Abstract
Imiquimod is a topical immune response modifier and stimulator used in humans to treat a number of cutaneous neoplasms. This case report describes a cat with actinic keratoses and squamous cell carcinoma of the pinnae. The pinnal lesions were treated with topical 5% imiquimod three times per week. Treatment was discontinued after 82 days of therapy. Twelve weeks of topical imiquimod application resulted in clinical resolution of the pinnal lesions. Although no post-treatment biopsies were performed, there was no relapse of the pinnal lesions in 5 months of clinical follow-up. Expected side effects were limited to erythema, crusting, alopecia, and mild discomfort at the sites of application during the first 3 weeks of application. These results suggest that topical imiquimod, although unproven, might be a therapeutic option or adjunct to therapy for cats with actinic keratoses and squamous cell carcinoma, especially those cats for whom surgery and radiation therapy are not an option.
Squamous cell carcinoma is one of the most common skin tumors in cats, especially in climates that are warm and sunny (Rogers 1994, Fox 1995, Lana et al 1997, Gomes et al 2000). Chronic exposure to ultraviolet light is a major predisposing factor. Lesions often begin as actinic keratoses, which are characterized by cellular atypia and dysplasia of the epidermis and superficial follicular epithelium (Scott et al 2001). Actinic keratoses are premalignant lesions that are capable of becoming invasive squamous cell carcinomas. In cats, both actinic keratosis and squamous cell carcinoma are common on sparsely haired and poorly pigmented skin. The most common sites are the nasal planum, pinnae, and eyelids (Dorn et al 1971, Miller et al 1991, Fox 1995, Lana et al 1997, Thomson 2007). Early cutaneous features of actinic keratosis include erythema, scales, and crusts. Plaque formation and ulceration usually indicate the development of squamous cell carcinoma (Lana et al 1997, Scott et al 2001).
Early actinic keratoses can regress with avoidance of sunlight and photoprotection. Systemic retinoids, surgical excision, or cryosurgery may be recommended for more persistent or advanced lesions (Kwochka 1989, Bensignor and Carlotti 1995). A variety of treatment modalities including surgery, radiation therapy, strontium plesiotherapy, cryosurgery, laser surgery, photodynamic therapy, and chemotherapy have been used to treat feline squamous cell carcinoma with variable success (Withrow and Straw 1990, Clarke 1991, Shelley et al 1992, Ogilvie et al 1993, Peaston et al 1993, Theon et al 1996, Fidel et al 2001, Stell et al 2001, Goodfellow et al 2006). Surgery can be expensive, disfiguring, and requires general anesthesia. Most of the other treatment options mentioned above are expensive, require general anesthesia, are only available at selected referral centers, or some combination of these.
Imiquimod (Aldara; 3M), an imidazoquinolinamine, is an immune system modulator and possesses both potent antiviral and antitumor activity in animal models and humans (Stanley 2002, Schon et al 2003, Smith et al 2004). Current theories suggest that imiquimod acts both directly by inducing apoptosis and by inducing secretion of pro-inflammatory cytokines (Stanley 2002, Schon et al 2003, Lysa et al 2004, Smith et al 2004). Imiquimod activates macrophages and other cells via binding to cell surface receptors, such as Toll receptor 7, and thereby induces secretion of pro-inflammatory cytokines such as interferon-α (IFN-α), tumor necrosis factor-α (TNF-α), and interleukin-12 (IL-12). These cytokines bias towards a Th1-dominant immune response, which in general is associated with inflammation and tissue injury through activated inflammatory leukocytes and with cytolytic activity through CD8+ lymphocytes (Abbas et al 1996). Imiquimod can also induce apoptosis directly in a Bcl-2-family-dependent manner by down-regulating anti-apoptotic genes such as hurpin and HAX-1 (Schon et al 2003, Lysa et al 2004).
In the United States, imiquimod 5% cream (Aldara; 3M) is approved for the treatment of actinic keratoses and external anogenital warts (viral papillomas) in humans. It also has been used with variable degrees of success in humans with cutaneous neoplasms such as basal cell carcinoma, Bowen's disease, invasive squamous cell carcinoma, epitheliotropic lymphoma, and keratoacanthoma (Stanley 2002, Drummer et al 2003, Schon et al 2003, Bhatai 2004, Geisse et al 2004, Hengge and Schaller 2004, Navi and Huntley 2004, Oster-Schmidt 2004, Smith et al 2004). More recently, imiquimod 5% cream has been used successfully in treating horses with sarcoids (Nogueira et al 2006) and epitrichial sweat gland ductal carcinoma (Cihocki et al 2007).
