Abstract
Using paediatric devices to collect venous blood from a cephalic vein in cats offers numerous practical advantages over traditional jugular venepuncture and vacuum closed systems: minimal restraint is required; there is minimal risk of serious injury to the cat; the discomfort associated with venepuncture is reduced by the use of small diameter (25 gauge) needles; very small volumes (200 μl) of blood are extracted; and the risk of vein collapse or haematoma is low. The aim of this study was to compare the haematological and plasma chemistry results obtained from six healthy cats using the two sampling techniques. Five plasma biochemical analytes were measured and a complete haematological examination was performed on each specimen. No clinically relevant difference between the two blood sampling techniques was observed for any variable, indicating that paediatric devices provide a useful alternative to vacuum tubes for venous blood collection in the cat.
Blood sampling can be difficult in cats because of their natural reluctance to be restrained. The current reference technique involves direct sampling from the jugular vein using a vacuum system (Else and Kelly 1998). However, this technique is often uncomfortable and stressful for the animal and could potentially cause serious injury to the patient due to the close proximity of major vessels, trachea and nerves (Godfrey 1997). The vacuum effect commonly increases blood extravasation as the needle is removed, risking haematoma formation, especially when blood is repeatedly sampled from the same vein. The minimum blood volume required using traditional sampling methods is 2–3 ml, causing blood loss which may be of clinical relevance in cats when repeated clinical pathological examinations are required over a limited period of time (Henry et al 2002).
For all the above-mentioned reasons, alternatives to vacuum systems are frequently used by practitioners. Blood may also be sampled from the jugular vein with syringe and needle to limit an excessive vacuum and to better control the volume collected. Nevertheless, as for any blood sampling technique, the risk of vein collapse and haematoma persists and the blood may coagulate if mixing with the anticoagulant is delayed. Cephalic or saphenous venepuncture with syringe and needle seems to be even better tolerated by cats, but in this species the peripheral veins are especially small and may, sometimes, prevent collection of the minimum blood volume required for routine haematology and plasma biochemistry. Also, as these veins often collapse when negative pressure is applied, a needle diameter of no less than 22 gauge is usually recommended, especially when haematological analyses are to be performed (Norsworthy 1993).
The aim of this study was to assess a new technique for sampling blood from the cephalic vein, based on a capillary system (Microvette 200 μl Sarsdedt, Nümbrecht, Germany) which can be used by veterinary practitioners in field conditions and overcomes many previously encountered problems with feline venepuncture. We compared haematological and biochemical results, practicability and stress evaluation for the capillary technique with a vacuum system (Venoject 3 ml Terumo Europe Laboratory System, Leuven, Belgium) technique.
Materials and methods
Six client-owned, clinically healthy, domestic shorthair cats (three females and three males, all neutered) aged between 9 months and 10 years and weighing 3–7 kg were used.
Venepuncture was attempted in each cat four times on the same day. In the morning, the cats were first sampled from a jugular vein by the vacuum system (V), then 5 min later from a cephalic vein by the capillary system (M). In the afternoon the cats were sampled in the same order, first from a cephalic vein by M and 5 min later from a jugular vein by V.
All samples collected were assayed within 30 min. Two tubes (one ethylene diamine tetra acetic acid (EDTA) tube for haematological analyses and one lithium heparin tube for plasma biochemical analyses) were taken at each sampling time with each sampling device (M and V).
For the V technique, samples were taken from the jugular vein with a 22 gauge needle and 3 ml vacuum tubes (Lithium heparinate and EDTA Venoject 3 ml Terumo Europe Laboratory System, Leuven, Belgium).
