Abstract
To evaluate the clinically normal feline cornea for the presence of virulent feline herpesvirus-1 (FHV-1), corneas from 31 cats (25 with normal eyes and six with active disease or corneal scarring) euthanased at a shelter were collected. Corneas from two specific pathogen-free cats were included as negative controls. Virus isolation (VI), fluorescent antibody (FA) staining and real-time polymerase chain reaction (rt-PCR) were performed on all samples. The presence or absence of dexamethasone in the media was evaluated for its effect on VI. VI was positive for FHV-1 in six corneas from five cats, all with clinically normal eyes. One cornea was positive for feline calicivirus (FCV) in addition to FHV-1, but only in media that included dexamethasone. Eight corneas were positive on rt-PCR for FHV-1, all from cats with clinically normal eyes. All positive VI samples were confirmed with FA staining. VI and rt-PCR were negative for FHV-1 and FCV in cats with active disease or corneal scarring. Data from this study indicate that virulent FHV-1 and FCV can be present in feline corneas that are clinically normal. Dexamethasone may enhance viral spread through a cell receptor mechanism.
Feline herpesvirus-1 (FHV-1) is a common pathogen of cats that causes upper respiratory and ocular disease. The virus is a member of the subfamily Alphaherpesvirinae, and as such develops neuronal latency following primary infection (Jones 1998). The trigeminal ganglion is a known site of FHV latency and recrudescent ocular infections may occur by reactivation of the virus and travel via the axons of the ophthalmic branch of the trigeminal nerve to the eye (Gaskell et al 1985). Herpesvirus DNA has been detected using polymerase chain reaction (PCR) assays in feline ocular tissues, both diseased and clinically normal (Stiles et al 1997a,b, Nasisse et al 1998, Burgesser et al 1999). Recovery rates of viral DNA from the corneas of asymptomatic cats have ranged from 5.9 to 49% (Stiles et al 1997a, Nasisse et al 1998, Townsend et al 2004). Recovery rates of viral DNA from the conjunctiva of asymptomatic cats have ranged from 2.6% to 31% (Stiles et al 1997b, Burgesser et al 1999, Low et al 2007). FHV-1 isolation from conjunctival swabs of asymptomatic cats was negative in two studies (Stiles et al 1997b, Holst et al 2005) and 10.7% in another study (Burgesser et al 1999). To the authors' knowledge, FHV-1 isolation from the normal feline cornea has not been reported.
The presence of FHV-1 DNA in the cornea of cats may represent active infection (actively replicating virus – usually associated with clinical disease), low level persistent infection (virus replicating at low levels but not causing clinical disease), latent infection (the classic definition being that cell free infectious virus cannot be recovered from fresh homogenized tissue but virus can be induced to reactivate through explantation or cocultivation), or remnants of viral DNA fragments (not capable of replicating) (Stevens and Cook 1973, Stroop and Baringer 1982, Abghari and Stulting 1988, O'Brien and Taylor 1989, Townsend et al 2004). The findings of several studies suggest that herpesviruses may develop corneal latency in rabbits, mice and humans (Cook et al 1987, Abghari and Stulting 1988, O'Brien and Taylor 1989, Kaye et al 1991). If the same is true for cats, it could explain the propensity of some cats to develop recurring corneal ulceration associated with FHV-1 without the virus being activated in the trigeminal ganglia and traveling to the eye.
A previous study from our laboratory sought to demonstrate latency associated transcripts (LATs) in the feline cornea (Townsend et al 2004). Although LATs, which represent viral transcripts of small strands of RNA and are considered to be molecular markers of latency, were identified in the trigeminal ganglia of some clinically normal cats, LATs were not documented in the cornea. Although several studies have documented FHV-1 DNA in the normal feline cornea (Stiles et al 1997a, Nasisse et al 1998, Townsend et al 2004) the replicative status of the virus has not been determined. This information is important in understanding the biologic behavior of FHV-1 and its ability to cause clinical disease in the cat. The purpose of the current study was to identify FHV-1 DNA and the replicative status of FHV-1 in the corneas of clinically asymptomatic cats, as well as in a group of cats with ocular disease that was clinically consistent with FHV-1 infection.
