Abstract
Although cats and their arthropod parasites can sometimes be important sources of zoonotic diseases in humans, the extent of exposure among various cat populations to many potential zoonotic agents remains incompletely described. In this study, 170 domestic cats living in private homes, feral cat colonies, and animal shelters from California and Wisconsin were evaluated by serology to determine the levels of exposure to a group of zoonotic vector-borne pathogens. Serological positive test results were observed in 17.2% of cats for Rickettsia rickettsii, 14.9% for R akari, 4.9% for R typhi, 11.1% for R felis, and 14.7% for Bartonella henselae. Although vector-borne disease exposure has been documented previously in cats, the evaluation of multiple pathogens and diverse cat populations simultaneously performed here contributes to our understanding of feline exposure to these zoonotic pathogens.
Although cats and their arthropod parasites can sometimes be important sources of zoonotic diseases in humans, the extent of exposure among various cat populations to many potential zoonotic agents remains incompletely described. Bacteria in the genera Rickettsia, Bartonella and Coxiella are the causative agents of a number of potentially serious infectious diseases of humans and domestic animals including murine typhus-like disease caused by R felis, murine typhus from R typhi, Rocky Mountain spotted fever (RMSF) caused by R rickettsii, Q-fever caused by C burnetii and cat scratch disease (CSD) caused by B henselae (Rolain et al 2003). The cat flea (Ctenocephalides felis), the Oriental rat flea (Xenopsylla cheopis), the house mouse mite (Liponyssoides sanguineus) and various tick species (Dermacentor andersoni, D variabilis, Rhipicephalus sanguineus and Amblyomma americanum) carry and transmit these agents (Comer et al 2001a). In humans, clinical sequelae of vector-borne disease can include fever, endocarditis, encephalitis, and more rarely death (Gorbach et al 1998). In cats, these agents often are not associated with illness or clinical signs (Shaw et al 2001).
Despite some studies of exposure to vector-borne disease in cats (Chomel et al 1995, Matthewman et al 1997), there remains insufficient information regarding the prevalence of these pathogens in cats to fully evaluate human risk. Several important questions include the degree to which infection circulates within feline populations; whether or not cats maintain or amplify infection after sporadic introduction of disease from sylvatic or other reservoirs; whether cats may function as sentinels that could potentially aid in the detection and prevention of human disease; and whether different cat population structures, including feral cat populations, shelters, breeding catteries, and private homes, impact the likelihood of exposure to various vector-borne pathogens among cats.
The aim of this study was to provide data for specific pathogens within several domestic and feral cat populations at risk for vector-borne diseases. The pathogens chosen represent the flea-transmitted zoonoses for which cats may be involved as reservoirs, vector-borne ‘select agent’ (ie, possible agents of bioterrorism) bacterial pathogens which could circulate in cat populations and for which cats could serve as sentinels of human risk, and pathogens which could cause test cross-reactivity. To accomplish this, 170 cats from private homes, feral cat colonies, and animal shelters were evaluated by serology to determine the frequency of exposure to a panel of zoonotic vector-borne pathogens comprising the cat flea-transmitted zoonoses (B henselae and R felis), feline agents of possible biological warfare (C burnetii, and Rickettsia species) and likely serologically cross-reacting agents (R akari and R rickettsii).
Materials and methods
Experimental animals and populations
Blood samples were obtained as convenience samples from the Dane County Wisconsin Humane Society, the Solano County California Animal Shelter, the Orange County California Animal Shelter, and from among cats that had been admitted to the University of California, Davis Veterinary Medical Teaching Hospital (VMTH) between 2001 and 2003. The shelters and the individual samples were selected solely on the basis of being readily available. Cats from shelters were described as feral (untamed, typically from a wild colony not directly maintained by humans) or general population (previously owned, tame, and either owner-relinquished or found roaming). In all three shelters, feral and general population cats were kept separately in different areas of the shelter, although feral cat samples were only available from the Solano County facility. The blood samples from the VMTH were from pet cats residing in Yolo and Sacramento Counties and the San Francisco Bay area. Whole blood samples were collected in EDTA tubes, centrifuged to separate the plasma from the packed red blood cells, and frozen until ready for shipping. Once the samples reached the laboratory they were maintained in a −70°C freezer until the assays were performed.
