Abstract
A 10-year-old female neutered domestic shorthair (DSH) cat and a 6-year-old female neutered Siamese cat were presented following a peracute onset of decerebellate rigidity and a cerebellar vestibular syndrome, respectively. In both cats, physical examination and routine blood tests were unremarkable, as was routine analysis of cerebrospinal fluid obtained from the DSH cat. Based on the magnetic resonance imaging (MRI) features – focal wedge-shaped lesion in the cerebellum characterised by hyperintensity in T2-weighted, T2∗-gradient echo and fluid attenuated inversion recovery (FLAIR) images – a presumptive diagnosis of cerebellar infarct was made in both cases. In the DSH cat, the post-mortem examination confirmed the diagnosis of cerebellar infarct and additionally found acute renal infarcts and a pulmonary neoplasia. In the Siamese cat, ultrasonographic evaluation of the heart revealed a probable low-grade chronic valvular endocarditis which was thought to be a potential source of thromboembolism. This paper describes the first two cases – one confirmed and the other suspected – of cerebellar infarct in the cat. The in vivo potential diagnostic value of the MRI study is highlighted. Cerebellar infarcts should be included in the differential diagnosis of cat with a peracute onset of cerebellar signs regardless of the severity of neurological deficits.
An indoor only, 10-year-old neutered female domestic shorthair (DSH) cat developed overnight peracute non-ambulatory tetraparesis. The cat did not appear to be in pain and on physical examination was tachypnoeic and tachycardic, but otherwise alert and responsive. Routine haematology and serum biochemistry were unremarkable. No treatment was given and the cat was referred approximately 4 h after the event to The Queen Mother Hospital for Animals, The Royal Veterinary College. The neurological examination revealed normal mental status with decerebellate posture and non-ambulatory tetraparesis characterised by opisthotonus, with increased extensor tone of the thoracic limb and flexion of the pelvic limbs' joints with normal pelvic limb muscle tone. The postural reactions were reduced and the segmental spinal reflexes were increased in all four limbs. Bilateral mydriasis with reduced pupillary light responses was also noted and menace responses were absent bilaterally. The neuroanatomical localisation was consistent with a peracute ‘cerebello-medullary lesion’ with involvement of the cerebellar nuclei and brainstem (DeLahunta 1983a, b). Differential diagnoses were restricted to ischaemic or haemorrhagic vascular accident, trauma, tumour or infectious/inflammation disease of the central nervous system (CNS).
An MRI study, performed using a 1.5 T machine (Gyroscan NT, Philips Medical System), approximately 6 h after the onset of the clinical signs, showed a single, focal cerebellar lesion that was isointense on T1-weighted images, hyperintense on T2-weighted images, on T2-weighted fluid attenuated inversion recovery (FLAIR) images and on T2*-gradient-echo images, with no uptake of contrast agent. There was a sharp demarcation between the lesion and adjacent normal cerebellum and no apparent mass effect. On sagittal midline images, the lesion involved the rostral, approximately 60% of the cerebellum (Fig 1). On transverse images, the lesion was centred on the midline and had an upturned wedge-shaped appearance, radiating outwards dorsally and extending to the dorsal surface of the cerebellum (Fig 2). A hyperintense lesion was also detected in the brainstem and forebrain on T2-weighted and FLAIR images (Fig 3). Based on the MRI appearance (hyperintensity in T2-weighted, T2*-gradient-echo and FLAIR images) history and neurological examination, a presumptive diagnosis of rostral cerebellar and multiple lacunar brain infarction was made (Garosi et al 2005, 2006).

T2-weighted MRI on sagittal midline. The ischemic lesion involving approximately 60% of the rostral cerebellum (arrows) appears hyperintense.
