Abstract
This study evaluated the effect of two combination products containing febantel, pyrantel, and praziquantel (FPP) for the treatment of Giardia species in experimentally infected kittens. In experiment 1, five kittens were administered the United States (US) formulation of FPP at doses of 37.8 mg/kg, 7.56 mg/kg, and 7.56 mg/kg, respectively, PO, q24h, for 5 days and four kittens remained as controls. In experiment 2, five kittens were administered the European formulation of FPP at the doses of 12.5 mg/kg, 12 mg/kg, and 4.16 mg/kg, respectively, PO, q24h, for 5 days and four kittens remained as controls. In experiment 3, six kittens were administered the US formulation of FPP at 56.5 mg/kg, 11.3 mg/kg, 11.3 mg/kg, respectively, PO, q24h, for 5 days and five kittens remained as controls. Thirteen days after treatment, kittens testing negative for Giardia species cysts were administered 20 mg/kg methylprednisolone acetate, IM, weekly for a maximum of two injections. Feces were analyzed for Giardia species cysts using a direct immunofluorescence test. After experiment 3, four of the six treated kittens, but no control kittens, remained negative for Giardia species after the administration of methylprednisolone acetate.
Giardia lamblia is a flagellate parasite that can cause significant gastrointestinal disease in a wide variety of mammals including kittens and humans. G lamblia is a species complex comprising at least eight major assemblages which can be host specific, or not. To date, only the genetic group I from Assemblage A has been demonstrated to infect both humans and animals, thus proving zoonotic transmission (Monis and Thompson 2003). In the United States (US), Giardia species infection was detected in 5 of 206 (2.4%) adult kittens tested in Colorado (Hill et al 2000) and in 19 of 263 (7.2%) kittens tested in New York State (Spain et al 2001). In Europe, Giardia species infection was detected in 228 of 2234 (10.2%) kittens tested in Germany (Barutzki 2001) and 18 of 81 (22.2%) kittens tested in Serbia (Nikolic et al 2002). In Australia, 2 of 40 (5%) and 32 of 40 (80%) kittens tested positive for Giardia species by microscopy and by polymerase chain reaction (PCR), respectively; suggesting that highly sensitive techniques are required for the diagnosis of giardiasis in kittens (McGlade et al 2003). Thus, it is possible that even larger numbers of kittens are infected by Giardia species than previously estimated.
Albendazole, metronidazole and fenbendazole have been used most commonly for the treatment of feline and canine giardiasis. Albendazole was effective in the treatment of canine giardiasis, but was ineffective in one study of Giardia species infected kittens (Barr et al 1993) and has been associated with bone marrow suppression in kittens (Stokol et al 1997). Metronidazole has been showed to be effective in eliminating Giardia species cyst shedding in some naturally and experimentally-infected kittens (Nesvadba 1979, Shatto 1981, Zimmer 1987, Scorza and Lappin 2004). However, gastrointestinal and central nervous system toxicity has been associated with the administration of metronidazole in some dogs (Dow et al 1989) and kittens (Caylor and Cassimatis 2001). Administration of fenbendazole at a dosage five times greater than the approved dosage was well tolerated by healthy, adult, non-pregnant kittens (Schwartz et al 2000). When fenbendazole was administered to eight kittens concurrently infected with Giardia species and Cryptosporidium parvum, only four stopped shedding Giardia species cysts (Keith et al 2003). A vaccine containing inactivated Giardia species trophozoites, originally developed for the prevention of infection, has proven beneficial in the treatment of some naturally infected dogs (Olson et al 2001). However, the administration of three Giardia species vaccinations did not lessen or eliminate cyst shedding in treated kittens when compared to untreated control kittens (Stein et al 2003).
The administration of a combination of febantel, pyrantel, and praziquantel (FPP), has been evaluated for the treatment of giardiasis in dogs (Barr et al 1998, Barutzki et al 2001, Payne et al 2002). The US and the European formulations of FPP are available. When FPP was developed for the US, efficacy data on roundworms, hookworms and whipworms were submitted with a dose of at least 25 mg/kg to achieve highly effective whipworm control (personal communication, Bayer Animal Health 2005). For Europe, studies have shown that a lower dosage of febantel was sufficient for gastrointestinal nematode control including whipworm and thus, a different formulation was licensed (personal communication, Bayer Animal Health 2005).
To our knowledge, this drug combination has not been evaluated for the treatment of giardiasis in kittens.
