Abstract
Antibody microarrays are gaining popularity as a high-throughput technology to investigate the proteome. However, protein extracts from most body fluid or biopsy samples are available in very small volumes and are often unsuitable for large-scale antibody microarray studies. To demonstrate the potential for protein analysis with as little as a few nanoliters of sample, we have developed a new technology called NanoProbeArrays based on piezoelectric liquid dispensing for non-contact printing and probing of antibody arrays. Instead of flooding the protein sample on the antibody microarray surface, as in conventional microarray screening, a piezoelectric inkjet printer is used to dispense nanoliters of fluorescently labeled proteins over the antibody spots on the array. The ability of NanoProbeArrays to precisely identify and reliably distinguish between test proteins from different sources, without any loss of sensitivity and specificity as compared with conventional antibody microarrays, is illustrated here. The utility of NanoProbeArrays for biomarker identification in a complex biological sample was tested by detecting the cytokine interleukin-4 in serum. The significant reduction in volume of sample during NanoProbeArray analysis, as compared with conventional antibody microarrays, offers new opportunities for basic and applied proteomic research.
Keywords
Introduction
Proteomics, the global analysis of proteins, is currently one of the fastest growing areas of biomedical research. A quantitative study of the entire complement of proteins (or the proteome) provides a more accurate picture of the physiological state of a cell than the transcriptome, because mRNA levels do not always correlate with protein amounts. 1 Applications of proteomics range from basic research on the regulation of gene expression, protein networks, and biochemical activities, to applied research on drug discovery, biomarker identification, and clinical profiling of samples for detection of biomarkers. Although recent proteomic studies have provided a wealth of information in these areas, proteomics faces many challenges compared with genomic studies. Major protein differences can occur between organs, tissues, or even cells of an individual because of cellular processing of zymogens, post-translational modifications (such as glycosylation and phosphorylation), protein turnover, and cellular localizations. 2 The major technologies driving proteomics to address these challenges are mass spectrometry, two-dimensional gel electrophoresis, and protein microarrays. Microarrays, in particular, are gaining popularity as a high-throughput protein profiling technology. Protein microarrays consist of a library of proteins immobilized as discrete spots in an ordered manner on an array surface, such as a glass slide. A mixture of fluorescently labeled molecules (proteins, DNA/RNA, sugars, or other molecules) from a test sample is used to ”probe“ the microarray. Information obtained from the binding of these molecules to the specific proteins on the microarray is used to identify protein-protein, protein-DNA, protein-small molecule, and other molecular interactions. Antibody microarrays are a type of protein microarray where antibodies spotted on a substrate are used to detect protein antigens from serum, cell lysates, or other body fluids, and are particularly useful for protein expression profiling and biomarker identification. 3
Antibody microarrays are fabricated using specialized positioning robots designed to deposit antibody solutions at specific spots on microscope slides. Depending on whether the print head of the microarrayer makes physical contact with the microarray substrate, the method of printing can be classified as contact or non-contact printing. 4 The enabling technology for non-contact printing is piezoelectric ”inkjet“ dispensing. Piezoelectric inkjet dispensing has been around for more than 60 years, starting with Clarence Hansel's patent of 1950, which describes a “means of spraying liquids in fine jets by making use of the phenomenon which takes place when supersonic vibrational waves strike an exposed surface of the liquid.” 5 By first filling the inkjet device with a liquid and then applying a short voltage pulse to the piezoelectric element, an acoustic pressure wave is impulsively imparted to the liquid, causing a drop of liquid to be ejected from the nozzle. The voltage pulse applied to the piezoelectric element is typically less than 40 μs long. Negative pressure during the first half of the pulse actually pulls fluid slightly into the pipette, and then, positive pressure pushes it out with a velocity of the order of 1 m/s. Depending on the size of the nozzle and the voltage applied, drop volumes range from less than ten to hundreds of picoliters. 6