For the M technique, blood was sampled from the cephalic vein with a 25 gauge needle and 200 μl capillary tubes (Lithium heparinate and EDTA Microvette 200 μl, Sarstedt, Nümbrecht, Germany). The needle was inserted in the cephalic vein and when a drop of blood appeared through the hub of the needle, the free end of the capillary tube was positioned and maintained in contact with the blood drop until the tube was totally filled (Fig 1). The duration of each procedure (time A from the start of restraint to completion of the sample procedure and time B to fill the two tubes once the needle was inserted in the vein) was measured. The compliance of the cats with each procedure was evaluated by an observer and stress reactions were assessed using a scoring system established by the investigators (Table 1). Heart rates were also measured before and after each procedure.

Procedure for blood sampling with capillary tubes. (A) Position and handling of the cat for blood sampling. (B) A 25 G needle has been inserted in the cephalic vein and a drop of blood appears through the hub. (C) The capillary tube is filled.
Criteria and scoring scale used to assess the stress induced in the cat by the blood sampling procedure

Biochemical assays were performed using a commercially available analyser (Vitros 250 chemistry system, Ortho-Clinical Diagnostics, Raritan, NJ). For plasma analyses, heparinised tubes were centrifuged (10 min, 3000 g) and five parameters (glucose, creatinine, alanine aminotransferase, alkaline phosphatase and potassium) were measured. The haematological analyses (blood cell count, haemoglobin concentration, haematocrit) were performed using a commercially available analyser (Scil Vet abc analyzer, Grayslake, IL). In addition, a microhaematocrit was performed for manual packed cell volume measurement and a blood smear was made for manual determination of cell counts.
Statistical analyses were performed using a commercially available software package (Systat version 8.0, SPSS Inc, Chicago, IL). Results are expressed as mean±SD. Haematological values, biochemical values, duration of procedures (times A and B), stress scores and heart rates obtained with the M technique were compared to those obtained with the V technique using a general linear model (Dobson 2001). A P-value <0.05 was considered significant.
