Abstract
Pyothorax was diagnosed in 27 cats between 1983 and 2002. In 21 (78%) of the cases, pleural fluid culture and/or cytology was consistent with a mixed anaerobic bacterial infection of oropharyngeal origin. In six cases (22%), infection was caused by unusual pathogens or pathogens of non-oropharyngeal origin, including a Mycoplasma species, Cryptococcus gattii, Escherichia coli, Salmonella typhimurium and Staphylococcus aureus. The overall mortality rate was 22%. Treatment was successful in 18 of 19 cases (95%) where closed thoracostomy tubes were inserted. One case resolved only after thoracotomy. Actinomyces species were isolated in three cases and in contrast to dogs where thoracotomy is recommended, they were resolved with tube thoracostomy. Mechanical complications occurred in 58% of the cats with indwelling chest tubes. Probable mechanisms of pleural space infection were identified in 18 cats (67%) including haematogenous infection (n=1), direct inoculation of bacteria into the pleural space (n=1), intrathoracic oesophageal rupture (n=1) and parapneumonic extension of infection (n=15; 56%). Of the latter, perioperative aspiration was suspected in two cats, parasitic migration in two and antecedent upper respiratory tract infection was implicated in seven. Parapneumonic spread of infection after colonisation and invasion of lung tissue by oropharyngeal flora appears to be the most frequent cause of feline anaerobic polymicrobial pyothorax and contests the widespread belief that direct inoculation of pleural cavity by bite wounds is more common.
Pyothorax, or thoracic empyema, describes infection of the pleural space characterised by accumulation of a purulent exudate. Mechanisms of infection include haematogenous or lymphatic spread from a distant site (systemic sepsis), extension from an adjacent structure (bronchopneumonia, parapneumonic spread, oesophageal rupture, mediastinitis or subphrenic infection) or direct inoculation (penetrating trauma, foreign body, thoracocentesis or thoracic surgery). Most cases of feline pyothorax involve polymicrobial infections with obligate and facultatively anaerobic bacteria similar in composition to those found in subcutaneous bite abscesses (Love et al 1979a, 1979b, 1981) and to the bacterial flora of the normal feline oropharynx (Love et al 1982, 1989, 1990, 2000). Feline pyothorax is thus usually the result of contamination of the pleural space with oropharyngeal flora. Organisms involved include Bacteroides species, Fusobacterium species, Peptostreptococcus species, Clostridium species, Actinomyces species, Eubacterium species, Propionibacterium species, Prevotella species, Filifactor villosus, Porphyromonas species, Pasteurella multocida and Streptococcus species. In most cases, the mechanism of pleural space infection is not determined antemortem (Sherding 1979, 1994, Bauer 1986, Demetriou et al 2002, Waddell et al 2002).
The aims of this study were to review cases of feline pyothorax diagnosed at the University of Sydney Veterinary Centre, Sydney (UVCS) between 1983 and 2002, to establish demographic, haematological, cytological and microbiological features of pyothorax, clinical signs, response to treatment and outcome. In addition, predisposing factors and underlying diseases were identified to elucidate the aetiopathogenesis of this disease syndrome in cats.
Materials and methods
Medical records for cases of feline pyothorax, diagnosed by thoracic radiography and/or thoracic ultrasonography, thoracocentesis, cytology and culture of pleural fluid, were obtained through a manual search of the medical records and microbiology reports of the UVCS. A total of 27 cases identified for the period between February 1983 and February 2002 were included in the study. Pyothorax was diagnosed by identifying septic inflammation in cytological preparations of pleural effusion and/or growth of microorganisms from culture of pleural fluid. Organisms were cultured on 5% v/v sheep blood agar (Oxoid Base 2) incubated aerobically and anaerobically in a Gas Pak Anaerobic System (Becton Dickinson: BBL) at 37°C. In some cases where anaerobes were isolated, genus identification was performed using standard anaerobic identification procedures (Holdeman and Moore 1977). Identification and antimicrobial susceptibility testing of anaerobic species was not performed routinely. Criteria used to identify Filifactor villosus were as described by Love et al (1979a). Organisms growing on anaerobic plates only were identified as obligate anaerobic bacteria. In one case where narrow-necked, budding, capsulate yeasts were identified on pleural cytology, organisms were cultured on Sabaroud's dextrose agar, birdseed agar containing antibiotics and subsequently on canavanine–glycine–bromthymol blue agar (Dimech 1990).