Materials and methods
Corneas were obtained from 31 cats euthanased at the local animal shelter, and from two healthy specific pathogen-free research cats euthanased for reasons unrelated to this study. Of the 31 cats from the shelter, 16 were males and 15 were females. Ages were estimated, with 17 classified as young (≤3 years), 13 classified as middle aged (4–8 years), and one classified as old (≥9 years). No medical history was available for the shelter cats. The research cats were both 2-year-old neutered males. Of the 31 cats from the animal shelter, 25 had a normal ocular examination using slit lamp biomicroscopy and had no evidence of respiratory tract disease. Six cats had evidence of active or prior ocular disease that could be consistent with FHV-1 infection (ocular discharge, conjunctivitis, keratitis, corneal ulceration or corneal scarring). These cats were included as probable FHV-1 positive cats. The research cats had normal ocular examinations and no evidence of systemic disease. These cats were included as negative control cats.
The corneas from each cat were harvested using sterile instruments within 1 h of euthanasia. The corneas were rinsed in sterile saline and placed in sterile cryo vials containing Dulbecco's modified essential medium (DMEM) and placed on ice. Within 1 h tissues were transferred to a −80°C freezer where they were stored until processing.
Cell culture and virus propagation
Crandell Reese feline kidney (CRFK) cells were grown in DMEM with 8% fetal calf serum, bicarbonate buffer, gentamicin, streptomycin and amphotericin B. A virulent field strain of FHV-1 was propagated on CRFK cells, harvested and used for positive controls in this study. The virus titer was determined to be 107 TCID50 (50% tissue culture infective dose)/ml.
Virus isolation (VI)
Corneas were thawed and cut into pieces with a sterile scalpel blade. The pieces were then placed in a sterile disposable tissue grinder (Kendall Precision Disposable Tissue Grinder, The Kendall Company, Mansfield, MA, USA) with DMEM and homogenized. The homogenate was spun at 4°C at 3000 RPM for 10 min. The supernatant was removed and separated into two aliquots. The aliquots for VI were passed through a 2 μm syringe filter (Millipore Corporation, Ballerica, MA, USA).
CRFK cells were grown in 96 and 48 well plates until approximately 90% confluent. The corneal filtrate samples were added to the wells (50 μl/sample for the 96 well plates and 150 μl for the 48 well plates). All samples were run in duplicate. Two additional replicates from each sample had 0.1% ophthalmic dexamethasone added to each well (dexamethasone sodium phosphate ophthalmic solution, Falcon Pharmaceuticals, Ft. Worth, TX, USA). The final concentration of dexamethasone was 4×10−8 M. This concentration was determined to have no detrimental effect on CRFK cells (data not shown). The plates were incubated in 5% CO2 at 37°C for 7 days. Positive controls consisted of FHV-1 added to the wells and negative controls consisted of CRFK cells in media only. All plates were examined daily for 7 days for cytopathic effect (CPE). The supernatants from the initial inoculations were passed one time on to fresh CRFK cell monolayers. All VI samples that were positive for CPE also had supernatant frozen at −80°C for evaluation by PCR. All plates were stained with a direct FHV-1 fluorescent antibody (FA).
FA staining
Plates were fixed with 80% acetone for 10 min and then air dried at 25°C. The plates were then stained with a direct polyclonal FHV-1-specific FA (American BioResearch, Pullman, WA, USA) labeled with fluorescent isothiocyanate and examined with an inverted microscope.
Real-time PCR (rt-PCR)
A previously published rt-PCR protocol (Vogtlin et al 2002) was used to amplify a conserved 81-bp sequence within the open reading frame of the glycoprotein B (gB) gene of FHV-1 using a Stratagene Mx3000P detection system.
