Abstract
A vulvar leiomyosarcoma was diagnosed in an 8-year-old mixed-breed, sexually intact cat. The cat had a history of vulvar swelling, bleeding and stranguria. The mass was located at the ventral commissura of the vulva. The cut surface of the mass was lobulated and greyish-white, with areas of necrosis and haemorrhage. Histologically, it was a spindle-cell sarcoma consisting of interlacing bundles of very elongated neoplastic smooth muscle cells and variably dense collagenous matrix separated by individual cells or streams. It also contained areas of focal necrosis and haemorrhage. The mitotic index ranged from seven to 12 mitoses per 10 high-power fields. Immunohistochemically, the spindle-shaped neoplastic cells were strongly positive for smooth muscle actin (SMA) and vimentin, whereas no immunoreactivity was obtained for desmin. The vascular elements of the internal control tissue also exhibited strong SMA and vimentin immunoreactivity. Based on the morphological and immunohistochemical features, the tumour was classified as a leiomyosarcoma.
Leiomyosarcomas (LMSs) are commonly encountered mesenchymal neoplasms. Of all LMSs the one located in the genitalia is the most common in dogs and has been well documented (Cooper and Valentine 2002). However, only one case of genital LMS had been reported in the uterus of a domestic cat (Miller et al 2003). Five cases were, however, reported in the uteri of 219 (2.2%) zoo felids (Chassy et al 2002). In addition, there are also a few reports of feline leiomyosarcoma presented in other body sites (Burk et al 1975, Cooper and Valentine 2002, Miller et al 2003).
In November 2004, an 8-year-old mixed-breed, sexually intact cat was referred to a private clinic with the complaint of enlargement and bleeding of a vulvar mass, which was noticed by the owner 2 months previously. According to the findings of the complete blood count and physical examination, the mass was diagnosed as an abscess and the patient was administered medical therapy for 7 days. About 2 weeks later, the cat was submitted to the department of Obstetrics and Gynecology with difficulty in urination. Examination revealed a firm, round restricted and ulcerative mass located at the ventral commissura of vulva, obstructing the vaginal canal (Fig 1). The mass was evaluated as a tumour and removed surgically.

Perineal region; cat. The initial vulvar mass, almost closing the entrance.
Forty-seven days later, the animal was presented again, with local perineal recurrence of the initial mass, and difficulty in defecation, lethargy and anorexia (Fig 2). Thoracic and abdominal radiographies revealed no abnormality. Because of the probability of the recurrence of the tumour, ovariohysterectomy was performed with the surgical removal of the recurred masses. The animal was euthanased on request of the owner in a private clinic after the second surgery. Necropsy was not performed.

Perineal region; cat. Local recurrence in the perineal region.
Macroscopically, the cut surface of the initial mass (35×10×12 mm) and the recurred masses (multiple, ranging from 3–6 mm in diameter) were lobulated and greyish-white, with areas of necrosis and haemorrhage. For microscopic examination specimens were initially fixed in 10% buffered formalin, embedded in paraffin wax, sectioned at 4 μm and then stained with haematoxylin and eosin and Masson's trichrome.
Immunohistochemical (IHC) labelling was performed with the streptavidin–biotin peroxidase complex technique using primary antibodies provided by Neomarkers (Lab Vision, 47790 Westinghouse Drive, Fremont, CA 94539, USA). Briefly, endogenous peroxidase activity was quenched by incubation with 0.3% hydrogen peroxidase in methanol for 15 min at room temperature (20–25°C). Sections were then incubated in a humid chamber with the primary antibodies for 1 h at room temperature. The primary antibodies used were as follows: mouse anti-vimentin (Clone V9) monoclonal antibody (mAb) at a dilution of 1:400, mouse anti-smooth muscle actin (SMA/Clone 1A4) mAb at 1:200, and mouse anti-desmin (Clone D33) mAb at 1:75. Prior to anti-vimentin labelling sections were pretreated in 10 mM citrate buffer solution (pH 6.0) in a microwave oven. No special pretreatment was performed for anti-SMA and anti-desmin. The procedure was followed by immunoperoxidase staining using a commercially available kit (Lab Vision, TP-125-HL, ready-to-use), which contained a protein blocking solution (Ultra UV Block), a link antibody (biotinylated goat anti-polyvalent) and enzyme-labelled streptavidin (streptavidin peroxidase). Development of reaction product was carried out with peroxidase-compatible chromogen, 3-amino-9-ethylcarbazole (AEC Substrate system, Lab Vision). PBS was used as the rinsing solution within each step during the staining procedure. Then the slides were counterstained with Mayer's haematoxylin and mounted with Aqueous-Mount (Scytec).
