Abstract
The three calcitropic hormones, parathyroid hormone (PTH), 1,25-dihydroxycholecalciferol and calcitonin are together responsible for calcium homeostasis in the mammal. Feline PTH is an 84 amino acid, single chain polypeptide with a molecular weight of 9449, which is secreted by the parathyroid glands. The principle secretagogue for PTH is a low plasma ionised calcium concentration, although both 1,25-dihydroxycholecalciferol and phosphate have significant roles in regulating PTH secretion. The ability to accurately measure circulating PTH in the cat has simplified the evaluation of disorders of calcium metabolism in this species. In primary parathyroid disorders the lesion is located within the parathyroid gland, with parathyroid secretion being inappropriate to the prevailing mineral balance. By contrast, in secondary conditions a pathological state out with the parathyroid gland alters mineral homeostasis and the parathyroid gland responds in an appropriate manner. The measurement of circulating PTH may then be used to determine if PTH secretion is appropriate to the prevailing calcium concentrations to differentiate primary from secondary disorders. Although primary hyper and hypoparathyroidism are generally considered rare endocrine conditions of the cat, the ability to measure PTH has led to their increasing recognition.
Disorders of the parathyroid glands are generally considered to be uncommon feline endocrine disorders. However, despite the paucity of clinical reports of spontaneous parathyroid disease in the cat, the ability to measure PTH has led to their increasing recognition.
The role of the parathyroid glands in the regulation of calcium metabolism was first recognised by MacCallum and Voegtlin, who in 1909 (MacCallum and Voegtlin, 1909) demonstrated that the tetany caused by extirpation of the parathyroid glands was associated with a fall in the calcium content of the blood. Over the last decade improvements in the laboratory assessment of calcium homeostasis in the cat, principally the availability of parathyroid hormone assays, have greatly increased our understanding of parathyroid disorders and simplified their diagnosis.
The parathyroid glands
There are two pairs of parathyroid glands in the cat, the external and internal parathyroid glands. Both pairs of glands are generally associated with the thyroid gland, which in the cat consists of two lobes with no connecting isthmus. The description of external and internal refers to whether the parathyroid glands are located external to the connective tissue capsule of the thyroid or are within the capsule embedded in the parenchyma of the thyroid gland.
There are conflicting reports regarding the exact position of the parathyroid glands in the cat. Some authors referring to the external parathyroids as being at the cranial pole of the thyroid gland (Waters, 1993) and others to their location at the caudal end of the thyroid (Dyce et al., 1996). The internal parathyroid glands occupy a variable position within the thyroid parenchyma, but can generally be appreciated from external examination of the thyroid glands. Ectopic parathyroid glands are also frequently seen, often at some distance from the thyroid altogether (although generally still paratracheal) (Flanders, 2003).
As with all endocrine glands the parathyroid glands are ductless. Histologically their parenchyma is surrounded by a dense irregular connective tissue capsule. The chief (or principal) cells form the major cell type and are arranged in clusters or cords and secrete parathyroid hormone (PTH). Chief cells are divided into two types on the basis of their histological appearance, dark and light cells. Whether this division represents differences in functional activity, dark cells being more actively secreting cells, or whether these differences in appearance represent an artefact of fixation is a matter of debate (Larsson et al., 1984; Yamaguchi et al., 1997). The chief cells contain a limited number of PTH containing secretory granules and, in the cat, may also contain glycogen inclusions (Waters, 1993). A second cell type the oxyphil cell, is considered rare in the cat, as in other carnivores (Waters, 1993).
Parathyroid hormone
Feline PTH is an 84 amino acid polypeptide with a molecular weight of 9449 (Toribio et al., 2002). Parathyroid hormone is highly conserved at the amino acid level amongst mammals, with the amino acid structure of feline PTH having the greatest identity with canine PTH. Based on the deduced amino acid sequence mature feline PTH is 84% identical to human PTH, with 88% identity in the N-terminal region which is responsible for the hormonal activity (amino acids 1–34) (Toribio et al., 2002).
The translational product of PTH mRNA is a 115 amino acid polypeptide called preproPTH. The “pre” region is a signal sequence, which is cleaved cotranslationally to the intermediate 90 amino acid precursor, proPTH. This then moves from the lumen of the rough endoplasmic reticulum to the Golgi apparatus where enzymatic cleavage of the “pro” sequence occurs, with little proPTH being stored in the chief cell. PTH is then packaged into secretory granules for secretion or storage. However, the cells of the parathyroid gland contain few secretory granules and up to 70% of the PTH synthesised is proteolytically degraded within the cell (Morrissey et al., 1980). The parathyroid gland appears to synthesise excessive quantities of PTH, the amount of PTH available for secretion being controlled by Ca2+dependent degradation. Therefore, the secretory product of the parathyroid glands consists of intact hormone and fragments, principally carboxyl terminal fragments (Morrissey et al., 1980).