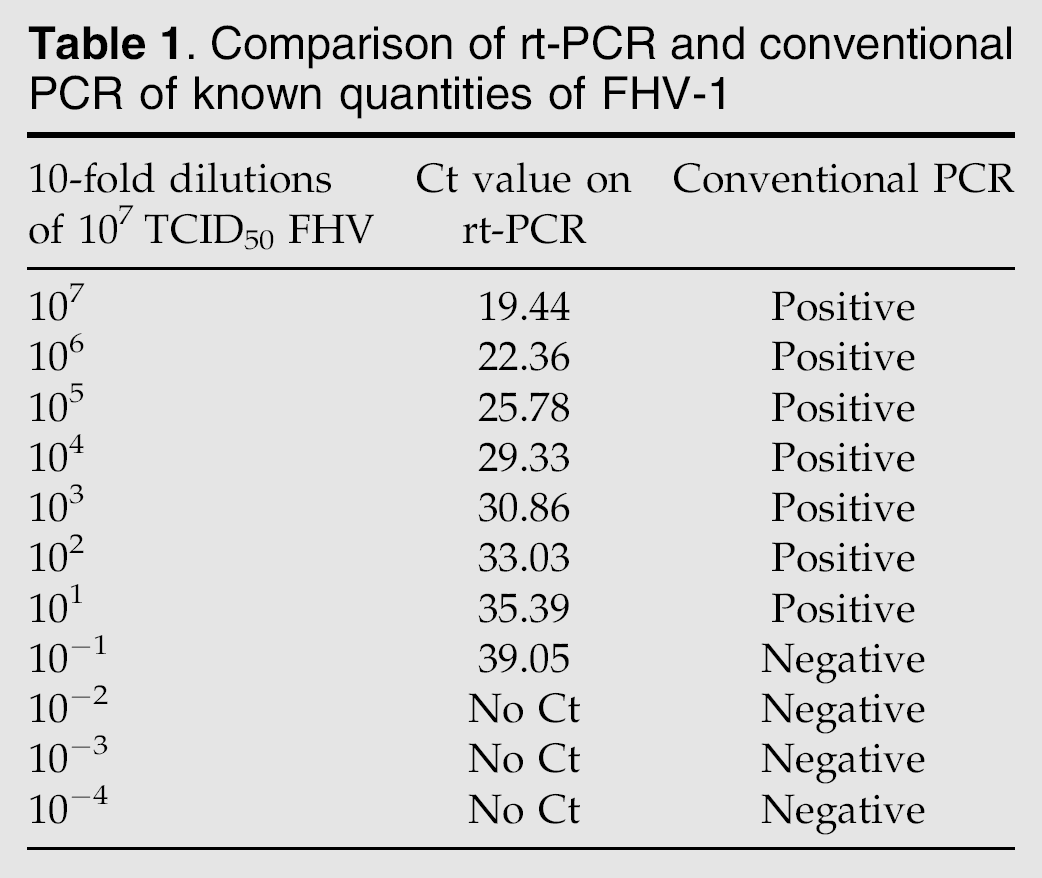
Ten-fold dilutions of FHV-1 stock (107TCID50/ml) in DMEM were used to establish a standard curve to quantify virus and compare virus titration values to the Ct values of rt-PCR.
Unfiltered aliquots of the corneal homogenate supernatant were used for rt-PCR. Viral DNA was extracted from 80 μl of each sample using a commercial extraction kit (QIAamp DNA Mini Kit, Qiagen, Valencia, CA, USA) and following the tissue preparation protocol. Positive controls consisted of DNA extracted from FHV-1 propagated in cell culture. Negative controls consisted of extraction and PCR controls in which water was the template. All samples were run in triplicate. Samples with a Ct value less than ≤40 were considered positive.
Conventional PCR
A previously published conventional PCR protocol (Vogtlin et al 2002) was used to amplify a 737-bp fragment of the gB portion of the FHV-1 genome. Ten-fold dilutions of FHV-1 stock (107 TCID50/ml) in DMEM were used to evaluate the detection limit of conventional PCR using an Applied Biosystem detection system (Applied Biosystem, Foster City, CA, USA). The PCR detection limit was compared to the rt-PCR detection limit.