The following information was obtained for each case where available: age, sex, breed, patient history, duration of illness before treatment, physical findings, radiographic findings, pleural fluid cytological findings, aerobic and anaerobic culture results, haematological data, feline immunodeficiency virus (FIV)/feline leukaemia virus (FeLV) serostatus, underlying or concurrent disease, treatment and outcome. Pyrexia was defined as rectal temperature >39.2°C, hypothermia as rectal temperature <37.7°C, tachypnoea as respiratory rate >40 breaths per min, tachycardia as heart rate >200 beats per min and bradycardia as heart rate <140 beats per min. Thoracic radiographs were reviewed by a veterinary radiologist (GSA). Comparative statistics were performed using a two-tailed Fisher exact test and χ2 approximation with Yates' correction for continuity, and significance was set at P<0.05.
Results
Clinicopathological findings and thoracic radiology
Pyothorax was diagnosed in 27 cats between 1983 and 2002. The age of affected cats ranged from 2 months to 11 years (mean 5 years, median 5 years). Six cats (22%) were less than 1 year (Fig 1), which was not significantly different from a hospital reference population (P=0.51). No gender or breed predispositions were identified. A male preponderance (17 cats; 63%) was not significant compared with the reference hospital population (P=0.25). Of the male cats, 15 were neutered and two were entire. Of the 10 female cats, seven were neutered and three were entire and less than 6 months of age (Table 1). Historical and clinical findings are presented in Table 2. Partial or complete anorexia were the most common historical findings (81%) followed by dyspnoea (59%) and lethargy or weakness (56%). Dyspnoea (81%) and tachypnoea (78%) were the most common physical findings. Thirteen of the 26 cats (50%) in which rectal temperature was recorded were pyrexic and four (15%) were hypothermic. Approximately one-third of all cats examined were in poor body condition, dehydrated or had muffled heart sounds. Heart rate was recorded in 24 cats; five were tachycardic (21%) and none were bradycardic. Four cats (15%) had clinical signs of upper respiratory tract infection (oculonasal discharge, and/or third eyelid prolapse) at presentation and an additional four (15%) had a history of recent upper respiratory tract infection. Apparent duration of illness before presentation ranged from 1 day to 8 weeks (mean 12 days, median 8 days).
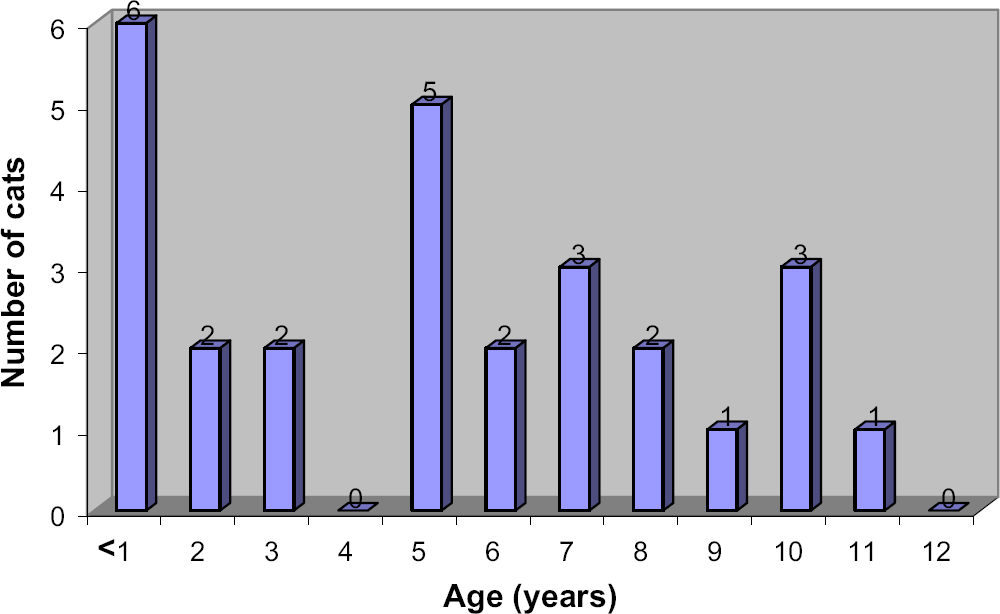
Age distribution of 27 cats with pyothorax.
Predisposing causes of pyothorax and outcome of 18 cats with pyothorax

MN/FN=male/female neutered, DSH=domestic shorthair, NI=not identified, E=euthanased, R=recovered, GA=general anaesthesia, D=died, MGOFA=mixed growth of obligate and facultative anaerobes, UP-L=unilateral pyothorax, left, UP-R=unilateral pyothorax, right, MGOA=mixed growth of obligate anaerobes, URTI=upper respiratory tract infection.
Historical and clinical findings in 27 cats with pyothorax

URTI=upper respiratory tract infection.
Respiratory rate not recorded in four cases.
Temperature not recorded in one case.
Heart rate not recorded in three cases.