Circulating PTH is heterogeneous, composed principally of intact hormone and middle and carboxyl terminal fragments of PTH. The ratio of large carboxyl fragments to intact PTH in the circulation has been shown to vary dependent on the plasma calcium concentrations, the proportion of intact hormone increasing in hypocalcaemia (D'Amour et al., 1992). The various immunoreactive forms of PTH present in plasma result from the proteolytic metabolism of PTH both within the parathyroid glands and in peripheral tissues.
Actions of parathyroid hormone
Parathyroid hormone is one of the three calcitropic hormones, PTH, calcitonin and 1,25-dihydroxycholecalciferol (calcitriol or 1,25-(OH)2D3, the active form of vitamin D), responsible for the regulation of extracellular fluid calcium concentration. The principal function of PTH is to maintain plasma calcium concentration within a narrow range, via actions on bone and kidney. Secondary to this function PTH regulates plasma phosphate concentration. The broad functions of PTH are: to stimulate the release of calcium and phosphate from bone; to increase the reabsorption of calcium and inhibit the reabsorption of phosphate from the glomerular filtrate; and to stimulate the synthesis of 1,25-(OH)2D3 by the proximal tubules of the kidney, thereby indirectly increasing the intestinal absorption of calcium and phosphate. The synthesis of 1,25-(OH)2D3 is also influenced by phosphate concentrations, phosphate inhibiting the 1α-hydroxylase enzyme that catalyses the reaction.
The actions of PTH are mediated by a specific cell membrane receptor which when activated triggers a second messenger cascade within the target cell. The PTH receptor binds and is activated by both PTH and PTH-related peptide (PTHrP) and is therefore termed the PTH/PTHrP receptor. This receptor has a widespread distribution but is particularly abundant in PTH target tissues (Ureña et al., 1993) and is a member of the G protein-coupled receptor superfamily (Jüppner et al., 1991). More recently a second G protein-coupled receptor, termed the PTH2 receptor has been identified (Usdin et al., 1995). The PTH2 receptor is activated by PTH only and, although it would have seemed logical that this receptor would selectively mediate the endocrine actions of PTH, its limited distribution, predominately in the brain and pancreas, appears to preclude this role.
Parathyroid secretion
Regulation of the quantity of PTH entering the circulation is dependent on control at four levels; gene transcription, synthesis, intracellular catabolism and finally secretion. In the longer term, the rate of parathyroid cell proliferation will also influence the secretory capacity of the parathyroid glands. As PTH regulates plasma concentrations of calcium, phosphate and 1,25-(OH)2D3 it is not unexpected that there is a well defined feedback loop between these parameters and the parathyroid glands.
The principal secretagogue for PTH is a low extracellular ionised calcium concentration. In contrast to most cells, those of the parathyroid gland respond to changes in extracellular calcium with parallel alterations in intracellular calcium concentrations. By secreting in response to a hypocalcaemic stimulus, the parathyroid gland differs from most other secretory systems in which a high intracellular calcium level acts as a second messenger to stimulate exocytosis. This link between extra and intracellular calcium concentrations is brought about by the calcium sensing receptor (CaR), which permits the chief cells to act as extracellular ionised calcium sensing cells. The CaR is a member of the superfamily of G protein-coupled receptors and is present in the chief cells, C cells, intestine and the kidney, all systems pivotal in calcium homoeostasis (Chattopadhyay, 2000).
An inverse sigmoidal relationship exists between the extracellular calcium concentration and PTH secretion. There is a non-suppressible element to PTH secretion even at very high serum calcium concentrations. A detailed analysis of this sigmoidal relationship by Brown (1983) demonstrated the importance not only of the maximum and minimum rates of secretion, but also of the so called set-point in regulating PTH secretion. The set-point is defined as the calcium concentration required to produce half of the maximum inhibition of PTH secretion, and as such represents the sensitivity of the gland to the suppressive effect of calcium. In a number of disease conditions, including secondary renal hyperparathyroidism (RHPTH) it is a shift of this set point to the right (to a higher ionised calcium concentration) which contributes to the over-secretion of PTH, even in the presence of normal plasma calcium concentrations.
At normal plasma calcium concentrations, the rate of PTH secretion is only slightly greater than that at maximally suppressed levels. This reflects the regulatory effect of calcium concentrations on PTH gene expression, where a similar inverse sigmoidal relationship also exists. In vivo studies have demonstrated that small decreases in serum calcium concentration produce large increases in PTH mRNA, whereas a high serum calcium has no effect (Naveh-Many et al., 1989). The effect of calcium on PTH gene expression has been shown to be post-transcriptional, by altering the stability of PTH mRNA (Moallem et al., 1998). Since PTH synthesis is almost maximally suppressed at normal serum calcium concentrations, the parathyroid gland is therefore primed to respond to even small hypocalcaemic challenges with a marked increase in the rate of PTH synthesis.
