Abstract
A randomized double blind and placebo controlled design was used to investigate the hemostatic, biochemical, gastrointestinal and behavioral effects of pre- and postoperative administration of vedaprofen 0.5 mg/kg PO (V), tramadol 2 mg/kg SC (T), their association (VT) or placebo (P) in 40 adult female cats (3.0±0.32 kg; 1.8±0.7 years) distributed in groups of 10. Platelet aggregation and bleeding time were measured before and 52 h after ovariohysterectomy. Serum urea, creatinine, alanine aminotransferase, alkaline phosphatase and gamma-glutamyl transferase concentrations were measured before and 7 days postoperatively. The occurrence of vomiting, frequency and consistency of feces, and behavior were observed for 7 days postoperatively. Morphine (0.5 mg/kg, IM) was used as rescue analgesic. Laboratory variables did not change. Vomiting was observed only after morphine administration. Mild euphoria was observed in T and VT. The perioperative use of vedaprofen and/or tramadol did not modify the hemostatic, biochemical and gastrointestinal function in cats.
Non-steroidal anti-inflammatory drugs (NSAIDs) and opioids are the most commonly used substances for multimodal treatment of acute pain. However, their use may be limited in cats due their countercurrent side effects, 1–4 such as opioid-induced excitement, and hemostatic, renal and gastrointestinal disorders produced by NSAIDs. 5 The metabolism and excretion of NSAIDs in cats are also slow due to the impaired hepatic glucuronization. 6 The adverse effects of NSAIDs include anorexia, vomiting, diarrhea, nephrotoxicity leading to acute renal failure 7 and impaired platelet function with increased bleeding. 8 These changes are mainly related to the inhibition of cyclo-oxygenase-1 (COX-1) enzyme, responsible for the production of prostaglandins and A2 thromboxane (TXA2). Prostaglandins regulate renal blood flow and protect gastric mucosa, 9 and TXA2 is a potent platelet aggregation substance. 8
The therapeutic action of NSAIDs is mediated by COX-2 inhibition, the major isoenzyme responsible for prostaglandin production which is related to inflammation and hyperalgesia. 10 It is suggested that the adverse effects of NSAIDs are minimized when they are more specific COX-2 inhibitors. 9 However, recently it has been shown that COX-1 may also produce prostaglandins that contribute to inflammation, 11 and COX-2 is also expressed in normal conditions at the kidney, brain, and reproductive tract. 6
According to the comments above, the NSAIDs toxicity is dependent on COX specific inhibition. Vedaprofen is a preferential COX-2 inhibitor in dogs. 12 It is apparently safe in these species, as there was a low incidence of gastrointestinal changes, like vomiting and diarrhea after a long-term treatment for 30–56 days. 13–15 Although there is no information on selectivity of COX inhibition of vedaprofen in cats, 16 the administration of this drug for 3–5 days was well tolerated. 17
The analgesic and adverse effects of opioids are mediated by both peripheral and central nervous system receptors. 5 The adverse effects include bradycardia, respiratory depression, nausea, vomiting, reduced gastrointestinal motility and urinary retention. 18 Cats are more susceptible than dogs to the excitement effects of opioids, particularly when large doses are used. 5 Tramadol is an opioid analgesic with an unusual mechanism of action, involving μ agonism and activation of monoaminergic system (by inhibition of noradrenaline and serotonin reuptake). 19,20 Tramadol might be an alternative to the use of classic opioids, as this double mechanism of action could enhance the analgesic effect and improve tolerability. 21,22
This study aimed to investigate the safety of perioperative administration of vedaprofen, tramadol or their combination by studying the effects of these drugs on behavior, platelet aggregation, bleeding time and biochemical variables in cats undergoing ovariohysterectomy.
Materials and Methods
This study was approved by the animal owners and the Institutional Animal Research Ethics Committee under protocol number of 123/2003. Forty mixed breed cats were used (1.8±0.7 years; 3.0±0.32 kg). They were docile, not pregnant, and considered healthy by physical examination and hematologic, hepatic and renal function tests.
