Abstract
A 9-year-old intact male cat was presented for vomiting and straining to defecate. A large abdominal mass was palpated. The urinary bladder was full and non-expressible. Exploratory laparotomy revealed that the mass was compressing the colon and encircling the urethra caudal to the bladder. The mass was removed, the urethra transected, and the urinary bladder marsupialized to the ventral abdominal wall to allow urine drainage. Histopathologic examination of the mass revealed a prostatic carcinoma. The cat died approximately 6 weeks after removal of the mass. This is the first reported case of a prostatic carcinoma causing urethral obstruction and obstipation in a cat.
Neoplasms of the prostate gland are uncommon in domestic animals except dogs (MacLachan et al., 2002). Only a few cases of prostatic carcinomas in cats have been reported, (Hawe, 1983; Hubbard et al., 1990; Caney et al., 1998) none of which resulted in obstruction of the urinary or digestive tract. We describe a case of prostatic carcinoma in a male cat that resulted in obstruction of the urethra and colon, leading to blockage of the urinary tract and severe obstipation.
A 9-year-old intact male domestic shorthair cat with a 2-year history of diabetes mellitus and weight loss was evaluated for diarrhea with large amounts of mucus of several weeks duration. Upon rectal examination, a mass approximately 2 cm in diameter was palpated on the floor of the pelvis. A biochemical profile revealed mild hyperglycemia (8 mmol/l; reference interval 4–6 mmol/l) and azotemia (creatinine 141.4 μmol/l; reference interval 53–124 μmol/l). The serum sodium and chloride concentrations were also slightly increased (sodium 152 mmol/l; reference interval 132–146 mmol/l; chloride 121 mmol/l; reference interval 100–110 mmol/l). Results of a complete blood count were within reference intervals. Due to financial constraints, the owner elected medical treatment for the colitis and did not allow further diagnostic tests to better characterize the pelvic mass.
The cat was re-evaluated 3 months later for inappetence, vocalization during urination, and excessive licking of the perineal region. Rectal palpation revealed significant growth of the mass in the pelvic canal. The cat was anesthetized and a urinary catheter was placed and the bladder drained of urine. Due to clinical suspicion the pelvic mass may have been an enlarged prostate gland, and an orchiectomy was performed, and the cat was returned to its owner. Six weeks later, the owner reported that the cat had been urinating normally and eating better. However, re-examination revealed that the pelvic mass had not diminished in size in response to the castration.
Eight months later, the cat was presented for evaluation of lethargy, vomiting, continued weight loss, and straining to defecate. Physical examination and a retrograde urethrogram revealed a large, soft-tissue dense mass occluding the rectum, severe obstipation, and a markedly dilated, non-expressible urinary bladder. An indwelling urinary catheter was placed and the bladder drained. An intravenous catheter was placed and the cat was re-hydrated using lactated Ringer's solution. The cat was prepared for surgery, and an exploratory laparotomy was performed. The location of the mass in the pelvic canal necessitated performing a pubic osteotomy. The mass was found to completely encircle the urethra in the location of the prostate gland. The owner was contacted and informed of the poor prognosis, but refused to allow euthanasia of the cat. As a salvage procedure, the urethra was transected and the mass removed. The remaining abdominal urethra was transected free from the bladder and the bladder was sutured. To allow expulsion of urine, the bladder was marsupialized through the abdominal wall. Surgical biopsies of the mass were obtained and submitted for histopathologic evaluation. Recovery from anesthesia was uneventful, and the cat was discharged on enrofloxacin (Baytril; Bayer) (15 mg once daily) and cephalexin (Cefa-tabs; Wyeth) (100 mg every 12 h), in an attempt to prevent infection of the urinary tract through the marsupialization site. A recheck urinalysis 1 month after surgery revealed marked hematuria and pyuria, likely indicating a urinary tract infection (bacterial culture and sensitivity testing were not performed). The cat lived for approximately 2 months following surgery before dying from unknown causes. A necropsy was not permitted.
Histopathologic examination of the biopsies revealed a well-circumscribed, encapsulated mass. The mass was composed of highly cellular lobules of moderately to markedly anaplastic epithelial cells separated by a poorly cellular, dense fibrous connective tissue stroma. The lobules were composed of neoplastic epithelial cells arranged in solid sheets and cords and occasionally forming acinar or papillary structures (Fig. 1). The carcinoma cells contained round to ovoid nuclei with an open chromatin pattern and prominent multiple nucleoli. Marked anisocytosis and anisokaryosis were present, with many cells containing giant megaloblastic nuclei. The cytoplasm often contained large, 5–30 μm, non-staining vacuoles. Mitotic figures were commonly seen (4 per high-power 400× field). Capsular invasion was noted, but there was no evidence of intravascular spread of the cancer. A densely cellular stromal cell proliferation that occasionally surrounded the neoplastic acini was interpreted to be a scirrhous reaction induced by the carcinoma cells. Several areas of the neoplasm were undergoing coagulation necrosis. A portion of the urethra present in the biopsy was ulcerated. Hemorrhage and edema were commonly seen in the connective tissue stroma. The histologic diagnosis of the mass was a prostatic adenocarcinoma.