Serum 1,25-(OH)2D3 and phosphate concentrations both regulate parathyroid secretion indirectly via their influence on calcium concentrations, however both also have a direct effect. The second major regulator of PTH secretion is 1,25-(OH)2D3. Studies demonstrated a decrease in PTH mRNA concentrations in response to 1,25-(OH)2D3, this regulatory effect occuring at the level of PTH gene transcription (Silver et al., 1986). In contrast phosphate acts as calcium at a post transcriptional level, hypophosphataemia decreasing the stability of PTH mRNA and decreasing PTH synthesis (Moallem et al., 1998).
Diagnosis of parathyroid disorders
The availability of assays to measure PTH has revolutionised the diagnosis of disorders of the parathyroid gland. Although the presence of insufficient or excess PTH may be predicted from a combination of parathyroid pathology and serum calcium concentrations the definitive diagnosis of hyperparathyroidism or hypoparathyroidism by definition requires the measurement of PTH. On a more practical level the ability to measure PTH reduces the need for exploratory cervical surgery and protracted diagnostic investigations to rule out non-parathyroid causes of hypercalcaemia.
Total and ionised calcium
Clinical signs of disorders of the parathyroid glands are usually non-specific and as such are usually first suspected on finding hyper or hypocalcaemia on screening biochemistry. Calcium in plasma is distributed between three fractions. In the normal cat, approximately 50% is present as free hydrated calcium ions, the remainder being bound. This bound fraction is further divided into protein bound, comprising approximately 40% of the total calcium, with complex bound calcium making up the final 10%. The majority of the calcium bound to plasma proteins is bound to albumin. The calcium in the complex bound fraction is associated with low molecular weight ligands such as bicarbonate, lactate, citrate and phosphate. It is generally considered that the ionised component alone is responsible for the physiological actions of calcium and is therefore the fraction that is subject to homeostatic control.
The distribution of calcium between these various fractions is controlled by the law of mass action and is therefore dependent upon the concentration of calcium and binding ligands, as well as the pH. Alterations in plasma albumin or protein concentrations can have a major influence on the total calcium concentration. Investigation of this phenomenon in the cat has shown that although there is a linear relationship between total calcium and albumin, it is too variable to allow the derivation of correction equations (Flanders et al., 1989). Therefore, to fully assess calcium homeostasis it is vital that ionised calcium concentrations are measured directly. This is particularly important in conditions such as renal failure, where there is concurrent alteration in a number of these parameters, and total calcium is likely to be a poor reflection of calcium status. As it is the ionised fraction of calcium that directly controls PTH secretion, measurement of ionised calcium is the ideal in the investigation of parathyroid activity.
Most laboratory assessment of serum calcium involves the measurement of total calcium, which includes the ionised, bound and complexed calcium fractions. Accurate measurement of ionised calcium concentrations is possible using ion selective electrode (ISE) systems, which measure the electrochemical potential of the calcium ions and, therefore, probably best reflect their biological activity. Given the temperature and pH dependence of ionised calcium concentrations, samples should be handled as blood gas samples. Hand held and bench top ISE systems for ionised calcium measurement are available.
Parathyroid hormone
Currently, two PTH assays 1 have been validated for use in the cat (Barber et al., 1993; Provencher Bolliger et al., 2002). Both are two-site immunoradiometric assays (IRMA) for human intact PTH. Development of an assay specifically for feline PTH may improve the assay sensitivity, however such an assay is unlikely to be economically viable and the adoption of human assays provides the most effective means of providing a widely available PTH assay.
Given the heterogeneity of immunoreactive PTH in plasma, it is important that the PTH assay selected is specific only for the biologically active hormone, namely intact PTH. Two site IRMA overcomes many of the limitations associated with region selective assays and is specific for the biologically active polypeptide. Specificity for intact PTH is achieved by the use of a double antibody technique (hence “two site”), employing antisera which recognise epitopes at both terminals of the peptide. Recent studies have shown that the intact assay not only reacts with 1–84 PTH but also with large PTH fragments, the majority of which is 7–84 PTH. This has resulted in the development of a “whole” PTH assay, which although validated for the dog (Estepa et al., 2003), has not yet been assessed in the cat.