Histologically, it was a spindle-cell sarcoma consisting of interlacing bundles of elongated neoplastic smooth muscle cells and variably dense collagenous matrix separated by individual cells or streams. The neoplastic cells had cigar-shaped central nuclei and strongly eosinophilic abundant cytoplasm (Fig 3). The same areas displayed moderate cellular pleomorphism, including anisocytosis, anisokaryosis, multinucleation, hyperchromasia and bizarre mitoses. The mitotic index ranged from seven to 12 mitoses per 10 high-power fields (HPF). In addition, neoplastic tissue contained areas of focal necrosis and haemorrhage. Histological criteria of the tumour supported the diagnosis of leiomyosarcoma.

Vulvar mass; cat. General view of leiomyosarcoma. Rounded to spindle cells, exhibiting marked cellularity and nuclear pleomorphism. Haematoxylin and eosin. Bar=100 μm.
Immunohistochemically, the spindle-shaped neoplastic cells were strongly positive for SMA and vimentin. No immunoreactivity was demonstrable for desmin despite the positive immunoreactivity detected in the tunica media of blood vessels, which served as internal positive controls for desmin antibody. The vascular elements of the internal control tissue also exhibited strong SMA and vimentin immunoreactivity (Fig 4). This was consistent with a case of primary leiomyosarcoma.
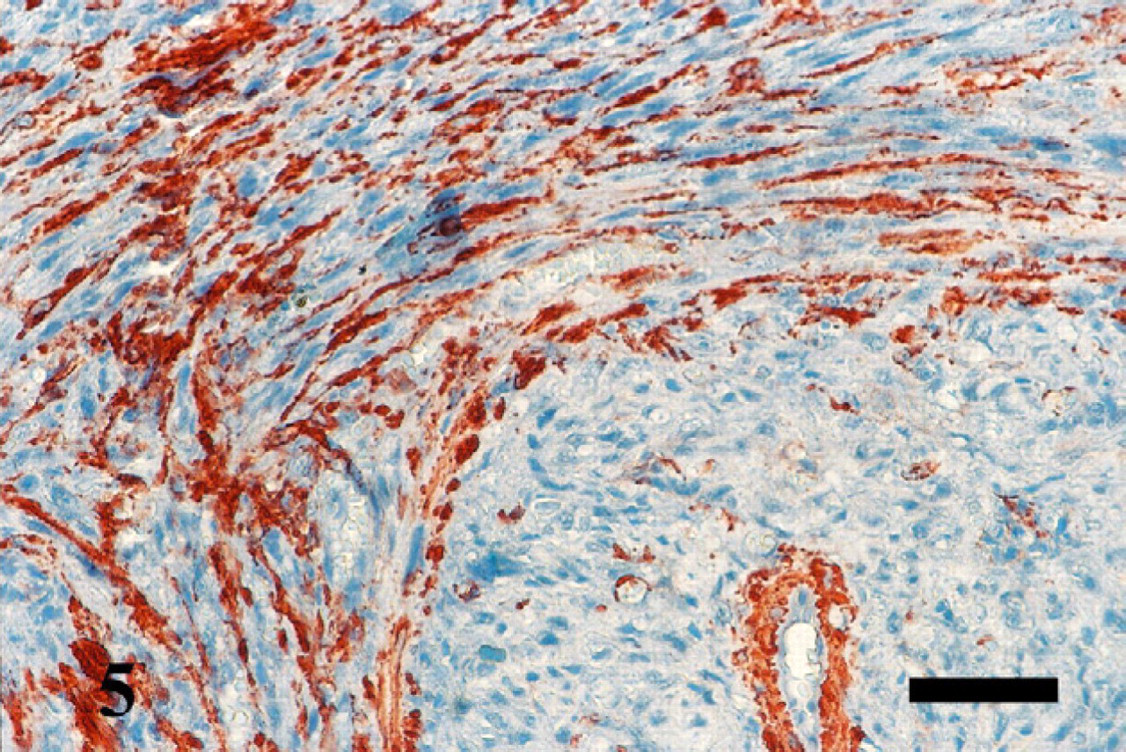
Vulvar mass; cat. Neoplastic cells, demonstrating dense intracytoplasmic immunoreactivity for smooth muscle actin, located up left side of the figure. Streptavidin-biotin method, Mayer's haematoxylin counterstain. Bar=50 μm.