Electron microscopy
The supernatant from six VI samples that were positive for FHV-1 were evaluated by electron microscopy using a negative staining technique with 2% phosphotungstic acid. An additional two samples that were positive for CPE but in which other diagnostic tests were negative were also examined by electron microscopy.
Results
VI
Six of 66 (9%) corneas from five cats were positive for FHV-1 isolation. These were confirmed by FA staining and rt-PCR. Samples that were positive for CPE, but in which FA staining and rt-PCR were negative were not counted as positive samples. All of the positive samples were from the shelter cats with normal eye examination findings. All cats were from the young aged group and all were females. One cornea had positive feline calicivirus (FCV) isolation in addition to FHV-1 isolation. This result was confirmed by FCV indirect monoclonal FA staining (Custom Monoclonal Antibodies, Sacramento, CA, USA) and reverse transcriptase PCR using a previously published protocol (Scansen et al 2004). The opposite cornea from this cat was negative for VI. Of the six corneas positive for FHV-1 isolation, three were positive on the initial culture and three were positive on the first passage. All corneal samples from shelter cats with clinical signs of ocular disease were negative for VI, as were the corneal samples from the specific pathogen-free research cats.
Dexamethasone
Of the six corneal samples positive for FHV-1 isolation, one was initially positive only in wells that included dexamethasone. In the first passage, this cornea was positive for FHV-1 isolation in wells that did and did not include dexamethasone. The one cornea that was positive for FCV was only positive, on both initial culture and first passage, in wells that included dexamethasone.
rt-PCR
Serial 10-fold dilutions of the stock virus correlated well with the Ct values from rt-PCR (Table 1). Over a range of six logs (107–102) a linear regression curve was calculated, using the formula Y=−3.397×log(X)+44.48 (R2=0.996, efficiency=97%).
Comparison of rt-PCR and conventional PCR of known quantities of FHV-1
Eight of 66 (12%) corneas were positive for FHV-1 on rt-PCR. Six of these eight positive samples were positive for FHV-1 isolation, including two corneas from one cat. Of the six corneas positive for VI, two were positive on rt-PCR on the tissue homogenate and four were positive on rt-PCR on the initial VI supernatant. Two corneas from two cats were positive on rt-PCR and were negative on VI (Fig 1). All samples that were positive on rt-PCR were from shelter cats with normal eye examination findings. All cats were from the young aged group, with six females and one male represented. All corneal samples from shelter cats with clinical signs of ocular disease were negative for FHV-1 rt-PCR, as were the corneal samples from the specific pathogen-free research cats.

Correlation of Ct value to copy number of FHV-1 on samples positive for rt-PCR on corneal tissue. FHV-1 copy numbers are expressed as the log value of TCID50. The two samples with the higher FHV-1 copy number were positive for FHV-1 isolation, whereas the two samples with lower FHV-1 copy number were negative for VI.
Conventional PCR
Serial 10-fold dilutions of the stock virus (107 TCID50/ml) were evaluated for detection limit on conventional PCR. The 737-bp band could be detected on a 1.5% agarose gel stained with ethidium bromide at a dilution that correlated to 10 TCID50/ml (Table 1).
Electron microscopy
In two of six (33%) samples that were positive on VI and evaluated by electron microscopy FHV-1 particles were identified. Two additional samples that had been suspicious for CPE but in which other diagnostic tests did not confirm either FHV-1 or FCV were negative for viral particles on electron microscopy.
Discussion
This study documents the presence of FHV-1 capable of replicating in the corneas of asymptomatic cats. Whether this finding represents latency of FHV-1 in the cat cornea or low level persistent infection is not possible to determine. The latter is more likely given the fact that CPE was observed within a few days of either inoculation or first passage in cell culture. In some studies of rabbit corneas, in which herpes simplex virus (HSV) latency has been suggested based upon recovery of viable virus during a period of latency, CPE was observed from 1 to 6 days (O'Brien and Taylor 1989), 7 to 26 days (Cantin et al 1992) and 15 to 35 days (Cook et al 1987). In mice HSV was isolated from the corneas of latently infected animals from 10 to 24 days after being placed in cell culture (Abghari and Stulting 1988).