Although there are no published reports on the use of topical imiquimod in cats, it has been used anecdotally with some success in treating squamous cell carcinoma in situ (Bowen's disease) and invasive squamous cell carcinoma (Veterinary Information Network, www.vin.com/Members/BoardsMain/Boards.plx?Read=2&ld=4*121*547141, Gill 2006). The purpose of this case report is to describe the use of topical imiquimod for the treatment of actinic keratoses and squamous cell carcinoma in a cat.
A 15-year-old, spayed female, white and gray, European shorthair cat presented to our clinic for dermatological evaluation. The cat had a history of slowly progressive, non-pruritic, bloody, crusted, and ulcerated lesions on the tips and lateral margins of both pinnae, a focal ulcerated and crusted lesion on the nasal planum, and mildly thickened and erythematous lower eyelids. Beginning 8 years earlier, the ear tips appeared to be ‘melting’ and became more blunted and rounded over time according to the owners. In the ensuing years, the pinnal lesions became plaque-like and ulcerated and tended to be exacerbated in warm weather (summer) and less so in cold weather (winter). The nasal planum lesion, which the owners described as a scab, was first noted 2 months prior to presentation. The scab exfoliated about 1 month prior to presentation revealing a deeply ulcerative lesion over the left nostril. In the last year, the lesions on both pinnae had worsened, and in the last 2 weeks, the nose lesion had become more crusted and bloody. None of the skin lesions were reported to be pruritic.
The cat was an indoor/outdoor cat, spending about 75% of the spring/summer outdoors, but most of the winter indoors. The patient received annual examinations, routine vaccinations, and deworming. The cat tested negative for feline leukemia virus (FeLV) as a kitten. The patient was reported to be systemically healthy. The diet consisted of a dry commercial kibble. No medications or topical treatments had been instituted for the skin lesions prior to presentation.
On general physical examination, the cat appeared in good health. The cat was slightly thin with a body condition score of 2 out of 5. Dermatological examination revealed a focal 12 mm (long)×6 mm (wide) erythematous and ulcerated lesion (about 2 mm deep) on the nasal planum extending from just dorsal to the left nostril into the haired skin. The tips of both pinnae were moderately blunted and erythematous with alopecia and multiple large crusts and ulcerations, ranging from 3 to 10 mm, along the lateral margins (Fig. 1). The largest crust on the left pinna was overlying a palpable nodule measuring 8×10×4 mm (Fig. 1). There was also a fine scale over the affected skin between the ulcerated and nodular areas. The lower eyelids were mildly thickened, erythematous and alopecic. The lower conjunctiva of the left eye was moderately swollen and erythematous. All skin lesions were present in white-haired and hairless areas. The history, signalment, and location of the skin lesions were strongly suggestive of solar-induced actinic keratoses and squamous cell carcinoma.
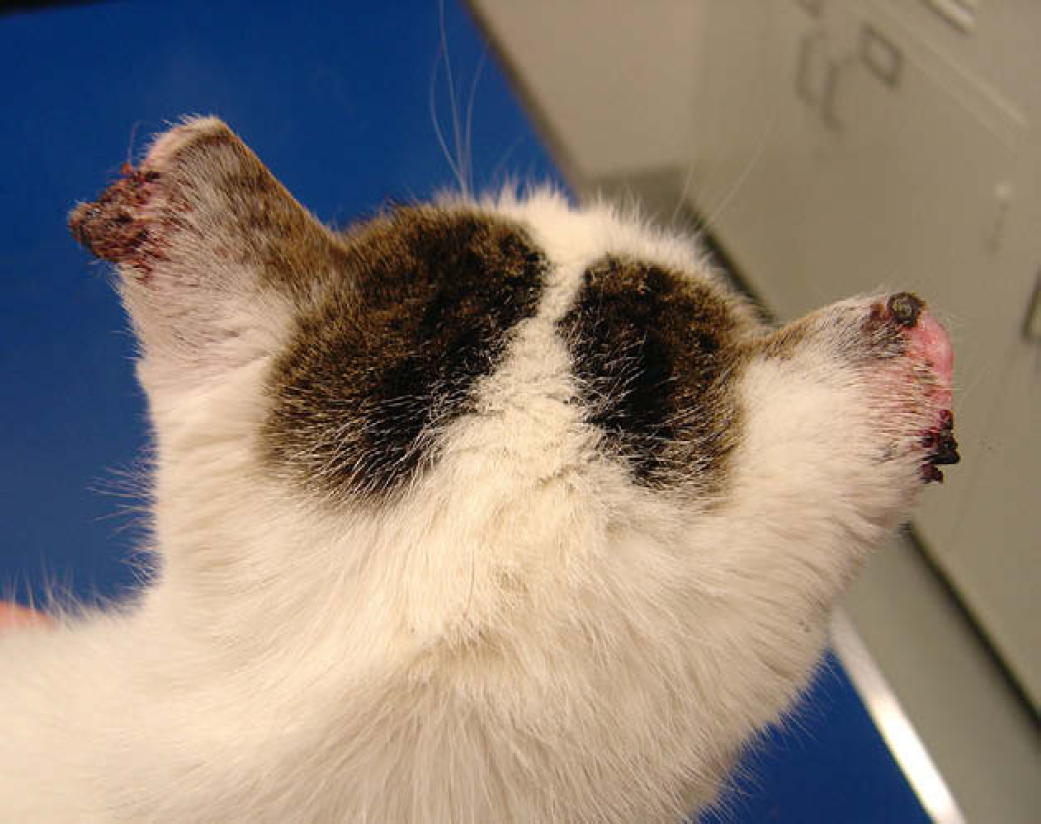
Photograph of the 15-year-old, spayed female, European shorthair cat with actinic keratoses and squamous cell carcinoma of the right pinna and a presumed spindle cell squamous cell carcinoma of the left pinna.