Haematology was performed at presentation in 11 cases. Eight cats had a neutrophilic leukocytosis (mean leukocyte count 29×109/l, median 26×109/l, range 5–39×109/l; reference range 8–14×109/l), of which six had left shifts (mean band neutrophil count 2.4×109/l, median 1.4×109/l, range 0–8.8×109/l; reference range 0–0.42×109/l) and four also had a monocytosis (range 0.7–3.5×109/l; reference range 0.08–0.6×109/l). All three cats in which neutrophilic leukocytosis was not present had increased circulating band neutrophils (range 0.5–1×109/l). Eosinophilia (3.1×109/l) was present in one cat (cat 18) that had concurrent Aelurostrongylus abstrusus infection (Table 1). Two cats were anaemic, one with non-regenerative anaemia (PCV 0.23 l/l, absolute reticulocyte count 0) and the other with a regenerative anaemia (PCV 0.21 l/l, absolute reticulocyte count 174×109/l). Serological tests for FIV (Witness; AGEN) and FeLV (Synbiotics; ViraCHEK) were performed in nine cats. One cat with concurrent cryptococcal rhinitis (case 9) was FIV-positive and one kitten (case 25) was FeLV-positive (Table 1).
Thoracic radiographs were obtained in 25 cases. Nineteen cats (76%) had bilateral pleural effusion, two (8%) had unilateral right pleural effusion and four (16%) had unilateral left pleural effusion at presentation. One unilateral effusion became bilateral after 4 days. Radiographic evidence of concurrent pneumonia was present in 10 cats (40%, Table 1) including three with upper respiratory tract infections, two with suspected pulmonary parasitic migration, one with suspected perioperative aspiration and four with no identified predisposing cause. Findings included diffuse and focal alveolar infiltrates, air bronchograms within consolidated lung lobes, bronchointerstitial disease and diffuse interstitial disease. In all cases where pneumonia was identified, pulmonary changes were present in at least two sets of radiographs taken on more than one occasion post-pleural drainage. Atelectasis was documented in three cats. In four cases it was not possible to distinguish between recoil atelectasis, lobar consolidation and encapsulated pleural fluid.
Pleural fluid cytology and culture
Gram- and Diff Quik®-(Lab Aids Pty Ltd, Narrabeen NSW) stained smears prepared from pleural fluid were examined in all cases. In 21 cats (78%) cytology was suggestive of polymicrobial anaerobic bacterial infection, characterised by a large population of predominantly degenerate, polymorphic inflammatory cells, a small proportion of mononuclear cells and large numbers of pleomorphic, intracellular and/or extracellular bacteria. Cell types less commonly identified included erythrocytes, mesothelial cells and epithelial cells. Low numbers of eosinophils were present in the pleural fluid of one cat with concurrent aelurostrongylosis (case 18). Culture results were consistent with cytological findings except in one cat (case 21) treated with antibiotics before culture, from which no bacteria were isolated and in two cats from which pure growths of an obligate anaerobe (Gram-positive cocci or pleomorphic Gram-positive bacilli; case 13) and a Pasteurella species (case 27) were obtained. In these two cats a technical, laboratory or transport problem was suspected to account for the disparity between culture results and cytology. Pleural fluid from case 7 was not cultured and anaerobic culture was not performed for case 2. In all other cases both anaerobic and aerobic cultures were performed routinely.
Unusual pathogens including a Mycoplasma species (n=1), Salmonella typhimurium (n=1), Cryptococcus gattii (n=1), Staphylococcus aureus (n=1) and Escherichia coli (n=2) were isolated from six cats of which four were less than 6 months of age. In two cats no bacteria were observed in pleural fluid (cases 10 and 17). In case 10, inflammatory cells were predominantly small lymphocytes with low numbers of non-degenerate neutrophils and mononuclear cells; many capsulate, narrow-necked budding yeasts were seen and Cryptococcus gattii was subsequently cultured. In case 17 numerous non-degenerate neutrophils were present and a Mycoplasma species was cultured.
In four cats a single population of bacteria was identified on cytology of pleural fluid; short Gram-negative bacilli were seen in three cats (cases 9, 18 and 25) and Gram-positive cocci in the other (case 19). Subsequent culture yielded S typhimurium (in one cat with concurrent lungworm infection; case 18), E coli (in a cat with concurrent cryptococcal rhinitis and FIV infection; case 9) and E coli in an FeLV-infected kitten with septic E coli arthritis of the hock (case 25). In case 19, culture yielded a heavy growth of S aureus and a lighter growth of a facultative Gram-negative coccobacillus.
Treatment and outcome
Of the 27 cats, five cats died and one was euthanased because of severe concurrent congenital cardiac disease (Table 1). One kitten with FeLV infection died shortly after presentation (case 25). Two cats died in hospital prior to insertion of thoracic drains (cases 2 and 10) and two died during general anaesthesia for tube thoracostomy (cases 4 and 19). The mortality rate for cats >1 year (3/21 cats, 14%) was lower than for cats <1 year (3/6 cats, 50%) but the difference was not significant (P=0.1).