Studies in man have demonstrated pulsatile secretion of PTH with a biphasic circadian rhythm (Calvo et al., 1991; Kitamura et al., 1990; Markowitz et al., 1988). However, no studies have been performed in the cat to determine the daily pattern of PTH secretion and the author found no evidence of day to day or periodic variation in plasma PTH concentrations on repeated sampling of normal cats (Fig. 1). Midmorning is the recommended time for sampling in man to correspond to the nadir in PTH secretion, so allowing improved differentiation of marginally hyperparathyroid from normal subjects. In the absence of any evidence to the contrary it seems sensible to adopt this approach in the cat.

Results of a study to determine the degree of fluctuation in plasma PTH in nine normal cats (Barber, 1998). Midmorning, fasting blood samples were taken daily for two periods of three consecutive days with an intervening 18 day rest period. Analysis of variance demonstrated no significant variation with time (either daily or periodic) but significant variation in PTH concentrations between individual cats (P = 0.028).
Parathyroid hormone is relatively heat labile and samples for PTH assay require appropriate handling to prevent erroneously low results (Barber et al., 1993). Once obtained, plasma or serum should be harvested with the minimum exposure of samples to room temperature (less than two hours), and, unless assayed immediately, samples should then be frozen. Ideally, samples should be posted to outside laboratories packaged on ice, to arrive in a frozen state. Long term storage of samples should be at −80°C.
Parathyroid imaging
In the cat it is possible to palpate some enlarged (either hyperplastic or neoplastic) parathyroid glands in a similar manner to the palpation of a thyroid goitre. Ultrasonography and radionuclide scans may also be used to evaluate the parathyroid glands.
A number of reports are now available describing the ultrasonographic evaluation of the parathyroid glands in the cat, usually as part of the diagnostic evaluation of hypercalcaemia (Savary et al., 2000; Sueda and Stefanacci, 2000; Wisner and Nyland, 1998). Imaging is best carried out using a 7.5 to 10 MHz transducer, linear array transducers giving improved visualisation of the near field and superficial structures such as the thyroid region. Due to their small size parathyroid glands are not routinely seen on ultrasonographic examination but if seen appear as hypoechoic or anechoic structures of less than 2 mm in diameter (Wisner and Nyland, 1998). It has been suggested that the ultrasonographic identification of a parathyroid gland exceeding 4 mm in diameter is highly suspicious for parathyroid adenoma or carcinoma in the cat as in the dog (Sueda and Stefanacci, 2000). However, the author's experience of parathyroid pathology does not support this finding (Barber and Elliott, 1998). Differentiation of hyperplastic and neoplastic lesions on the basis of parathyroid size and number is likely to be difficult given that hyperplasia can lead to marked and asymmetric parathyroid enlargement and that multiple neoplastic lesions are not uncommon.
Parathyroid scintigraphy has proved a useful technique in human medicine in the diagnostic evaluation of recurrent primary hyperparathyroidism. The preferred radionuclide is technitium-99m-sestamibi (99mTc-sestamibi), which is taken up by both the thyroid and parathyroid glands. Therefore a dual isotope subtraction (using 99mTc-pertechnetate or 123I to identify thyroid tissue) or a single isotope dual phase (making use of the persistence of the isotope in abnormal parathyroid tissue) scan are used. However, the use of dual phase 99mTc-sestamibi scintigraphy had a poor sensitivity and specificity when used in hypercalcaemic dogs (Matwichuk et al., 2000) and the author is not aware of a study of this technique in the cat.
Parathyroid hormone related protein
Parathyroid hormone-related protein (PTHrP) was first isolated as the primary factor responsible for humoral hypercalcaemia of malignancy (HHM), having effects analogous to those of PTH. Both PTH and PTHrP utilise the same PTH/PTHrP G protein coupled receptor, however, the physiological role of PTHrP appears to be more complicated. PTHrP is produced in many tissues and appears to function locally in an autocrine or paracrine manner, to regulate cell growth and differentiation amongst other functions (Strewler, 2000).
The hypercalcaemic effects of PTHrP are due to homology of the amino acid sequence in the first part of the amino terminal to that of PTH. Beyond this region the two molecules are very dissimilar and hence PTHrP is not detected in assays for PTH. A human IRMA for PTHrP has been used successfully in the cat to diagnose HHM (Provencher Bolliger et al., 2002).
Vitamin D
Although initially classed as a vitamin, vitamin D in man is a conditionally essential nutrient, since exposure to ultraviolet radiation negates the dietary requirement. Most cats have highly pigmented, furred, skin and recent studies have reported that cats are unable to synthesise adequate amounts of vitamin D (How et al., 1994; Morris, 1999). It would therefore appear that in the cat vitamin D is a true vitamin being an essential dietary requirement. Both vitamin D2 and D3 are ingested in the diet and absorbed in a similar manner to other fat soluble compounds.
