Abstract
The quality and duration of anaesthesia, cardiorespiratory effects and recovery characteristics of a morphine, medetomidine, ketamine (MMK) drug combination were determined in cats. Six healthy, adult female cats were administered 0.2 mg/kg morphine sulphate, 60 μg/kg medetomidine hydrochloride, and 5 mg/kg ketamine hydrochloride intramuscularly. Atipamezole was administered intramuscularly at 120 min after MMK administration. Time to lateral recumbency, intubation, extubation and sternal recumbency were recorded. Cardiorespiratory variables and response to a noxious stimulus were recorded before and at 3 min and 10 min increments after drug administration until sternal recumbency. The time to lateral recumbency and intubation were 1.9±1.2 and 4.3±1.2 min, respectively. Body temperature and haemoglobin saturation with oxygen remained unchanged compared to baseline values throughout anaesthesia. Respiratory rate, tidal volume, minute volume, heart rate, and blood pressure were significantly decreased during anaesthesia compared to baseline values. One cat met criteria for hypotension (systolic blood pressure <90 mmHg). End tidal carbon dioxide increased during anaesthesia compared to baseline values. All but one cat remained non-responsive to noxious stimuli from 3 to 120 min. Time to extubation and sternal recumbency following atipamezole were 2.9±1.1 and 4.7±1.0 min, respectively. MMK drug combination produced excellent short-term anaesthesia and analgesia with minimal cardiopulmonary depression. Anaesthesia lasted for at least 120 min in all but one cat and was effectively reversed by atipamezole.
Injectable or inhalant anaesthetics are used to produce short-term anaesthesia in cats. Inhalant anaesthesia provides control of anaesthetic depth at relatively low cost but requires special equipment and adjunct medications. Adjunctive medications provide added sedation and analgesia thereby decreasing inhalant anaesthetic requirements and potentially improving cardiorespiratory status. Injectable anaesthetic drugs or drug combinations are relatively easy to administer, require minimal resources (ie, syringe, needles) and are capable of producing surgical anaesthesia, however, once administered the drug effect cannot be controlled and may require respiratory support.
Cats are a sympathetically active, fight or flight response species. Their small stature and agility may make them more difficult to restrain than other species and poses a threat to handlers (Karas 1999). Many cats respond adversely to physical restraint, hence the use of nets, boxes, bags and other forms of capture and restraint. While generally effective, these methods of control may exacerbate a stressful situation for the animal.
The ideal injectable short-term anaesthetic technique should produce excellent hypnosis, muscle relaxation and analgesia, act rapidly, produce predictable and minimal cardiorespiratory effects, be reversible and maintain analgesia after recovery from anaesthesia. Ketamine and tiletamine–zolazepam (Telazol) are the only short-acting injectable anaesthetics that may be administered intramuscularly and are approved for use in cats in the United States. Although ketamine has been demonstrated to be safe and effective in cats, the doses required to produce surgical anaesthesia also produce catalepsy, muscle rigidity and involuntary muscle movements and a prolonged time, generally hours, for recovery (Evans et al 1972). Ketamine is not reversible and is not noted as a particularly effective postoperative analgesic unless combined with opioids or α2 agonists (Verstegen et al 1990, 1991, Robertson and Taylor 2004). Ketamine–sedative drug combinations reduce the dose of ketamine required to produce anaesthesia thereby shortening the duration of recovery but most have fallen short of producing a clinically ideal anaesthetic experience. Acepromazine–ketamine drug combinations, for example, reduce the dose of ketamine required to produce anaesthesia and shorten the duration of recovery thereby improving the quality of anaesthesia but do not markedly improve analgesia and provide poor muscle relaxation (Verstegen et al 1991). Opioids–ketamine drug combinations provide analgesia during ketamine anaesthesia but do not effectively improve muscle relaxation (Robertson et al 2003). α2 Agonist (xylazine, medetomidine)–ketamine drug combinations enhance muscle relaxation and the quality of anaesthesia and, in addition, produce analgesia facilitating short elective surgical procedures (castration, onychectomy; Verstegen et al 1990, 1991, Lemke 2004). In Europe the drug combination of medetomidine, butorphanol and ketamine is licensed for anaesthesia in the cat. Recovery, however, can be prolonged (hours), if the α2 agonist is not reversed (eg, with atipamazole), during which time cats can develop hypothermia, bradycardia and respiratory depression (Verstegen et al 1990, Williams et al 2002, Selmi et al 2003).
