Abstract
A 9-month-old cat was presented for routine vaccination before rehoming. Physical examination revealed a palpable mass in the cranial abdomen. The right kidney was severely enlarged (6 cm×4 cm) on plain abdominal radiographs, and failed to opacify normally during intravenous urography. Ultrasonography demonstrated a hydronephrotic right kidney. During exploratory coeliotomy, a retroperitoneal mass was identified, adherent to the caudal edge of the right kidney, enveloping the ureter and blocking urine outflow. The ureter caudal to the mass was of normal size. Right ureteronephrectomy was performed; the mass was subsequently freed from adhesions to the caudal vena cava and sublumbar muscles and excised. Histopathological examination revealed the mass to be composed of both normal and necrotic adipose tissue and fibrous tissue surrounding the ureter and a thrombosed, recanalised vessel. This appearance was consistent with an area of infarction and fibrosis with obstruction of the ureter. The cat was clinically well 3 months postoperatively.
Hydronephrosis is most commonly seen in association with ectopic ureters (Kyles et al 1996, Rawlings et al 2003, Steffey and Brockman 2004). Other causes of urinary outflow obstruction, and consequently of hydronephrosis, have been described in small animal medicine. Congenital causes have been described, such as torsion, kinking, stenotic or atretic ureters (North 1978), or presence of aberrant renal vessels encircling the ureter (Osborne et al 1972). Causes of ureteral obstruction can also be acquired such as urinary calculi, ligation of the ureter during ovariohysterectomy (Thun et al 1975), external compression of the ureters by masses (neoplasms, haematomas, cysts, and abscesses) (Speakman et al 1983, Ochoa et al 1999) or defined as idiopathic in absence of an evident explanation (Robotham 1978, Weller et al 1979).
Retroperitoneal masses as a cause of hydronephrosis have rarely been described in cats. When they occur, they are usually fibrous masses consequent to renal transplantation (Aronson 2002), urinomas resulting from urinary tract disruption (Moores et al 2002), or due to unknown causes (Leib et al 1988). In the present case ureteral obstruction resulted from retroperitoneal fibrosis associated with vascular thrombosis. The animal had no previous history of abdominal surgery or trauma, and the cause of thrombosis remains unknown.
A 9-month-old, 4.4 kg, neutered male domestic shorthair cat was in the process of being rehomed. When presented for check-up and routine vaccination, abdominal palpation of the animal revealed the presence of a painless mass. This was located in the dorso-cranial abdomen, on the right hand side, and was approximately 6 cm×4 cm. Clinical examination of the cat was otherwise unremarkable.
Blood and urine samples were obtained, and an ultrasound scan of the abdomen was performed.
Haematology was unremarkable; serum biochemistry revealed a mild elevation in urea (12 mmol/l; reference range 6.5–10.5 mmol/l), mild hypercalcaemia (2.77 mmol/l; reference range 2.30–2.50 mmol/l) and mild hyperphosphataemia (1.81 mmol/l; reference range 0.95–1.55 mmol/l). Feline immunodeficiency and feline leukaemia virus tests proved negative.
Urinalysis of a cystocentesis-derived urine sample revealed a small number of struvite crystals and squamous epithelial cells in the deposit, as well as large numbers of transitional epithelial cells and bacteria. Urine specific gravity was 1031 (normal–above 1035). Bacteriological culture of urine revealed a scant growth of Escherichia coli.
Although the serum creatinine level was within reference ranges, the increase of urea, calcium and phosphate together with mild impairment in urine concentrating ability, was suggestive of mild renal dysfunction. Antimicrobial therapy was consequently instituted using amoxyclavulanate (Synulox; Pfizer Animal Health, Tadworth, UK).
Ultrasonography, performed with a 7.5 MHz sector transducer (Megas; Esaote Bio-medica, Huntingdon, UK) demonstrated the mass to be a severely hydronephrotic right kidney; the left kidney appeared unremarkable. An associated dilated ureter was not found, and an area of increased echogenicity was apparent caudal to the right kidney.
