Abstract
The objectives of this study were to prospectively identify and characterize weight loss and changes in body condition in feline cancer patients and to investigate the prognostic significance of these findings. Fifty-seven cats with neoplasia were evaluated. Body condition was assessed with a nine-point scoring system (BCS) and multiple sites were assessed for muscle and fat mass using four-point scoring systems. Feline cancer patients had a mean BCS of 4.4±2.1 kg (1=cachectic, 5=optimal, 9=obese). Fat mass was reduced in both sites assessed in 60% of the patients. Muscle mass was reduced at all three sites assessed in 91% of the patients. Feline cancer patients having a BCS <5 had a median survival time (MST) of 3.3 months compared to that of 16.7 months for cats with a BCS of ≥5 (P=0.008).
It is well recognized that weight loss is a common finding in human cancer patients and one which has been shown to have associations with clinical outcome (Tisdale 1997). There has been speculation that this is also the case with companion animal cancer patients (Ogilvie 1993). Weight loss in association with cancer can occur for a number of reasons. A cancer patient may experience effects attributable to the location or degree of infiltration of a tumor or may be anorexic as a consequence of the side-effects of therapy. However, the weight loss seen in many human cancer patients does not appear to be attributable to decreased food intake alone. Patients with neoplasia can experience loss of both lean and adipose tissue rather than principally adipose tissue as would occur with simple starvation (Moley et al 1987). Furthermore, often the degree of the weight loss does not correspond to the amount of food consumed, and again, unlike simple starvation, this weight loss cannot be reversed by simply increasing caloric intake (Costa et al 1980, Oveson et al 1992). This phenomenon has been called cancer cachexia. There is some debate as to the definition of the term cachexia in medical circles, however, cancer cachexia in human patients has generally been accepted to refer to an involuntary loss of both lean body mass and adipose tissue.
This paraneoplastic syndrome of cancer cachexia is hypothesized to result from metabolic alterations that exist as a consequence of the underlying tumor. Investigations in both dogs and humans with cancer have revealed derangements in carbohydrate, lipid, and protein metabolism that may contribute to the weight loss that develops in these patients (Shapot and Blinov 1974, Nixon et al 1980, Nolop et al 1987, Shaw and Wolfe 1987, Vail et al 1990, Tayek 1992, McMillan et al 1994, Ogilvie et al 1994, Vail et al 1994, Ogilvie et al 1997, Dworzak et al 1998). There is also evidence that cytokines, including TNFα, IL-1, and IL-6, could play a role in these metabolic alterations (Gelin et al 1991, Moldawer and Copeland 1997).
Cancer cachexia has been implicated as a negative prognostic factor for survival, surgical risk, response to chemotherapy, and tolerance of treatment in human cancer medicine (Daly et al 1979, DeWys et al 1980, McCaw 1989). While weight loss or poor body condition may be taken into consideration when staging companion animals with neoplastic disease, the impact of either of these conditions on prognosis has not yet been reported. Investigations have found that anywhere from 31% to 87% of human cancer patients develop cachexia (DeWys 1982, Moldawer and Copeland 1997). While there has been one study looking at body condition and weight loss in canine cancer patients (Michel et al 2004), there have been no similar investigations in feline cancer patients published to date.
The aims of this study were (1) to evaluate body condition and investigate the occurrence of weight loss in cats presenting to the Oncology Service at the Matthew J. Ryan Veterinary Hospital of the University of Pennsylvania (MJR-VHUP) and (2) to determine the prognostic significance of weight loss and body condition in feline cancer patients.
Materials and methods
Selection criteria
Between September 1999 and January 2001, all cats seen by the MJR-VHUP Oncology Service were evaluated sequentially. Information collected for all cats included signalment, date of diagnosis, weight at diagnosis, weight at time of body condition scoring (BCS), body condition scores, remission status, tumor type and stage.