The lingering sedative effects of α2 agonists can be antagonised by α2 antagonists (yohimbine, tolazoline, atipamezole) thereby hastening recovery although postoperative analgesia may be compromised and many cats show signs of excitement, irritability and discomfort most likely caused by pain and stress (Hsu and Lu 1984, Mama 1998). Atipamezole may be preferred compared to other α2 antagonists based on its selectivity for the α2 adrenoceptor (Lamont et al 2001).
Based upon this background and our clinical experiences using multiple preanaesthetic protocols in combination with ketamine we developed an α2 agonist, opioid, ketamine drug combination for intramuscular administration to cats. Our clinical objective was to produce anaesthesia characterised by excellent muscle relaxation and analgesia that was reversible without eliminating postoperative analgesic effects. The purpose of this study was to determine the quality of anaesthesia, cardiorespiratory effects and analgesic activity of morphine sulphate, medetomidine hydrochloride, ketamine hydrochloride drug combination administered intramuscularly to healthy cats. The antagonistic effects of atipamezole hydrochloride were also investigated.
Materials and methods
Animal selection and instrumentation
This study was reviewed and approved by The Ohio State University Institutional Laboratory Animal Care and Use Committee. Six adult female cats weighing between 3.5 and 4.8 kg were the subjects of this study. Each cat was judged to be in excellent health based on a physical examination, electrocardiogram, negative faecal floatation, blood haematology and serum chemistry. The cats used for these experiments had been surgically implanted with telemetry transmitters (TL11M2-D70-PCT, Data Science Co, St. Paul, MN) several months before the experiment (Miller et al 2000). The telemetry units permitted the recording of the electrocardiogram (ECG, lead II), direct aortic blood pressure and core body temperature (Dataquest ARTT Version 3.1, Data Science Co, St. Paul, MN).
Experimental procedures
Pilot and published studies suggested the doses of drugs we selected based upon the duration of non-responsiveness to a noxious stimulus (toe and tail pinch) was approximately 120 min which was selected as the time to administer the α2 antagonist atipamezole (Verstegen et al 1990, 1991, Lamont et al 2001, Robertson and Taylor 2004). Food but not water was withheld from each cat for at least 12 h before anaesthesia. All evaluations were carried out by the same investigator (AJW). Each cat was administered 0.2 mg/kg morphine sulphate (Elkins-Sinn), 60 μg/kg medetomidine hydrochloride (Domitor; Pfizer) and 5 mg/kg ketamine hydrochloride (Ketaset; Fort Dodge) drug combination (MMK) administered as a single injection in either the right or left quadriceps muscle and placed on a circulating warm water heating pad (Gaymar Industries Inc, Orchard Park, NY) set at 39°C. Data were recorded during stable conditions once the cats were acclimatised to the laboratory environment, 5 min before drug administration, 3 and 10 min after drug administration and every 10 min thereafter for a maximum of 120 min or until the cat was administered atipamezole (Antisedan; Pfizer) and responded to a noxious stimulus or became conscious.