Piezoelectric inkjet printing is gaining importance as a tool used to create various kinds of immunoassays and for other biological applications, because it has significant advantages over conventional pin spotters for printing protein microar-rays. 7 Although pin spotters are mechanically simpler, there are major inherent technical limitations resulting in relatively slow printing speed, satellite spots, rings or “coffee mug” effects, inhomogeneous spots, misplaced or even absent spots, and spot contamination because of carryover. 4 Piezoelectric pipetting technology for printing protein microarrays overcomes many of the shortcomings of pin spotters, including mitigated evaporation issues because of sample-fluid confinement, faster dispensing because of non-contact ”on-the-fly“ dispensing, the ability to dispense on complex surfaces, and precision control of high-density spot deposition at defined positions. 7
We have developed unique capabilities for piezoelectric pipetting technology that enable variable programmable spot sizes using burst-mode (multiple drops for each round of printing) dispensing and precise positioning of the print head for targeting of defined coordinates on the microarray. 6 The latter capability enables reprinting spots with the same molecule to increase concentration or with a different molecule to study intermolecular interactions. In this article, we describe the application of these capabilities to address a major challenge for the protein microarray analysis of clinical samples, that is, the limited volume of protein samples. Protein extracts derived from biopsies or body fluids are very small in volume and often unsuitable for large-scale antibody microarray studies where, typically, a few milliliters of sample are necessary. We recently reported a new probing technology called NanoProbeArray, where just a few nanoliters of sample is adequate to screen an entire lectin microarray during glycosylation analysis. 8 Instead of flooding the probe sample on the microarray surface, as in conventional microarray screening, a piezoelectric printer is used to dispense nanoliters of labeled probe directly onto the protein spots on the microarray. Here, we describe the use of NanoProbeArray technology for screening of antibody microarrays with just nanoliters of antigen probe sample. Because low sample abundance is a major challenge for the realization of the clinical potential of protein arrays, 2,9 the NanoProbeArray technology proposed here overcomes this limitation and offers new opportunities for protein microarray research.
Materials and Methods
Overview
To develop a new technology to significantly reduce the volume of probe required for screening antibody arrays, we leveraged the precise, non-contact printing capabilities of a piezoelectric inkjet printer. Antibody microarrays printed using the piezoelectric inkjet printer were probed and analyzed by the newly developed NanoProbeArray method where just a few nanoliters of antigen is directly printed onto the antibody spots. For comparison, the same type of antibody microarray was also probed and analyzed by the conventional method of flooding the entire microarray surface with a few milliliters of antigen. As proof of concept for the identification of biomarkers from complex protein samples, we analyzed interleukin-4 (IL-4) in human serum using the NanoProbeArray method.
Fabrication of Antibody Microarrays Using Piezoelectric Inkjet Printing
The antibodies, goat anti-rabbit IgG, rabbit anti-goat IgG, rabbit anti-chicken IgY (AnaSpec, San Jose, CA), and anti-human IL-4 (R&D Systems Inc, Minneapolis, MN), at 1 mg/mL, were mixed with Triton X-100 at 0.05% (v/v) and printed on a nitrocellulose substrate (Oncyte Film Slides, Grace BioLabs, Bend, OR) using the PiXY piezoelectric inkjet printer (Engineering Arts, Tempe, AZ; Fig. 1A) and the ActivePipette (Engineering Arts; Fig. 1B) piezoelectric pipettes. Using the burst-mode dispensing (Fig. 1C), three separate rounds of printing were done for each spot, with each round consisting of 10 individual 150 pL droplets per round. After the three rounds, each spot was 200 μm in diameter (Fig. 1D) and received a total of 4.5 nL of 1 mg/mL antibody (4.5 ng). As shown later, printing the antibody in three separate rounds concentrates the antibody in a smaller spot than if it is printed all at once. Printing was performed at ambient temperature and humidity. Spots dried out in approximately 30 s after each round of printing. There was approximately 1 min time delay between each round to allow the spots to dry out completely before reprinting. The microarray pattern was configured to use the Proplate Multi-Array Slide System (Grace BioLabs) to form 12 individual well subarrays, each consisting of four identical rows of three spots per row. The piezoelectric dispensing parameters are similar to those described previously. 8 A surface consisting of watersensitive paper (Syngenta, Basel, Switzerland) was printed along with the nitrocellulose slides to visualize the spots and confirm that they were dispensed correctly. Printed slides were allowed to dry at room temperature, blocked with SuperBlock blocking buffer (Thermo Fisher Scientific, Waltham, MA) for an hour and stored dry at 4 °C before probing.

