Abstract
A 10-year-old male castrated domestic shorthair cat was evaluated for coughing and lethargy. Thoracic radiographs revealed a soft tissue lung mass and diffuse peribronchial infiltrates. Bronchoscopy was performed and Capnocytophaga cynodegmi was cultured from bilateral bronchoalveolar lavage samples. Clinical signs and bacterial colonization resolved following treatment with enrofloxacin. A lung lobectomy was performed to remove the lung mass, which was diagnosed as pulmonary carcinoma. C cynodegmi is most frequently isolated from localized wound and corneal infections in humans. Specialized growth characteristics of C cynodegmi may result in low sensitivity for bacterial culture. To the authors' knowledge, this case represents the first report of C cynodegmi infection in a veterinary patient and only the second case in human or veterinary medicine where the organism has been isolated from a bronchoalveolar lavage sample. Based on this report, Capnocytophaga species should be considered as potential opportunistic pathogens.
A 10-year-old, male castrated domestic shorthair cat weighing 9 kg was evaluated at the University of California Veterinary Medical Teaching Hospital (UCD-VMTH) for a 2-month history of coughing and lethargy. Thoracic radiographs taken 1 month before admission revealed a consolidated right middle lung lobe. Treatment with enrofloxacin and amoxicillin–clavulanate (doses unknown, PO) was initiated. Due to presumed antibiotic-related vomiting, antibiotics were changed at 1-week intervals to enrofloxacin and amoxicillin (doses unknown, PO) and then enrofloxacin and metronidazole (doses unknown, PO). Ten days before admission, recheck radiographs revealed resolution of the consolidated lung and the appearance of a right caudal–dorsal lung field mass. The owners reported that the cat displayed decreased frequency but persistent coughing while on antibiotics. Antibiotics were discontinued for 7 days before admission and the cat was referred for further evaluation of the lung mass.
At the time of presentation, the cat was mildly febrile (rectal temperature, 39.2 °C) and normal bronchovesicular sounds were ausculted in all lung fields. Complete blood count (CBC), serum biochemistry, urinalysis, abdominal ultrasound and heartworm antibody serology did not reveal any abnormalities. Thoracic radiographs were similar to those taken 10-days previously with a discrete (2.4×4.2 cm) soft tissue mass visible within the caudal aspect of the right caudal lung lobe and diffuse peribronchial infiltrates throughout all lung fields (Fig 1).

Right lateral chest radiograph revealing a discrete soft tissue mass within caudal lung lobe (large arrows) and diffuse peribronchial infiltrates throughout all lung lobes.
Abnormalities noted on thoracic radiographs were investigated by bronchoscopy and bronchoalveolar lavage (BAL). Bronchoscopy with a 2.5 mm outer diameter×100 cm fiberoptic endoscope (Karl Storz Veterinary Endoscopy, Goleta, CA) revealed an intraluminal mass in the right caudal dorsal bronchus and mild to moderately edematous and hyperemic mucosa throughout all airways. Bronchoalveolar lavage was performed through the endoscope in the region of the intraluminal mass and was also performed at a separate site in the left cranial lobe to investigate the etiology of the diffuse peribronchial infiltrate. The external sheath and sampling channel of the endoscope were cleansed with sterile saline before and between lavages. Lavage from the mass region on the right side revealed increased cell numbers (600 cells/μl, normal 200–300/μl) and moderate to marked neutrophilic inflammation (55%) with normal percentages of lymphocytes (3%), eosinophils (11%), and macrophages (31%). Mildly increased cellularity (400 cells/μl) with normal percentages of neutrophils (7%), lymphocytes (10%), eosinophils (22%), and macrophages (61%) was evident in the left cranial lobe. No organisms were noted on cytology from either site, although the cytology from the right side was reported as suspicious for sepsis based on cellular characteristics. A sample of the mass lesion was retrieved during the lavage procedure of bronchoscopy and histologic examination of the mass lesion revealed severe, neutrophilic inflammation with a single raft of atypical epithelial cells.