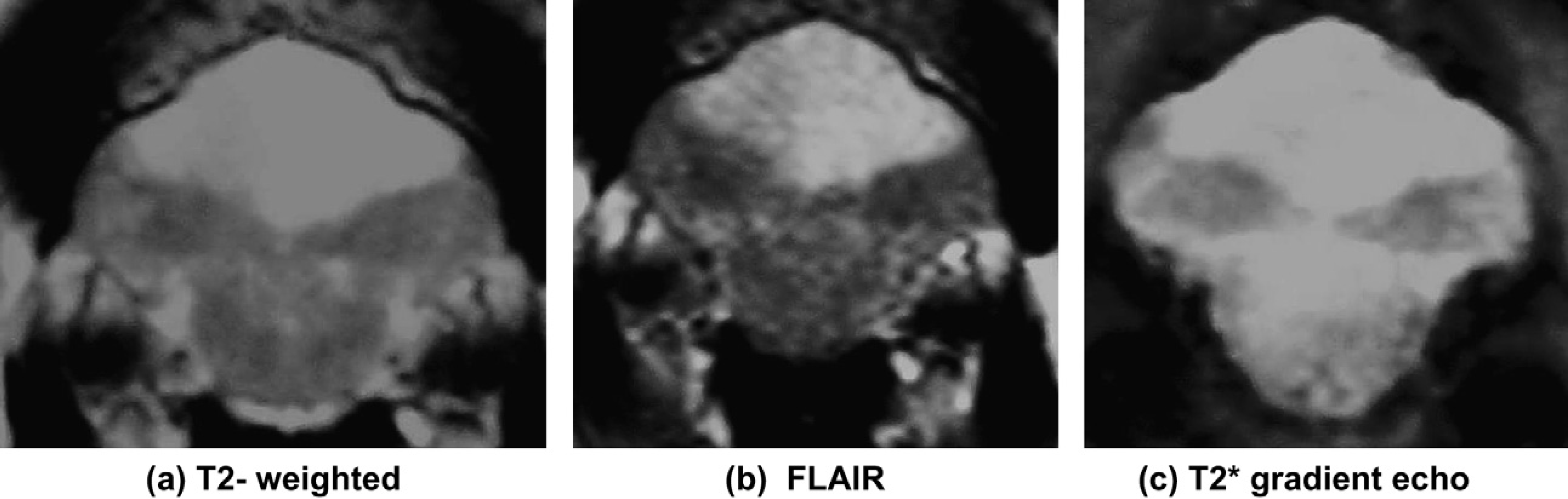
Transverse T2-weighted (a), FLAIR (b) and T2* gradient- echo (c) MRI scan. The lesion is centred on the midline and has a wedge-shaped appearance, radiating dorsally and extending to the dorsal surface of the cerebellum.
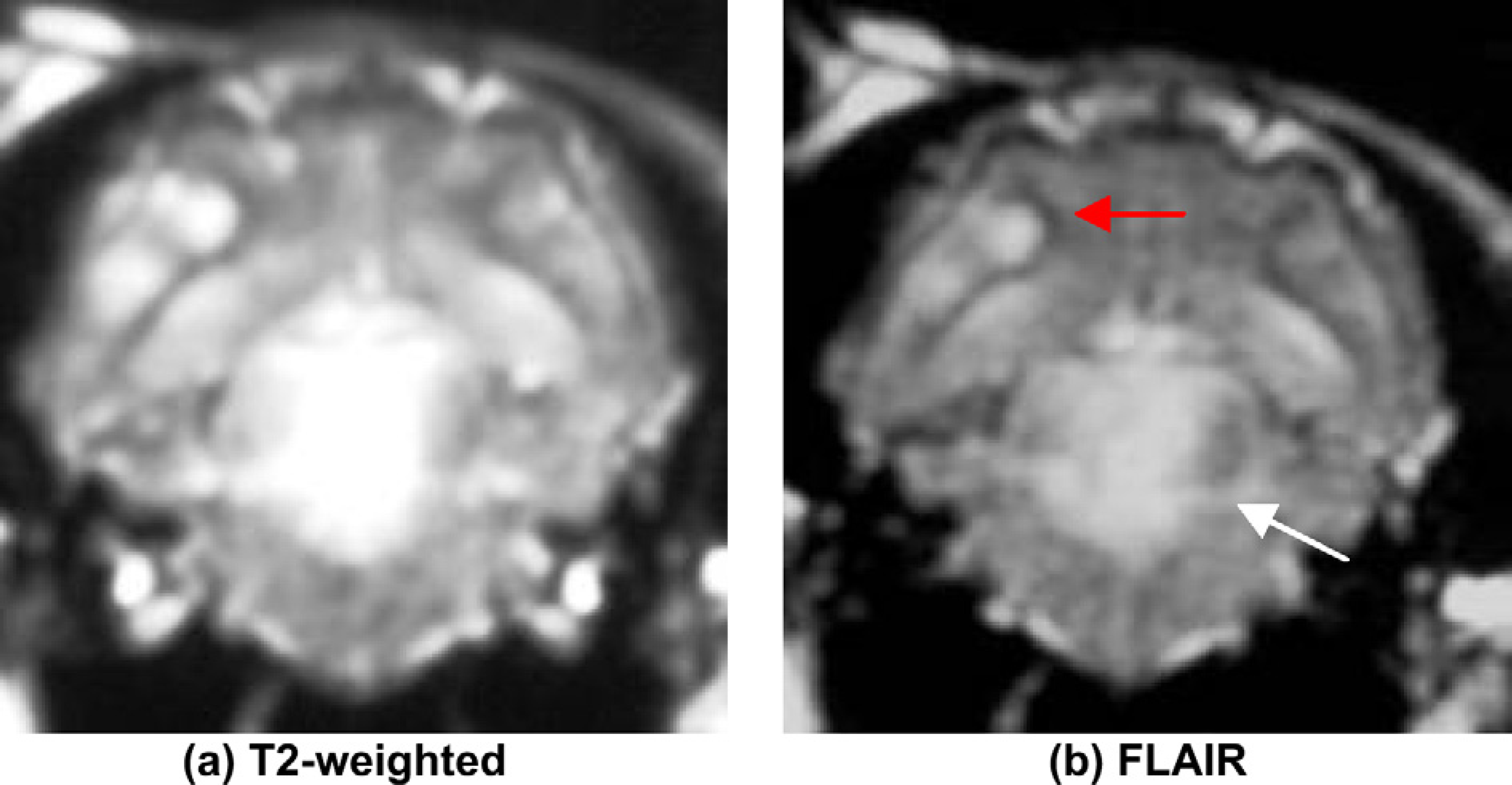
Transverse T2 (a) and FLAIR (b) weighted-MRI images of the DSH. Note the lesion on the brainstem (white arrow) and forebrain (red arrow).
As brain infarction has been reported to be a manifestation of some other underlying disease, a diagnostic work-up to rule out possible causative aetiological disorders was made (Thomas 1996, Chung and Caplan 1999, Garosi et al 2005, 2006, Garosi and McConnel 2005). Thus cerebrospinal fluid analysis, complete blood count (CBC), biochemistry, electrolytes, thyroxine (T4), basal cortisol, prothrombin time (PT), activate partial thromboplastin time (aPTT) electrocardiogram (ECG), non-invasive arterial blood pressure, thoracic radiographs and abdominal ultrasound analyses were all conducted soon after the MRI. All test results were unremarkable. The cat developed cardiorespiratory deterioration, probably secondary to an involvement of the respiratory brainstem centres, during the recovery period following the general anaesthesia. The owner elected for euthanasia.
The body was submitted for a complete post-mortem examination. Macroscopically, no lesions were visible in the brain and spinal cord but on histopathological examination, the rostral cerebellum contained a focal acute infarct (Fig 4) which corresponded in location to the affected area diagnosed by MRI. The area of infarction involved the grey and white matter