BCS was done using a nine-point system developed by Laflamme (1997) and two scoring systems developed by the authors: a four-point fat score and a four-point muscle mass score. The BCS system ranks cats numerically from 1 to 9 where 1 represents an extreme underweight condition and 9 an extreme overweight condition. In addition, as this body scoring system does not specifically address muscle wasting in cats with a BCS >2, body fat and muscle mass were assessed separately using a scoring scale of 0–3 (Table 1) that was adapted from a previously published investigation of body condition of canine cancer patients (Michel et al 2004). Fat mass was assessed at two sites: over the ribs and the abdominal fat pad area. Muscle mass was assessed at three sites: the temporal muscles, the scapulae, and the hind limbs. All scoring was performed by one investigator (JLB).
Fat and muscle mass scoring systems

Cats recruited in the study had a confirmed diagnosis of a neoplasm at the time of scoring. Some of these cats had already started treatment for their cancer. Patients were followed until death or last known follow-up to investigate the impact of weight loss and body condition on clinical outcome.
Statistical analysis
The paired t-test was used to evaluate change in body weight between diagnosis and treatment. Fisher's exact test was used to assess whether BCS groupings (score <5 and ≥5) were different by tumor type or prednisone use. The effect of prednisone on body weight and BCS was evaluated with the t-test. The Kaplan–Meier product limit method was used to estimate the portion of cats that were alive or had died in the two BCS groupings. Survival time was calculated from the time of diagnosis to the date of death or last follow-up. Animals that were still alive at last follow-up were considered censored at that time. Statistical differences in survival between these BCS groupings were assessed by the log rank test. To adjust for possible confounders such as age and tumor type, the proportional hazards model was used. Unless otherwise indicated, data are presented as means±standard deviation for continuous data and frequencies and percent for categorical data. All analyses were performed using SAS statistical software (Version 9.1, SAS Institute, Cary, NC). A P-value <0.05 was considered statistically significant.
Results
During the enrollment period, 57 cats with neoplasia were evaluated. The breed distribution of the study population included 49 domestic shorthairs, four domestic longhairs, and four other breeds including Himalayan (two), Persian, and Siamese. There were 28 (49%) castrated males, and 29 (51%) spayed females. The mean age was 11.1 years with a range of 1–19 years. The diagnoses of these patients were as follows: lymphoma (32), mammary gland adenocarcinoma (seven), squamous cell carcinoma (SCC) (seven), fibrosarcoma (six), adenocarcinoma (ceruminous gland) (two), and one each of mast cell tumor, nasal carcinoma, renal carcinoma, and spindle cell tumor (one case was diagnosed with both lymphoma and SCC). Fifty-three percent (17/32) of the cats with a diagnosis of lymphoma had involvement of the gastrointestinal tract.
Thirty-nine percent of the cats (22/57) were evaluated at the time that their cancer was diagnosed and before the commencement of any form of therapy. Of the 35 cats that begun therapy at the time of evaluation, 22 were receiving prednisone and five had undergone or were undergoing radiation therapy although only two of these had tumors involving the head and neck region. Even though body condition was evaluated in the majority of these cases 10 days or longer after the start of treatment, no difference was found when body weight at the time of diagnosis (4.4±1.3 kg) was compared to body weight at the time of body condition evaluation (4.2±1.3 kg; mean difference=0.2 kg, P=0.17). When cats that were not receiving prednisone at the time of body condition evaluation were compared to cats that were, no difference in mean body weight was found between these two groups (body weight: 4.3±1.3 vs 4.3±1.0, P=0.9).
Body condition scoring
The mean BCS of these cancer patients was 4.4±2.1 (Fig 1). Forty-four percent of the cats had a BCS of <5. The percentage of cats with a BCS <5 did not differ between those with a diagnosis of lymphoma and those with solid tumors (45% vs 42%, P=0.8). There was no difference in mean BCS detected between cats that were not receiving prednisone at the time of body condition evaluation and cats that were (4.0±2.2 vs 5.0±1.7, P=0.1). However, fewer cats who were receiving treatment with prednisone had a BCS <5 compared to those who were not, although the difference did not achieve statistical significance (27% vs 54%, P=0.06).

BCS of 57 cats with neoplasia. The interquartile range is represented by  , the median by ▪▪▪▪▪, and the mean by •.
, the median by ▪▪▪▪▪, and the mean by •.