All 21 cats (78%) successfully treated were given intravenous fluids and antimicrobial therapy. Nineteen of these were treated using closed tube thoracostomy. Two cats were treated with antimicrobial therapy alone after initial thoracocentesis yielded only a small volume of pleural fluid. Of the cats in which thoracostomy tubes were successfully inserted, treatment was successful in 18 of 19 (95%) cases. In one cat only (case 23) was thoracotomy and debridement necessary for resolution of infection. Bilateral thoracic drains were inserted in 16 cats while unilateral thoracic drains were placed in three cats with unilateral effusions at presentation. Duration of thoracic drainage in cats ranged from 3 to 15 days (mean 7 days, median 6 days). Paediatric thoracic trocar catheters ranging in size from 10 to 18 Ga were inserted under general anaesthesia in 18 cats and under local anaesthesia in one moribund cat with concurrent paracetamol toxicity.
Pleural cavities were lavaged extensively at the time of drain insertion with warmed physiologic sterile saline (PSS). Thereafter, twice daily aspiration and lavage was performed using 40–250 ml of PSS or Hartmann's solution. In adult cats a total volume of 200 ml, instilled in 50–100 ml aliquots, was typically used for pleural cavity lavage twice daily. One cat became hypokalaemic (potassium 2.3 mmol/l) after twice daily pleural lavage with 250 ml of PSS. Mechanical complications associated with closed tube thoracostomy included failure of drainage due to tube kinking or poor positioning in four cats. Two of these developed subcutaneous oedema associated with leakage of lavage fluid and one developed a subcutaneous abscess at the site of tube insertion. There was radiographic evidence of poor thoracic drain positioning of one drain (too dorsal, cranial or caudal within the thorax or kinked) in a further seven cats although clinical complications were not reported in these cases. In total, mechanical drain complications occurred in 58% of the cats with indwelling chest tubes.
Of the 21 cats successfully treated, 19 had polymicrobial anaerobic bacterial infections, one (case 9) had a pure growth of E coli and one (case 18) had a pure growth of S typhimurium. Antimicrobial susceptibility testing was performed in cases 9 (E coli) and 18 (S typhimurium). Based on the results, these cats were treated with amoxicillin–clavulanate and enrofloxacin, respectively. Of the other 19 cats, selection of antimicrobial therapy was empiric and at the discretion of the prescribing clinician. The majority of cats were treated with benzyl penicillin intravenously (IV) (11 cats) at 30 mg/kg every 6–8 h or with amoxicillin–clavulanate subcutaneously (SC) at 12.5–20 mg/kg every 12 h (n=5). Oral antibiotics were prescribed either when cats resumed eating in hospital or when they were discharged from hospital and included amoxicillin–clavulanate (n=9), phenoxymethylpenicillin (n=2), clindamycin (n=2), or combination therapy with amoxicillin–clavulanate and clindamycin (n=3), amoxicillin–clavulanate and enrofloxacin (n=1), amoxicillin–clavulanate and metronidazole (n=1) or clindamycin and enrofloxacin (n=1). Duration of oral antimicrobial therapy ranged from 1 to 12 weeks (mean 5 weeks, median 4 weeks).
Follow-up thoracic radiographs were taken at recheck examinations in 14 of the 21 surviving cats (67%) from 1 to 6 weeks after diagnosis. In one cat (case 23) there was recurrence of pyothorax in the right hemithorax 7 days after discharge from hospital. An exploratory thoracotomy was performed and abscessed areas of lung were decorticated and debrided and bilateral thoracostomy tubes were placed; the infection resolved without further complication. In another cat (case 27) radiographs taken during treatment and 2 weeks after discharge from hospital showed pneumothorax and complete atelectasis of the left lung. It was considered unlikely that the lung would expand after drainage of the pneumothorax because of residual pleural fibrosis and no further treatment was attempted. In the remaining 12 cats there was no evidence of recurrence of pyothorax. In total, 21 cats (78%) were successfully treated with no residual clinical signs of disease at recheck examinations from 2 to 6 weeks after diagnosis. Long-term survival data were not available.
Aetiology and predisposing disease conditions
Probable mechanisms of pleural space infection were identified in 18 cats (67%; Table 3). Haematogenous spread of infection from a septic hock (E coli) occurred in one kitten (4%) with FeLV infection (case 25, Table 1). Direct inoculation of bacteria into the pleural cavity occurred in one cat with unilateral left-sided pyothorax (case 2); a thoracic-wall puncture wound was identified at necropsy.