The difference plot method as described by Bland and Altman was used to illustrate the difference in biochemical and haematological results between the two techniques: the X-axis shows the mean of the results of the two methods ([M+V]/2), whereas the Y-axis represents the absolute difference between the two methods ([M−V]) (Bland and Altman 1986).
Results
All samples were taken as initially planned except that one cat refused V once, and another cat refused M on one occasion and V on another. Thus, 42 tubes (10 EDTA and 10 heparinised vacuum tubes; 11 EDTA and 11 heparinised capillary tubes) were collected instead of the expected 48.
There were no statistically significant differences between the two techniques for plasma creatinine, glucose, alanine aminotransferase and potassium. Alkaline phosphatase was the only biochemical parameter to differ according to the sampling technique. The values for V and M were 52±19.5 U/l and 61±23.9 U/l, respectively (P=0.021) and the largest difference between the two techniques observed for any single cat was 26 U/l. Plasma alkaline phosphatase values for the two techniques were compared using the Bland–Altman method (Fig 2).
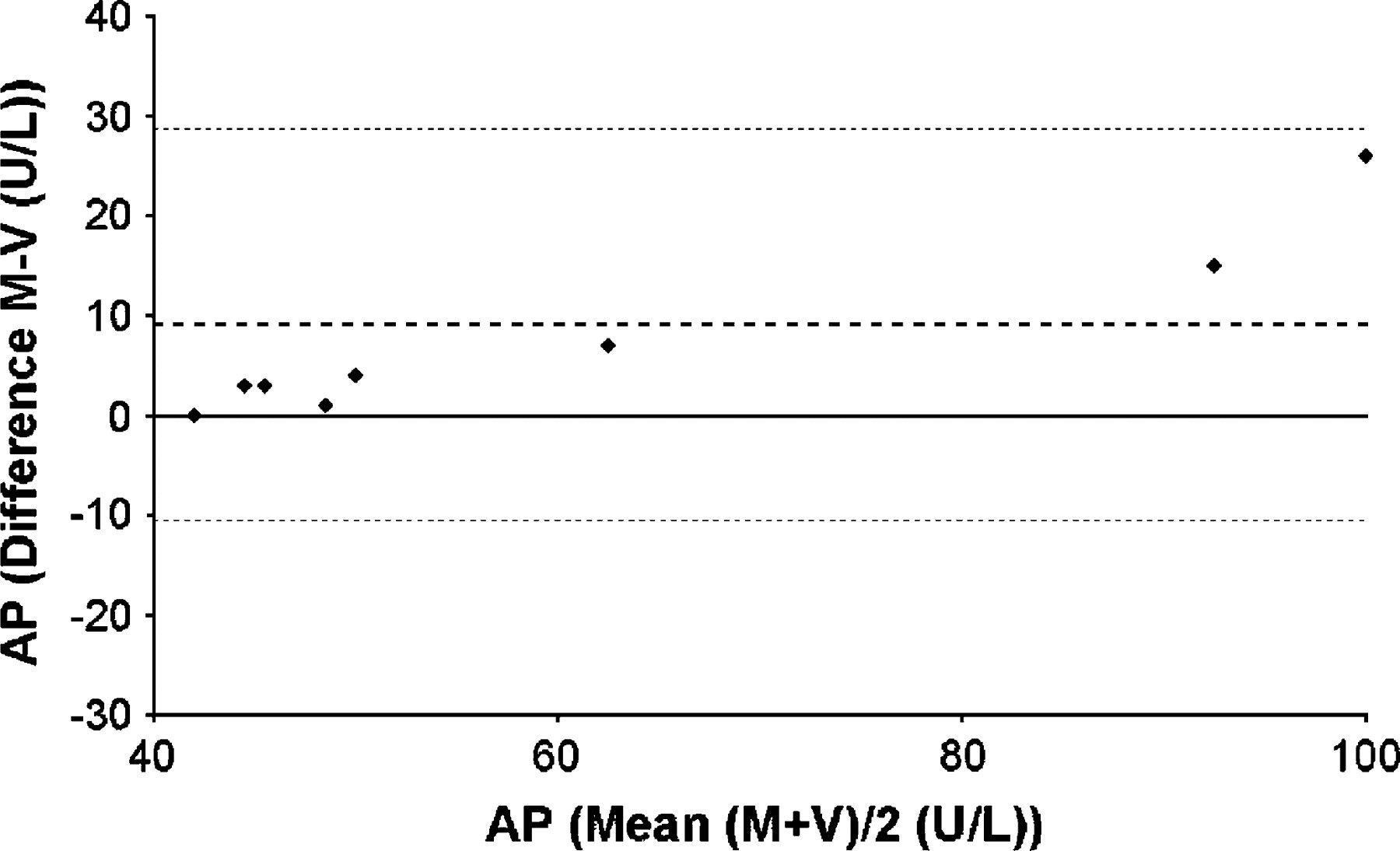
Bland–Altman plot – plasma alkaline phosphatase. This plots the difference between plasma alkaline phosphatase concentration obtained by capillary (M) and vacuum (V) techniques on the Y-axis and the mean value for plasma alkaline phosphatase using both techniques on the X-axis. Thick dotted line represents the mean of the differences between M and V. Thin dotted lines represent mean+2SD and mean−2SD of the differences between M and V.
Only three of the haematological parameters assessed showed statistically significant differences according to the blood sampling technique. The values for V and M were 9.3±0.99 and 10.1±1.29×1012/l for red blood cells (P=0.002), 12.3±1.19 and 13.4±1.45 g/dl for haemoglobin (P=0.001), and 42±4.7 and 46±5.5% for haematocrit (P=0.004), respectively. The two techniques were compared for these three parameters using the Bland–Altman method (Fig 3).

Bland–Altman plot – (A) red blood cell count; (B) haemoglobin; (C) haematocrit. This plots the difference between a haematological analyte results assessed by the analyser obtained by capillary (M) and vacuum (V) techniques on the Y-axis and the mean value for each haematological analyte using both techniques on the X-axis. Thick dotted line represents the mean of the differences between M and V. Thin dotted lines represent mean+2SD and mean−2SD of the differences between M and V.