A study of human corneal buttons removed during penetrating keratoplasty for prior herpetic disease, as well as non-herpetic disease, found that LATs could be identified in eight of eight corneas from patients with prior herpetic disease and three of 10 corneas of patients with non-herpetic disease (Kaye et al 1991). This finding represents considerable evidence that HSV can develop corneal latency in humans. In this same study only one cornea from a patient with prior herpetic disease was positive for HSV isolation.
The definition of corneal herpesvirus latency at this point is a functional one, with the precise state of viral DNA in the cornea still unknown (Cook et al 1987). In a prior study from our laboratory (Townsend et al 2004), LATs could not be identified in the feline cornea. This finding does not imply that LATs are never present in the feline cornea, or that FHV-1 cannot develop corneal latency in the cat, but we were unable to verify it.
The finding of neither positive VI samples (FHV-1 or FCV) nor any positive FHV-1 rt-PCR samples from the shelter cats with active ocular disease or corneal scarring suggestive of prior FHV-1 ocular infection was surprising. These cats were included in this study in order to serve as probable FHV-1 positive control cats. Other potential causes of ocular disease such as Chlamydophila felis were not investigated as that was not the purpose of this study. The finding of FCV in one cornea was unexpected. Although known to be a conjunctival pathogen (Stiles and Townsend 2007), FCV has not been reported as a corneal pathogen of cats. This finding warrants further investigation before any clinical significance can be determined.
Dexamethasone was included in the VI portion of this study as a direct effect of dexamethasone on corneal cell–HSV interaction has been documented (Weinstein et al 1982). In that study of rabbit corneal cells and HSV, dexamethasone increased cell susceptibility to the virus and facilitated the spread of virus through the corneal cell culture, even at concentrations of dexamethasone as low as 10−9 M. In the absence of exogenous immunologic factors, this activity was thought to be mediated by a corneal cell glucocorticoid receptor. Although the presence of glucocorticoid receptors in the feline kidney cell has not been reported, two VIs in the current study, one FHV-1 and one FCV, occurred during the initial inoculation only in wells containing dexamethasone suggesting the same receptor may be present.
Dexamethasone applied topically to the eyes of rabbits promotes HSV shedding in a reactivated model of latent infection (Gordon et al 2003). Dexamethasone has been suggested to exacerbate HSV keratitis in humans following penetrating keratoplasty surgery and in a rabbit model there was a higher prevalence of corneal ulceration, stromal keratitis and VI in animals treated with topical dexamethasone compared to controls following penetrating keratoplasty (Beyer et al 1989). A prospective study of 38 humans undergoing corneal transplantation confirmed that some patients develop HSV related keratitis following corneal transplantation and that the use of postoperative topical dexamethasone may trigger clinical disease (Robert et al 2003). In an experimental model of FHV-1 keratitis in cats, corneal sequestra developed in cats that received subconjunctival betamethasone (Nasisse et al 1989). In feline patients, the use of ophthalmic corticosteroids such as dexamethasone and prednisolone has often been associated with a worsening of herpetic disease, or emergence of herpetic disease when these agents are used to treat a different problem (Stiles 2003).
The finding in this study of virulent FHV-1 in the normal feline cornea has important clinical implications. Although no history was available for the shelter cats in this study, the finding of virulent FHV-1 in the corneas of some cats does not necessarily imply previous herpetic keratitis. Based on findings in human medicine, viral DNA as well as positive VI may be found in the corneas of individuals with no history of clinical disease (Robert et al 2003). Recurrences or emergence of ocular herpetic disease during periods of stress or other illness in cats may be more likely if the virus is present within the cornea rather than only within the trigeminal ganglia. The presence of virulent FHV-1 within the cornea of some asymptomatic cats means that corneal transplants from one cat to another should probably be avoided. Lastly, the use of topical corticosteroids may play a role in activating corneal FHV-1 in some cats through a cell receptor mechanism.
Footnotes
Acknowledgment
The authors thank Pamela Kirby, RVT and Melinda Anderson, RVT for technical assistance.
