Abstract
A 14-year-old neutered male domestic shorthaired cat was presented to the University Veterinary Centre Sydney for evaluation and treatment of dental disease. This cat developed an unusual bradyarrhythmia under anaesthesia. The possible causes and treatment of the dysrythmia are discussed.
A 14-year-old neutered male domestic shorthaired cat (6.3 kg) was presented to the University Veterinary Centre Sydney for assessment of dental disease. The cat had been diagnosed with chronic kidney disease (International Renal Interest Society (IRIS) stage 2) 2 years previously, and systemic hypertension (systolic pressure 200 mmHg before therapy, measured by Doppler sphygmomanometry) 1 year prior to presentation. In addition, the cat had experienced an undiagnosed dysrhythmia during a previous anaesthetic although no dysrhythmia had been detected at any time during physical examination. The cat did not have palpable thyroid lobes and its serum total thyroxine concentration (23 nmol/l) was within the reference range (5–39 nmol/l). Echocardiography had been performed 11 months previously (see Table 1).
Echocardiograph results

IVS, intraventricular septum; LVI, left ventricular inflow.
The results of these investigations were considered most consistent with acquired left ventricular hypertrophy secondary to systemic hypertension associated with chronic kidney disease. A standard electrocardiogram (ECG) performed on the same day as echocardiography was normal.
At the time of presentation, the patient was receiving amlodipine (1.25 mg in the morning; 0.625 mg at night) and benazepril (2.5 mg once daily) orally for treatment of hypertension.
On physical examination, the cat had severe periodontal disease and weight loss (7.2–6.3 kg over 11 months). Both kidneys were small on palpation, the heart rate was 160 beats per minute (bpm) and systolic blood pressure was 140 mmHg (using the Doppler method). Abnormalities on serum biochemistry included azotaemia, with serum concentrations of creatinine 270 μmol/l (reference range 90–180 mmol/l) and urea 12.59 mmol/l (reference range 7.2–10.7 mmol/l).
Together with the progression of chronic kidney disease, severe periodontal disease was considered to be contributing to the weight loss, and dental extractions under general anaesthesia were recommended to the owner.
The patient was premedicated with butorphanol (1.3 mg intravenously), which provided mild sedation. The heart rate was 140 bpm after premedication. General anaesthesia was induced with alphaxalone (Alfaxan® CD-RTU; 12.8 mg slowly IV), the cat was intubated with a cuffed endotracheal tube (5 mm internal diameter) and connected to a Bain breathing system with the isoflurane vaporiser set on 2%.
The cardiac rhythm was initially sinus, with a rate of 150 bpm, but periods of bradycardia began to appear until, 5 min after induction, the heart rate was 90 bpm.
A standard six lead ECG was performed after the ECG monitor revealed abnormal ECG tracings (Fig 1).
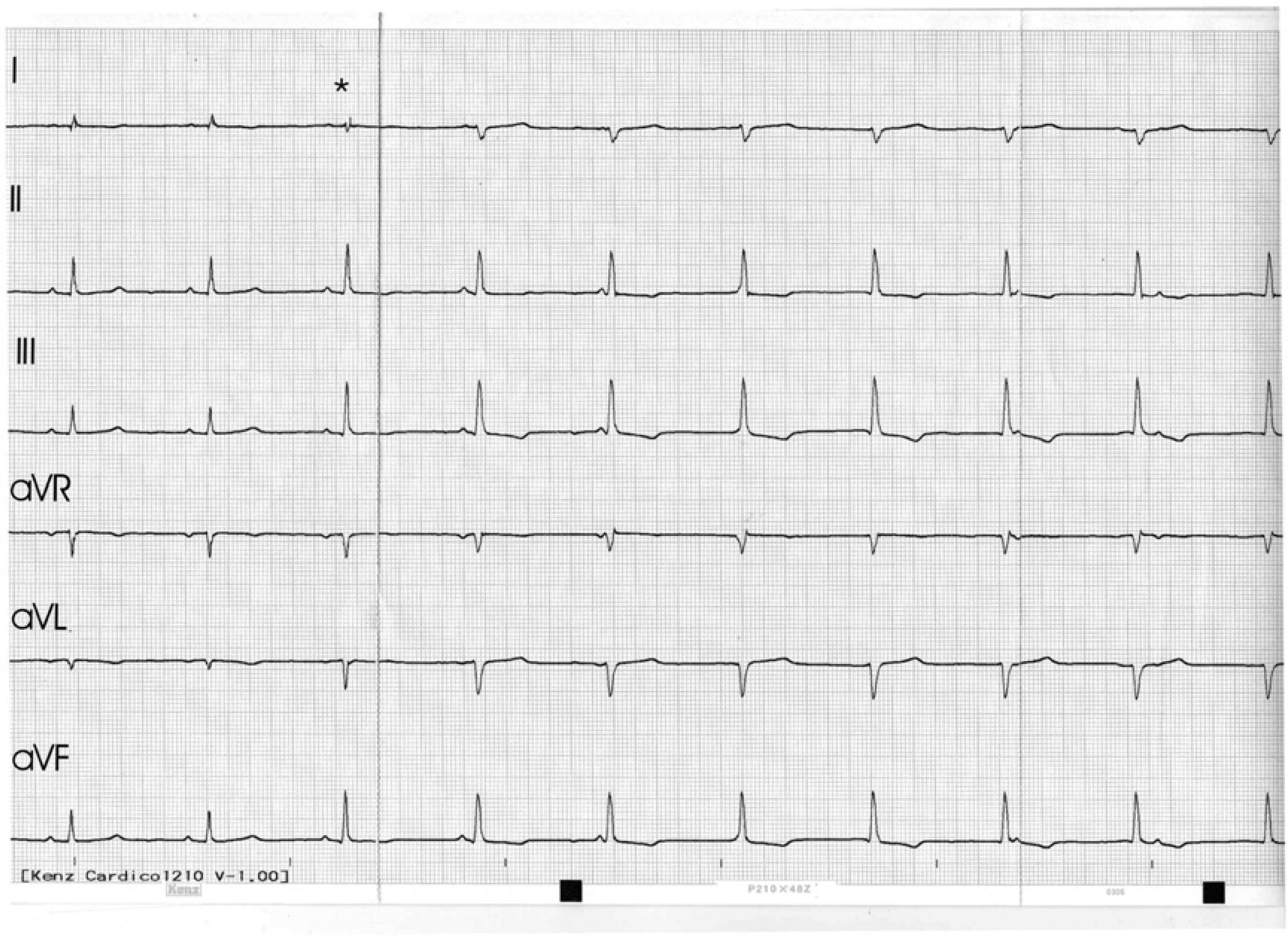
Simultaneous six lead ECG tracings from a controlled hypertensive cat anaesthetised with alphaxalone and isoflurane (10 mm/mV, 50 mm/s).
Questions
What is your electrocardiographic diagnosis?
How would you manage this rhythm disturbance under anaesthesia?
Speculate as to how and why this dysrhythmia may have occurred.
ECG of the month (Answers from page 522) Bradyarrhythmia in an anaesthetised, elderly, hypertensive cat