The purpose of this study was to evaluate the US and European formulations of FPP for the treatment of Giardia species infections of experimentally infected kittens.
Materials and methods
Animals
Twelve-week-old, mixed sex kittens (n=26) were purchased from a commercial vendor. The kittens were vaccinated subcutaneously (SQ) with a feline panleukopenia virus, herpesvirus 1, and calicivirus vaccine on 2 occasions 4 weeks apart and administered pyrantel pamoate PO twice before shipping to Colorado State University. On arrival, kittens were shown to be FeLV antigen and FIV antibody negative (SNAP Combo, IDEXX Laboratories, Portland, ME) and normal on complete blood cell count, serum biochemical panel, and urinalysis. The kittens were housed separately, fed a commercial feline diet ad libitum, and were observed daily for attitude, stool consistency, and signs of drug toxicity including anorexia, vomiting and diarrhea.
Fecal assays
Prior to inoculation with Giardia species, three fecal samples from each cat collected within a 10-day period, and assessed for the presence of enteric parasites by microscopic examination of feces after zinc sulfate centrifugation, following standard procedure for Colorado State University. In addition, each fecal sample was diluted 1:4 in 0.01 M phosphate buffered saline solution (pH=7.2) and a thin fecal smear made on microscope slides supplied in an in vitro direct immunofluorescence assay (IFA) capable of simultaneous detection of Giardia species cysts and Cryptosporidium species oocysts (Merifluor Crypto/Giardia IFA kit, Meridian Diagnostic Corporation, Cincinnati, OH). After slides were stained as instructed by the manufacturer, the number of cysts per slide was counted by use of a fluorescence microscope. A cyst score was then assigned from 0 to 4 based on the following: 0=0 cysts per slide; 1=1 to 250 cysts per slide; 2=250 to 500 cysts per slide; 3=greater than 500 cysts per slide; 4=TNTC (“too numerous to count”, defined as >100 cysts per 10× objective field).
Experimental design
Each of the three fecal samples collected from the 26 kittens were shown to be negative for enteric parasites prior to infection.
Experimental inoculation with Giardia cysts
Giardia species cysts from a naturally infected cat, and identified by IFA, were administered to two of the kittens to produce enough giardia cysts to infect the 24 kittens to be used in the treatment study. After withholding food for 12 h, each cat was given 2×104 giardia cysts in water by stomach tube while sedated with ketamine (5–10 mg/kg, IV) and acepromazine (0.002 mg/kg, IV).
Our original study design included the infection of 24 kittens that would be randomized into three treatment groups and one control group, with treatment beginning on day 10 post infection. However, only 17 of 26 kittens shed giardia cysts in consistently detectable numbers and the prepatent period varied between kittens. Therefore, the following three experiments were performed sequentially.
Experiment 1
On day 28 post-inoculation (PI), there were nine kittens shedding giardia cysts. The US formulation of FPP (Drontal Plus, Bayer Animal Health, Shawnee Mission, KS) at the approximate dosages of 37.8 mg/kg, 7.56 mg/kg, and 7.56 mg/kg, respectively, was administered to five kittens, PO, q24h, for 5 days. The four remaining kittens served as the untreated control group.
Experiment 2
On day 42 PI, an additional five, previously untreated kittens were persistently shedding giardia cysts. The European formulation of FPP (Drontal Plus, Bayer Vital GmbH, Leverkusen) at the approximate dosages of 12.5 mg/kg, 12.0 mg/kg, and 4.16 mg/kg, respectively, was administered to these five kittens PO, q24h, for 5 days. The untreated kittens from experiment 1 had continued to shed large numbers of giardia cysts and were used as the control group.
Experiment 3
After completion of experiments 1 and 2, 11 kittens were persistently shedding giardia cysts. The US formulation of FPP at the approximate dosages of 56.5 mg/kg, 11.3 mg/kg, and 37.8 mg/kg, respectively, was administered to six kittens, PO, q24h, for 5 days. The other five kittens served as the untreated control group. Three of the six treated kittens had been control kittens in the previous experiments and three had previously been administered FPP but were still persistently infected. Three of the five control kittens had never been treated and two had previously been administered FPP but were still persistently infected.