Serological tests
Preparation of antigen slides
Antigens for C burnetii and all rickettsiae were provided by one of the authors (WN), prepared by using standard protocols at the Centers for Disease Control (CDC) Atlanta, Georgia. All rickettsial and C burnetii antigens were propagated in chicken egg-yolk sac except for R felis, which was propagated via cell culture. Lyophilized R rickettsii and C burnetii antigens were re-suspended in sterile saline at UCD. Antigens of R felis, R typhi, and R akari were in frozen suspension upon arrival at UCD. Glass slides for R felis indirect immunofluorescence assay were coated with 1% bovine serum albumin (BSA) in water solution before antigen application, in order to prevent excessive downstream loss of R felis antigen. Immediately following application of the 1% BSA solution, the fluid was aspirated and the slides were allowed to dry. For all antigens, 3 μl aliquots were applied to 12-well (4 mm diameter) HTC slides (Cell-line/Erie Scientific Company, Portsmouth, NH) and allowed to air dry at room temperature within a biological safety cabinet. When dry, the slides were fixed in acetone for 15 min at room temperature, and the acetone was allowed to evaporate. The slides were then stored within sealed plastic bags in a −70°C freezer. B henselae slides were prepared as described previously, with modifications (Chomel et al 1995). Mixed B henselae strains', Houston I and U4, whole cell preparations were used as the source of antigen, 40 μl of antigen applied to wells, and slides incubated overnight. After two washes in phosphate-uffered saline (PBS), slides were fixed in acetone, air-dried and stored at −20°C.
Indirect immunofluorescence assay (IFA)
The positive controls used for C burnetii and Rickettsia species consisted of plasma or serum from known-positive human patients and obtained from the CDC. For B henselae, positive controls were obtained from experimentally infected (strains Houston I and U4) cats with confirmed positive titers. All negative controls were from specific-pathogen free cats.
Frozen plasma samples, as well as the positive and negative control samples, were diluted with PBS–1% BSA solution at a pH of 7.4. Twenty-five microliters (C burnetii and Rickettsia species) or 30 μl (Bartonella species) of plasma diluted in PBS containing 5% milk was applied to the wells of the respective antigen slides. Screening for C burnetii and Rickettsia species was performed at a 1/25 dilution and for Bartonella species at 1/64. Slides were incubated at 37°C in humid conditions for 35 min, then washed three times in PBS (5 min per wash). Fluorescein-conjugated goat anti-cat or anti-human heavy and light chain IgG (Kirkegaard and Perry Laboratories Gaithersburg, Maryland) were diluted in PBS at 1/800. Then 30 μl aliquots of the secondary antibody in PBS were spotted onto slide wells. The slides were again incubated at 37°C for 35 min, washed in PBS, and then counter-stained with Eriochrome Black T (Sigma, St. Louis, MO). A small drop of 10% glycerol in PBS was applied to each well followed by a cover slip. Slides were examined via ultraviolet epifluorescence by two examiners independently and scored as negative, positive, or weak (suspect) positive. For the purposes of statistical analysis, weakly reacting samples were deemed as negative. All serum samples that demonstrated positive serological reactions to multiple rickettsial species were further diluted to obtain individual endpoint titers and to attempt to distinguish among cross-reacting antigens.
Experimental design and statistical analysis
Data were maintained in Excel (Microsoft, Redmond, WA) and analyzed in ‘R’ (The R-Development Core Team, http://www.r-project.org). For all tests, data were considered statistically significant if P≤0.05. Cats were grouped into the following age classes: less than 1 month, greater than 1 month but less than 6 months, and greater than 6 months. A χ 2 test was employed to analyze the differences in serological positivity to each pathogen among cats based on age, on feral, general population, and privately owned status, and by shelter. Agreement between suspected cross-reacting antibodies (R rickettsii and R akari, and R typhi and R felis) was evaluated using the Kappa statistic.
Results
One hundred and seventy cats from four populations were evaluated by serology for exposure to vector-borne pathogens. The 80 Solano County Animal Shelter cats comprised five cats less than 1 month of age (6.3%), 28 (35%) between 1 and 6 months of age, and 47 (8.9%) greater than 6 months of age. Thirty-six cats (45%) were feral and 44 (55%) were from the shelter's general population. The 50 Wisconsin cats were from the general population but information regarding age was not available. Of the 20 Orange County cats, four (20%) were between 1 and 6 months of age, eight (40%) were greater than 6 months of age, and eight (40%) were of unknown age. All 20 cats were housed in the shelter's general population. The 20 cats from the UCD VMTH were privately owned and greater than 6 months of age.