Systolic, diastolic, and mean arterial blood pressures (mmHg), heart rate (beats/min) and rhythm (ECG), percent saturation of haemoglobin with oxygen (%, SpO2), respiratory rate (breaths/min), tidal volume (ml), minute volume (l), end tidal carbon dioxide concentration (mmHg) and core body temperature (°C) were recorded. Heart rate was obtained from the arterial pressure waveform. The pulse oximeter sensing probe was placed on the ear in conscious cats and tongue in anaesthetised cats. Respiratory variables were obtained by face mask (for conscious animals) or after orotracheal intubation in anaesthetised cats (Nico Cardiopulmonary Management System model 7300, Novametrix Medical Systems Inc, Wallingford, CT). The face mask (LEI Medical, Boring, OR) was visually and manually inspected to insure a tight fit. Lidocaine hydrochloride (Phoenix Scientific, St Joseph, MO), 0.1 ml 2%, was sprayed on the larynx to minimise laryngospasm when required. Cats breathed room air spontaneously for the duration of anaesthesia. Times to lateral recumbency, intubation, extubation (non-stimulated swallowing; end of anaesthesia) and recovery to sternal recumbency were recorded. Cats were observed for adverse events defined as apnoea for greater than 1 min, SpO2 less than 90% or a systolic arterial blood pressure less than 90 mmHg. Atipamezole, one-half the volume of medetomidine, was administered intramuscularly to all cats at 120 min. Respiratory variables other than respiratory rate were not determined after atipamezole administration to minimise stress during recovery.
Analgesia and anaesthesia
A noxious stimulus was applied, in the same location, to a rear limb metatarsus and the base of the tail. The noxious stimulus was applied after recording cardiorespiratory data at 3 and 10 min after MMK administration and every 10 min thereafter until the cat responded or was administered atipamezole. A 22 cm Pean intestinal clamp was used to produce a mechanical noxious stimulus by closing it to the first ratchet on the metatarsus or base of the tail for 10 or 60 s, respectively, or until the cat responded with a purposeful movement score of 2 or greater. Response to the noxious stimulus was categorised as no response (score of 0), minimal movement (1), limb/tail withdrawal (2), or limb/tail withdrawal and lifting of the head (3). Anaesthesia was assessed at approximately the same time points and categorically defined (see Fig 1). Anaesthesia was considered adequate if the score was 4 or greater.

Sedation/anaesthesia scoring system.
Statistical analysis
Data are reported as mean±SD. A one-way analysis of variance for (ANOVA) for repeated measures was used to analyse continuous numerical data (GraphPad Prism Version 4.0). A Dunnet's post test was used to detect differences from baseline. When appropriate a Tukey's post test was performed to detect differences between time points. Significance was considered to be P<0.05.
Results
Drug injection-associated vocalization and resistance to physical restraint occurred during the intramuscular administration of MMK. The onset of sedation and anaesthesia was less than 1 min and uneventful. Vomiting, excitement, apnoea, and abnormal behaviour were not observed in any cat. All but one cat (ANE5) required the administration of lidocaine to ablate laryngeal spasm and facilitate tracheal intubation.
Anaesthesia and analgesia
All but one cat (VD7) had an anaesthesia score of 4, 3 min after MMK administration. All but one cat had an anaesthetic score of 4 or greater from between 10 and 120 min after MMK administration. One cat (HS6) had an anaesthesia score of 3 from 100 to 130 min after MMK administration. All but one of the cats (HS6) were non-responsive to noxious stimuli from 3 to 120 min after MMK administration. This same cat responded to the toe pinch at 80 min and tail pinch at 115 min and was extubated at 115 min after responding (chewing, swallowing) to the noxious stimulus applied to the toe. All cats regained consciousness and were extubated at 2.9±1.1 min, after atipamezole administration. All cats had an analgesic score of 2 or less at the time of sternal recumbency.