Impression smears from the ulcerated regions on the pinnae and nose were negative for microorganisms. Fine-needle aspirates from the nodular lesion on the left pinna showed a few abnormal keratinocytes with variably sized nuclei and nucleoli suggestive of neoplasia. No significant abnormalities were noted on a complete blood count, serum biochemistry profile, or urinalysis. Feline immunodeficiency virus and FeLV testing was negative. Thoracic radiographs were negative for metastasis.
Superficial incisional skin biopsies from both pinnae were obtained under mild chemical sedation and local lidocaine block for histological examination. Because of the assumed risk associated with the cat's age, the owners requested that the biopsies be performed without general anesthesia. Therefore, the nose lesion, presumed to be a squamous cell carcinoma, could not be biopsied. The right pinnal lesions were characterized by a moderately hyperplastic epidermis with multifocal areas of basal layer dysplasia and mild surface orthokeratotic to parakeratotic hyperkeratosis (actinic keratosis, Fig. 2a). In multiple areas there was invasion through the basement membrane into the underlying dermis with neoplastic epithelial cells forming fingers and nests (Fig. 2b). These cells were polygonal with mild to moderate anisocytosis and anisokaryosis. They had abundant eosinophilic cytoplasm and round to oval nuclei with prominent nucleoli. Within some neoplastic nests, central swirls of keratinization (keratin pearls) were noted. Individual cell keratinization was also seen. In some areas, there were up to four mitotic figures per 400× field. These histopathological changes were consistent with actinic keratosis and focal invasive squamous cell carcinoma and the neoplastic cells extended to all examined specimen margins. The nodule on the left pinna (Fig. 2c) was characterized by a single focal densely cellular infiltrative neoplasm extending from the epidermis through the dermis to the deep margins of the specimen and effacing the dermoepidermal junction. It was covered by a large serocellular crust. The neoplasm was composed of bundles of haphazardly interweaving spindled cells. The cells were moderate in size and distinct with a moderate amount of pale eosinophilic cytoplasm. The nuclei were centrally located and round to oval with finely stippled chromatin and one to two prominent central nuclei. The mitotic rate was moderate to high (12 in eight 400× fields) with some unusual mitoses and moderate anisokaryosis and karyomegaly. There were occasional keratin pearls and rare individual keratinized cells. Neoplastic cells of this nodule also extended to all the examined margins of the specimen.

Photomicrographs of the cat's skin showing features of (a) actinic keratosis (right pinna) including epidermal hyperplasia, dysplasia (D) and parakeratosis (hematoxylin and eosin 400×), (b) squamous cell carcinoma (right pinna) including fingers (F) and nests (N) of neoplastic epithelial cells that have invaded through the basement membrane and keratin pearl (KP) formation (hematoxylin and eosin, 200×), (c) presumed spindle cell squamous cell carcinoma (left pinna). Note the densely cellular bundles of haphazardly interweaving spindle cells. There are rare individual keratinizing cells (K), some unusual mitotic figures (M), and karyomegaly (KM) seen occasionally (hematoxylin and eosin 200×).