of the cerebellar vermis and extended to the adjacent cerebellar hemispheres. The infarct was characterised by a focal, extensive, well-demarcated area of pale staining with disruption of the normal architecture of the parenchyma, fragmentation of neuropil, presence of multiple confluent clear spaces (oedema) within grey and white matter, degeneration and necrosis of Purkinje cells and swelling of astrocytes (Figs 5 and 6). The histopathological absence of macroscopic cerebellar lesions together with the observed microscopic findings were consistent with an acute cerebellar infarct of approximately 8 h duration (Storts and Montgomery 2001). In addition to this brain lesion, a poorly demarcated, white spherical mass was present within the left cranial lung lobe. Microscopically, this mass was consistent with a pulmonary adenocarcinoma; the tumour contained very rare small-sized blood vessels and lymphatics which were plugged by tumour cell emboli. Also, the cortices of both kidneys contained multiple, depressed, reddish areas, which measured up to 0.4×0.4 cm in diameter, extended wedge-shaped in the underlying parenchyma and were microscopically consistent with acute infarcts.

Cerebellum of case 1 (H&E staining). The cerebellar vermis shows a focal area of pale staining and disruption of the parenchyma (acute infarct, delineated by arrows). This is the location of the lesion identified in Fig 2.

Cerebellum of case 2 (H&E staining). Infarcted areas had pale staining, oedema and fragmentation of neuropil (arrows). Compare infarcted area with non-affected areas including granular cell layer (a) Purkinje cell layer (b) and molecular layer (c).