Probable mechanisms of pleural space infection in 27 cats with pyothorax

Extension of infection from an adjacent intrathoracic structure was the most likely mechanism of pleural space infection in 16 cats (59%). Intrathoracic oesophageal perforation occurred in case 19. The kitten had a history of acute haematemesis and gagging followed by dyspnoea and cervical subcutaneous emphysema. At necropsy there was evidence of mural oesophageal foreign plant debris, oesophageal ulceration and perforation leading to sclerosing pleuritis and a unilateral, left pyothorax. The pleural cavity also contained a large amount of hair and plant material. Parapneumonic extension of infection to the pleural cavity was suspected in the remaining 15 cats (56%). Perioperative aspiration of oropharyngeal flora resulting in bronchopneumonia and pyothorax was suspected in two cats; case 15 developed pyothorax 6 days after being anaesthetised for an ultrasonic tooth scaling procedure and case 27 was presented 7 days after general anaesthesia for castration. In both cats, pleural fluid cytology indicated polymicrobial anaerobic infections, consistent with aspiration rather than haematogenous infection. Bronchopneumonia and pyothorax secondary to parasitic migration were suspected in two cats; one with Aelurostrongylus abstrusus infection and concurrent salmonellosis (case 18), the other with Toxocara cati and pleural fluid cytology consistent with polymicrobial anaerobic infection (case 21). Eight cats (30%) had a recent history or clinical signs of upper respiratory tract infection (Table 1). Pyothorax was suspected as a sequela to bronchopneumonia, after colonisation of the lower respiratory tract with oropharyngeal flora, in seven of these cats. Case 17 developed pneumonia and pyothorax due to a Mycoplasma species infection 2 weeks after upper respiratory tract infection (Malik et al 1991). Necropsy findings confirmed bronchopneumonic extension of infection. In six other cats, including three with radiographic signs of pneumonia (Table 1), infections of facultative anaerobes and/or obligate anaerobes occurred, of likely oropharyngeal origin. Two cats in this cohort had severe concurrent periodontal disease. Upper respiratory signs were recorded in one other case (case 9), which was FIV-positive and from which E coli was isolated. However, the mechanism of pleural space infection was not identified since upper respiratory signs could have been due to concurrent cryptococcal rhinitis, which had been responding to therapy, and E coli is an uncommon isolate from pyothorax. The cat had a cryptococcal antigen titre of 8. In a further four cats with mixed anaerobic infections, there was radiographic evidence of concurrent pneumonia. Pneumonia was confirmed at thoracotomy in one cat (case 23) and another (case 7) was diagnosed with severe inflammatory, corticosteroid-responsive bronchial disease 3 months after resolution of pyothorax. In nine cats (33%) no mechanism of pleural space infection was identified.
Discussion
No breed or gender predisposition to develop pyothorax was identified in this study and none has been identified previously. The mean age of affected cats in our study (5 years) was similar to means of 4–6 years reported previously (Davies and Forrester 1996, Demetriou et al 2002, Waddell et al 2002). Pyothorax is more common in younger cats although cats of any age may be affected. The historical and clinical findings of cats with pyothorax have been well characterised (Sherding 1994, Waddell et al 2002). However, little emphasis has been placed on a prior history of upper respiratory tract infection. In an early study of feline pyothorax, upper respiratory tract infection was recognised as a predisposing event in three of 20 cases (Jonas 1983). In the present study, 26% of the cats with pyothorax involving polymicrobial anaerobic infections or a Mycoplasma species, had a history or clinical signs suggestive of antecedent upper respiratory tract infection, likely viral in origin. Mycoplasma species have been identified as a cause of infectious bronchopulmonary disease in both kittens and cats (Bart et al 2000, Foster et al 2004). Pyothorax caused by a Mycoplasma species and a facultative anaerobe (Arcanobacterium pyogenes) has been documented in a 1-month-old kitten (Gulbahar and Gurturk 2002).
Coughing was a common historical finding and was present in almost a third of the cats; in this setting, coughing is indicative of pleuritis and/or concurrent pneumonia (Sherding 1994). Fifty percent of the cats in our study were pyrexic but the true incidence may be higher as some cats had received prior antimicrobial therapy from referring clinicians. Hypothermia, which occurred in 15% of the cats, is indicative of severe sepsis particularly when accompanied by bradycardia (Brady et al 2000). No cats were bradycardic. However, in the largest retrospective study of 80 cats with pyothorax, non-survivors had significantly lower heart rates than survivors, although not all cats with bradycardia were hypothermic (Waddell et al 2002).
Although the duration of clinical signs before diagnosis was typically 1–2 weeks, the range of up to 8 weeks demonstrates that pyothorax can progress as an insidious infection before cats are presented for investigation (Davies and Forrester 1996, Demetriou et al 2002). Dyspnoea was the most common clinical finding but it can be surprisingly subtle and was not observed by 40% of the owners.