Intravenous urography was performed in order to evaluate function in the contralateral kidney. Intravenous fluid therapy was instituted using Hartman's (lactated Ringer's) solution (Isolec; Ivex Pharmaceuticals, Millbrook Larne, Northern Ireland). General anaesthesia was induced using intravenous propofol (Propoflo; Abbott Animal Heath, Queenborough, UK), and maintained with a mixture of isoflurane in oxygen (Isoflo; Abbott Animal Heath, Queenborough, UK).
On the survey films, the right kidney was enlarged. It was impossible to clearly evaluate the perirenal area, due to the superimposition of other abdominal viscera. The left kidney appeared normal.
Intravenous urography was performed by injecting iohexol (Omnipaque; Nycomed, Marlow, UK) intravenously as a bolus at the dose of 400 mg of iodine per kg body weight. Exposures were taken at 5-min intervals, starting immediately after the injection of the contrast agent. These showed a normal nephropyelogram in the left kidney. It was possible to follow the left ureter along its course and entering the bladder in a normal ‘J’ shape. A slow and faint uptake of contrast material was apparent in the right kidney. Opacification of the parenchyma started 5 min and was maximal 10 min after injection. No excretion from the right kidney was evident, with consequent lack of visualisation of the right ureter. An irregular shaped structure of mixed opacity was evident on both views immediately caudal to the right kidney (Fig 1).
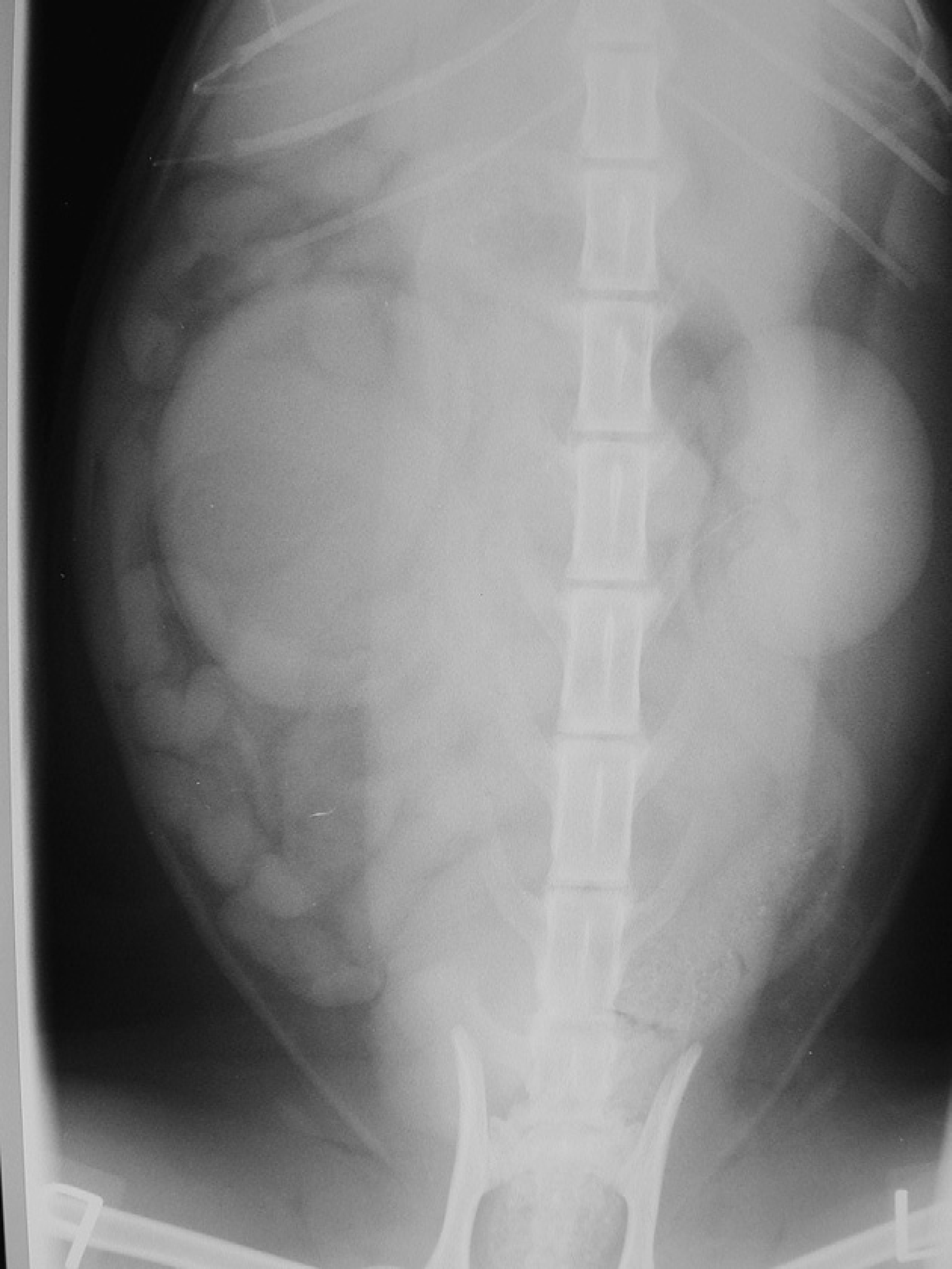
Ventrodorsal view of the abdomen after contrast injection, showing a faint enlarged right kidney.
Two days later, a ventral midline coeliotomy was performed. Following institution of intravenous fluid therapy using Hartman's (lactated Ringer's) solution (Isolec; Ivex Pharmaceuticals, Millbrook Larne, Northern Ireland), the cat was anaesthetised by means of the same regime used for the investigation.
On abdominal exploration the omentum was found to be adherent to the right perirenal area. A firm ovoid mass approximately 4 cm×3 cm enveloped the proximal ureter, immediately caudal to the hydronephrotic right kidney (Fig 2). The mass was beige to yellow, and had an irregular contour. Caudal to it, the ureter appeared to be of normal size, and entered the bladder neck in a normal position.