Cats were randomly and equally distributed into four groups: vedaprofen (V), tramadol (T), vedaprofen and tramadol combination (VT) or placebo (P). Vedaprofen (Quadrisol; Intervet) at 0.5 mg/kg (V and VT) or the same volume of placebo gel (T and P) was administered orally using a 1 ml syringe 1 h before anesthesia induction and 24 and 48 h after first treatment. Tramadol (Tramadon; Cristália) at 2 mg/kg, diluted in sterile saline solution to 0.3 ml (T and VT) or 0.3 ml sterile saline solution (V and P) was administered subcutaneously 1 h before anesthesia induction and every 8 h up to 72 h after surgery. Drug administration, platelet aggregation and bleeding time were performed by an examiner blind to the treatment.
Behavioral changes were evaluated pre- and postoperatively. Animal posture and mental alertness were investigated during rest and interaction activities as well as the ability to stand and walk. Sedation score was registered by visual analogue scale, ranging from zero, without any signs of sedation to 100, maximal sedation. Signs of excitement, suggestive of dysphoria and euphoria, were observed.
Animals underwent an initial 60 h adaptation period to experimental conditions. In the 24 h before surgery cats were anesthetized with 8 mg/kg propofol IV (Propovan; Cristália) for insertion of a 20-gauge catheter into the jugular vein for later blood sampling. Catheter patency was maintained by heparinised saline solution (20 UI/ml), the catheter was fixed to the skin with cyanoacrylate glue and covered with an elastic bandage. Before each sampling, 0.5 ml of blood was removed from the catheter and after blood sampling three times the volume of lactated Ringer's solution replaced the volume of blood. The catheter was maintained for 52 h postoperatively. For surgery, anesthesia was induced with propofol (8 mg/kg, IV), and lactated Ringer's solution was administered at 10 ml/kg/h. Anesthesia was maintained by isoflurane (Isoforine; Cristália) at 1.8±0.35% expired in 100% oxygen (500 ml/kg/min) using a non-rebreathing system. Animals underwent ovariohysterectomy through midline incision performed by the same experienced surgeon. Animals scoring 33% of pain numerical rating scale received rescue analgesia with morphine (Dimorf; Cristália) IM at 0.5 mg/kg.
Platelet aggregation was performed in eight animals per group. Blood was collected from the jugular catheter at 2 h before and 4, 28 and 52 h after surgery. When the catheter patency was not available at the last sampling, 0.05 mg/kg acepromazine (Acepran 0.2%; Univet) and 2 mg/kg ketamine (Ketamin; Cristália) were administered IV and blood was removed by jugular puncture. Blood (2 ml) was homogenized and stored at room temperature in sterile siliconized tube containing 3.2% of sodium citrate (Vacuette; Greiner Bio-One). Laboratory assay for measurement of platelet aggregation started 30 min after blood sampling. Blood was centrifuged at 500 rpm for 10 min and 400 μl of platelet rich plasma (PRP) was placed in an Ependorf tube, protected against light by aluminium paper and left for 30–40 min. The remaining blood was centrifuged at 3000 rpm for 10 min to obtain 400 μl of platelet poor plasma (PPP). Blood platelet and PRP count were performed in Neubauer chamber, using 1% ammonium oxalate. When the platelet count was above 400×103/μl in PRP it was diluted with PPP.
Platelet aggregation measurements were completed 2 h after sampling using a double channel aggregometer (Net Lab 2020) at 37°C. Maximal (PPP) and minimal (PRP) light transmittance calibration were performed for each sample. The sample was incubated for 1 min, before addition of 50 μl of the agonist adenosine diphosphate (ADP), at a fixed concentration of 10 μM. A maximal aggregation time of 5 min was allowed and the maximal platelet aggregation percentage registered. The morphology of the aggregation curve was evaluated.