Thirteen days after each treatment period, all kittens that were negative for giardia cysts were administered 20 mg/kg methylprednisolone acetate, IM weekly for a maximum of two injections. Kittens shedding cysts after the first injection of methylprednisolone were considered giardia-infected and did not receive the second injection. Kittens that remained negative for cysts on the last day of each experiment (day 32 after beginning treatment) were considered not to have Giardia species infection.
Clinical monitoring
Each cat was examined daily throughout the study. Fecal samples were collected daily and analyzed by IFA. Consistency of feces was classified daily as: normal=1; soft=2; watery=3. Appetite was assessed as: normal=1; less than normal=2; and anorexia=3. Complete blood cell count, serum biochemical panel and urinalysis were performed 5 and 19 days after completion of the FPP treatment period in experiments 1 and 2.
Statistical analysis
For each cat group, the mean giardia cyst score, percentage of giardia-positive fecal samples, and percentage of soft stools were calculated within time periods; pre-treatment period (7 days [experiments 1 and 2] or 14 days [experiment 3] prior to drug administration), intra-treatment period (5 days), post-treatment period 1 (7 days post-treatment) and post-treatment period 2 (from days 8 to 14 post-treatment). Differences between treated and control cat groups within each period were evaluated using ANOVA appropriate for a repeated measure experiment (the MIXED procedure of SAS, SAS Institute, Cary, NC).
Treatment, period (time), and the treatment by time interaction were considered fixed effects. For the percentage of giardia-positive fecal samples and percentage of soft stool samples, an arcsine transformation was used in the statistical analysis. In addition, Wilcoxon's rank sum test was also used to assess for mean giardia cyst score differences between groups during the intra-treatment and post-treatment periods. Statistical significance was defined as P<0.05.
Results
Experiment 1
Over the course of the experiment, a total of 159 fecal samples were assessed by IFA. Overall, positive results were detected in 56 of 86 samples (65%) in the treatment group and in 72 of 73 samples in the control group (98%). When percentages of positive samples were compared between the treatment and control groups (Table 1), statistical significance was detected for the