Immunohistochemistry was undertaken to help further define this unusual spindle cell tumor. Neoplastic cells were negative for cytokeratin, sarcomeric actin and melan A. Neoplastic cells were diffusely positive for vimentin and about 30% were positive for S100. The morphology of this neoplasm coupled with the results of the immunohistochemistry suggested that the most likely differential diagnoses for this neoplasm were fibrosarcoma and malignant melanoma. However, the multifocal areas of keratinization and early keratin pearl formation, taken together with the history, signalment and location of the lesions suggested the possibility of spindle cell squamous cell carcinoma (Sigal et al 2001). These neoplasms may be cytokeratin negative and only diagnosable by electron microscopy, which was not available to us in this case.
To reduce the inflammation associated with the presumed actinic blepharitis, 0.1% dexamethasone ophthalmic drops were prescribed at one drop in each eye twice per day. Inflammation was mild after 2 weeks, at which time the frequency of dosing was decreased to one drop on the lower eyelids once per day. A tapering dose of 0.1% dexamethasone drops were discontinued after 53 days of therapy.
Treatment options for feline cutaneous squamous cell carcinoma and actinic keratosis such as surgery, radiation therapy, strontium plesiotherapy, cryosurgery, photodynamic therapy and chemotherapy (Withrow and Straw 1990, Clarke 1991, Ogilvie et al 1993, Peaston et al 1993, Theon et al 1996, Fidel et al 2001, Stell et al 2001, Goodfellow et al 2006) were discussed with the owners. Because of the extent of this cat's lesions, surgical excision of both pinnae and the nasal planum was recommended and pre- or post-surgical radiation was also suggested. Because the owners were only interested in a less invasive and less expensive treatment option and were willing to try a novel form of therapy, treatment with imiquimod 5% cream was initiated instead. Avoidance of sun exposure was also recommended as a preventative for additional lesions (Rogers 1994). The owners were instructed to wear gloves and rub a small amount of topical 5% imiquimod into the lesions of the nasal planum and pinnae three times per week, which is the commonly used protocol in humans. The skin lesions looked worse after 15 days of treatment (Fig. 3). This was not an unexpected finding as topical imiquimod is known to produce a local inflammatory response and erythema and crusting are common side effects reported in human patients (Hengge and Schaller 2004, Navi and Huntley 2004).

Photograph of the skin lesions after 15 days of topical imiquimod 5% cream. The tips and lateral margins of the pinnae were completely covered by dark brown to black adherent crusts. The crust over the nasal planum was 15×15×3 mm.
After 29 days of treatment, the crusting noted on day 15 was markedly reduced. The ulcerative nasal planum lesion was 10×10×2 mm and it was partially covered by a hemorrhagic crust. A thin rim of alopecia and some crusting were present around the ulcer. Most of the crusts on the pinnae were gone. No plaque or nodular lesions were present, but there was some focal thickening on the left pinna. After 54 days of treatment, the pinnal lesions were continuing to improve, but there was limited improvement of the nasal planum lesion since day 29. Imiquimod therapy was continued for a total of 82 days. During the course of therapy, the owners reported some ingestion of the topical medication as the cat would lick the nasal planum after application. Numerous attempts were made to curb this behavior such as feeding immediately after application, holding the cat and using an E-collar, but it still occurred to some extent. A hemogram and serum biochemistry were repeated at this time (day 82) and no significant changes were noted.
An examination undertaken about 33 days after discontinuing imiquimod therapy showed both pinnae were alopecic at the tips and lateral margins but no other lesions were present other than a small hemorrhagic crust on the left pinna (Fig. 4a and b). There was an 8×8 mm hemorrhagic crust over the nasal planum lesion. The skin around the crust was moderately swollen, rounded, and erythematous.

Photographs showing (a) the clinical resolution of the pinnal lesions after 82 days of imiquimod therapy. (b) There is only a small crust remaining on the left pinna.