The gross examination of the ovaries and the uterus revealed polycystic changes and hydrometra, respectively. Additionally, histological changes included glandular dilation and endometrial atrophy with haemorrhagic and cystic changes in the ovaries.
Almost all cases of LMS of the genital system have occurred in female animals and are most common in intact females, and in dogs (Brodey and Roszel 1967, Thacher and Bradley 1983, Cooper and Valentine 2002). One case of feline genital LMS was reported in the uterus. It was discovered during an elective ovariohysterectomy (Miller et al 2003). Clinically, these lesions can easily be mistaken for a benign process such as an infectious granuloma, dermoid or Bartholin cyst, fibroma or lipoma (Tjalma and Colpaert 2005). This was in agreement with the present case where the preliminary clinical diagnosis was an abscess.
Histological features were similar to those of leiomyosarcomas at other sites. But the initial diagnosis of soft tissue sarcomas reflected the fact that it could be difficult to distinguish fibrosarcoma, rhabdomyosarcoma and leiomyosarcoma (Cooper and Valentine 2002). In the current case, it was a spindle-cell sarcoma, consisting of interlacing bundles of elongated neoplastic smooth muscle cells and variably dense collagenous matrix separated by individual cells or streams. Moderate cellular pleomorphism, including anisocytosis, anisokaryosis, multinucleation, and hyperchromasia were observed in the neoplastic cells with cigar-shaped central nuclei and strongly eosinophilic abundant cytoplasm, typical of LMS. Mitoses are generally present and may be numerous in LMS in animals. According to the literature, an average mitotic index of moderate malignancy was greater than 10 mitoses per 10 HPF in vaginal smooth muscle tumours in women (Tavasoli and Norris 1979, Behzatoğlu et al 2003). On the other hand, the mitotic rate was reported to be 34 mitoses per 10 HPF in a case of human vulvar myxoid LMS (Tjalma and Colpaert 2005) which was apparently higher than the former. In the current case, the mitotic index ranged from seven to 12 mitoses per 10 HPF. In addition, the neoplastic tissue contained areas of focal necrosis and haemorrhage. These findings supported the diagnosis of a leiomyosarcoma.
The histogenesis of the tumour is not totally understood. It has been suggested that the tumour arises from either leiomyomas or de novo from transformed myometrial cells. Immunohistochemical demonstration of small and intermediate filaments has proved increasingly useful in the diagnosis of soft tissue tumours in veterinary pathology. In the current case, the tumour was strongly positive for vimentin, confirming its mesenchymal origin, and positive for SMA, supporting the diagnosis of leiomyosarcoma (Cooper and Valentine 2002). No immunoreactivity was detected for desmin despite the positive immunoreactivity detected in the tunica media of blood vessels, which served as internal positive controls for desmin antibody. The lack of desmin immunoreactivity was considered to be consistent with the literature suggesting that desmin might be a less reliable antibody than muscle specific actin in the diagnosis of tumours of smooth muscle origin (Anderson and Mahaffey 1987). The vascular elements of the internal control tissue also exhibited strong SMA and vimentin immunoreactivity.
Age and breed predisposition is unclear due to the very limited number of reported cases of feline genital LMS (Cooper and Valentine 2002). In the current case, the patient was an 8-year-old, mixed-breed cat. The role of the gonadal steroids in the development of the tumour is unknown, but it is believed that the gonadal hormones promote the growth of leiomyomas. Previous reports of a history of polycystic ovaries and cystic endometrial hyperplasia (Cooper and Valentine 2002) concurred with our findings in the ovaries and the uterus in this case. Other reasons to believe the hormonal basis of these tumours are as follows: ovariectomy causes the regression of these tumours, leiomyomas only rarely occur in ovariectomised dogs and occurrence of the leiomyoma increases with pregnancy but decreases after menopause in women (Chassy et al 2002).
In animals, the tumour may cause reproductive failure or urinary or colonic obstruction. Moreover, it has been indicated that these tumours might show marked infiltrative growth within the vaginal or uterine wall and could occlude the pelvic canal. These were observed in our case. No metastases were reported in any canine or feline genital leiomyosarcomas which was in accordance with our thoracic and abdominal radiographic findings (Cooper and Valentine 2002). To our knowledge this is the first case of vulvar leiomyosarcoma occurring in a domestic cat, which was evaluated both histopathologically and immunohistochemically.