Haematological data were available for a small number of cases and, as expected for pyogenic infections, neutrophilic leukocytosis with a left shift was the most common finding. Serum biochemical data were not available for most cats. The most common abnormalities reported in pyothorax are hypoalbuminaemia, hyperglobulinaemia, hypo- or hyperglycaemia, hyponatraemia, hypochloraemia, hypocalcaemia and mild elevations of aspartate aminotransferase (AST) and bilirubin (Demetriou et al 2002, Waddell et al 2002). Hypoalbuminaemia is a common finding in sepsis, attributed to increased vascular permeability and decreased hepatic synthesis due to a shift towards synthesis of acute phase reactants (Brady et al 2000). In one study, cholesterol concentrations were significantly lower in survivors than in non-survivors, although the significance of this finding was unclear (Waddell et al 2002).
The direct inoculation of oral flora into the thorax from cat bite wounds is regarded as a common route of infection of the pleural space in cats, particularly in young, free-roaming, entire male cats. This widely held belief is based on the findings of an earlier report (Jonas 1983). However, it may be that, given the current demographic in Australia of more neutered cats with restricted territories, this route of infection is now less common. In the present study, thoracic puncture wounds were identified in only one case (4%). Likewise, in a large retrospective study of 80 cats with pyothorax, recent history of a bite or other external wound was documented in only 14.5% of cats. Of 25 cats necropsied in that study, thoracic puncture wounds were identified in only four cats (Waddell et al 2002). Also, sexually intact males were not over-represented compared to a control population of 212 cats. Despite this evidence, it was postulated that the most likely route of transmission of oral cavity flora to the pleural space of other cats is by bite wounds to the thorax or environmental contamination from penetrating thoracic injuries. The same study showed that cats with pyothorax are 3.8 times as likely to have come from multi-cat households compared to control cats (Waddell et al 2002). The authors proposed that cats from multi-cat households are more likely to incur thoracic bite wounds than control cats. However, behavioural studies do not support the notion of this type of aggression in stable multi-cat households (Crowell-Davis et al 2004) and an alternative explanation is that cats from a multi-cat household are at greater risk of developing viral upper respiratory tract infection. In addition, if pyothorax occurred secondary to environmental contamination of thoracic wounds, a higher isolation rate of saprophytic bacteria such as Nocardia species, Pseudomonas species and mycobacteria would be expected.
We consider that the most common mechanism of infection in feline polymicrobial pyothorax is aspiration of oropharyngeal flora and subsequent colonisation of the lower respiratory tract. Direct extension of infection from the bronchi and lungs subsequently results in pyothorax. This mechanism of infection is the most common cause of human anaerobic pyothorax and equine pleuropneumonia (Bartlett 1993, Racklyeft et al 2000). In human anaerobic lung infections, tissue necrosis results in abscess formation and/or bronchopleural fistula with subsequent extension to the pleural space (Bartlett 1993). Pleuropneumonia is a common sequel to transportation in horses. Mucociliary clearance of lower respiratory secretions is impaired when horses are restrained with their heads elevated; this results in accumulation of aspirated oropharyngeal flora and increased risk of pleuropneumonia (Racklyeft et al 2000). Viral upper respiratory tract infection may also temporarily impair the mucociliary escalator in cats, humans and horses (Carson et al 1985, Willoughby et al 1992, Gaskell et al 2004), predisposing them to pleuropneumonia.
In our study, pleural space infection secondary to aspiration of oral flora, was most likely in 14 of 18 (78%) cats in which probable mechanisms of pleural space infection were identified, including two with likely perioperative aspiration of oropharyngeal flora, four with polymicrobial anaerobic infections and radiographic signs of pneumonia, one with T cati infection, six with suspected viral upper respiratory tract infection and one with severe Mycoplasma species upper respiratory infection. Larval migrans may have caused pulmonary damage, a lowered oxidation/reduction potential and conditions favourable for proliferation of oropharyngeal anaerobes and pyothorax in case 21, which vomited a heavy burden of T cati nematodes and had radiographic evidence of pneumonia.
In necropsies of cats with pyothorax, pneumonia or focal pulmonary abscessation was identified in seven of 15 (46%) of cats in one study (Davies and Forrester 1996) and diffuse or focal pulmonary lesions were the most common findings in another (Waddell et al 2002). Such lesions are not compatible with aetiology of penetrating trauma and instead support a pathomechanism of parapneumonic spread. Detection of the route of pleural space infection antemortem can be difficult. In a recent study no initiating aetiological factors were identified (Demetriou et al 2002). Our findings emphasise the importance of obtaining both a thorough history to aid in identification of predisposing factors, and post-drainage imaging for the detection of pulmonary disease. In radiographs taken 24 h after thoracostomy tube placement, pulmonary densities are more likely to be referable to pulmonary disease rather than recoil atelectasis or loculated effusion (Bauer 1986).