treatment by time interaction (P=0.0006). In the pre-treatment period, there were no differences between the groups for percentage positive fecal samples or cyst scores (Tables 1 and 2). Compared to the untreated kittens, kittens treated with FPP had significantly fewer positive fecal samples in post-treatment periods 1 and 2 and significantly lower cyst scores in post-treatment period 1. One of the five treated kittens was still cyst negative on day 13 post-treatment but resumed cyst shedding after one dose of methylprednisolone acetate. Overall, diarrhea was detected in 6 of 86 samples (6.9%) in the treatment group and 4 of 73 samples (5.5%) in the control group; differences between treated and control cat groups were not detected.
Percentage of fecal samples positive for giardia cysts in untreated kittens and kittens treated with a combination product containing febantel, praziquantel, and pyrantel
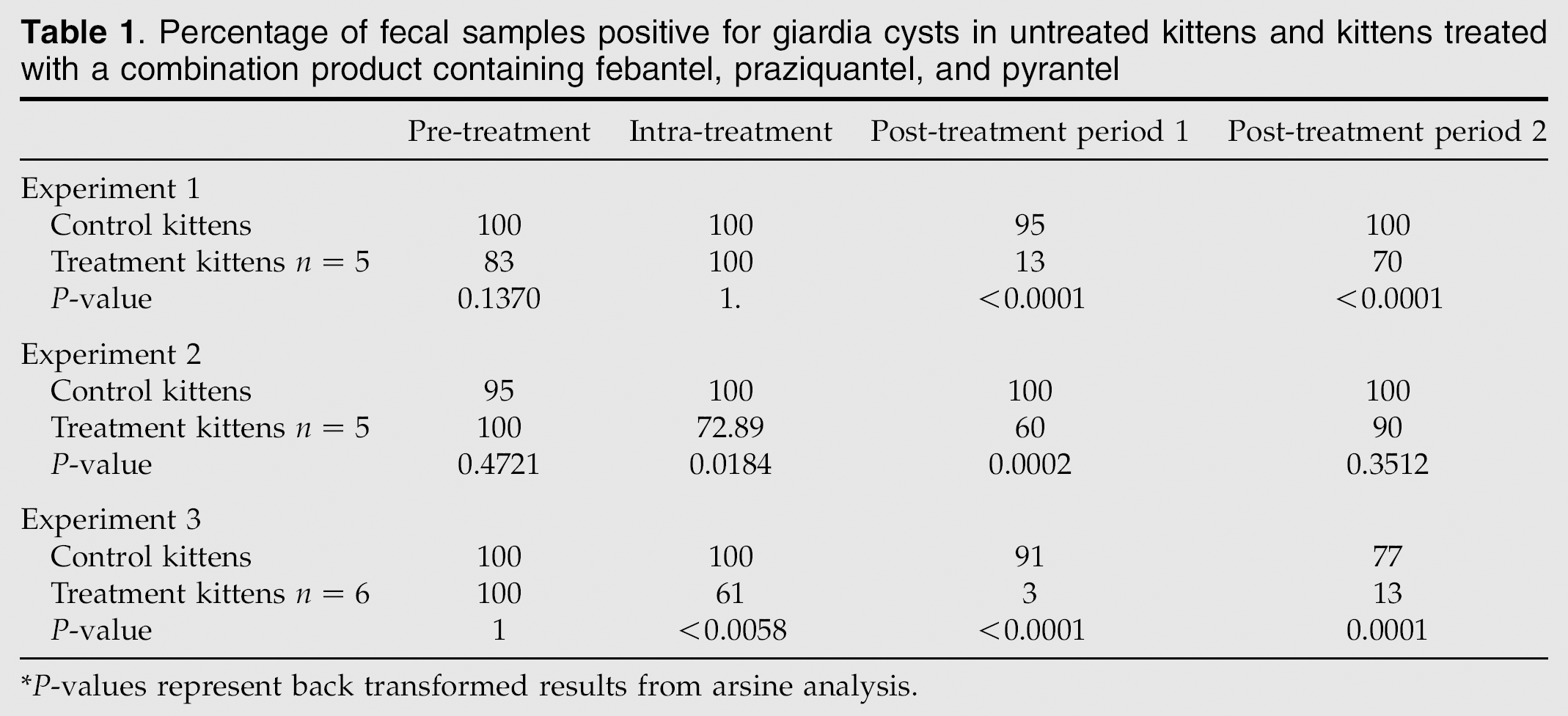
P-values represent back transformed results from arsine analysis.
Cyst score for giardia cysts in untreated kittens and kittens treated with a combination product containing febantel, praziquantel, and pyrantel
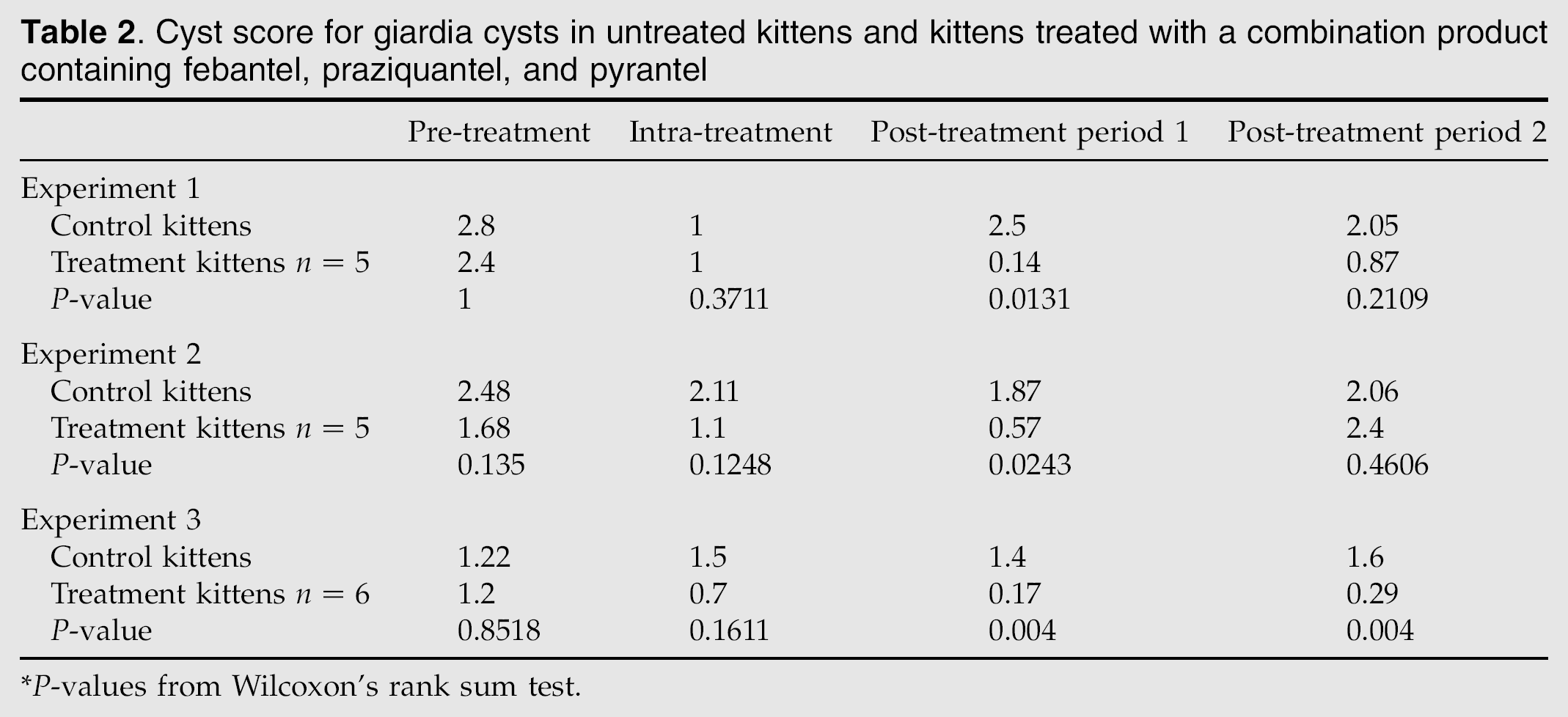
P-values from Wilcoxon's rank sum test.
Experiment 2
Over the course of the experiment, a total of 135 samples were assessed by IFA. Overall, positive results were detected in 61 of 76 samples (80%) in the treatment group and in 58 of 59 samples in the control group (98%). When the percentage of positive samples were compared between the treatment and control groups (Table 1), statistical significance was detected for the treatment by time interaction (P=0.0004). In the pre-treatment period, there were no differences between the groups for percent positive fecal samples or cyst scores (Tables 1 and 2). Compared to untreated kittens, treated kittens had significantly fewer positive fecal samples in the intra-treatment period and post-treatment period 1 and significantly lower cyst scores in post-treatment period 1. None of the kittens became cyst negative in post-treatment periods 1 or 2. Overall, diarrhea was detected in 5 of 76 samples (6.5%) in the treatment group and 5 of 59 samples (8.7%) in the control group; differences between treated and control cat groups were not detected.
Experiment 3