Piezoelectric liquid dispensing for printing and probing of NanoProbeArrays. (A) PiXY arrayer from Engineering Arts, Tempe, AZ. (B) Engineering Arts ActivePipette for piezoelectric liquid dispensing. (C) Dispensing ofdroplets from the ActivePipette orifice in burst mode. (D) Schematic illustration of NanoProbeArray spots and spot sizes.
Conventional Probing of Protein Microarrays
Printed antibody microarrays were probed by the conventional method of flooding the entire microarray surface with a few milliliters of antigen. Protein antigen probes, namely, normal rabbit IgG, goat IgG, chicken IgY (AnaSpec), and recombinant human IL-4 (BioClone Inc, San Diego, CA), were fluorescently labeled using the AnaTag HiLyte Fluor 555 dye (AnaSpec) according to the manufacturer's instructions. Labeled antigen probes were diluted in a range of concentrations from 0.25 μg/mL down to 0.0125 μg/mL in phosphate-buffered saline (PBS). Printed and blocked slides were washed twice with tris-buffered saline with 0.05% Tween (TBST) for 10 min each and attached to the Proplate Multi-Array Slide System. Each dilution of the probe solution was added to one Proplate well of the microarray, and the slide was incubated at room temperature for 1 h with gentle shaking. After incubation, the probing solution was carefully removed by pipetting, and the wells were rinsed once with TBST. The Proplate attachment was removed, and the slides were washed two more times with TBST. The slides were allowed to dry in the dark and scanned using the Axon 4000A scanner (Molecular Devices, Sunnyvale, CA) with an excitation wavelength of 532 nm. Data acquisition and analysis were done using the GenePix Pro 6.0 software (Molecular Devices). Local median intensity was used for background subtraction.
Non-contact Piezoelectric Probing of Protein NanoProbeArrays
The printed antibody microarrays were probed by the newly developed NanoProbeArray method (Fig. 2). Nitrocellulose slides containing printed antibodies were used for depositing the labeled protein antigen probes directly on the antibody spots using the piezoelectric liquid dispenser. Printed and blocked slides were carefully replaced at the same location on the microarrayer platform and checked for alignment. Using the PiXY microarrayer (Engineering Arts), 4.5 nL (30 drops of 150 pL) labeled probe was dispensed onto each antibody spot on the microarray. The diameter of the protein probe spot was 500 μm and completely overlapped the 200 μm antibody spot. Spot overlapping was confirmed using the previously printed water-sensitive paper control slide. The relative humidity during printing was maintained at 75%, the air temperature was 26 °C, and the temperature of the microarrayer printing surface was 18 °C. After printing the probe, slides were incubated inside the microarrayer for 30 minutes in the dark to prevent photobleaching of the fluorescent tags. The protein/antibody spots remained moist during incubation with no condensation. The slides were later washed with TBST, imaged with the Axon 4000A scanner, and analyzed as described earlier.