A statistical comparison of the duration of each procedure did not show any significant difference, for either time A (3.3±1.9 and 2.2±1.3 min for V and M, respectively) or time B (2.0±1.4 and 1.4±1.2 min for V and M, respectively). The stress scores (7.9±4.0 and 7.7±4.1 for V and M, respectively) and the heart rates (173±25 and 164±22 beats/min for V and M, respectively) for the two blood sampling techniques were not statistically different.
Discussion
This study examined the effect of sampling technique on feline plasma biochemical and haematological parameters, using a new, minimally invasive technique, compared with a more commonly used, traditional method. Previous similar studies examining the effect of collection site showed that anatomical site made no practical difference to the values of a variety of these parameters in cats (Coates et al 1992, Jacobsen and Jensen 1998, Thomson et al 2002). Our results reflect those of previous work, in that while some statistically significant differences were observed when sampling techniques were compared, their absolute differences were small enough to make their clinical relevance negligible.
In a previous study, alkaline phosphatase concentration was found to be significantly higher in jugular than in cephalic blood (O'Brien et al 1998). The opposite was observed here as well as smaller amplitude in the differences. No explanation could be found for this discrepancy.
The placement of indwelling jugular catheters or vascular access ports is a valuable alternative to repeated venepunctures when serial blood sampling is required, as in dynamic functional testing, or in some clinical situations when extended follow-up is mandatory, and it provides reliable laboratory data (Henry et al 2002). However, these techniques are invasive; they require sedation or anaesthesia and strict asepsia during placement; and a blood volume equal to the dead space of the device needs to be discarded at each sampling time (Martin and Rand 1999, Henry et al 2002, Albarellos et al 2003). These factors could lead to infectious complications or anaemia (Blaiset et al 1995). Capillary blood can be used with specific techniques for serial blood glucose concentration measurements (Wess and Reusch 2000), but is not appropriate for routine haematology and biochemistry, because it is not easily handled, labelled and stored for delayed analyses. Our study is the first one to propose a minimally invasive alternative blood sampling method for routine plasma and haematological examinations in the feline species. When haematological examination is not required, a second 200 μl lithium heparin capillary tube can be filled thus allowing a more complete plasma panel of up to 10 different variables depending on the analyser.
Jugular venepuncture induces a stress response in cats which can have noxious consequences for the patient and/or modify drug pharmacokinetics (Albarellos et al 2003). Although no statistically significant difference in stress scores between the two blood sampling techniques was observed in the particular conditions of this study, the M technique seemed easier to perform for the operators and more comfortable for the cats. One of the limits is that the scoring scale used to assess the stress induced by the blood sampling procedure has not been validated. However, the assessment of stress levels in felines is a difficult issue as stressed cats can react in different ways depending notably on the stressful stimulus, and no validated scale for blood venepuncture in the cat has been published to our knowledge. For example, stress hyperglycaemia can be suspected in cats that are overtly aggressive and struggle during restraint and venepuncture, but may also occur in cats that are easily removed from the cage and do not resist the sampling process (Nelson 2002). The heart rate was used as a secondary criterion, but this indicator was shown not to be sensitive enough in some previous studies assessing surgery-induced stress and pain (Smith et al 1999, Glerum et al 2001). Therefore, the lack of effect of the sampling procedure on heart rate does not mean that the methods were similarly tolerated. To complete their observation, the authors used the M technique to obtain samples for yearly check-ups from 148 client-owned cats, all animals having been sampled within the same day by the same investigator. Only nine cats could not be sampled using this new technique (data not shown).
In conclusion, the Microvette® capillary system offers a rapid, minimally invasive and safe alternative to previously used vacuum collection systems for routine haematology and plasma biochemistry in the cat especially when serial sampling is needed.
Footnotes
Acknowledgments
This study was in part supported by Sarstedt, Nümbrecht, Germany. The authors wish to thank JC Meauxsoone for photographs.