Vitamin D itself is not biologically active and undergoes conversion to more polar metabolites prior to biological activity. The majority of the initial conversion occurs in the liver to produce a very weakly active metabolite, 25-(OH)D3, which is the major circulating form of vitamin D. Subsequently, 25-(OH)D3 is further hydroxylated in the proximal tubular cells of the kidney, by a mitochondrial 1-α-hydroxylase enzyme, to the active metabolite 1,25-(OH)2D3. This final phase of activation of vitamin D is very closely regulated and is the rate limiting reaction of this synthetic process.
Both 25-(OH)D3 and 1,25-(OH)2D3 have been successfully assayed in the cat (Barber and Elliott, 1998; Morris, 1999), although limited availability means such assays are likely to remain research tools for the foreseeable future.
Classification of parathyroid disorders
Parathyroid disorders may be broadly categorised into those where parathyroid secretion is reduced below normal levels, hypoparathyroidism, and those where secretion is increased, hyperparathyroidism. These diseases may then be further divided into primary or secondary categories. In primary disorders the lesion is located within the parathyroid gland, with parathyroid secretion being inappropriate to the prevailing mineral balance. In secondary conditions a pathological state out with the parathyroid gland alters mineral homeostasis and the parathyroid gland responds in an appropriate manner.
Note on parathyroid pathology
In some cases, direct examination and biopsy may be required to completely evaluate parathyroid pathology. Classically parathyroid hyperplasia implies an abnormality involving all parathyroid tissue, presumably as a response to a secretory stimulus affecting all the glands, such as would be present in cases of secondary hyperparathyroidism. Autonomous secretion of hormone generally involves neoplastic tissue with the remaining normal tissue becoming atrophied. Unfortunately, the division of hyperparathyroidism into primary and secondary disease on the basis of parathyroid pathology is not this simple. Firstly, abnormal tissue may appear normal on gross examination and secondly the criteria for distinguishing adenomas from hyperplasia on the basis of gland histology are not well established. Indeed, pathologists tend to rely on the gross description of the glands, a tentative diagnosis being based on the number of glands affected (one enlarged gland implies adenoma, whereas multiple gland enlargement implies hyperplasia) (Verdonk and Edis, 1981; Lucke, V.M., personal communication). Although hyperplasia affects all parathyroid glands, the increase in the size of each gland is not necessarily uniform, even when the stimulus to enlargement is the same for each gland (Verdonk and Edis, 1981). It is possible for an apparently normal sized gland to be hyperplastic or contain areas of hyperplastic tissue, which must then be distinguished on histology. At the other extreme hyperplasia can become nodular in nature, terms such as nodular hyperplasia or adenomatous hyperplasia are then utilised. In the human field some of these nodules have been demonstrated to be monoclonal in nature, presumably resulting from a single cell with a higher innate proliferative potential than its neighbours, blurring further the distinction between hyperplasia and neoplasia (Arnold et al., 1995). Although hyperplasia is generally associated with secondary disease, parathyroid hyperplasia has been recorded as a cause of primary hyperparathyroidism in the cat (Savary et al., 2000).
Primary hyperparathyroidism
Primary hyperparathyroidism is considered a relatively rare condition in the cat but is being recognised with increasing frequency. However, due to the sporadic nature of the condition only one small case series has been reported (Kallet et al., 1991), the remainder of the literature consisting of case reports (den Hertog et al., 1997; Kaplan, 2002; Marquez et al., 1995; Savary et al., 2000; Sueda and Stefanacci, 2000). In a retrospective review of 71 cats with hypercalcaemia a diagnosis of primary hyperparathyroidism was made in only four cases (Savary et al., 2000). A review of the clinical presentation, diagnosis and treatment of the 17 cases reported in the literature, where the relevant information was available, is given below.
Primary hyperparathyroidism typically affects older animals, the age range being 8 to 20 years, with no sex predisposition. Although five of the seven cases first reported were Siamese suggesting a predisposition in this breed, subsequent cases have been in mixed breed cats. The signalment of four cases was not reported (Savary et al., 2000). The most frequent clinical signs reported were lethargy, anorexia and vomiting (Table 1). Clinical signs were generally non-specific and in at least one case none were reported. The polydipsia and polyuria noted in dogs is less commonly reported in the feline. Physical examination was generally unremarkable, the only consistent findings being a palpable cervical mass (7 of 17 cases) and dehydration.
Clinical signs and parathyroid pathology reported in 17 cats with primary hyperparathyroidism

One cat with a unilateral parathyroid adenoma (right side) which was surgically removed later represented with hypercalcaemia due to a concurrent parathyroid carcinoma (right side) and adenoma (left side).