Frequencies of exposure among the pathogens ranged from 0 to 24%, with antibodies most commonly detected against B henselae, R rickettsii, R akari, R felis and R typhi (Table 1). Weak positive reactions to C burnetii phase II were detected in 23% of cats, up to 50% in Orange County, but these were scored negative according to the protocol described in Materials and methods. No serological evidence for exposure to C burnetii phase I antigen was demonstrated.
Fraction of cats seropositive (and 95% confidence intervals) for bacterial pathogens based on shelter or owned status

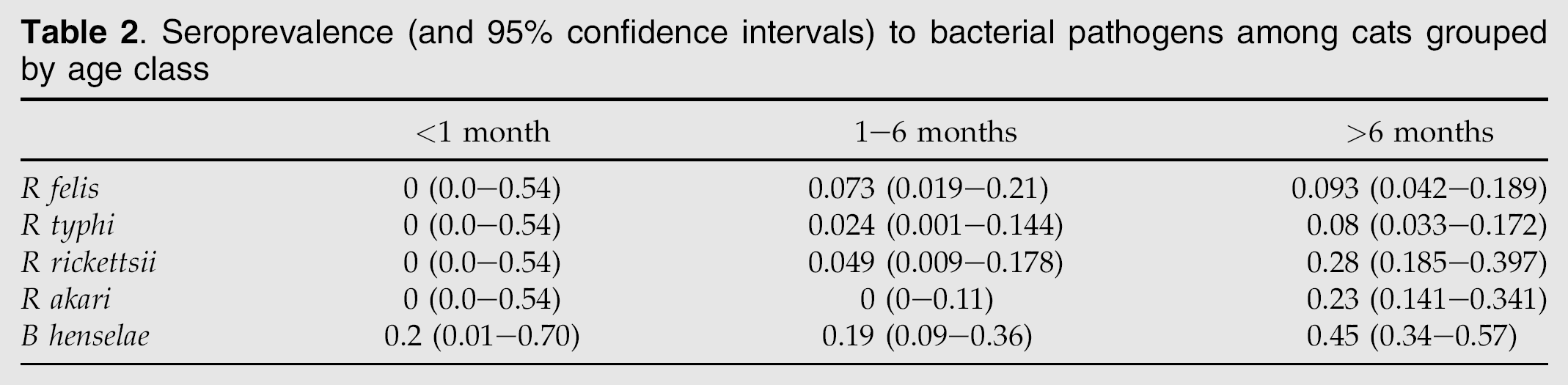
There was no single shelter or cat population that had the highest rates of exposure to all pathogens, although Solano County had the highest rates of exposure to three of the five pathogens. B henselae was significantly more common in Solano and Orange counties, when compared to Dane County, Wisconsin (Table 1) (P=0.8×10−5). Differences in exposure among shelters for the other pathogens were not statistically different. There was evidence of increasing exposure to all pathogens as a function of age, particularly in R rickettsii, R akari and B henselae (Table 2.) There were no significant differences in exposure between feral or general-shelter population cats in the Solano shelter (Table 1). When feral and general-shelter population cats from all sources were grouped and contrasted with privately owned cats, B henselae exposure seemed most common in feral cats, while sheltered cats appeared over-represented for R akari exposure although these differences were not statistically significant (Fig 1).

Comparison of seroprevalence to feline vector-borne pathogens among feral cats, privately owned cats, and cats in shelter general populations.
Seroprevalence (and 95% confidence intervals) to bacterial pathogens among cats grouped by age class
Between R rickettsii and R akari serology, the Kappa statistic was 0.16 (poor test agreement). Of cats with seropositive test results for both spotted fever group antigens, the titer to R akari was higher than to R rickettsii 17.5% of the time. For R felis and R typhi, a Kappa statistic of 0.18 was calculated and there was cross-reactivity in 18.2% of the samples that were positive for either R felis or R typhi. Titers to R felis tended to be higher than to R typhi.
Discussion
Although vector-borne disease exposure has been documented in cats many times, few studies particularly target cats at high risk or evaluate multiple pathogens simultaneously, making interpretation of possible cross-reaction and coinfection difficult. Flea exposure is very common among cats, depending on the environment and flea density, with one study in Florida documenting infestation in 93% of feral cats (Akucewich et al 2002). Thus understanding feline and human risk of these zoonotic diseases is an important goal and this study provides useful information regarding exposure in North American cats to five vector-borne zoonotic bacterial pathogens.