Histopathologic appearance of the feline prostatic carcinoma. The carcinoma was composed of numerous acini filled with highly anaplastic epithelial cells proliferating in papillary to solid patterns. The acini were surrounded by prostatic stromal tissue containing collagen fibers, fibroblasts and smooth muscle cells. (Hematoxylin and eosin stain, 40×; bar = 15 μm.)
In order to identify the proportion of the neoplastic cells actively in the growth fraction, Ki-67 immunohistochemistry (murine anti-human Ki-67 monoclonal antibody, clone MIB-1, 1:160 dilution; DakoCytomation, Carpinteria, CA) was performed on the prostatic carcinoma (Fig. 2) and on a prostate gland from a normal cat. A Ki-67 labeling index (LI) was calculated to reflect the percentage of immunoreactive cells present out of 500 randomly selected epithelial cells. The LI of the normal feline prostate gland was 2.6%, while the LI of the prostatic carcinoma was 55%.
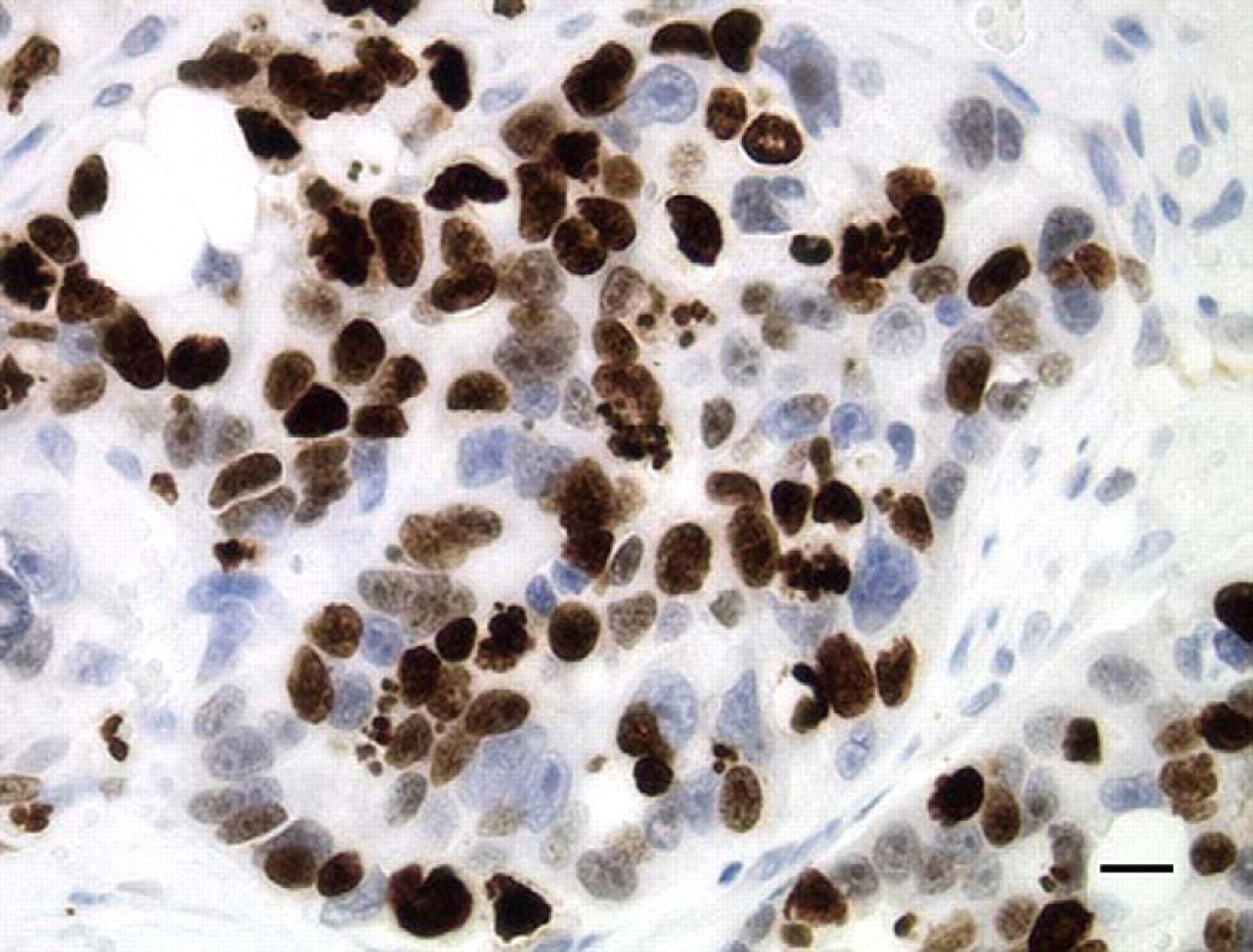
Ki-67 immunoreactivity of the feline prostatic carcinoma. The brown, granular staining present in the nuclei of most of the carcinoma cells in this acinus demonstrates their rapid growth rate. (Diaminobenzidine [DAB] chromogen with hematoxylin counterstain, 40×; bar = 19.2 μm).