Aliquots of BAL fluid from left and right lung lavages were plated separately onto both 5% sheep blood agar and MacConkeys agar and incubated at 37 °C in air with 5% CO2. Aliquots were also plated on to pre-reduced Brucella plates and incubated under anaerobic conditions at 37 °C. Heavy growth of Capnocytophaga cynodegmi was obtained from the right side with light growth from the left. The organism was a catalase and oxidase positive, slightly branching Gram-negative rod. Indole, urease and hydrogen sulfide were negative. The organism hydrolyzed esculin but did not hydrolize gelatin or reduce nitrate, and did not assimilate glucose, mannitol, arabinose, maltose, malate and galactose. A very light growth of mixed Gram-positive and -negative anaerobic bacteria was also obtained from the right. No Mycoplasma species organisms were isolated from either BAL sample; however, due to the difficulties in isolating Mycoplasma species, an infection cannot be ruled out. C cynodegmi was considered significant largely because of its presence bilaterally, with heavy growth from the right lung. The anaerobic bacteria were considered likely contaminants because of the very light growth. It is unusual for infectious bronchitis that there was such extensive growth of C cynodegmi from the right in the absence of intracellular organisms detected on cytology; however, the BAL fluid cytology was interpreted as possibly septic based on cellular characteristics and the results may have been influenced by recent antibiotic administration.
Pending culture results, the patient had been discharged on enrofloxacin (2.5 mg/kg, PO, q12hr) and this was continued for 4 weeks based on good clinical response. Thirty days later (3 days after finishing enrofloxacin), the owner reported a marked decrease in cough frequency with an improvement in appetite and activity level. Results of CBC and serum biochemical analysis were unremarkable. Thoracic radiographs revealed progressive enlargement of the right caudal lung lobe mass (2.8×5.2 cm) with unchanged diffuse peribronchial infiltrates. Bronchoscopy was repeated for follow-up bacterial cultures to assess efficacy of therapy for Capnocytophaga cynodegmi bronchitis and to obtain a minimally invasive biopsy of the mass lesion, prior to definitive surgery.
Bronchoscopy revealed persistent, diffuse, mild to moderately edematous and hyperemic airways, and the intraluminal mass was again noted. A diagnostic biopsy of the mass lesion could not be obtained because of the small diameter (1.8 mm) of the biopsy forceps. Cytology of BAL fluid from four separate sites within the lung revealed normal cell counts (100–300 cells/μl) with mild–moderate neutrophilic inflammation (11, 13, 26, and 72% neutrophils; the 72% neutrophil count was likely associated with iatrogenically induced hemorrhage). Bacterial culture of BAL fluid from the right caudal lung lobe distant to the mass lesion had light growth of actinomyces-like species, while culture of BAL of the segment containing the mass lesion had very light growth of Pasteurella species (two colonies) and coagulase negative Staphylococcus species (one colony). The left caudal lung lobe BAL grew a very light growth of Streptococcus viridans sensitive to all antibiotics tested except enrofloxacin. No BAL site grew Capnocytophaga cynodegmi, anaerobic bacteria, Mycoplasma species or fungi. Because of light growth of mixed bacteria and the lack of obvious septic changes on cytology, bacterial growth was not considered clinically significant.
The patient re-presented 10 days following the bronchoscopy for surgical removal of the lung mass. Pre-surgical thoracic CT scan revealed a large (2.9×5.4×3.3 cm) soft tissue dense mass with air trapping and mineralization involving the dorsal aspect of the right caudal lung lobe (Fig 2). Following the CT, right caudal dorsal and accessory lung lobectomies were performed. Histopathologic diagnosis of the mass lesion was an anaplastic carcinoma with severe pyogranulomatous, necrotizing pneumonia and chronic bronchitis with bronchiectasis in the surrounding lung. Although bacterial cultures of BAL fluid were negative, additional tests were performed on the mass lesion to confirm resolution of Capnocytophaga species infection. No organisms were seen using Brown and Brenn, GMS, Fites histologic stains, or BCG. In addition, a 5 μm-section paraffin-embedded tissue was deparaffinated with xylene and DNA extracted using a commercial kit (DNA-Easy kit, Qiagen, Chatsworth MA). ‘Universal’ polymerase chain reaction (PCR) was performed using conserved primers targeting bacterial 16S rRNA-gene as previously described (Foley et al 1998). No amplicon was produced indicating the absence of bacterial DNA. A 355 base pair amplicon was obtained from positive control tissue run concurrently with the mass lesion. Considering the lack of histopathologic or PCR evidence of infectious organisms, the inflammation was presumed to be secondary to necrosis from the anaplastic carcinoma. Subsequent to a 3-month recheck, the cat developed pericardial effusion and was euthanased by the referring veterinarian. No post-mortem examination was performed.