Intraoperative view of the mass located caudal to the right kidney. The kidney has already been removed, and a stay suture is in place to ease manipulation.
After ligation of the right renal blood vessels, the proximal ureter was dissected free to the level of the mass, and removed together with the kidney. The mass was cautiously dissected off the caudal vena cava medially and the sublumbar muscles dorsally. Dissection of the ureter then continued to the ureterovesicular junction. The distal ureter was ligated, sectioned, and removed. All ligatures were 3/0 polydioxanone (PDSII; Ethicon, Livingston, UK).
In view of the fact that the left kidney was ultrasonographically and macroscopically normal, it was decided not to collect biopsies from it at surgery.
Closure of the abdomen was achieved by means of a simple continuous suture using similar material. Suture of the subcutaneous tissue and of the skin was routine.
The cat made an uneventful recovery. Pain relief was provided for the first 24 h by means of intramuscular administration of buprenorphine (Vetergesic; Alstoe Animal Health, York, UK). Postoperative amoxyclavulanate (Synulox; Pfizer Animal Health, Tadworth, UK) was also administered for 5 days, pending tissue culture results. As these, as well as urine culture from a sample submitted 3 days after surgery, were negative, the antibiotics were discontinued.
Macroscopic examination confirmed severe hydronephrosis of the right kidney (Fig 3A), with thinned cortex. On histological examination interstitial fibrosis and inflammation were observed in the kidney. The excised periureteral mass (Fig 3B) consisted of normal and necrotic adipose tissue, and fibrous tissue. A fibrosed, recanalised thrombus was present in a small artery, likely to be the cranial ureteral artery, within the mass. Evidence of a thrombotic episode in the vessel (Fig 4A) was supported by presence of necrotic adipose tissue and fibrosis (Fig 4B). Bacteriological culture of the excised kidney and retroperitoneal tissue yielded no organisms and foreign material was not detected in the excised tissues.

(A) Excised right kidney, sectioned. Evident the thin cortex and the completely altered architecture. (B) Excised mass, sectioned. Histopathological evaluation revealed it to be composed by normal and necrotic adipose tissue and fibrotic tissue.