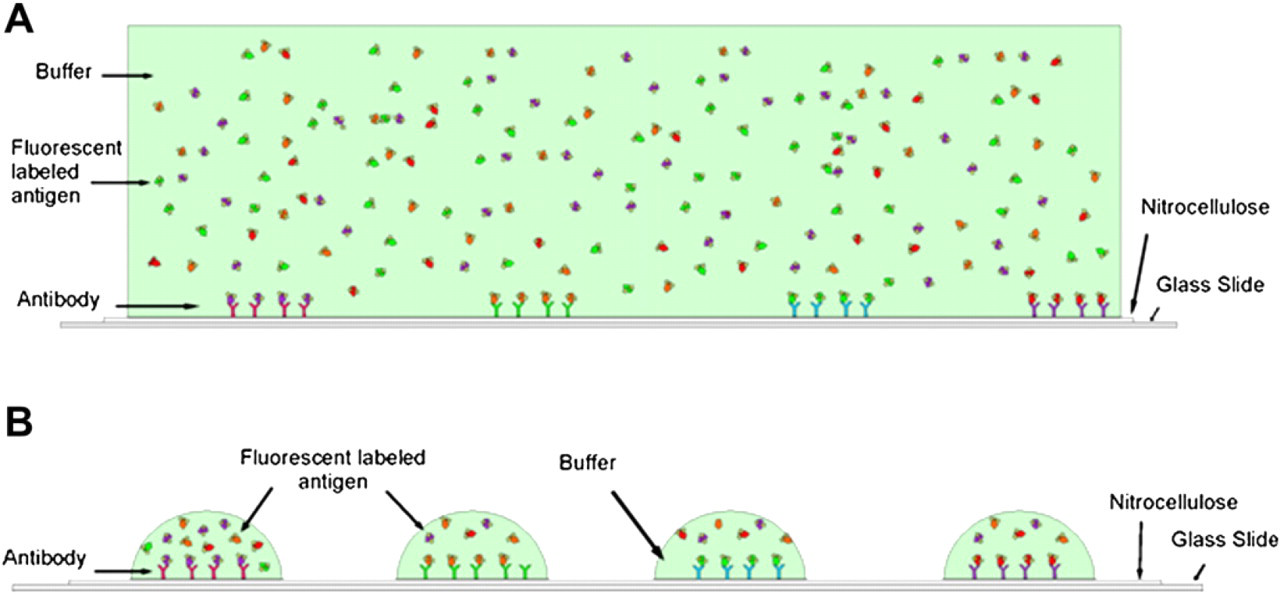
Cross-sections of (A) conventional microarray versus (B) NanoProbeArray. (A) Microarrays are fabricated by spotting antibodies in an ordered fashion on an array surface, such as nitrocellulose. Fluorescently labeled antigen solution is flooded on the surface of the array. After incubation to allow binding of the antigens with their corresponding antibodies, the slides are washed and scanned using a microarray scanner. (B) During NanoProbeArray analysis, instead of flooding the surface of the slide, a piezoelectric liquid dispenser is used to deposit 4.5 nL of labeled probe directly onto each antibody spot on the array. After incubation in controlled humidity and temperature conditions, the slides are processed in a similar manner as conventional microarrays.
Analysis of Interleukin-4 in Human Serum
Human Serum was obtained from United States Biological (Swampscott, MA). The serum was diluted 1:1 with PBS containing various amounts of fluorescently labeled IL-4. Printed antibody microarrays were probed with the IL-4 spiked serum by the conventional flooding method and by the NanoProbeArray method as described earlier.
Results and Discussion
The ability of protein microarrays to detect small changes in protein expression has made it a powerful, high-throughput screening technology to analyze the proteome. 1 Recent advances in the field of protein microarrays, such as improved surface chemistry for protein conjugation, microarray printing methods, sample processing, fluorescent labeling, slide imaging, and data analysis, have led to highly sensitive microarrays for proteomic research. One of the major constraints for broader applicability of protein microarrays is the inability to use low sample volumes for analysis. The development of efficient protein microarrays that use low sample volumes without compromising sensitivity and specificity is important for their use as high-throughput tools for the discovery, validation, and routine clinical identification of biomarkers. Currently available technologies for low-volume immunoassays for biomarkers use microfluidic platforms that require significant expertise and specialized laboratories. 10 By using the piezoelectric liquid dispensing technology to both print and probe antibody arrays, we devised a simple method to achieve several orders of magnitude reduction in the volume of sample required for microarray analysis.
Piezoelectric Inkjet Printing of Antibody Microarrays
Nitrocellulose has been traditionally used for studying antigen-antibody interactions using western blots and is also an excellent substrate for printing antibody microarrays. We used a commercially available nitrocellulose-coated glass slide with low fluorescence background for printing our antibody microarrays. Antibodies were dispensed on the nitrocellulose slide surface by non-contact printing using the PiXY microarrayer (Fig. 1). The antibodies irreversibly adsorb to the surface upon printing so that there is no need for any special conjugation chemistry. Fabrication of antibody microarrays with high precision is critical for the quantitative analysis of target analytes in the sample.