Over the course of the experiment, a total of 170 samples were assessed by IFA. Overall, positive results were detected in 52 of 109 samples (47%) in the treatment group and in 56 of 61 samples in the control group (92%). When percentages of positive samples were compared between the treatment and control groups (Table 1), statistical significance was detected for the treatment by time interaction (P=0.0012). In the pre-treatment period, there were no differences between the groups for the percent positive fecal samples or cyst scores (Tables 1 and 2). Compared to untreated kittens, treated kittens had significantly fewer positive fecal samples in the intra-treatment period and post-treatment periods 1 and 2 and significantly lower mean cyst scores in post-treatment periods 1 and 2. On day 13 post-treatment, five of the six treated kittens remained negative for Giardia species by IFA, therefore were administered methylprednisolone acetate as described. Four kittens remained giardia cyst negative for the duration of the study. Overall, diarrhea was detected in 7 of 109 samples (5.5%) in the treatment group and 0 of 61 samples (0%) in the control group; differences between treated and control cat groups were not detected.
Other than salivation by some kittens, signs of toxicity were not recognized and CBC and serum biochemical panel results were normal after the three experiments.
Discussion
In this study, it would have been optimal for all kittens to have become infected with Giardia species, to have had the same prepatent period and have shown clinical signs of giardiasis. Nevertheless, in the kittens that developed detectable infections, chronic, persistent shedding did occur. During the 7–14 day pre-treatment time periods in each of the three experiments, there was no difference in Giardia species cyst shedding between the treated and control kittens. In addition, the untreated control kittens continued to consistently shed giardia cysts through each of the intra-treatment and post-treatment periods of each experiment. Thus, we believe that the statistical differences detected between treated and control kittens are most likely a treatment effect rather than due to chance. Because of the variable prepatent period and the sequential design of the three experiments, we believe the results should be interpreted cautiously. However, we also believe that the results should be reported and suggest that further experiments assessing the use of FPP for treatment of feline giardiasis be pursued using clinically affected cases.