1. Interpretation of the ECG tracings
In Fig 1, the first two P–QRS–T complexes are normal except for a prolonged PR interval (100 ms; reference range 50–90 ms) and QT interval (260 ms; reference range 70–200 ms) (Tilley 1992) with a rate of 93 bpm. The third beat (marked with an asterisk) has a slightly shorter PR interval (90 ms) and a different R wave (taller in lead II (0.7 mV vs 1.2 mV) and differing in morphology in lead I). Subsequent complexes have progressively shorter PR intervals (60 ms, then 0 ms, then disappearing into the QRS complex), with the P–P interval being longer than the R–R interval. This is indicative of artioventricular (AV) dissociation, ie, the atrial and ventricular rhythms are independent of each other. The rhythm is described as AV dissociation rather than third degree AV block as, in the latter, the atrial rate is much faster than the ventricular rate and in this case, the atrial rate is slower than the ventricular rate.
For the complexes after the asterisk, the R–R interval is invariable (600 ms), suggesting that an idioventricular rhythm is present with the AV dissociation. Because the rate of spontaneous firing of the sinoatrial (SA) node has been reduced, the ectopic focus discharges at a rate sufficiently rapid to just capture the cardiac rhythm periodically. The P–P interval is longer than the R–R interval, thus the P–R interval initially shortens until the P wave is superimposed on and finally falls after the QRS complex. Occasionally, during this process, a point is reached where the atrial impulse (originating from the SA node) is able to recapture the cardiac rhythm. When this occurs, a capture beat is evident (Fig 2, star), associated with a prolonged PR interval.

Simultaneous six lead ECG tracings from controlled hypertensive cat under isoflurane anaesthesia (10 mm/mV, 50 mm/s).
2. How would you manage this rhythm disturbance under anaesthesia?
The primary problem in this case was the slowing of SA nodal discharge rates although there is also evidence of slowing of AV conduction (prolonged PR intervals). This may have been the result of myocardial injury secondary to chronic hypertension, increased vagal tone as a consequence of the opioid premedication, an effect of amlodipine on calcium channels in SA and AV nodal cells, hypothermia or an effect of the isoflurane.
The dysrhythmia occurred during early anaesthesia (5 min after connection to isoflurane) and disappeared completely approximately 15 min after disconnection from isoflurane. The cat was anaesthetised for 57 min and warming was provided in the form of a heating pad, hot water bottles and covering of the animal. Ideally, the cat's temperature should have been taken at the time of the dysrhythmia to definitively exclude hypothermia as a cause but this was not undertaken. The cat's postextubation temperature was 37.1°C. Despite the fact that hypothermia is one of the causes of bradycardia in an animal under anaesthesia, it was considered to be an unlikely aetiology in this case because of the onset of the dysrhythmia so early in the anaesthetic (the cat was unlikely to be hypothermic at this time) and the fact that the temperature at extubation was 37.1°C, a temperature not low enough to result in this type of dysrhythmia. It has been reported that a sinus bradycardia occurs initially at temperatures of around 32°C (Solomon et al 1989).