Primary hyperparathyroidism is characterised by an excessive, autonomous secretion of PTH causing hypercalcaemia. Serum total calcium will obviously be elevated and if ionised calcium is measured this will also be raised. Of the 17 cases, PTH was measured in nine cats and was elevated in four of these. One case had variable (high and normal) results and in the other four cases, PTH concentrations were normal. Normal PTH concentrations are inappropriate in the face of hypercalcaemia, when PTH secretion should be suppressed, and so can be considered diagnostic for primary hyperparathyroidism. Due to the phosphaturic effect of PTH, cats with primary hyperparathyroidism would be predicted to have low or low normal serum phosphate concentrations, unless renal function is impaired, when the ability of the kidney to excrete the increased phosphate load will decrease and hyperphosphataemia may develop. In those cases where results were recorded approximately two thirds were azotaemic, hence phosphate concentrations were extremely variable with only 38% of cats being hypophosphataemic. Calcium oxalate urolithiasis causing dysuria was reported in one case.
Surgical resection of the abnormal parathyroid tissue is the current treatment of choice for primary hyperparathyroidism. In the dog primary hyperparathyroidism has also been successfully treated using percutaneous ultrasonographically guided techniques of chemical ablation (ethanol injection) (Long et al., 1999) or radiofrequency heat ablation (Pollard et al., 2001). To the authors knowledge the use of these techniques has not been reported in the cat, although ethanol ablation has been used unsuccessfully in the treatment of hyperthyroidism in cats (Wells et al., 2001). Chronic hypercalcaemia leads to atrophy of normal parathyroid glands and surgical removal of the autonomously secreting gland will lead to a rapid decline in PTH levels and relative hypoparathyroidism. Therefore post-operatively it is essential that the patient is monitored closely for clinical signs of hypocalcaemia. Opinion varies as to whether routine preventive therapy for hypocalcaemia should be considered or whether treatment should be reserved for individuals showing clinical signs. Initiating treatment for hypocalcaemia before the onset of clinical signs removes the hypocalcaemic stimulus to reverse parathyroid atrophy and actively inhibits PTH secretion.
In one reported case (Kallet et al., 1991), despite successful surgery to remove a parathyroid adenoma, hypercalcaemia recurred 569 days later due to bilateral parathyroid neoplasia (carcinoma and adenoma). Concurrent hyperparathyroidism and hyperthyroidism (Kaplan, 2002) in one case and hyperparathyroidism due to hyperplasia of all four parathyroid glands with metastatic adrenal carcinoma have also been reported. Multiple endocrine neoplasia (MEN) has been reported in cats (Feldman and Nelson, 1996). The MEN syndromes are characterised by autonomous and excess secretion of hormones by more than one endocrine gland. In man MEN type 1 and MEN type 2A may involve parathyroid hyperplasia or neoplasia, although the condition has not been well characterised in the cat.
Primary hypoparathyroidism
Similar to primary hyperparathyroidism, primary hypoparathyroidism is a relatively rare condition in the cat, which is being recognised with increasing frequency. Again cases are sporadic with only one small case series reported (Peterson et al., 1991), the remainder being single case reports (Bassett, 1998; Forbes et al., 1990; Parker, 1991; Ruopp, 2001). The following is a review of the clinical presentation, diagnosis and treatment of nine cases reported in the literature, where the relevant information was available.
Primary hypoparathyroidism appears to be a disease of young cats with a mean age of 1.8 years (range 0.5 to 6.7 years) and four of the reported cases being in cats one year old or less. At least one case showed clinical signs from two months of age. Based on the small number of cases available there appears to be no sex predisposition. The majority of cases have been in domestic short haired cats although one Himalayan and one Siamese have also been affected.