11
We first tested the ability of PiXY microarrayer to dispense quantifiable amounts of proteins with high accuracy, consistency, and concentration (Fig. 3). Figure 3A compares the actual spot diameter with the theoretical diameters as a function of the number of rounds of printing. The theoretical diameters assume that the entire volume is dispensed all at once without repeated printing and drying out between rounds. Calculation of the theoretical diameter, d, is based on the volume relationship—
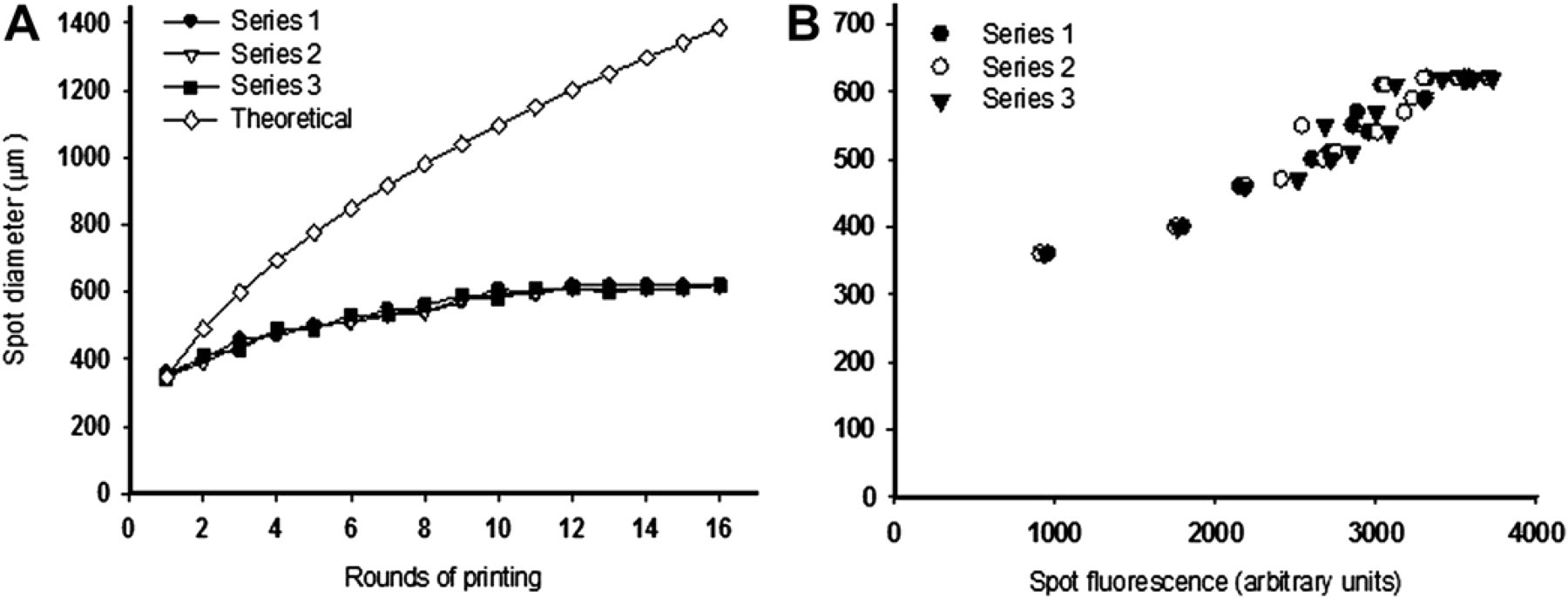
Programmable spot size and protein concentrations using piezoelectric liquid dispensing. Three nanoliters of fluorescently labeled rabbit immunoglobulin (1 mg/mL) was spotted on the nitrocellulose-coated glass slide multiple times (1–16). Spots were allowed to dry before reprinting. (A) Theoretical versus actual spot diameters as a function of the number of rounds of printing and (B) fluorescence intensity of spots as a function of the number of the rounds of printing. The experiments were done in triplicate to evaluate repeatability.