There was overall substantial exposure to B henselae, and lower but important levels of exposure to the spotted fever group and typhus group rickettsiae. Younger kittens under 6 months of age tended to have less evidence of exposure compared to cats greater than 6 months of age, likely because of an accumulated increasing post-weaning risk of exposure. Kittens under 1 month of age have had little time to make antibodies and seropositivity would commonly suggest maternal antibody transfer. Interestingly, neither feral cats nor those from animal shelters appeared to be more commonly exposed to vector-borne disease than privately owned cats. These results mirror those of an earlier study that showed feral cats to be at no greater risk of exposure to bartonellosis (as well as feline retroviruses) than owned pet cats (Luria et al 2004), although one study in North Carolina found the opposite (Nutter et al 2004). We had hypothesized that cats in animal shelters (housed at high density with high turnover) and in feral cat colonies would have higher rates of exposure to vectors than privately owned cats, and thus our results and those of Luria et al (2004) initially were unexpected. However, many cats only remain in animal shelters for a few days to a few weeks, which is probably not enough time to seroconvert after vector exposure. Exposure of cats in shelters to hard ticks (which vector some rickettsiae) would be unusual, although it might be expected in feral cats and has been reported for feral cats from Florida (Akucewich et al 2002). All three groups could be expected to suffer flea infestations. The fact that our data documented significant exposure to vector-borne pathogens even among pet cats emphasized the importance of surveillance and vector control even for these cats.
Cats are the main reservoir species for Bartonella henselae infection, with reported seroprevalence varying from 4% to 81%, including 24% in cats in the present study and 36% in catteries (Koehler et al 1994, Childs et al 1995, Chomel et al 1995, Foley et al 1998, Shaw et al 2001). Among breeding catteries, the most important risk factor for infection was flea infestation, with non-significant risk factors including cattery size, presence of rescued cats, presence of dogs, and history of travel (Foley et al 1998). In a study from the UK, 22% of C felis flea pools from cats were B henselae polymerase chain reaction-positive (Shaw et al 2004). In the present study, age was significantly associated with B henselae exposure, with a higher rate in cats over 6 months old, yet feral cats did not have significantly higher exposure to B henselae when compared to cats from a shelter's general population, or to privately owned cats. However, French cats adopted from shelters or found as strays were at greater risk for bacteremia compared with other cats (Gurfield et al 2001). Infection is important in cats because they can serve as reservoirs for the pathogen and the fleas, and can sometimes suffer clinical consequences of infection themselves (Guptill et al 1998, Kordick et al 1999, Guptill 2003).
Seropositivity to C burnetii could not be confirmed in the present study because of the finding of only weak positive phase II titers in up to 50% of cats, most commonly in cat populations in the Orange county area. Previously reported seroprevalence has ranged from 9 to 20% in populations of cats from the US, Japan, and Canada (Willeberg et al 1980, Shaw et al 2001). In nature, virulent C burnetii organism expresses a lipopolysaccharide; in culture this phase is described as phase I. After serial laboratory passages, the organism fails to express lipopolysaccharide and enters phase II. Paradoxically, although only phase I is virulent, cases of acute Q-fever in humans are characterized by rising phase II and not phase I titers; chronic Q-fever is characterized by elevated titers to both phases I and II (Raoult et al 2000). In contrast, wild and domestic animals have varying titers to the two phases, with higher phase II, compared to phase I, titers reported for cats, goats, sheep, deer, and raccoons, but not hares, moose, or cattle (Marrie et al 1985, 1993). In this study, there were no phase I reactions and only weak reactions to phase II. Although cats that are subcutaneously inoculated with C burnetii can become febrile and lethargic, exposure in cats is most significant because cats may constitute an important source of infection to humans (Chomel 1997).
There was evidence in cats in this study of exposure to typhus- and spotted fever group rickettsiae. However, interpretation of these results, and information from the literature, is difficult. R typhi and R felis have significant serological cross-reactivity, making it sometimes difficult to know which pathogen is responsible for seroreactors without confirmatory evidence of active infection and the identification of the agent, eg, by polymerase chain reaction (PCR), cross-adsorption, or some other technique. Moreover, because R felis was only relatively recently described, older literature referring to ‘murine typhus’ may sometimes be including R felis.