Cardiovascular parameters
The intramuscular administration of MMK significantly decreased heart rate (P<0.01; Fig 2). Heart rate remained relatively stable from 10 to 120 min after MMK administration. Heart rate was not different from baseline values after the administration of atipamezole but was significantly increased compared to the 10–120 min values. Arterial blood pressure was increased 3 min after MMK administration compared to baseline values (Fig 3). The increase in diastolic blood pressure was significantly (P<0.01) different from baseline values while systolic and mean blood pressure increased, but not significantly. Maximum arterial blood pressures at 3 min were systolic blood pressure 181±13.2 mmHg, diastolic arterial pressure 145±14.1 mmHg, and mean arterial pressure 158±8.7 mmHg. Arterial blood pressure decreased after 3 min. There were no significant differences in arterial blood pressure between 60 and 120 min. One cat (ANE5) experienced hypotension (systolic blood pressure 83 mmHg) following administration of MMK. The cat became hypotensive (systolic blood pressure between 76 and 89 mmHg) approximately 4.5 min after the administration of MMK and remained below 90 mmHg for 30 min. No other adverse events occurred in this cat. Blood pressure decreased after the administration of atipamezole and was significantly less than baseline values (Fig 3). The SpO2 (93±6%) did not change from baseline values.

Effects of the intramuscular administration of MMK on heart rate (beats/minute) in six cats (mean±SD). The −5 min recording is before drug administration. *=significantly different from baseline (−5 min). P<0.05.
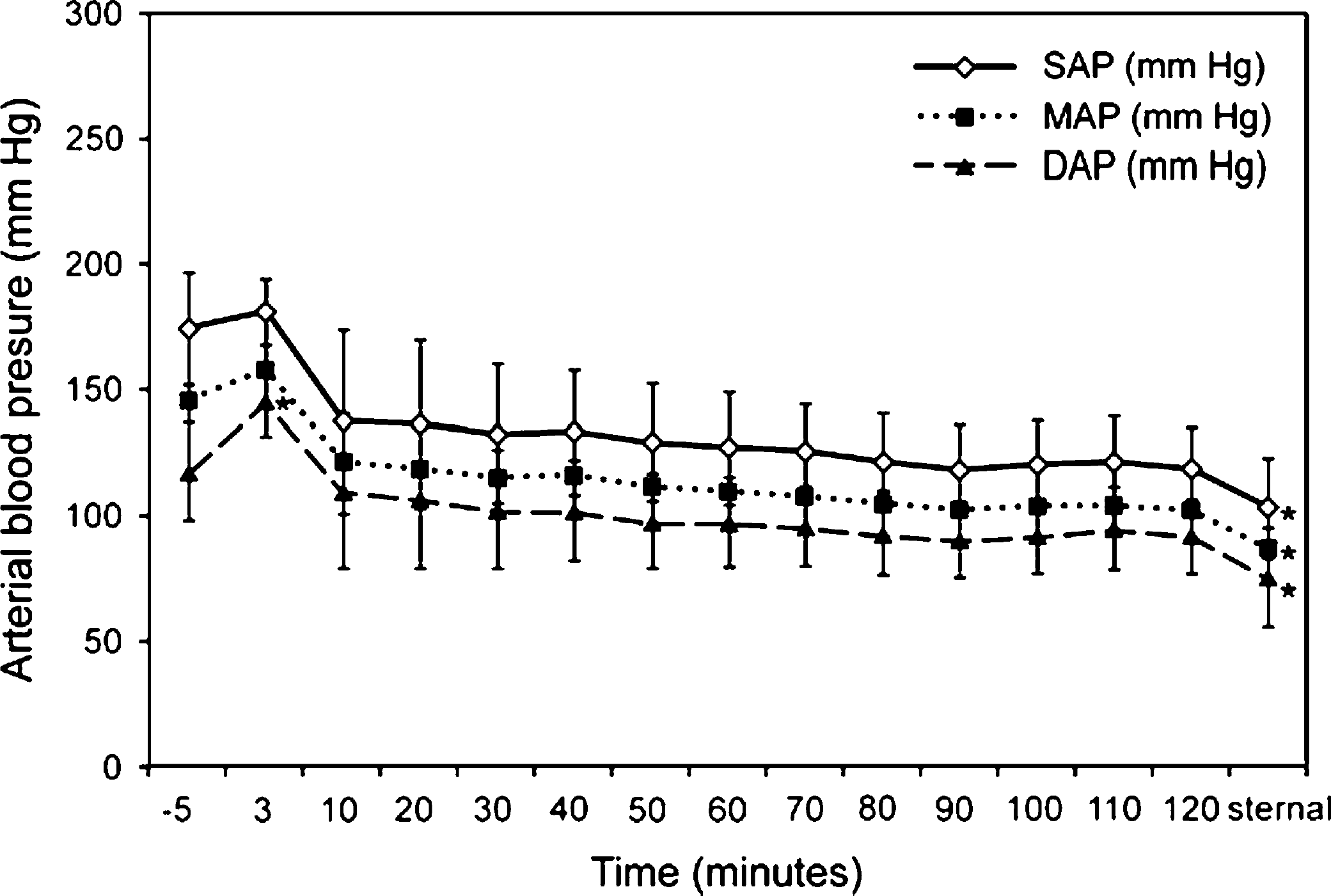
Effects of the intramuscular administration of MMK on systolic (SAP), mean (MAP) and diastolic (DAP) arterial blood pressure in six cats (mean±SD). The −5 min recording is before drug administration. *=significantly different from baseline (−5 min). P<0.05.