Comparison between non-infarcted (a, b) and infarcted area of the cerebellum (c, d). Grey matter with outer molecular layer (M), Purkinje cell layer (*) and inner granular cell layer (G), and white matter (•). a. Non-infarcted area of the cerebellar cortex (2×). b. Close-up of non-infarcted area of the cerebellar cortex (40×). Viable Purkinje cells have piriform perikarya, pale eosinophilic cytoplasm and vesicular round nuclei (arrowhead). c. Infracted area of the cerebellar cortex (2×) characterised by pale staining and disruption of the parenchyma. d. Close-up of infarcted area of the cerebellar cortex (40×). Necrotic Purkinje cells (arrowhead) are shrunken and have hypereosinophilic cytoplasm and pyknotic nuclei.
A 6-year-old female neutered Siamese cat was presented to Stone Lion Veterinary Centre 12 days after peracute onset neurological signs that included turning to the right and inability to stand. An intention tremor was also described. The cat was treated with corticosteroids and supportive care and made some improvement before referral. Five years prior to the neurological event, the cat had a road traffic accident suffering extensive injuries to both pelvic limbs, requiring amputation of the left pelvic limb and a hock arthrodesis and amputation of the two middle digits of the right pelvic limb. Recovery had been complicated by wound infection.
On referral, a neurological examination was performed. The cat was unable to walk, although this was in part caused by the pre-existing disability. When the cat made attempts to walk, it veered and had a head tilt to the right. There was a brief period of horizontal nystagmus after moving the head. Hopping and tactile placing was slower on the right but correction of a knuckled paw was normal. Cranial nerve function was normal aside for positional horizontal nystagmus. There was a mild intention tremor. When the cat was sedated (prior to anaesthesia for an MRI examination) a spontaneous horizontal nystagmus (fast phase to the right) and a left head tilt developed. The neurological examination suggested a right cerebellum-medullary anatomic diagnosis. Serum biochemistry and haematology were unremarkable. An MRI scan was performed using a 1.0 T machine (Siemens Impact Expert, 20 mT/m gradients) and revealed a hyperintense wedge-shaped area within the right rostral cerebellum on T2-weighted images (Fig 7). These changes were considered to be consistent with infarction in the territory of the right rostral cerebellar artery. As a cause of cerebellar infarction in man is embolism via the vertebrobasilar arteries; the cat was investigated for possible cardiac disease and other causes of thromboembolic disease (Chung and Caplan 1999, Kumral et al 2005). Auscultation was normal. Cardiac ultrasound revealed hyperechoic aortic valves consistent with a low-grade chronic valvular endocarditis. There was spectral evidence of aortic regurgitation and it was considered that this was a potential source of embolism and infarction. However, the possibility of a thrombus adjacent to the mitral attachment could not be excluded. There was also a significantly enlarged left atrium and poor compliance of the myocardium consistent with a subclinical, mild, restrictive-type cardiomyopathy. A coagulation panel was normal with a PT of 8.0 s (control 8–13), an activated partial thromboplastin time of 11.8 s (control 12–25) and a normal buccal mucosal bleeding time. Routine haematology and biochemistry were also normal. The cat was managed with general supportive care including physiotherapy and was prescribed acetyl salicylic acid at 18.75 mg twice weekly (Aspirin Angettes 75; Bristol-Myers Squibb, 75 mg). It was suspected that the endocarditis was inactive and related to the previous orthopaedic injury and infection, but as a precaution the cat was prescribed a 2-month course of cephalexin (Ceporex; Schering Plough) at 50 mg twice daily. In addition, the cat's diet was supplemented with l-carnitine (Conval S; Genetrix, 200 mg once daily) and vitamin E and omega 3 fatty acids (Viacutin; Boehringer Ingelheim, 0.6 ml twice daily). On re-examination 4 weeks after the original event, the cat was only able to walk if the hindquarters were supported, and had poor balance and a left sided head tilt. The owner persevered with supportive care and approximately 10 months after the neurological event the owner reported that the cat was able to walk without assistance. Twelve months following the neurological event the cat required dental work which was performed at the primary veterinary surgery. The cat recovered poorly from the anaesthetic and, unfortunately, died within a few hours of the procedure. The details of the anaesthesia and postoperative recovery were not available. A post-mortem examination was conducted by independent general practitioner, who felt that there were renal lesions suggestive of neoplasia.

Transverse T2-weighted of the Siamese cat. Note the sharp demarcation between the normal and abnormal (arrow) parenchyma.