Two doses of atropine (250 μg each) were administered intravenously to determine the extent to which the low rate of sinus node discharge was attributable to increased vagal tone. Despite waiting 5 min after each dose there was no discernable effect on the cardiac rate or rhythm. We considered the possibility of reversing the effects of butorphanol using intravenous naloxone, although butorphanol alone had not affected the cardiac rhythm and naloxone could not be expected to fully reverse the effects of the butorphanol. Instead, it was decided that the rate and rhythm were sufficiently stable to permit the expeditious completion of the dental procedures, while carefully monitoring the patient's ECG tracings, peripheral perfusion and oxygenation (using pulse oximetry readings and Doppler blood pressure determinations).
The isoflurane was discontinued 57 min after intubation, and by the time of extubation, 9 min later, the cardiac rhythm was largely sinus with a heart rate of 113 bpm, although still with periods where the dysrhythmia recurred. In the recovery room following extubation, the heart rate was 140 bpm with a sinus rhythm. It remained at this rate throughout the night and the further recovery of the patient was uneventful. The cat's body weight had returned to 6.7 kg 6 months after the affected teeth were extracted.
3. How and why did this arrhythmia develop?
AV dissociation usually results from abnormalities of impulse formation or conduction, rather than a primary disturbance of rhythm. Possible causes include depressed automaticity of the SA node, increased automaticity of the AV junction or ventricles and disturbed AV conduction (Tilley 1992).
We considered that the primary physiological process in this patient was suppression of the normal automaticity of the SA node under anaesthesia. An auxiliary pacemaker developed in the ventricles, and competition between this site and the SA node was responsible for the rhythm disturbance encountered in this patient.
Volatile anaesthetic agents can be considered to be non-specific calcium channel blockers (Aguilera and Vaughan 2000, Yamakage 2002) and calcium channels are known to be involved in the pacemaker depolarisation of the SA node action potential (Zeljko et al 1983). Halothane, isoflurane and enflurane have been shown to decrease heart rate by altering the rate of phase 4 and phase 0 depolarisation of pacemaker cells within the SA node, thereby favouring the emergence of ectopic pacemaker formation (Zeljko et al 1983).
The dihydropyridine calcium channel blockers have also been shown, through their effects on L-type calcium channels, to prolong the action potential duration in the SA node (Masumiya et al 1997) although the anti-hypertensive action of these agents is mediated by peripheral vasodilatation (Poelaert and Roosens 2000).
Inhalational anaesthetic agents have been shown to prolong AV nodal conduction time in the dog (Atlee et al 1986, Yokota 1989) and in isolated heart preparations in vitro (Raatikainen et al 1998), as well suppressing calcium channel function in canine cardiac Purkinje cells (Eskinder et al 1991). This effect has been shown to be unrelated to the depth of anaesthesia (Atlee et al 1986). Isoflurane has been shown, however, to slow AV conduction the least of the available agents (Atlee et al 1986, Raatikainen et al 1998) or not to slow conduction at all (Nakaigawa et al 1995).
Amlodipine is a dihydropyridine calcium channel blocker that, in humans, has been reported to have a minimal effect on AV nodal conduction time at therapeutic doses (Dunlap et al 1989, Vetrovec et al 1991, Freysz et al 1996, Yamakage 2002). The dihydropyridine calcium channel blocker nifedipine also does not affect AV conduction (Yokota 1989, Yamakage 2002) but when administered concurrently with a volatile anaesthetic agent causes significant prolongation of the sinus cycle length (Yokota 1989). Experimentally, calcium channel blockers and volatile anaesthetic agents have been shown to interact in such a way as to increase AV conduction time additively when isoflurane is used (Marijic et al 1988, Yokota 1989, Raatikainen et al 1998).
It has been recommended that caution be taken when calcium channel blockers and volatile anaesthetic agents are administered concurrently (Marijic et al 1988, Yokota 1989, Raatikainen et al 1998) because of the possibility of additive effects on AV conduction.
Given the experimental data and the clinical presentation of this case, including the fact that a higher than average dose of amlodipine was being used, it seems possible that the bradyarrhythmia which occurred under anaesthesia, could have been the result of an interaction between the calcium channel blocker used to control blood pressure and the volatile agent used to maintain anaesthesia. Failure of atropine to reverse the bradycardia suggests that increased vagal tone was not a key pathomechanism and the occurrence of the arrhythmia very early in the anaesthetic as well as the fact that the postextubation temperature indicated only mild hypothermia makes this an unlikely cause of the arrhythmia.