Treated kittens in experiment 3 were administered higher doses of FPP than treated kittens in experiments 1 and 2. These kittens also had the best intra-treatment and post-treatment results, including failure to detect cyst shedding in four of six treated kittens even after administration of glucocorticoids. While Giardia species infection can spontaneously resolve (Barr 1998), we believe it is more likely the FPP played a role in organism clearance at this dose because each of the control kittens was persistently infected. However, it is possible that the four kittens that became giardia-negative after treatment were still infected but at a level below the sensitivity limit of the IFA and that the glucocorticoid protocol we used was not high enough to induce shedding. We believe that if FPP is studied further for the treatment of feline giardiasis, minimum doses of 56.5 mg/kg, 11.3 mg/kg, and 37.8 mg/kg, respectively, should be used. This dose is similar to that reportedly effective for the treatment of giardiasis in dogs (Barr et al 1998, Barutzki et al 2001). In addition, when fenbendazole was administered to kittens with Giardia and Cryptosporidium species co-infections at 50 mg/kg, PO q24h, for 5 days, Giardia species infection was eliminated in four of eight kittens (Keith et al 2003). The anti-giardia activity of FPP is thought to be from the febantel component, which is metabolized into fenbendazole, oxfendazole and other compounds after oral administration (Barr et al 1998). Administrations of the two formulations of FPP were well tolerated by the kittens; the only side effect that was observed in these kittens was transient salivation after the administration of the tablets. Nevertheless, when FPP was first launched in Australia, there were reports of side effects (transient neurotoxic signs characterized by uncoordinated movements) and it was decided not to continue with FPP, but to launch Drontal (Pyrantel/Praziquantel) for cats. At that time it was believed that the side effects were caused by febantel, while pyrantel is generally not absorbed. However, after launching Drontal there were some neurotoxic signs in cats still recognized, suggesting the toxicity was not related to febantel. Today, after many years of monitoring, there are no correlations to either cats' breed or age. Thus, the reasons for the first findings remain unclear (personal communication, Bayer Animal Health 2005). Diarrhea was uncommon in the kittens in this study; therefore we could not determine whether the administration of the FPP lessened the clinical signs of giardiasis.
The failure of all 24 inoculated kittens to develop detectable patent infections could be from an insufficient Giardia species cyst dose, the administration of a minimally virulent strain, innate resistant of some kittens, specific immunity in some kittens, or failure to detect Giardia species cysts because of low level infection. While there is no standardized Giardia species infection protocol reported for kittens; the infective dose in humans is thought to range from 10 to 1000 cysts (Flaubert 2000). Thus, we assumed, 2×104 cysts of a feline isolate should be sufficient to cause infection in kittens. The giardia cysts were concentrated following the instructions of a published protocol (O'Handley et al 2000), stored at 4°C for a maximum of 10 days, and were inoculated into all kittens at the same time. Thus, each kitten should have received a similar number of viable giardia cysts. We infected the kittens with giardia cysts derived from a naturally infected cat with clinical signs of giardiasis. However, it may be possible that the strain that we administered was not virulent enough to produce infection and clinical signs of giardiasis in all the kittens. Kittens as well as children are thought to be more susceptible to giardia infection than adults (Barr 1998). However, protection against giardiasis can be acquired passively by neonates from immune mothers (Farthing 1990). It is possible that maternal antibodies against Giardia species were still present in some kittens at the time of inoculation, blocking infection. giardia cyst shedding by kittens may fluctuate from undetectable levels to concentrations of >1,000,000 cysts/g of feces and the duration between any two given peaks of cysts excretion in kittens ranged from 2 to 7 days in one study (Kirkpatrick and Farrell 1984). Thus, it is also possible that some of the kittens were previously infected by Giardia species at undetectable levels and so were immune to infection. However, we believe that this hypothesis is unlikely because results were consistently negative in uninfected kittens through the study and the IFA used here is one of the most sensitive tests for the diagnosis of giardiasis. When used with cervine feces, the detection limit of this IFA is 500 cysts/ml of feces (Deng and Cliver 1999). In addition, we also tested one sample from each kitten by a commercially available antigen test (Prospect Giardia Microplate Assay, Alexon Trend, Ramsey, MN) and all the samples tested were negative.
Acknowledgments
The authors would like to thank Jason Eberhardt, Melissa Brewer, and Jennifer Hawley for their help in collecting samples and with animal care. This project was supported by a grant from Bayer Animal Health.