The mean rib body fat score was 1.2±0.9, indicating that on average these patients had a minimal amount of fat over their ribs (Fig 2A). Sixty-eight percent of the cats had rib fat scores <2. The mean abdominal fat pad score for the cases was 1.1±0.9 (Fig 2B). Sixty-one percent had abdominal fat pad scores <2. Fat mass was reduced in both sites assessed in 60% of the cancer patients.
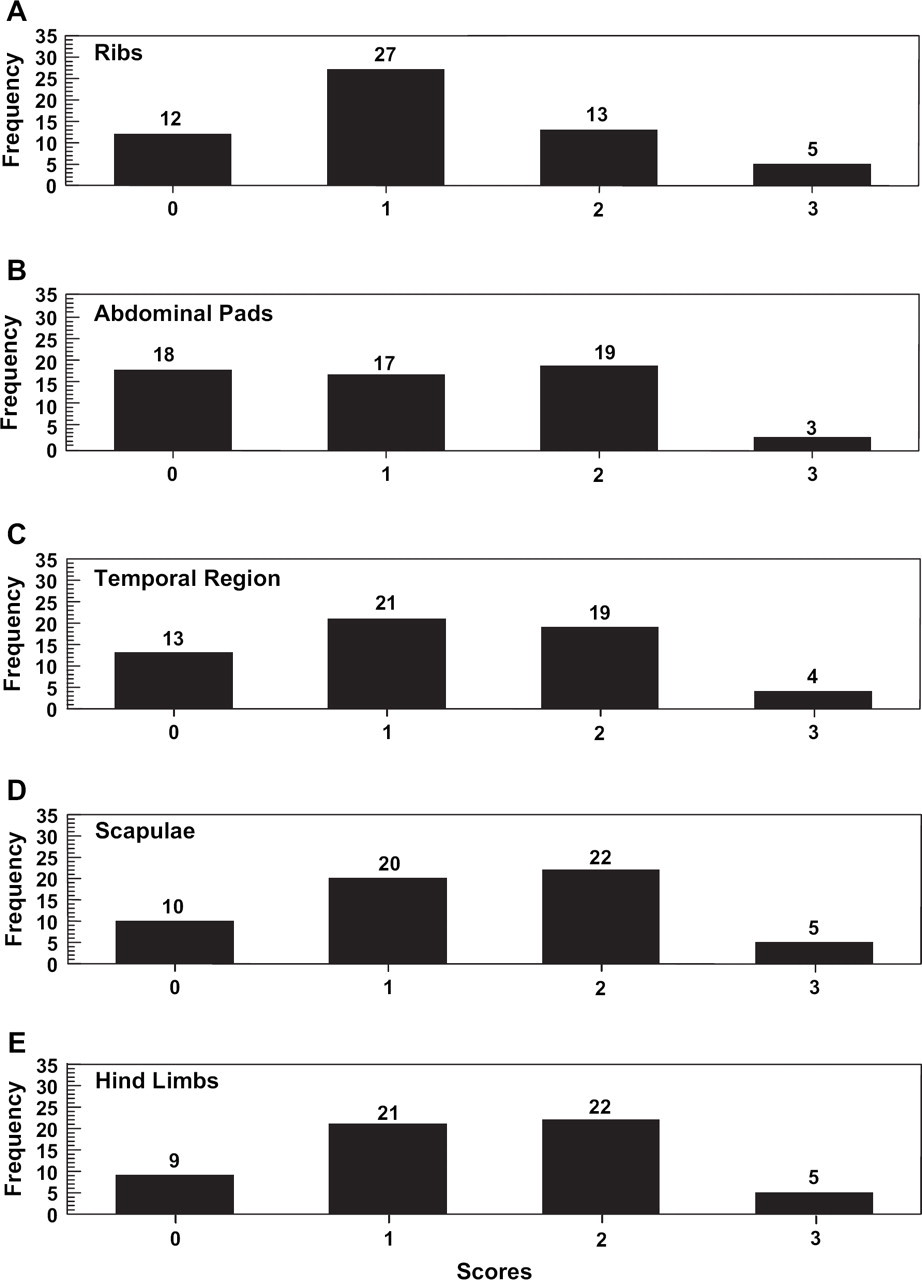
Fat mass and muscle mass scores of 57 cats with neoplasia. (A) Rib body fat score; (B) abdominal fat pad score; (C) temporal muscle score; (D) scapular muscle score; and (E) hind limb muscle score.