Bleeding time was measured 24 h before and 52 h after surgery by puncture with a specific scalpel in the vein located on the external surface of the ear, due to difficulty related to assessment at the oral mucosa in the conscious cat. A tissue absorbent paper was used to dry the blood, without pressing at the puncture site. Time from puncture to the end of bleeding was measured by a chronometer.
Serum urea and creatinine concentrations were determined by the colorimetric and kinetic enzymatic methods, respectively. Serum alanine aminotransferase (ALT), alkaline phosphatase (ALP) and gamma-glutamyl transferase (GGT) were determined by the kinetic method. Blood samples (2 ml) were collected 18 h before surgery by the jugular catheter and 7 days after surgery by jugular puncture. The occurrence and frequency of vomiting and the consistency and frequency of defecation were monitored until 7 days after surgery.
Parametric data were analyzed by ANOVA for two factors (group and time) and repeated measurements with multiple comparisons by the Tukey test. Data were recorded as means and standard deviations. The pearson coefficient was used to evaluate correlation between platelet count and platelet aggregation. Significance level was considered as 5% (P<0.05). Analyses were performed using SigmaStat (Version 3.1; Systat Software).
Results
The jugular catheter was not patent at 52 h postoperatively in three animals from T, two from P and VT and one animal from V. There were no differences for platelet aggregation, blood platelet and PRP counts either between the groups or along the time in each group (Fig 1). Monophasic curves of platelet aggregation were observed in all treatments at the four time periods. The mean blood and PRP counts for all animals and times were 258±72×103/μl and 355±84×103/μl, respectively. A moderate correlation was observed (r=0.559; P<0.001) between the blood platelet and PRP counts (Fig 2). There was no correlation between the platelet aggregation percentage and blood platelet count (r=0.094; P=0.291) or PRP count (r=0.124; P=0.164) (Fig 3).

Platelet aggregation percentage (mean±SD) in response to ADP at the perioperative period of ovariohysterectomy in cats treated with tramadol (n=8), vedaprofen (n=8), vedaprofen and tramadol combination (n=8) or placebo (n=8).

Correlation between the blood platelet count and PRP count in cats treated with tramadol (n=8), vedaprofen (n=8), vedaprofen and tramadol combination (n=8) or placebo (n=8).

Correlation between the blood platelet count and percentage of platelet aggregation in cats treated with tramadol (n=8), vedaprofen (n=8), vedaprofen and tramadol combination (n=8) or placebo (n=8).
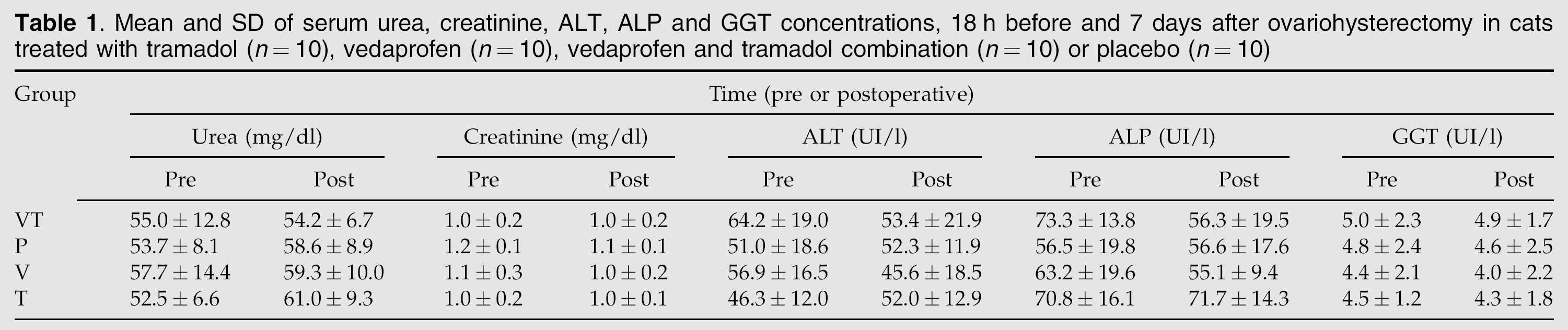
There was no difference for bleeding time either between the groups or over time for each group. Preoperative values were 68±15 s (VT), 59±12 s (P), 69±12 s (V) and 58±13 s (T) and 52 h postoperative values were 62±14 s (VT), 66±15 s (P), 69±21 s (V) and 63±21 s (T). There were no differences for serum urea, creatinine, ALT, ALP and GGT either between before and 7 days postoperatively or over time for each group (Table 1).