(A) Microscopic section of the excised mass: low power view of a portion of a perirenal mass from a cat, showing the ureter (small arrow) and a small artery (large arrow), the lumen of which is occluded by granulation tissue with several, small calibre vessels. Haematoxylin and eosin×4, bar=500 μm. (B) Microscopic section of the excised mass: low power view of one margin of a perirenal mass from a cat, showing fibrous tissue (upper left) and necrotic adipose tissue (lower right). Haematoxylin and eosin×4, bar=500 μm.
Blood pressure was evaluated, and an echocardiogram was performed, in order further to investigate possible primary cardiac disease, that may have given rise to an arterial embolus. The blood pressure was 138 mmHg (minimal risk of end organ damage if less than 150 mmHg) and echocardiography was unremarkable.
Fasted blood samples were taken 2, 3, 4, 6 and 10 weeks after surgery. On all occasions the blood urea concentrations were marginally elevated (between 13 and 17 mmol/l; reference range 6.5–10.5 mmol/l).
Serum creatinine concentrations were between 104 and 120 μmol/l (reference range 133–175 μmol/l); urine specific gravity, measured 3 and 10 weeks after surgery, was 1.038 and 1.044, respectively (normal–above 1.035). Urine culture performed on these occasions proved negative.
Three months postoperatively the cat was clinically normal.
Although the finding of E coli in the urine but not in the excised right kidney/mass, together with the mild renal dysfunction present preoperatively, raised the possibility of infection in the contralateral kidney, negative postoperative cultures suggested either the first culture to be falsely positive, or the elimination of the causative agent, and, therefore, determined the interruption of the antibiotic treatment after 10 days.
Furthermore, the fact that the urine concentrating ability was back to normal postoperatively suggests only a temporary impairment in the kidney function.
In the cat, trauma or neoplasia have been described as causing urinomas (Tidwell et al 1990, Moores et al 2002). The only documented cases of retroperitoneal fibrosis obstructing the ureter have been reported in feline renal transplant recipients (Aronson 2002). Histopathological characteristics of the mass found caudal to the right kidney in the present case were consistent with perirenal fat necrosis and retroperitoneal fibrosis. A thrombosed, recanalised artery was also found within the mass.
The cause of the thrombosis remains unclear, and it cannot be established whether the thrombus caused the necrosis with fibrosis in the adipose tissue, or was secondary to it.
Thrombosis and embolisation can occur because of local tissue or vessel injuries causing damage to the vessel wall and/or turbulent flow, or may be subsequent to systemic factors such as myocardial disease, thrombocytosis, or circulatory stasis (Ware 2003). Although emboli entering the peripheral circulation most commonly occlude the distal aortic trifurcation, they can also lodge within mesenteric, renal, pulmonary, cerebral or brachial arteries.
In this case, cardiac disease was excluded by further investigations (blood pressure measurement and echocardiogram), making arterial thromboembolism most unlikely.
Local trauma could also have been the initiating cause of altered circulation and consequent thrombosis in the area. However, in the excised mass there was no evidence of foreign material, as could have been expected in the case of penetrating trauma, or any associated infection or inflammation, and whilst there was no previous history of external trauma, and the animal had been always kept indoors, this aetiology cannot be excluded.
In dogs, an extrarenal arterial circle composed of aorta, renal artery, lumbar artery, internal spermatic and ureteral artery medially has been described (Christie 1980). In the horse, there can be many variations in the origin of the internal spermatic artery (Sisson and Grossman 1964). In the dog, both the proximal ureteral artery and the internal spermatic artery can arise directly from the aorta (Evans 1993). A collateral anastomotic branch could have been present between the internal spermatic artery and the ureteral artery in this cat. In that case it would have been possible that surgical trauma related to the neutering procedure of this cat could have injured the internal spermatic artery together with the local pararenal vasculature, especially if the technique had involved pulling rather than tying and cutting the spermatic cord.
Footnotes
Acknowledgements
The authors would like to thank Prof P.E. Holt and Prof G. R. Pearson for their comments on the manuscript and Mr Ivan Doran, for his help with the management of the case.