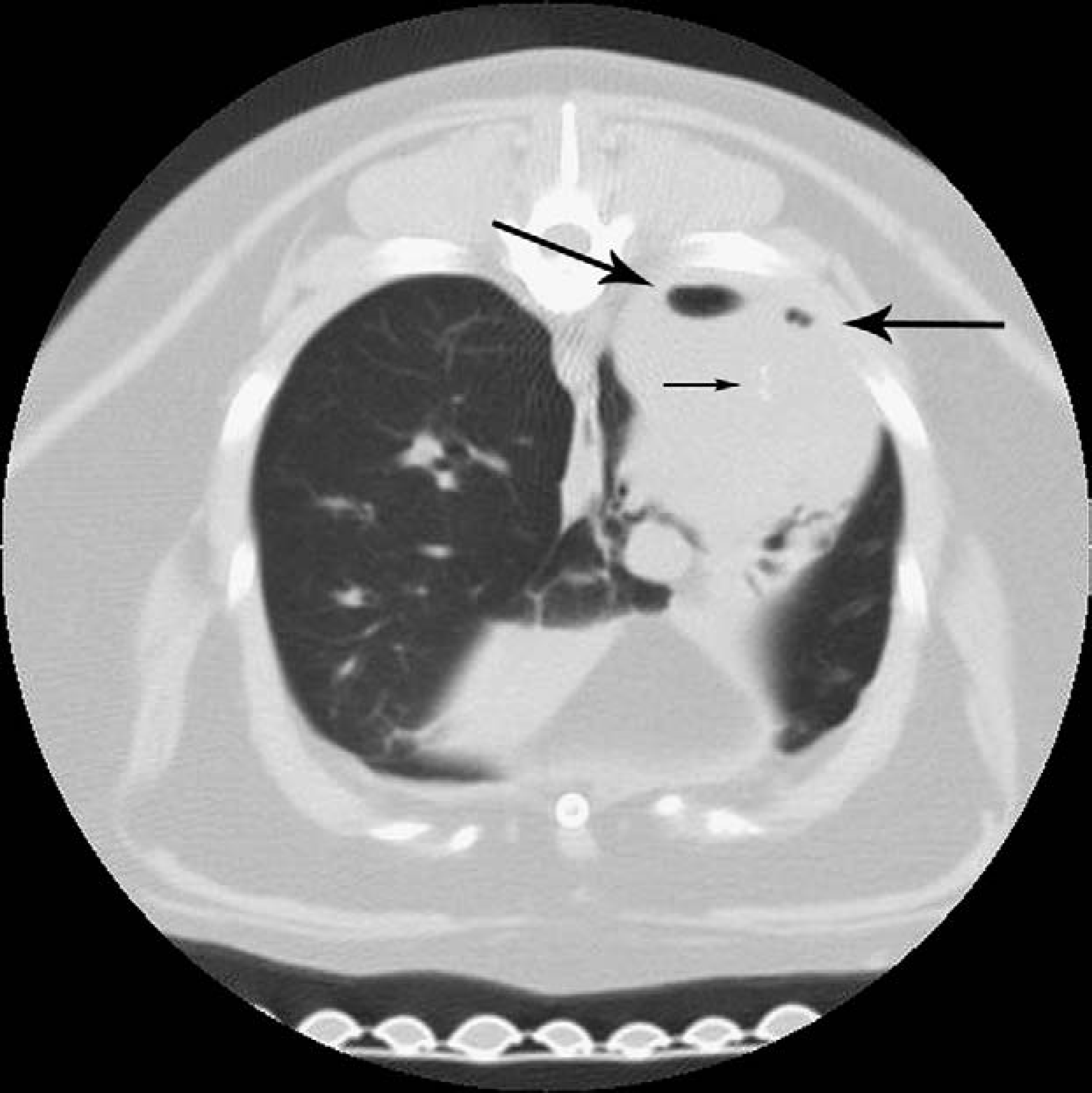
Contrast-enhanced thoracic computed tomography image revealing a soft tissue dense mass. Air trapping (large arrows) and mineralization (small arrow) involving the dorsal aspect of the right caudal lung lobe.