Non-oropharyngeal pathogens were isolated from five cats. Fungal aetiologies for feline pyothorax are rare and include, Cryptococcus species, Candida albicans and Blastomyces dermatitidis (McCaw et al 1984, Sherding 1994). It is likely that cryptococcal infection in case 10 began as a nasal cavity infection with subsequent lower respiratory tract involvement (Malik et al 2001). C gattii is a commensal of certain eucalypts in regions including Australia and California. Infection is typically restricted to the nasal cavity and adjacent structures. Pulmonary involvement in cats has been reported but is rare (Barrs et al 2000). In our study, non-oropharyngeal pathogens were more likely to be isolated from kittens with pyothorax. This may be due to increased susceptibility to other infectious conditions such as FeLV infection, ascarids or lungworm, compared with adult cats. S typhimurium, isolated from the pleural fluid of case 18, was most likely a gastrointestinal contaminant adhered to the surface of or excreted by migrating Aelurostrongylus abstrusus larvae, or alternatively was localised in lung tissue following bacteraemia. This case has been documented in detail elsewhere (Barrs et al 1999). Other non-oropharyngeal pathogens that may cause feline pyothorax but were not isolated in this study include Rhodococcus equi, Nocardia species and other enteric and non-enteric Gram-negative organisms (Klebsiella species, Proteus species, Pseudomonas species) (Sherding 1994).
Two cats with low volume effusions were successfully treated by thoracocentesis and antimicrobial therapy alone, without indwelling drain placement. However, mortality rates of 42–80% have been reported in cats with pyothorax when pleural drainage was by repeated thoracocenteses (Bauer 1986). We do not recommend this method of treatment unless euthanasia is the only other option or unless effusion is of small volume and pneumonia is the primary problem. In this study, the success rate of managing pyothorax using closed tube thoracostomy was very high (95%) and was simple and cost effective. Continuous water seal suction is not necessary for effective pleural drainage in most cases of feline pyothorax. In addition, water seal chest drainage units are more expensive and require continuous monitoring, as leakage between the pleural cavity and water seal can be fatal. Mortality may be reduced by use of pre-anaesthetic thoracocentesis before tube thoracostomy and reducing the time between diagnosis and thoracic drain insertion. Hypokalaemia occurred in one cat when 50 ml/kg of PSS was used to lavage the thoracic cavity. Use of a lavage solution containing potassium (eg, Hartmann's solution) or using volumes of lavage of fluid not exceeding 25 ml/kg may have prevented this complication (Sherding 1994, Greene 1998).
There was a high mechanical complication rate with thoracostomy tubes although resolution of infection occurred with this method of treatment in all but one case. Where pyothorax is bilateral, we recommend bilateral thoracostomy tube placement. Bilateral chest tubes are more likely to provide effective drainage in cases of persistent loculation of fluid or where the mediastinum is complete (Sherding 1994, Bauer and Woodfield 1995). Furthermore, if complications arise with the use of one tube it may be removed and drainage continued via the remaining tube. If unilateral tube thoracostomy is considered, thoracic radiographs should be taken immediately after drain placement and the removal of as much fluid as possible through the tube. If there is minimal residual effusion, one tube may be sufficient for treatment. Otherwise, a second thoracostomy tube should be placed immediately. The median duration of thoracic drainage was 6 days, similar to the findings of others (Demetriou et al 2002, Waddell et al 2002).
Thoracotomy was required in only one of 21 treated cats (5%). Similarly, five of 53 (9%) treated cats in another study required thoracotomy when medical management failed and surgery was curative in all cases (Waddell et al 2002). Although these case numbers are small, thoracotomy appears to offer a favourable prognosis for cats in the treatment of pyothorax refractory to medical management or where imaging studies reveal intrathoracic masses including pulmonary abscesses. In dogs, thoracotomy is recommended if Actinomyces species is isolated from pleural fluid, because of poor outcomes associated with medical therapy alone (Rooney and Monnet 2002). In this study Actinomyces species were identified in three cases of polymicrobial pyothorax and all resolved without thoracotomy. In both dogs and cats Actinomyces species are members of normal oropharyngeal flora (Love et al 1990, Edwards 1998). Pyothorax in dogs has often been associated with inhalation or penetration of grass awns or florets that are contaminated with this bacterium (Edwards 1998). In cats, aspiration of Actinomyces species in combination with other oropharyngeal flora is less likely to be associated with grass awn foreign bodies.
Because of time constraints, technical complexity and the low incidence of β-lactamase producing organisms, antimicrobial susceptibility testing of obligate anaerobic bacteria using agar dilution or broth microdilution was not routinely performed and selection of antimicrobials was empiric. The development of the E-test method for susceptibility testing of all types of anaerobic bacteria offers a simple, rapid alternative and results for rapidly growing organisms, such as the B fragilis group and Filifactor villosus, are available after overnight incubation (Citron et al 1991, Love and Wigney 1997).