‘Stroke’ is a non-scientific but commonly used term used to describe a clinical event characterised by a sudden onset of neurological deficits secondary to a cerebrovascular disease. It is defined as the clinical manifestation of a sudden onset of non-progressive, focal brain signs secondary to haemorrhage or ischaemic infarction due to a thrombus or thromboembolism (Garosi et al 2005). Stroke accounts for about 10% of all deaths in most industrialised countries and is a leading cause of adult disability in the USA (Chung and Caplan 1999). Approximately 80% of strokes are classified as ischaemic (Frankel and Chimowitz 1999). MRI findings of cerebellar infarcts have been reported in man where they represent approximately 3% of all brain infarcts (Amarenco et al 1993, Frankel and Chimowitz 1999). Cerebellar infarcts are uncommonly reported in small animals (Bagley et al 1988, Joseph et al 1988). However, the incidence of cerebellar infarction in dogs is increasingly being recognised, especially as MRI examination is becoming routine (Berg and Joseph 2003, Garosi et al 2005, 2006, McConnell et al 2005). Brain infarcts can be classified according to location of the brain involved; the type or the artery involved into: lacunar (superficial or deep perforating artery territory), territorial (main artery of the brain or one of their branches), watershed (boundary zone between two artery territories) or global; according to the pathogenesis as thrombotic or embolic; considering the presence (red infarct) or absence (pale infarct) of secondary haemorrhage within the infarcted tissue and finally idiopathic (Hommel and Besson 1998, Frankel and Chimowitz 1999, Panarello et al 2004, Garosi et al 2005). Possible predisposing factors in man and dogs include cardiac diseases, hypercoaguable states, vasculopathy, hypothyroidism, diabetes mellitus, hyperadrenocorticism, hyperlipaemia, hypertension, obesity and neoplasia (Joseph et al 1988, Thomas 1996, Benson and Sacco 2000, Garosi et al 2005, 2006). In each of the cats reported here, one of the known predisposing factors was present and, in case 1, may have been associated with infarction in more than one organ system. While neoplasia can predispose to tissue infarction either by vascular occlusion by tumour cell emboli or, rarely, by causing hypercoagulation state, neither was present in case 1 despite detailed examination of multiple sections of infarcted brain and renal parenchyma. This is not inconsistent with previous reports in the dog where, in about half of the cases of brain infarcts, no underlying cause could be detected (Garosi et al 2005). In case 2, the heart valve disease could be a possible source of emboli. However, pathological examination of the brain was not performed and thus the exact cause of the infarct remains uncertain.
The prevalence of cardioembolic strokes is reported to be high in man (Chung and Caplan 1999). Canine vascular encephalopathy associated with bacterial endocarditis have been reported and some breeds such as cavalier King Charles Spaniel appear to be predisposed to cerebellar infarction possibly due to a combination of valvular disease and platelet dysfunction (Cook et al 2005, Rusbridge 2005, Garosi et al 2006). No such reports exist for the cat. The location of the cerebellar infarcts in the two cats reported here is also of interest. Two arteries supply the cerebellum in cats: the rostral and the caudal cerebellar artery. The rostral cerebellar arteries supply the rostral cerebellum and the dorsolateral brainstem, while the caudal cerebellar arteries supply the caudal and ventral cerebellum and the lateral medulla (Jenkins 1978, King 1987). In case 1, the extension of the lesion involving grey and white matter, approximately 60% of the rostral cerebellar hemispheres and vermis, cerebellar nuclei and part of the brainstem could be accounted for by a territorial infarct of the rostral cerebellar arteries; in contract, in case 2, the lesion would appear to relate the right rostral cerebellar artery. This would be consistent with findings in man and in dogs where most cerebellar infarcts are reported to be territorial infarcts of the rostral cerebellar artery (Amarenco et al 1993, Hommel and Besson 1998, McConnell et al 2005). Cardiac embolism seems to be a common cause of bilateral infarcts in the territory of the superior cerebellar artery in man (Hyun-Ah et al 2006). However, the histopathological examination of the heart in cat 1 revealed just a minimal degeneration and fibrosis associated with myofibril hypertrophy.