Capnocytophaga species are fastidious, Gram-negative, capnophilic and facultatively anaerobic bacteria. They appear as thin, 2–4 mm long filamentous rods with slightly curved, tapered ends and gliding motility (Sarma and Mohanty 2001). Originally termed ‘dysgonic fermenter’ groups 1 and 2 (DF-1 and 2, respectively) by the Center for Disease Control, DF-2 was later reclassified as DF-2 (C canimorsus) and DF-2-like (C cynodegmi) (Sarma and Mohanty 2001). Little information is available in the literature on Capnocytophaga species. C canimorsus is the most frequently reported species and it is considered a commensal organism of the oral cavity of dogs and cats (Fischer et al 1995, Lion et al 1996, Blanche et al 1998, Sarma and Mohanty 2001). Human cases are reported in immunocompromized individuals; however, 40% of cases occur in patients with no known predisposing conditions (Hicklin et al 1987, Lion et al 1996).
C canimorsus is most frequently isolated in human cases with septicemia and meningitis, although it has also been isolated in a large spectrum of disorders (Fibbe et al 1985, Chan and Fonseca 1986, Waters et al 1986, Kiel et al 1987, Paton et al 1988, Krol-van Straaten et al 1990a, b, Malnick et al 1991, Chambers and Westblom 1992, Mahrer and Raik 1992, Lion et al 1996, Pers et al 1996). In contrast to the disseminated infections associated with C canimorsus, C cynodegmi is most frequently isolated from localized wound and corneal infections (Brenner et al 1989). In a single case report, C cynodegmi was isolated from BAL fluid, blood, purulent wound discharge and sputum of a diabetic man with cellulitis, bacteremia, and pneumonitis (Sarma and Mohanty 2001).
Antibody susceptibility testing was not performed on the C cynodegmi isolate in this case report. Standard disc diffusion methods of testing antibiotic susceptibilities for C canimorsus are difficult because of the organism's slow growth in culture. C canimorsus has previously been reported to be broadly susceptible to numerous antibiotics (including ciprofloxacin) (Verghese et al 1988). In the single report available on the antimicrobial susceptibility pattern for C cynodegmi, susceptibility was similar to C canimorsus (Sarma and Mohanty 2001). The cat in this report responded clinically to a 1-month course of therapy with enrofloxacin despite the presence of concurrent pulmonary malignancy. It is unclear why previous antibiotic therapy failed to provide better control of clinical signs in this cat. It is possible that the C cynodegmi infection occurred following treatment for consolidating pneumonia.
Infection with Capnocytophaga species has not been documented in the veterinary literature, although the zoonotic potential of DF-2 infections has been reported (August 1988). To the authors' knowledge, this case represents the first report of C cynodegmi infection in a veterinary patient and only the second case in human or animal where the organism has been isolated from a BAL sample. It is probable that the patient was immunosuppressed secondary to anaplastic lung carcinoma and thus was predisposed to C cynodegmi colonization. The origin of the bacteria in this case is unknown but may have been the cat's own oral cavity. It was considered possible but unlikely that the C cynodegmi growth occurred secondary to oral contamination of the bronchoscope during the procedure, which would be consistent with the lack of bacteria observed cytologically on BAL. Standard sterile procedures, including sterile saline rinses performed before and between lavages, were employed during bronchoscopy to minimize opportunities for microbiological contamination. Moreover, it is improbable that such heavy growth and particularly consistent growth from more than one site would occur in the context of neutrophilic inflammation if the source were contamination during the procedure.
Unfortunately, culture of C cynodegmi is not highly sensitive and some infections may not be appropriately attributed to this organism. The reasons for poor sensitivity include special requirements for CO2 enrichment and agar type and slow growth even under optimal conditions.
C cynodegmi will grow on blood agar but this medium does not provide any inhibition of miscellaneous contaminating bacteria, while growth on MacConkeys agar may be non-robust. Additionally, sensitivity can be affected by prior antibiotic therapy and post-antibiotic inhibitory effects on bacterial growth, and failure to identify the organism even when isolation is successful. Diagnostic sensitivity was adequate in this case because samples were rapidly processed microbiologically after collection, cultured under appropriate conditions and DNA sequencing was utilized for identification of suspected pathogens not identified by routine biochemical tests. Mixed infection with other organisms, as seen in this case, may result in failure to recognize this infectious agent. Based on this report, Capnocytophaga species should be considered as potential opportunistic pathogens in veterinary patients.