Conventional Antibody Microarray Analysis
To test the ability of the printed antibody microarrays to identify proteins and distinguish between similar proteins, we used immunoglobulin molecules from two different sources: rabbit and goat. We fabricated a microarray consisting of antibodies that recognize immunoglobulins from several different sources (see Materials and Methods). The microarray was probed with either fluorescently labeled rabbit or goat immunoglobulin. The probing was done using conventional methods where the microarray surface is flooded with the probe solution. As expected, we observed highly specific binding for both the rabbit and goat immunoglobulins with their corresponding antibodies (Fig. 4A and C). The lower limits of detection for both the immunoglobulins were less than 12.5 ng/mL, whereas all the concentrations tested were in the linear range of detection. We used a Proplate attachment on the antibody microarray to create wells and divide the slide into 12 blocks. A volume of 250 μL was used for each well, and about 4 mL of total probe volume was necessary for screening the entire microarray.
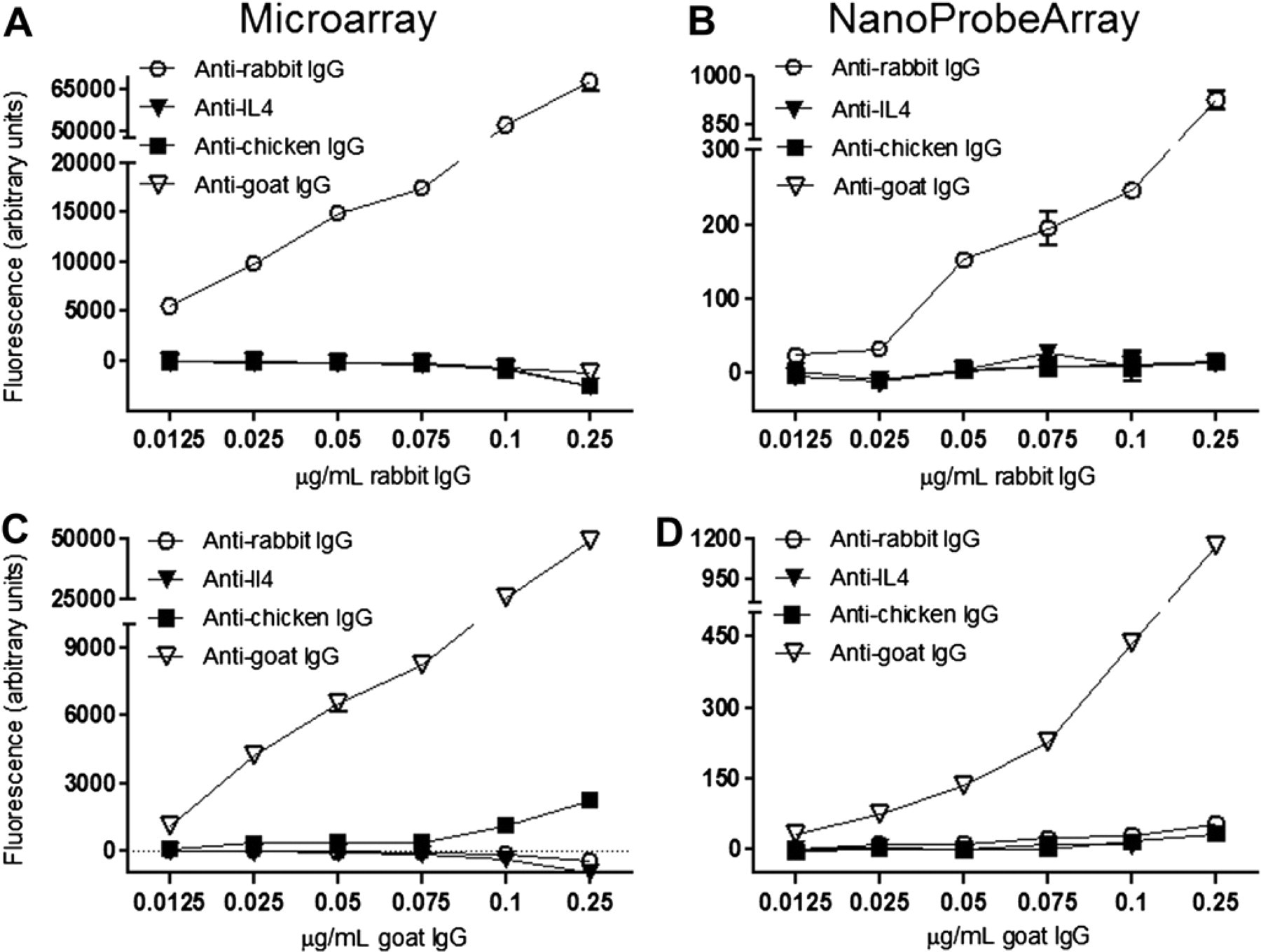
Comparison of conventional antibody microarray and NanoProbeArray analyses. Immunoglobulin proteins from rabbit and goat were fluorescently labeled with HiLyte Fluor 555 dye and used for probing the antibody arrays. The reactivity of antibodies (anti-rabbit, anti-IL-4, anti-chicken, and anti-goat antibodies) on the array with a range of concentrations for the rabbit and goat immunoglobulins was analyzed as described in Materials and Methods. All experiments were done in triplicates, and error bars represent standard error of the mean.
Piezoelectric Probing of Antibody NanoProbeArrays
We further exploited the unique non-contact printing capabilities of the piezoelectric liquid dispensing technology for probing the antibody microarray. Instead of flooding the protein probe on the microarray surface as in conventional microarray screening, the PiXY microarrayer was used to dispense the fluorescently labeled protein probe over the antibody spots on the microarray (Fig. 2). The ability of the PiXY microarrayer to precisely position and target probe samples toward predefined antibody spots on the microarray with less than 5 μm positional accuracy was found to be critical. A volume of exactly 4.5 nL (30 drops of 