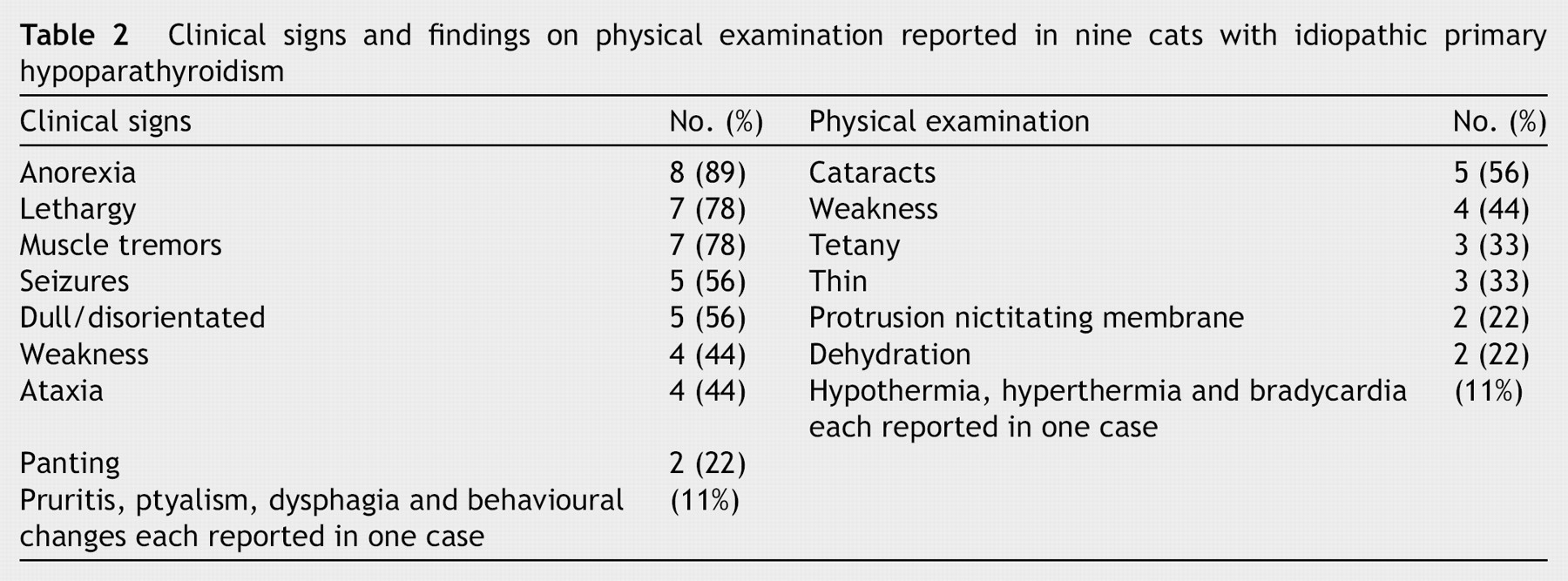
Clinical signs and physical examination findings are similar to those reported in the dog (Table 2). All cases were characterised by intermittent neurological or neuromuscular signs including generalised seizures, muscle tremors, or tetany. Cataracts were noted in over half of the affected cats and are typically bilaterally symmetrical, multiple, punctate opacities in the anterior and posterior subcapsular region of the lens (lenticular cataracts). Bilateral protrusion of the nictitating membrane was also reported in some cases. All cases were persistently hypocalcaemic, with eight of the nine having concurrent hyperphosphataemia. No cases were reported to be azotaemic. Elevated alanine aminotransferase concentrations were noted in one third of cases. Parathyroid hormone concentrations have only been measured in two cases and were normal. This would be inappropriately low in the face of a hypocalcaemia of sufficient severity to cause clinical signs.
Clinical signs and findings on physical examination reported in nine cats with idiopathic primary hypoparathyroidism
Histopathological examination of the parathyroid glands of affected cats has only been carried out in three cases. In all of these cases no parathyroid tissue (either internal or external) could be identified. However, one case showed a number of focal lymphocytic infiltrates within the excised thyroparathyroid complex suggestive of a lymphocytic parathyroiditis. The presence of clinical signs from two months of age in one case (Bassett, 1998) also raises the possibility of parathyroid agenesis as an aetiology. Given all these findings it would therefore seem likely that cases of feline hypoparathyroidism will be irreversible.
With life long therapy and careful monitoring primary hypoparathyroidism has an excellent prognosis. Initial management is usually carried out before the confirmation of the diagnosis, by PTH assay, as the majority of cases require emergency treatment for hypocalcaemic tetany or seizures. Calcium replacement, usually using calcium gluconate, may be carried out by intravenous (slow bolus or infusion) or subcutaneous routes. Suspected calcinosis cutis has been associated with the subcutaneous administration of calcium, even as diluted calcium gluconate (Ruopp, 2001). Given that 1,25-(OH)2D3 is necessary for the intestinal absorption of calcium and in turn PTH is required to activate vitamin D to 1,25-(OH)2D3, oral administration of calcium alone will not correct the hypocalcaemia. Therefore, as PTH replacement is not possible, long term management is with an active form of vitamin D, either 1,25-(OH)2D3 (calcitriol) itself, alphacalcidol (1α hydroxycholecalciferol, which only requires hepatic 25-hydroxylation, which is not PTH dependent) or dihydrotachysterol (a vitamin D analogue only requiring hepatic 25-hydroxylation). Calcitriol and alphacalcidol have a more rapid onset of action and shorter half life than dihydrotachysterol, but may be difficult to administer in the cat given the low doses required and the available preparations. Oral calcium supplementation is usually used while the cat is stabilised but can normally be tapered and discontinued as dietary calcium should be sufficient. Aluminium hydroxide was used as a phosphate binder to maintain normal phosphate concentrations in two of the nine reported cases (Bassett, 1998; Ruopp, 2001).