Mean and SD of serum urea, creatinine, ALT, ALP and GGT concentrations, 18 h before and 7 days after ovariohysterectomy in cats treated with tramadol (n=10), vedaprofen (n=10), vedaprofen and tramadol combination (n=10) or placebo (n=10)
Morphine was administered in all animals from P and V and in five animals from T. Nausea and vomiting were not observed in any animal until morphine was used. Ninety percent of P and all animals of V vomited after morphine. Nausea, lip liking and salivation were observed in 20% of the animals from T after morphine. Frequency and consistency of feces were not modified compared to the same animals during their adaptation period.
Sedation score was zero at all times. Signs of slight euphoria, like increased purring behavior and rubbing and scratching the front limbs were observed when animals were stimulated to interact with the observer in VT and T groups and after morphine analgesic rescue in all groups. Fixed staring, increased sensitivity to sound stimulation, anxiety, fright behavior and escape attempts were observed in two animals from T after morphine analgesic rescue and in two animals from P approximately 30 min after the second consecutive analgesic rescue with 0.5 mg/kg morphine IM (given 3 h after the first rescue).
Discussion
Perioperative use of vedaprofen, tramadol or their combination did not modify primary hemostasis and renal, liver or gastrointestinal function. This suggests that doses and administration intervals of these drugs were apparently safe in cats.
The main restriction of NSAIDs administration in healthy animals at the perioperative period is the potential nephrotoxicity associated with hypotension and/or hypovolemia. 10 Preferential COX-2 inhibitors apparently minimize bleeding and gastrointestinal disorders, however the risk of renal dysfunction remains. 11 Although serum urea and creatinine concentration are used routinely to investigate kidney function in cats treated with NSAIDs, 23–25 they have a low sensitivity to detect damage. 26 Renal clearance and glomerular filtration rate should be performed for a more complete evaluation of renal function in cats as performed previously in studies carried out in dogs. 27–30
Another factor of concern related to use of NSAIDs during the perioperative period is the risk of increased bleeding. These drugs inhibit platelet COX and reduce TXA2, a potent vasoconstrictor and platelet aggregation agent. 8 Primary hemostasis is mediated by interaction between the vascular endothelium and the platelets, where the pro-aggregation and vasoconstriction effects of TXA2 are balanced by the anti-aggregation and vasodilation effects of prostaglandin I2 and nitric oxide derived from the vascular endothelium. 31 Non-selective NSAIDs like ketoprofen, ketorolac, diclofenac 32,33 and naproxen 34 inhibit platelet function in humans. There is no consensus about the effect of ketoprofen 35,36 and carprofen 31,37 in primary hemostasis in dogs. Preferential COX-2 inhibitors, like meloxican and selective COX-2 inhibitors, like coxib-derivates, did not modify platelet function in dogs 36,38,39 and humans. 34,40,41 In cats, meloxican did not change the bleeding time. 24 In our study, vedaprofen alone or combined with tramadol did not inhibit platelet aggregation, or modified bleeding time.
