commenced (2.8 mEq potassium gluconate twice daily [Kalstat; Apex Laboratories]). However, hypokalaemia persisted.
Physical examination and initial investigations
On physical examination at referral the cat weighed 3.8 kg and had non-pruritic bilaterally symmetrical alopecia with marked, generalised thinning of the skin, comedones and dry flakes over the lateral thorax and abdomen, ventrum and dorsum. Cutaneous fragility was present, with skin tears measuring 1–3 cm in diameter noted on the lateral thorax (Fig 1). There was severe muscle wasting affecting all limbs and the spinal musculature, and the cat had a ‘pot-bellied’ appearance. On abdominal palpation, a cranial abdominal mass was suspected. A gallop rhythm and increased lung sounds were auscultated. Systolic blood pressure, measured by Doppler sphygmomanometry, was 173 mmHg.

Alopecia, dermal fragility, comedones and thinning of the skin of the lateral thorax and abdomen. (inset) Close-up view of the cutaneous changes
Putative diagnosis and differentials
An adrenal neoplasm causing endocrine alopecia and skin fragility was suspected and the cat was admitted for further investigation. Other differential diagnoses for the skin lesions such as paraneoplastic alopecia, hyper-sensitivities, ectoparasites and dermatophyto-sis were considered. Skin cytology and skin scrapes were performed and were negative for infectious agents. Dermatophyte culture was not performed and further investigation was declined.
Confirmatory tests
Abdominal ultrasound revealed diffuse hepatomegaly with hepatic venous congestion. The right adrenal gland was small, measuring 1.8 mm in diameter. A heteroechoic, lob-ulated, left adrenal mass, 46 mm in diameter, was identified adjacent the aorta and caudal vena cava, displacing the left kidney caudally (Fig 2). Free peritoneal fluid was present. Thoracic radiographs revealed a low volume pleural effusion, an alveolar-interstitial pattern in both ventral caudal lung lobes and an enlarged caudal vena cava. On routine echocardiography there was biatrial enlargement (left atrium 19.2 mm, right atrium 18.4 mm), right and left ventricular volume overload (left ventricular internal dimension [LVID] in end-systole 18 mm; RR 10–16 mm; LVID in end-diastole 10 mm; RR 6–10 mm) and normal left ventricular contractility (fractional shortening 44%). Myocardial wall thickness was within the reference range. On the basis of these findings, left- and right-sided congestive heart failure (CHF) was diagnosed.

Ultrasound image of the left adrenal gland mass showing caudal displacement of the left kidney
Plasma aldosterone concentration (PAC) was elevated (45,198 pmol/l; RR 110–540 pmol/l) while plasma renin activity (PRA) was normal (2.8 ng/ml/h; RR 0.3–3.0 ng/ml/h). The PAC:PRA ratio was elevated (16,142; RR 0.3–3.8). An adrenocorticotrophic hormone (ACTH) stimulation test was performed using 125 μg of cosyntropin (Synacthen; Novartis) administered intravenously. Progesterone, cortisol and androstenedione were measured before and at 1 and 2 h after administration. Progesterone was increased before ACTH (19 nmol/l; RR 0.19–2.22 nmol/l) and at 1 and 2 h post-ACTH (50 nmol/l on both occasions; RR 2.86–14.62 nmol/l). Cortisol was normal before ACTH (34 nmol/l; RR 25–75 nmol/l) and low at 1 and 2 h post-ACTH stimulation (78 and 77 nmol/l, respectively; RR 200–400 nmol/l). Androstenedione levels were below normal at all three measurements (1.83, 2.09 and 2.01 nmol/l at 0, 1 and 2 h post-ACTH, respectively; RR 3.84–12.56 nmol/l).
Based on these results, an aldosterone-and progesterone-secreting adrenal neoplasm was diagnosed. The owner declined further diagnostic investigations, including aspiration cytology of the adrenal mass.
Therapy and outcome
Therapy was commenced with benazepril (Fortekor; Novartis) 2.5 mg q24h orally (PO), furosemide (Lasix; Aventis Pharma) 10 mg q24h PO, aspirin 5 mg q72h PO and spirono-lactone (Aldactone; Pfizer) 6.25 mg q24h PO. Insulin glargine and potassium supplementation were continued as previously.
The cat was euthanased 8 weeks later by the referring veterinarian. Post-mortem examination was declined.
Discussion
In the case presented here, the constellation of clinicopathological abnormalities was suggestive of an adrenal tumour secreting both aldosterone and either cortisol or another adrenal steroid. The persistent hypokalaemia, metabolic alkalosis, elevations in CK and urea, and systemic hypertension in the presence of an adrenal mass were consistent with primary hyperaldosteronism (PHA).1–3 However, this could not account for the DM, polyphagia, polyuria, polydipsia, pot-bellied appearance and cutaneous fragility, which suggested concurrent hyperadrenocorticism or hyperprogesteronism.2,4,5 Measurement of PRA and PAC, and adrenal steroid profiling using an ACTH stimulation test, were undertaken to test for these endocrinopathies.