In the present study, exposure to both pathogens was evaluated concurrently and dual-reacting sera were titered to endpoint. Results suggested that there were approximately twice as many cats with R felis compared with R typhi exposure, although confirmation would have required cross-adsorption studies which were not feasible given the limited amounts of available sample and reagents. From 4 to 10% of cats from both California and Wisconsin had antibodies to R typhi. This pathogen occurs worldwide, particularly in areas with warm climate and is transmitted by rat and mouse fleas Xenopsylla cheopis and Leptopsylla segnis, respectively (Raoult and Roux 1997). Cats may be infected through exposure to rodent fleas or possibly by predation on rodents (Matthewman et al 1997). Several writers have claimed that cats can be ‘reservoirs’ for R typhi (Greene and Breitschwerdt 1998) or that R typhi can be ‘found’ in domestic cats (Kettle 1995) either without citation to primary literature or citing an early review monograph (Traub et al 1980). One study did not find an association between R typhi exposure and cat (or dog) ownership (Wiggers and Stewart 2002).
R felis is a spotted fever group rickettsia with documented serological cross-reactivity with R typhi (Adams et al 1990, Matthewman et al 1997). Disease in humans infected with R felis is murine typhus-like (Raoult et al 2001) but infection in cats is typically asymptomatic (Wedincamp and Foil 2000). The 11% R felis exposure rate was comparable to the 8% found in a previous study of cats in the northeastern US (Higgins et al 1996). R felis in humans, cats, and the primary reservoir, the opossum (Didelphis virginiana) is vectored by the cat flea Ctenocephalides felis (Chomel 1997, Raoult et al 2001). Twenty percent of C felis pools from cats in the UK were PCR-positive for R felis (Shaw et al 2004) and experimental exposure of cats to R felis-infected fleas resulted in PCR- and seroconversion but no clinical signs (Wedincamp and Foil 2000). Infection in humans directly from cats has not been described, but human murine typhus was described (including with a positive PCR test) in a German couple who were exposed to dogs with R felis titers of 128 (Richter et al 2002). During an investigation of human typhus cases in Los Angeles County, 90% of cats from an area close to the human cases were serologically positive for R typhus compared with 0% from a control area, and ownership of a cat was a significant risk factor for typhus in people (Sorvillo et al 1993).
There is also serological cross-reactivity among spotted fever group rickettsiae; thus samples were evaluated for multiple pathogens in the present study. Antibodies to R rickettsii were detected at about the same frequency as antibodies to R akari (17.2% and 14.9%, respectively), with likely cross-reactions 17% of the time. Although not definitive for identification of the pathogen, titers to R akari in cross-reacting cats tended to be higher than to R rickettsii. Cats, which could be infected with R rickettsii via exposure to Dermacentor species ticks, are described as incidental hosts for R rickettsii (Greene and Breitschwerdt 1998). The relevance of the titers to R akari is not known. R akari exposure has not been documented previously in cats, although dogs from New York City were exposed to R akari, probably via exposure to rat mites (Comer et al 2001b). The zoonotic significance of these findings is not known but worth future surveillance.
Because each of the vector-borne diseases described in this study can induce morbidity and in some cases mortality in humans, and yet typically is clinically silent in cats, evidence of population-level exposure often can be overlooked without deliberate surveillance. Cats in multiple-cat populations such as feral cat colonies, shelters, or pet stores may have high turnover and high rates of arthropod infestation. When adopted, such cats could present a risk for human disease, depending on whether they are actively infected and/or infested with fleas and ticks. Additional useful information in further studies would include more data regarding cat history, signalment, and clinical conditions at the time of testing. Randomized selection of cats or epidemiological study designs such as prospective cohort analysis might yield valuable information. However, this study shows that exposure to some vector-borne diseases is common in cats and requires ongoing investigation and intervention.
Footnotes
Acknowledgments
The authors thank the Dane County, Wisconsin Humane Society, the Solano County, California Animal Shelter, the Orange County, California Animal Shelter, and the University of California, Davis, Veterinary Medical Teaching Hospital for their contributions on this project. Niki Drazenovich and Rick Kasten assisted with laboratory assays. Funding was provided by the UC Davis Center for Vectorborne Diseases.