The neurological localisation was consistent with the MRI findings in both cases. The bilateral mydriasis poorly responsive to the pupillary light reflex (PLR) present in case 1 can be secondary to a lesion involving the fastigial nucleus (contralateral pupil dilation) and the interposital nuclei (ipsilateral dilation) or can be secondary to a swelling compressing the brainstem and in turn causing the compression of parasympathetic branch of the cranial nerve III at the initial presentation and the cardiorespiratory deterioration few hours later (DeLahunta 1983a). In severe cerebellar diseases, the animal may be in lateral recumbency, unable to right itself, with the head and neck extended in opisthotonus. The rostral lobe of the cerebellum is inhibitory to the stretch reflex mechanism of antigravity muscles; the red nucleus and the rubrospinal tract in the brainstem are predominantly facilitatory to motoneurons of flexor muscles, so a lesion in both areas may result in opisthotonus with rigidly extended thoracic limbs and increased muscle tone (DeLahunta 1983a). As reported in man, the rostral cerebellar artery stroke can present with cerebellar plus brainstem involvement and can have a relatively benign prognosis (Amarenco et al 1993).
Usually, in man, a brain infarct becomes clearly evident as an area of hyperintensity in T2-weighted images by 6 h after infarction. In the acute phase, the earliest changes occur within the neuron within 20 min to 4–6 h, while the remainder of the nervous tissue is normal. At 4–6 h, ischaemic cytological changes become more obvious, with loss of subcellular integrity. Cytotoxic oedema may be detected as mild increased hyperintensity in T2-weighted images as parenchyma swelling as quickly as 2 h post infarct. Vasogenic oedema secondary to the blood barrier breakdown (BBB) is best visualised in T2-weighed images (Scuotto et al 1997). However, in case of brain ischaemia, it is now recognised that the phases of brain oedema may overlap and thus the extent of oedema fluid depends in large part to the severity and the time of duration of the ischaemic accident (Tuor et al 1998, Neumann-Haefelin et al 2000, Ayata and Ropper 2002). FLAIR sequences have showed in human studies to increase the sensitivity of T2-weighted images in detecting acute brain cortical infarct (Brant-Zawadzki et al 1996).
T2*-gradient-echo images have been proven to be the most accurate of all MRI sequences in detecting the presence of brain haemorrhage characterised by hypointensity regardless the times, the location and if arterial or venous blood; where the hyperintensity is a non-specific sign (Atlas and Thulborn 2002). Thus, in case 1 T2*-gradient-echo images allowed to rule out the presence of blood. In early post-infarct lesions, histopathological lesions consistent with both cytotoxic and vasogenic oedema, hyperintensity on T2-weighted, FLAIR- and T2*-gradient-echo images and no ‘fogging’ effect can be observed (Brant-Zawadzki 1988, Scuotto et al 1997, Ayata and Ropper 2002, Marks 2002). An early temporary increase in hyperintensity in T2-weighted images has been found to correlate with the severity and the time of the vascular occlusion in rat models and be associated with the degree of vasogenic oedema and mortality (Neumann-Haefelin et al 2000). Diffusion-weighted imaging (DWI) has a higher sensitivity and specificity for detecting areas of ischaemia. The affected areas become hyperintense within 1 h of the ischaemic episode, and this appearance may persist for up to 72 days. When comparing with an apparent diffusion co-efficient (ADC) map, it becomes possible to differentiate an acute infarct from an old infarct. ADC becomes reduced (dark) within minutes of an ischaemic episode, and this persists for 4 days. ADC values then gradually increase with the affected area becoming bright by the 10th day (Geijer et al 1999, Marks 2002). DWI and ADC mapping were not performed on these cases.
The cause of the hyperintense areas on T2-weighted and FLAIR images in brainstem and forebrain could not be revealed by pathological examination. Vasogenic oedema appears to be by far the most important contributor to ischaemic brain swelling and consequently neurological deterioration; it is well recognised as a major factor that contributes to early death in human patients with severe brain infarct (Berroushot et al 1998). In case 1, despite medical treatment, the cardiac and respiratory deterioration were considered to be secondary to an ongoing compression over the brainstem cardiorespiratory centres consequent to a mass effect due to the increasing of a poorly medical responsive vasogenic oedema visible on MRI (Fig 3). In case 2, the mild extension of the lesion visible on the MRI, involving only the cerebellum, was correlated with a good, however slowly, progressive outcome.
To the authors' knowledge, this report represents the first published cases of cerebellar infarction in the cat. It illustrates that MRI is an invaluable diagnostic technique in the diagnosis of this condition and suggests that cerebellar infarction should be considered in the differential diagnosis in cats with a peracute onset of cerebellar signs. As in dogs and man, there may be an association between cerebellar infarction in the cat and either a cardiovascular disorder or a neoplasia elsewhere in the body.
Footnotes
Acknowledgements
The authors thank Merixtell Peris for referring the DSH cat, and Rob Thomas for the cardiological expertise and support in the clinical and diagnostic management of the Siamese cat.