Respiratory parameters
Respiratory rate and minute volume were significantly decreased compared to baseline values (57 breaths/min and 2.2 l/min), after the administration of MMK. Tidal volume was increased at 3 min but was decreased from 10 to 120 min compared to baseline. End tidal carbon dioxide was significantly increased at all time points compared to baseline (PaCO2=29±4 mmHg). Respiratory rate, minute volume, tidal volume and end tidal carbon dioxide were not significantly different between 3 and 120 min (Fig 4). Respiratory parameters returned towards baseline values after the administration of atipamezole. Body temperature (38.2+0.2°C) did not change from baseline values.
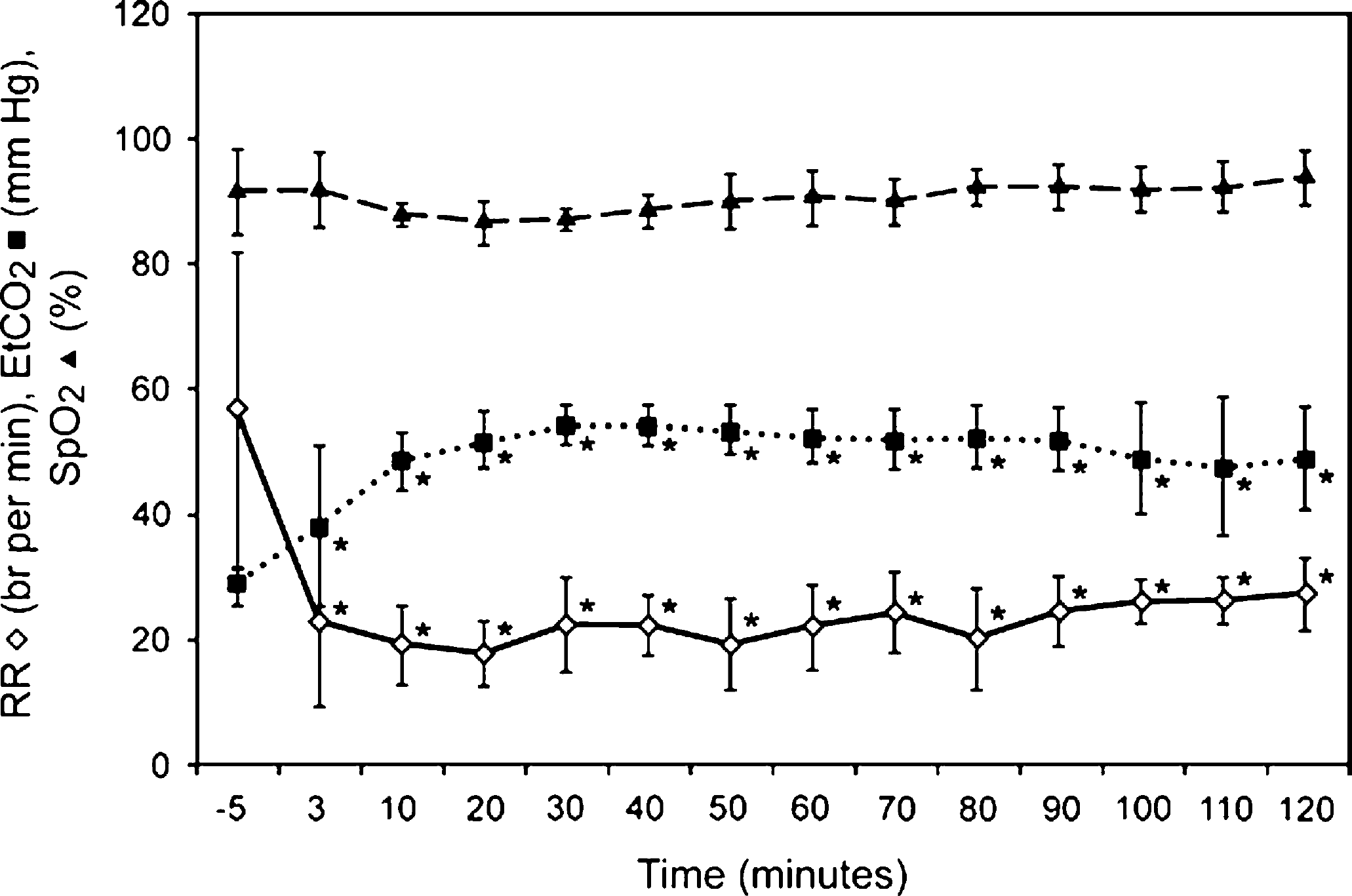
Effects of the intramuscular administration of MMK on respiratory rate (RR), end tidal carbon dioxide concentration (EtCO2) and saturation of haemoglobin with oxygen by pulse oximetry (SpO2) in six cats (mean±SD). The −5 min recording is before drug administration. *=significantly different from baseline (−5 min). P<0.05.
Timing of anaesthesia events
The average time to lateral recumbency following administration of MMK was 1.9±1.2 min. The average time to intubation was 4.3±1.2 min. The average time to extubation and sternal recumbency following administration of atipamezole were 2.9±1.1 and 4.7±1.0 min, respectively.
Discussion
Intramuscular administration of MMK to cats produced rapid and uneventful onset of sedation and anaesthesia, which persisted for a minimum of 90 min and was rapidly and uneventfully antagonised by atipamezole. Predictable and clinically unimportant changes in arterial blood pressure and haemoglobin saturation measured by pulse oximetry were observed in five of six cats. The duration of non-responsiveness to a noxious stimulus applied to the toe and tail persisted for a minimum of 90 min in one cat and for 120 min in the remaining five cats suggesting adequate analgesia. The quality of anaesthesia was good to excellent and produced only minor and clinically irrelevant changes in cardiopulmonary function. Recovery from anaesthesia was uneventful and excitement free. Each cat recovered quickly and smoothly and, although reluctant to move demonstrated minimal ataxia.