Prostatic carcinomas are extremely rare in all species except humans and dogs. This case is unusual because of the rarity of prostatic carcinomas in cats, and also because the prostatic carcinoma in this cat caused obstruction of the prostatic urethra and the descending colon, preventing the cat from urinating and defecating. This case demonstrates that, even though uncommon in cats, prostatic carcinoma should be considered as a differential for cats presenting with signs of urinary and/or colonic obstruction.
We were unable to evaluate this cat thoroughly for metastases, as the owner did not permit a post-mortem examination. Metastases occur frequently with prostatic carcinoma in other animals. Prostate cancer commonly metastasizes to lymph nodes and bones in human beings and dogs (Kennedy et al., 1998; Guise and Mundy, 1998; Young et al., 2000). The few cases of prostate cancer reported in cats have not provided an opportunity for extensive study of the metastatic behavior of these neoplasms in cats. We recognize, however, that metastases may have been found in this patient if a necropsy had been performed.
The histologic appearance of this tumor is consistent with previous reports from cats with prostatic carcinoma (Hawe, 1983; Hubbard et al., 1990; Caney et al., 1998). As this case illustrates, prostatic carcinoma in dogs and cats often results in marked, diffuse enlargement of the gland. As the prostate enlarges, it impinges on abdominal organs and may cause organ dysfunction. In contrast, in men with benign prostatic hyperplasia the prostate undergoes intra-capsular enlargement that compresses the prostatic urethra, leading to dysuria, but rarely impairing defecation.
It is likely that castration plays an important role in the low incidence of prostatic carcinomas in cats, but prostate carcinomas are still very rare in intact wild cats kept in zoos (personal communication, Donald S. Coffey, The Johns Hopkins University). It is interesting to speculate that, even in this intact male cat, testosterone may not have been an important growth factor for the carcinoma, as no reduction in size or growth of this neoplasm was noted following castration. One possibility is that the carcinoma in this cat had become androgen-independent, as occurs commonly in men with advanced prostate cancer (Pilat et al., 1998). Alternatively, it is also possible that this neoplasm was a transitional cell carcinoma that originated in the prostatic urethra and invaded the prostate gland. Transitional cell carcinomas would not be expected to regress following castration. However, we believe that this cancer was unlikely to be a transitional cell carcinoma, because the mass was confined to the prostate gland, and visual inspection of the bladder during surgery did not reveal any evidence of cancer cell growth. Currently, there are no markers useful for differentiating transitional cell carcinomas from prostatic carcinomas in domestic animals. Additional tools, such as ultrastructural analysis, may be necessary to accurately identify the cellular origin of these neoplasms.
The Ki-67 labeling index demonstrated that 55% of the neoplastic cells were in the growth fraction. This was markedly increased compared to the LI of a normal cat prostate gland (2.6%). While difficult to draw conclusions from a single case, the high Ki-67 labeling index of the carcinoma cells suggests that, unlike human prostate carcinoma, a high percentage of the cells in feline prostatic carcinomas may be actively dividing. Human prostate carcinoma is primarily a disease of neoplastic cell accumulation, in which the carcinoma cells maintain a low growth rate, but have acquired mutations in apoptotic death proteins which allows the neoplasm to continue to grow (Berges et al., 1995).
Footnotes
Acknowledgments
Special thanks to Diane Lawson and the staff of the Emory University Hospitals Histopathology Laboratory, Atlanta, GA for performing the Ki-67 immunohistochemistry.