The patient population had a mean temporal muscle score of 1.2±0.9, indicative of significant muscle wasting (Fig 2C). Ninety-three percent had some evidence of muscle wasting in the temporal region (score <3). The mean scapular muscle score was 1.4±0.9 (Fig 2D). These were very similar to the hind limb muscle scores, 1.4±0.9 (Fig 2E). The percent of cases with scapular or hind limb scores of <3 was 91%. Muscle mass was reduced at all three sites assessed in 91% of the cancer patients.
When cats with BCS of ≥5 were evaluated with respect to muscle mass scores, 72% of cases (23/32) had evidence of moderate to severe muscle wasting at one or more anatomical location (score <2).
Body weight and prognosis
When patients were subgrouped according to body weight at the time of evaluation, median survival time (MST) for cats weighing <3.3 kg was 3.9 months, 6.5 months for cats weighing ≥3.3 kg and ≤5 kg and 19.9 months for cats weighing >5 kg (P=0.025). However, when only the patients with lymphoma were subgrouped according to body weight at the time of evaluation, the difference in MST was not significant (<3.3 kg: MST=3.9, 3.3 kg: MST=12 months, >5 kg: MST=16.9 months; P=0.3).
Body condition scores and prognosis
The BCS of the patients was evaluated with respect to MST. Cats with a BCS <5 had a MST of 3.3 months in contrast to 16.7 months for cats with a BCS of ≥5 (Fig 3; P=0.01). Similarly, when only those cats having a diagnosis of lymphoma were considered, the MST for cats with BCS <5 was 3.3 months vs 16.9 months for cats with BCS ≥5 (Fig 4; P=0.01).

Kaplan–Meier survival curves comparing all patients with BCS <5 to all patients with BCS ≥5.

Kaplan–Meier survival curves comparing lymphoma cases with BCS <5 to lymphoma cases with BCS ≥5.
Remission status was known for 56/57 of the patients. Fifty-nine percent (33/56) were in remission from their cancer at the time of evaluation. Remission status was closely associated with BCS with a mean BCS of cats that were in remission of 5.2±1.7 vs a mean BCS of 3.5±2.2 for cats that were not in remission (P=0.0014). When patients with lymphoma were considered, the mean BCS of those in remission was 5.2±1.7 vs 2.8±1.9 for those not in remission (P=0.0008) whereas for patients with solid tumors there was no significant difference in BCS between the two groups (in remission: 5.2±1.8 vs not in remission: 4.0±2.4; P=0.19). The percentage of cats with a BCS <5 did not differ between those with lymphoma (45%) and those with solid tumors (42%; P=0.79).
A proportional hazards model adjusting for age, treatment with prednisone, and diagnosis with SCC did not alter the effect of BCS on survival. In addition to BCS, only having the diagnosis of SCC was independently associated with survival (hazards ratio=0.8, P=0.004 and 6.7, P=0.001 for BCS and SCC, respectively).
Discussion
The majority of feline cancer patients evaluated in this investigation showed evidence of weight loss and/or muscle wasting. In addition, a significant association between MST and both body weight and BCS in feline cancer patients was found.
There is limited information in the veterinary literature regarding the syndrome of cancer cachexia in companion animals. A previous study evaluated body condition and weight loss in dogs with neoplastic disease (Michel et al 2004). One hundred dogs with various neoplasms were examined and assigned a BCS, muscle mass score, and fat mass score, using similar criteria as were used for cats in this study. In contrast to our findings, where 44% of the cases had a BCS <5, only 15% of dogs received scores <5. Likewise, only 35% of the dogs were found to have evidence of muscle wasting, as opposed to 93% of cases in this investigation. Based on these preliminary studies, it appears that both weight loss and loss of lean body mass, the generally accepted criteria for defining cachexia in humans, may be more prevalent in cats with cancer than in dogs with cancer.