Antimicrobials most commonly used were high doses of synthetic penicillin (benzyl penicillin) or amoxicillin–clavulanate administered parenterally and when clinical condition improved, oral amoxicillin–clavulanate. Penicillin and its derivatives are reliably effective against non-β-lactamase producing obligate anaerobes (Jang et al 1997). Antibiotics effective against most β-lactamase producing anaerobes such as from the Bacteroides fragilis group include amoxicillin–clavulanate, ticarcillin–clavulanate and metronidazole. In a recent study of obligate anaerobes from dogs and cats, all isolates were susceptible to amoxicillin–clavulanate and 98% were susceptible to metronidazole (Jang et al 1997). Only 71% of Bacteroides isolates were susceptible to ampicillin, and only 83% were susceptible to clindamycin (Jang et al 1997). But in feline pyothorax, Bacteroides tectum is a more common isolate than Bacteroides fragilis and β-lactamase producing strains of this species are uncommon (Love et al 1989, Love and Wigney 1997). Also, polymicrobial infections are synergistic and concurrent facultative bacteria scavenge oxygen thus creating a more suitable environment for proliferation of anaerobes. Therefore, the combination of drainage and antibiotics effective against only non-β-lactamase producing anaerobes and facultative bacteria is often adequate.
It is often recommended that empiric therapy for the Gram-negative facultative bacterial component of pyothorax should include either an aminoglycoside (gentamicin or amikacin) or a fluoroquinolone (Hawkins and Fossum 2000, Walker et al 2000). Such recommendations have arisen erroneously from considering canine and feline pyothorax as a single entity. In canine, pyothorax enterobacteriaceae, especially E coli, are isolated relatively commonly (Walker et al 2000, Demetriou et al 2002, Rooney and Monnet 2002). In feline pyothorax enterobacteriaceae are uncommon, with E coli being isolated in 7% of the cases in this study and in 0–5% of cases in other studies (Love et al 1982, Jonas 1983, Walker et al 2000, Demetriou et al 2002). The most common facultative Gram-negative rod likely to be isolated from cats with pyothorax is Pasteurella species (Love et al 1982). Cephalexin and other first generation cephalosporins have poor activity against Pasteurella species and have only intermediate activity against many anaerobes (Dow et al 1986, Goldstein et al 1988). Their use in empiric therapy of feline pyothorax is therefore not recommended. Pasteurella species are susceptible to penicillin and its derivatives as well as to quinolones and aminoglycosides. Based on these findings, the addition of a quinolone or an aminoglycoside is unnecessary for initial empiric therapy. Furthermore, aminoglycosides are nephrotoxic, enrofloxacin is potentially retinotoxic and many isolates of E coli are not susceptible to quinolones. Of 50 E coli isolates obtained from cats examined at a veterinary teaching hospital, 75% were susceptible to amoxicillin–clavulanate whilst only 55% were susceptible to enrofloxacin (Walker et al 2000). Adjunctive antimicrobial therapy can be administered if indicated by the results of antimicrobial susceptibility testing of bacteria isolated from pleural fluid, or if Gram-negative rods only are seen in smears of pleural fluid.
The median duration of oral antimicrobial therapy in this survey was 4 weeks, similar to current recommendations of 4–6 weeks (Sherding 1994, Hawkins and Fossum 2000). In general, treatment of anaerobic infections requires high doses of antimicrobials administered for extended periods of time because these infections are associated with devitalised tissue and tend to relapse if therapy is discontinued prematurely.
Pyothorax in cats is most often caused by obligate and facultative anaerobes of oropharyngeal origin. However, in 22% of cats unusual pathogens or pathogens of non-oropharyngeal origin were found, including a Mycoplasma species, C gattii, E coli, S typhimurium and S aureus, emphasising the necessity for pleural fluid cytology and culture. This would appear to be the first clinical paper that has emphasised parapneumonic spread of infection after colonisation and invasion of lung tissue by oropharyngeal anaerobes as the apparent most frequent cause of feline pyothorax and contests the widespread belief that direct inoculation of pleural cavity by bite wounds is more common. Clinicians should consider routine antimicrobial prophylaxis in cats with viral upper respiratory tract infections and after dental procedures under general anaesthesia, utilising antibiotics such as amoxicillin–clavulanate or doxycycline. Closed tube thoracostomy, twice daily aspiration and lavage and antimicrobial therapy resulted in complete resolution of disease in 95% of cats in which thoracic drains were successfully placed.
Footnotes
Acknowledgement
This manuscript is dedicated to the memory of Daria Love, who was instrumental in its inception. The authors thank all clinicians at the University Veterinary Centre, Sydney who contributed cases. They also thank Denise Wigney, Chris Oxenford and Graeme Bailey for microbiological data.