Our study provides data that support reports suggesting that the combination of ketamine with analgesics improves anaesthesia in cats (Verstegen et al 1991, Selmi et al 2003). Morphine, the prototypic μ-opioid agonist, produces pronounced analgesia in cats by occupying μ receptors at pre- and postsynaptic sites both peripherally and in the dorsal horn of the spinal cord (Song and Zhao 1993). Despite historical reluctance to use opioids in cats (Lascelles et al 1999), due to inadvertent overdose and development of central nervous system (CNS) side effects, current recommendations suggest that clinically relevant doses of morphine in cats produce excellent analgesia for prolonged periods of time (Robertson and Taylor 2004). Morphine is metabolised by uridine diphosphoglucuronide transferase in the liver, a process which is relatively ineffective and partially absent in cats. Morphines slow metabolism in cats explains the 6.5 and 8 h duration of clinical effect (Taylor et al 2001, Robertson et al 2003). Medetomidine is a highly selective α2 agonist (α2:α1 selectivity 1620:1) that activates both pre- and postsynaptic α receptors in the peripheral nervous system (PNS) and presynaptic α receptors in the CNS thereby decreasing norepinephrine release and reducing CNS sympathetic outflow (Hsu and Lu 1984, Virtanen 1989, Allen et al 1986, Lamont et al 2001). Medetomidine is a very effective sedative producing dose dependent analgesia that may be profound, although opioids are still considered more potent analgesics (Verstegen et al 1990, Ansah et al 2002). Ketamine acts centrally to produce dissociative anaesthesia, and is a N-methyl-d-aspartate (NMDA) antagonist (Song and Zhao 1993, Bell et al 2005). Low dosages (less than 10 mg/kg IV, IM) of ketamine preserve cardiac function but do not provide surgically acceptable muscle relaxation or analgesia and may cause hypersalivation (Allen et al 1986).
Our studies provide data that demonstrate that clinically relevant dosages of MMK produced minimal changes in the measured cardiorespiratory variables compared to baseline values. The transient increase and then decrease in arterial blood pressure were attributed to an initial vasoconstriction typical of α2 agonists such as medetomidine that wanes with time (Lemke 2004). Blood pressure transiently dropped and heart rate increased after atipamezole administration due to blockade of peripheral α2 receptors, subsequent vasodilation and a compensatory increase in heart rate occurred (Lemke 2004). Although the analgesic and sedative effects of medetomidine were reversed by atipamezole, the residual and possibly synergistic analgesic effects of ketamine and morphine are unaffected (Nemmani et al 2004, Shulte et al 2004).
Ketamine probably prevented bradycardia from developing because both medetomidine and morphine are capable of producing bradycardia (Seth et al 1990). Hypotension occurred in one cat (systolic arterial pressure <90 mmHg) but this did not require administration of rescue drugs, indicating that this is a relatively cardiovascularly safe drug combination. This result is in contrast to a previous report where α2 agonist–ketamine drug combinations produced more significant hypotension (Allen et al 1986). Temperature did not change in any cat in our study following the administration of MMK. Others have reported an increase in body temperature in some cats administered opioids (Selmi et al 2003, Robertson et al 2003). Further studies are required to determine the dose response characteristics of opioids on body temperature in cats.
The decrease in respiratory rate and corresponding increase in end tidal carbon dioxide after MMK administration did not significantly affect the SpO2 at any time during anaesthesia. Adequate SpO2 during anaesthesia suggests good tissue perfusion and adequate oxygen tension was maintained.
Non-invasive assessment of haemodynamic variables in cats has proven to be a stressful and potentially unreliable method for monitoring arterial blood pressure (Branson et al 1997, Pedersen et al 2002). The cats in the current study required minimal handling or restraint because they had been instrumented with telemeter units that measured direct arterial blood pressure, the electrocardiogram and core body temperature. This helped to minimise stress produced by physical restraint or confinement. The only time the cats required physical restraint was during drug administration. We tested the anaesthetic, analgesia and cardiorespiratory effects of a single dose of the MMK combination in an analgesiometry model using a mechanical noxious stimulus. This dose combination did not cause clinically relevant disturbances in the measured cardiorespiratory parameters and provided good analgesia in this test model. We did not evaluate the anaesthetic or analgesic effects of MMK in cats subjected to surgery and pain from elective procedures or naturally occurring diseases. This will be the focus of future studies.
Our findings suggest that intramuscular injection of MMK doses investigated in the present study should be adequate for most elective surgical procedures but remains to be tested clinically. Sedation can be antagonised by atipamezole without significantly affecting the extended analgesic activity of morphine.
Footnotes
Acknowledgements
The authors wish to thank Dr Dan Smeak for performing transmitter implantations and Dr Anja Waselau, Ms Barbara Lang, and Dr Tokiko Kushiro for technical assistance.