ADP was chosen in this study as an aggregation agents, because it has been used in dogs 31,37–39 and humans 32,33,40,41 to investigate the effect of NSAIDs in platelet function. The ADP concentration used to induce platelet aggregation in cats range from 0.6 to 100 μM. 42–46 The concentration of 10 μM was used because this is the maximal concentration reported in studies in humans 32,33,40,41 and is the mean concentration reported in studies in dogs. 31,37 A fixed concentration was used in this study, therefore, there is no information whether different ADP concentrations would modify the response. The monophasic platelet aggregation curve observed in this study showed that the concentration of 10 μM of ADP is high to produce platelet aggregation in cats. 43
Platelet aggregation was measured about 6 h after administration of the NSAID. As in the first postoperative day, nine animals from P and V and four from T were treated with morphine 2 h before the test. The evaluation of platelet aggregation in these cases might have been compromised, due to the possible influence of morphine in this variable. In humans morphine potentiates agonist induced-platelet aggregation, by activation of α2 adrenoreceptors and mobilization of intracellular calcium. 47 However, in this study in cats, there was no significant difference in platelet aggregation after morphine administration when compared to the other times in any group.
There was no positive correlation between platelet aggregation percentage and either blood platelet count or PRP count. This supports the premise that platelet aggregation in to ADP is not influenced by platelet number. 42
Although direct jugular venepuncture has been used for blood collection to perform platelet aggregation test in cats, 42,44 this technique was not used here, as animal restraint, likely excitement and epinephrine release might increase platelet response to aggregation factors and produce clots in blood samples. 43 It is also known that cat platelets are more reactive when compared to other species. 44 For these reasons, blood was collected by jugular catheter, when possible. When the catheter was not patent at 52 h postoperatively, cats were anesthetized with low acepromazine and ketamine doses for atraumatic jugular venous puncture. These drugs are currently used for chemical restraint in studies on platelet aggregation in cats and do not appear to influence this variable. 43,48
Bleeding time is the most common in vivo method used to investigate platelet function abnormalities, and may be increased by the administration of NSAIDs, 8 however it is not reliable to predict bleeding disorders associated with the use of NSAIDs. 49 Bleeding time was measured at the external surface of the ear, unlike previous studies in cats 24 and dogs. 36,38 However, the results were comparable to tests performed at the oral mucosa in cats, 24 and this site could be considered an alternative location to avoid stress associated with major restraint in this species.
None of the drugs or their combination produced anorexia, vomiting, diarrhea or constipation, confirming previous studies with the use of vedaprofen in cats 17,50 and dogs. 13–15 Unlike cats, nausea and vomiting are the most common adverse effects produced by tramadol in humans. 22 This study also indicated that tramadol is related to a low incidence of constipation in cat. Short term use of tramadol and vedaprofen is apparently safe for hepatic function as there were no changes in the hepatic enzymes ALT, ALP and GGT.
Morphine analgesic rescue induced vomiting in animals given P and V and nausea in animals given T. Morphine and other opioids produce vomiting by activation of the vomiting center at the chemoreceptor trigger zone, probably due to stimulation of μ receptors. 51
Euphoria is the most common behavior observed after opioid administration in cats. 52 Dysphoria and excitement are usually related to opioid overdose. 5 Dysphoria was observed with the combination of tramadol and morphine and when morphine was administered at a short time interval of 3 h. Tramadol produced slight euphoria in cats at the first administration, which was reduced after repeated administration of this drug every 8 h. This finding has also been reported following the regular use of buprenorphine and levometadone in cats, 53 suggesting that tolerance is developed.
In conclusion, the perioperative use of vedaprofen, tramadol or their combination in cats according to the prescription protocol used in this study was not associated with significant changes in primary hemostasis, gastrointestinal, renal and hepatic function. Mild euphoria was associated with tramadol treatment.
Footnotes
Acknowledgements
This work was supported by the National Council of Technological and Scientific Development (CNPq). The authors thank Masterfoods South América (Effem Brazil) for supplying cat food.