A search of the literature revealed a single report of hyperaldosteronism and hyper-progesteronism in a cat secondary to an adrenocortical carcinoma. 6 Unfortunately, identification of tumour type was not possible in our case.
Synthesis of more than one adrenal hormone by an adrenal tumour is unusual. Mechanisms include enhancement of the biosynthetic pathway of aldosterone in an aldosterone-secreting tumour, resulting in increased serum concentrations of the intermediates of aldosterone production, such as progesterone (see box below). 6 Alternatively, there may have been increased secretion of aldosterone and progesterone from neoplastic cells within the zona glomerulosa and fasciculata/ reticularis, respectively. Production of more than one hormone by this mechanism has been documented in humans. 7 Measurement of the intermediates of aldosterone production could be performed to elucidate the mechanism involved.
Steroid synthesis in the adrenal cortes
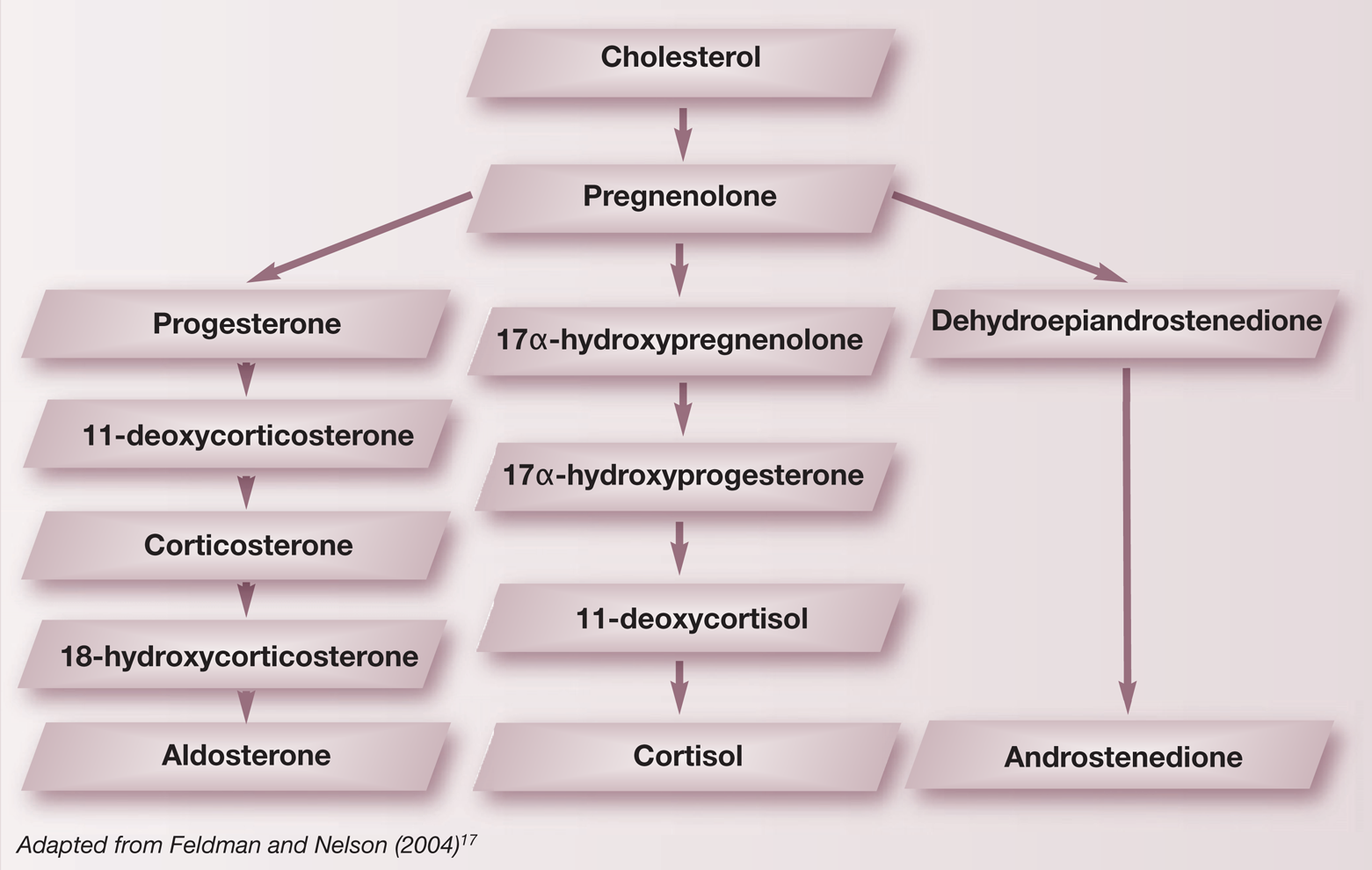
Hyperaldosteronism may be primary or secondary. In PHA, neoplastic or hyper-plastic cells in the zona glomerulosa secrete aldosterone autonomously. Negative feedback results in low or normal PRA. 8 Thirty-one cases of PHA in the cat have been described in the literature.1–3,9–13 In the majority of cases, PHA is caused by a unilateral adrenocortical carcinoma (10/31 cases), although unilateral (6/31) and bilateral (2/31) adrenal adenomas, and bilateral adrenal hyperplasia (3/31) have also been reported. In the remaining cases, the cause of PHA was not reported. Secondary hyperaldosteronism describes those conditions in which aldosterone is produced in response to activation of the renin-angiotensin-aldosterone system triggered by hypovolaemia or reduced effective circulating volume, and PRA is expected to be high.9,14 In the present case, despite concurrent CHF, PHA was confirmed based on elevated PAC in the presence of normal PRA and an elevated PAC:PRA ratio.