At this point, the pinnal lesions were clinically resolved. The nasal planum lesion was still present and there was no significant decrease in size. Because the nasal planum lesion was still present and likely to progress and eventually cause serious problems for the cat, other treatment modalities for this problem were recommended. Several options were discussed as before, including conventional surgery, cryosurgery, and/or radiation therapy, all of which were declined by the owners. Five months after imiquimod therapy was stopped, there was no recurrence of the pinnal lesions. The nasal planum lesion worsened about 4–5 months after discontinuing therapy. Palliative radiation therapy was recommended for the nasal planum lesion but was again declined by the owners. The cat was subsequently lost to follow-up.
Although surgical excision and irradiation are currently the best curative methods for treating squamous cell carcinoma in cats, this case demonstrates the potential use of a topical, easily applied and non-invasive therapy for this common feline neoplasm. Topical imiquimod may be especially useful for debilitated patients when surgery and radiation cannot be safely performed. In the cat reported here, no alterations were seen in the hemogram and serum biochemistry profile done 82 days after therapy started as compared to the blood work undertaken before the drug was initiated. The cat did not show any systemic side effects. The cat exhibited mild discomfort, transient erythema, and extensive crusting at the sites of application the first 2 weeks of imiquimod therapy. Alopecia also developed, especially at the tips and lateral margins of the pinnae. Beutner et al (1999) showed a dose-related severity of local reactions in people. Rest periods (days without treatment) were required by all patients applying imiquimod twice daily, half of those on a once-daily regimen, and one-quarter of those on a three times weekly protocol.
Extensive clinical trials in humans have shown that side effects of topical imiquimod are generally mild and not life-threatening. The most common side effects in humans from topical use include burning, itching and pain at the application site and surrounding erythema, scaling, pruritus, crusting, erosions, and edema (Geisse et al 2004, Hengge and Schaller 2004, Navi and Huntley 2004). Documented treatment toxicities of oral imiquimod in humans are dose-related and include flu-like symptoms, nausea, lymphopenia, fever, fatigue, increased alanine aminotransferase, hypotension, and depression (Savage et al 1996). Isolated accounts in people of drug-induced pemphigus foliaceus and pemphigus vulgaris have also been reported (Campagne et al 2003, Lin et al 2004).
In the cat reported here, the topical application of imiquimod caused clinical regression of the well-differentiated squamous cell carcinoma and the actinic keratoses of the right pinna, as well as the presumed spindle cell squamous cell carcinoma on the left pinna. Clinical resolution of the lesions was not supported by a post-treatment biopsy but, in the 5 months following therapy, there was no clinical recurrence of the pinnal lesions. Further, it is not believed that the neoplasms on either pinna were cured by biopsy excision as the biopsy specimens were very superficial and histopathologically neither neoplasm was completely excised.
There are several possibilities for the poor response of the nasal planum lesion to topical imiquimod. First, it could be attributed to the limited contact time with the imiquimod cream. As mentioned, the owners reported difficulty in preventing the cat from licking immediately after application of the cream. Second, the poor response may have been due to insufficient trial time. In humans, the treatment is generally given for 4–16 weeks. In this case, treatment was discontinued after almost 12 weeks. However, insufficient trial time is likely not the cause, as this particular lesion was not showing much improvement after day 29 of therapy. Third, if this presumed squamous cell carcinoma was poorly differentiated and deeply invasive, response to topical imiquimod may have been unlikely. Poorly differentiated and deep or invasive squamous cell carcinoma tends to respond less favorably to conventional therapies (Scott et al 2001). Fourth, the presumed squamous cell carcinoma may have been an entirely different type of neoplasia.
Topical imiquimod offers the option of a novel, relatively safe, non-invasive, conservative treatment for actinic keratosis and cutaneous squamous cell carcinoma. Although this case does not prove complete histopathological disappearance of the pinnal lesions, the apparent clinical resolution of the squamous cell carcinoma and actinic keratoses on both pinnae provides encouraging results for the potential use of topical imiquimod in treating this condition in other cats. Not only could this form of therapy be an alternative for sick or otherwise debilitated cats that cannot have surgery or radiation safely, but it might also prove useful as a post-surgery or post-radiation therapy in those cases where neoplastic cells have been left behind. Successful treatment of a squamous cell carcinoma by surgery followed by topical imiquimod has been reported in humans (Oster-Schmidt 2004). More clinical trials and carefully designed studies in cats are warranted to define the overall safety and effectiveness of topical imiquimod therapy for feline actinic keratosis and cutaneous squamous cell carcinoma.
Footnotes
Acknowledgment
The authors acknowledge the contributions of Dr Christine Miyasaki, 2005 graduate of Cornell University, College of Veterinary Medicine.