Cancer cachexia has been implicated as a negative prognostic factor for survival, surgical risk, response to chemotherapy, and tolerance of treatment in human cancer medicine (Daly et al 1979, DeWys et al 1980, McCaw 1989). This study showed that poor body condition in cats with various neoplasms was a negative prognostic factor with regard to survival, in cases with BCS <5 having a significantly shorter MST compared to cases with a BCS of ≥5. Similar survival times were seen when the lymphoma cases were looked at as a subgroup and BCS remained independently associated with survival in a multivariate analysis that adjusted for age, tumor type, and treatment with prednisone. We also found that cats that were in remission had higher BCS than cats not in remission. This is interesting in light of the finding that humans with cancer cachexia have a decreased response to treatment, leading to fewer patients in a clinical remission of their cancer who are cachectic (DeWys 1985).
In humans with cancer cachexia, there is an equivalent loss of both fat and muscle mass, which is in contrast to humans with anorexia nervosa, who tend to lose more fat than muscle mass (Moley et al 1987). In this study, both fat and muscle were assessed in different body regions to account for differences in conformation among individuals. The majority of patients evaluated in this investigation (91%) had evidence of muscle wasting at all three sites assessed. Even the majority cats with a normal or overweight BCS (72%) had evidence of significant muscle wasting at one or more anatomical location. The degree of lean body mass and visceral protein depletion is associated with reduced survival in human cancer patients (Nixon et al 1980) with the major site of the protein loss being the skeletal musculature (McMillan et al 1994). This is consistent with our findings that MST was associated with a lower BCS in feline cancer patients. However, in humans, as is likely the case in cats, not all cancer patients experience cachexia, and the syndrome of cachexia itself tends to be more severe in patients with specific types of cancer, such as pancreatic and gastric cancers (Tisdale 1999). Further investigations into whether weight loss or alterations in body condition are more or less associated with particular neoplasms in cats are needed.
Lastly, the finding that 72% of the cases with BCS ≥5 had significant evidence of muscle wasting bears further investigation. This may be, in part, a consequence of the obligatory use of amino acids as an energy substrate by this species. Compared to the report on canine cancer patients (Michel et al 2004), a greater percentage of the patients this study had evidence of muscle wasting.
In human cancer patients, loss of both muscle and adipose tissue occurs, and the degree of the loss does not correspond to the amount of food consumed (Costa et al 1980, Moley et al 1987, Oveson et al 1992). Investigations in both dogs and humans have revealed that derangements in carbohydrate, lipid, and protein metabolism could contribute to the development of cachexia (Shapot and Blinov 1974, Nixon et al 1980, Nolop et al 1987, Shaw and Wolfe 1987, Vail et al 1990, Tayek 1992, McMillan et al 1994, Ogilvie et al 1994, Vail et al 1994, Ogilvie et al 1997, Dworzak et al 1998). Among the aberrations already noted in dogs are hyperlactatemia and hyperinsulinemia (Vail et al 1990, Ogilvie et al 1997). Identification of similar abnormalities in dogs and humans warrants further investigation of feline patients particularly given the differences in intermediary metabolism between cats and other species.
There are several limitations to this investigation. First were the subjective nature of the BCS and the evaluation of fat and muscle mass. Having one individual perform the assessment of body condition and fat and muscle mass in all patients was an advantage in that it should have resulted in more consistent scoring and avoided the possibility of inter-observer error. This was of particular importance as the fat and muscle mass scores, in contrast to the method of BCS used, have not been evaluated for reproducibility between observers. The scoring system developed for this investigation may have some clinical utility and should be investigated further both in terms of repeatability and whether it can be applied consistently by multiple observers.
Second, this investigation was not designed to evaluate whether or not cats were losing weight in the face of adequate nutritional intake. We were also not able to determine the exact cause of weight loss in these patients. Therefore, we cannot categorically state that the cats exhibiting signs of weight loss were experiencing the syndrome of cancer cachexia. However, all patients with a BCS <5 also exhibited some degree of muscle wasting, so at least one of the hallmarks of this syndrome was identified in these patients.
In conclusion, the majority of feline cancer patients evaluated in this investigation showed evidence of weight loss and muscle wasting and a significant association between MST and both body weight and BCS was found. Further investigation of weight loss and body condition in feline cancer patients is warranted as it could lead to nutritional or pharmacological interventions to their benefit.