Iatrogenic hypoparathyroidism
Despite the rarity of naturally occurring primary hypoparathyroidism, iatrogenic hypoparathyroidism particularly after bilateral, surgical, thyroidectomy is well recognised. The assumption that this is solely due to hypoparathyroidism resulting from surgical trauma to the parathyroid glands or their vascular supply has recently been questioned (Graves, 1995). In a prospective study of human patients undergoing thyroidectomy, one of the factors most closely predicting post operative hypocalcaemia was the presence of an elevated free T4 concentration pre-operatively (as opposed to patients who were euthyroid at the time of surgery) and post operative decline in PTH concentrations was not found to be a significant risk factor (McHenry et al., 1994). No similar clinical studies have been conducted in the cat and calcium homeostasis in hyperthyroidism is significantly different from that in man, with hyperparathyroidism, presumably secondary in aetiology, occurring in 77% of hyperthyroid cats (Barber and Elliott, 1996). In contrast to surgical thyroidectomy, radioactive iodine therapy, although inducing a rapid decline in thyroid hormone secretion, has not been associated with clinical signs of hypocalcaemia. Hence, it is likely that parathyroid trauma during surgery may induce hypocalcaemia, but whether this is the sole mechanism remains unknown.
Secondary parathyroid disorders
In secondary parathyroid disorders a pathological state out with the parathyroid gland alters mineral homeostasis and the parathyroid gland responds in an appropriate manner. Initially these conditions are completely reversible if the underlying mineral imbalance is corrected. With chronicity however, atrophy or hyperplasia will limit the ability of the parathyroid glands to respond appropriately to acute changes in calcium homeostasis.
Suppression of parathyroid secretion will occur in hypercalcaemia, with eventual parathyroid atrophy if the hypercalcaemia is sufficiently severe and chronic. The vast majority (81.7% of 322 hypercalcaemic cats) of cats with hypercalcaemia have PTH concentrations in or below the lower half of the reference range (Provencher Bolliger et al., 2002), suggesting parathyroid independent hypercalcaemia (secondary hypoparathyroidism). The most frequent disorder associated with hypercalcaemia in the cat is neoplasia (Savary et al., 2000). Although HHM has been identified in cats (Provencher Bolliger et al., 2002) it is not known in what proportion of cats with malignancy associated hypercalcaemia this is mediated by PTHrP, direct bone lysis or other mechanisms. Other causes of hypercalcaemia in the cat include chronic and acute renal failure, vitamin D toxicosis and granulomatous disease.
Hyperparathyroidism occurs secondary to any condition that causes hypocalcaemia, vitamin D deficiency or hyperphosphataemia. However, many cases will have no or only mild hypocalcaemia as the increased PTH secretion acts to normalise plasma calcium concentrations. With a chronic stimulus varying degrees of parathyroid hyperplasia will occur. Parathyroid hyperplasia is of significance because each cell has a non-suppressible basal level of PTH secretion and so with hyperplasia, the non-suppressible component from gland as a whole will automatically increase. The two most commonly recognised conditions are renal and nutritional secondary hyperparathyroidism.
Idiopathic hypercalcaemia
An increasing number of cats are being identified with moderate hypercalcaemia with no obvious identifiable cause after routine investigation (Midkiff et al., 2000). Affected cats are young to middle-aged and long-haired cats appear predisposed. The hypercalcaemia is a true hypercalcaemia with increases in both total and ionised calcium. Clinical signs are either minimal or those of hypercalcaemia including anorexia, lethargy and polyuria and polydipsia. Some affected cats are azotaemic and evidence of nephrocalcinosis may be seen on imaging. Feline idiopathic hypercalcaemia has also been associated with calcium oxalate urolithiasis including ureteral uroliths. The response of the parathyroid glands is normal with plasma PTH concentrations being normal or low, and PTHrP usually undetectable.
Summary
A thorough understanding of the regulation of calcium homeostasis is required to interpret the pathophysiology of parathyroid gland disorders. The ability to measure circulating PTH, facilitates the differentiation between primary and secondary parathyroid disorders, allowing appropriate treatments to be instigated. Although primary parathyroid disease is still considered relatively rare, it is being recognised with increasing frequency in the cat. The availability of newer tools to investigate calcium homeostasis, including measurement of ionised calcium, assay of vitamin D and PTHrP and parathyroid imaging techniques, are likely to increase our understanding of these conditions and lead to the discovery of other more complex disorders of mineral balance.
Footnotes
1
Intact PTH, Nichols Institute Diagnostics. Intact PTH IRMA, Diagnostic Products Corporation.