The clinical signs of PHA result from systemic hypertension caused by blood volume expansion, and include hyphaema, mydriasis and retinal detachment, and/or polymyopathy due to hypokalaemia, manifesting as weakness and cervical ventroflexion. Hypokalaemia is the most consistent laboratory finding. The hind limb weakness in this case may have been due to hypokalaemic polymyopathy or diabetic polyneuropathy. In previous reports of PHA, the magnitude of hypokalaemia was variable, and clinical signs were not consistently present. Additionally, the severity of hypokalaemia does not always correlate with clinical signs.2,6
Other clinical findings in the present case are consistent with previous reports of feline hyperprogesteronism.4,6,15 Of particular interest are the results of endocrine testing. The fT4 and TT4 concentrations were subnormal in this cat, a finding not previously reported in cats with hyperprogesteronism. Subnormal T4 may have been caused by the effect of progesterone on serum cortisol. Some progestins have considerable intrinsic glucocorticoid effect. In addition, progesterone binds to cortisol-binding protein (CBP) and may mimic hypercortisolaemia by displacement of cortisol from CBP, thus increasing the amount of circulating active, unbound cortisol despite normal total serum cortisol.
In dogs, chronic hypercortisolism suppresses pituitary secretion of thyroid-stimulating hormone, may alter binding of thyroid hormone to plasma proteins and may reduce peripheral iodination of T3 to T4. 16 Approximately 70% of dogs with hyperadrenocorticism have low T4 and/or T3 concentrations. In contrast, serum TT4 was subnormal in only 9% of cats with hyperadreno-corticism. 17 It is possible that excessive free cortisol caused suppression of the thyroid axis. However, the effect of excess circulating progesterone on thyroid hormone concentration has not been evaluated in the cat.
A subnormal adrenal response to ACTH stimulation, resulting in low post-ACTH cortisol concentrations, has been reported in two other cases of hyperprogesteronism in the cat.4,15 Potential mechanisms include either a lack of enzymes within the tumour necessary to convert progesterone to cortisol or excessive unbound cortisol due to displacement from CBP, resulting in suppression of the hypothalamic-pituitary-adrenal (HPA) axis. A lack of ACTH receptors within the neoplastic cells, although possible in some cases, does not explain our results since progesterone was stimulated by exogenous ACTH here. Low post-ACTH cortisol has been documented in dogs with atypical hyperadrenocorticism. In two of these dogs hyperprogesteronism was postulated to play a role through suppression of the HPA axis.18,19
Our case presented with CHF, which has been reported in one other case of hyperprog-esteronism. 15 While Doppler studies were not permitted in this case to rule out underlying myocardial diseases, including restrictive cardiomyopathy, relative glucocorticoid excess may have contributed to CHF. In cats, the administration of exogenous glucocorticoids has been demonstrated to cause plasma volume expansion and CHF as a result of an intra- to extracellular fluid shift.20,21 Excess endogenous glucocorticoids may have the same effect. Certainly, in humans with hypercortisolaemia, excess circulating glucocorticoids predispose to cardiac structural changes, diastolic dysfunction and CHF.22,23
Plasma volume expansion and hypertension secondary to aldosterone excess is a possible, but less likely, cause of CHF in the case reported here. Left ventricular wall thickness was normal, and end-diastolic and end-systolic ventricular dimensions were increased. By contrast, typical echocardiographic findings in cats with hypertension include mild left ventricular hypertrophy and a reduction in diastolic left ventricular diam-eter. 24 To the authors' knowledge, there are no case reports of CHF associated with hyper-aldosteronism in cats.
Trilostane, a competitive inhibitor of 3β-hydroxysteroid dehydrogenase, may have been useful in the medical management of this case. 3β-hydroxysteroid dehydrogenase is the enzyme responsible for conversion of pregnenolone to progesterone, 17α-hydroxy-pregnenolone to 17α-hydroxyprogesterone and dehydroepiandrostenedione to andro-stenedione. Trilostane therefore blocks all pathways of steroid synthesis (see box on page 760). Trilostane has been used by us and others to treat cases of hyperadrenocorticism associated with excess Cortisol and excess sex steroids in the cat, but its use in the management of hyperprogesteronism has not been reported.25–27
In summary, this second reported case of hyperaldosteronism and hyperprogesteronism in the cat suggests clinicians should be alert to the possibility of concurrent disease in cats with adrenal tumours showing clinical signs referable to the two conditions.
Conflict of interest statement
The authors declare no conflict of interest.
Sources of funding
No external sources of funding were provided for this case report.