150 pL) containing antigens was dispensed to create a spot of 500 μm diameter over the 200 μm antibody spot. Both antigen and antibody spots received the same volume of 4.5 nL; however, the antigen spot was larger, because it was dispensed all at once instead of in three separate rounds. Precise control of air temperature and relative humidity along with a temperature-regulated microarrayer surface set to the dew point was used to ensure that arrayed spots remained wet during printing and subsequent incubation to create an aqueous environment for the proteins to interact with the antibodies.
The binding pattern of piezoelectric, liquid-dispensed protein probes to the antibodies during NanoProbeArray analysis correlated very well with the results from conventional microarray probing experiments without any loss of sensitivity or specificity (Fig. 4). However, the major difference was a significant reduction in the amount of probe required for probing the microarray. We used about 4 mL to probe a microarray with conventional probing methods, whereas the NanoProbeArray required less than 1 μL or 4000 times less volume. The amount of sample required by NanoProbeArrays is dependent on the number of spots on a microarray. For example, to screen a sample for reactivity to 1000 antibodies on a NanoProbe Array requires less than 5 μL volume using the current method. Because piezoelectric liquid dispensing allows accurate dispensing of less than 100 pL, the amount of probe can be further reduced for higher-density antibody NanoProbeArrays. Commercial desktop inkjet printers can dispense droplets as low as 4 pL, resulting in feature sizes of just a few microns. Because assay sensitivity and volumes are inversely related, 13,14 NanoProbeArray analysis on 4 pL spots can potentially lower the limits of detection to a single molecule.
Identification of Serum Biomarkers Using NanoProbeArray
Human serum is the most frequently used clinical sample for biomarker detection. 15 However, the detection of the biomarker of interest during immunoassays from serum is challenging, because serum is a complex biological mixture often containing a vast excess of molecules that can interfere with immunoassays. To test the ability of NanoProbeArrays to analyze complex samples we used 50% human serum (v/v in PBS) and spiked it with various amounts of recombinant human IL-4. Interleukins are signaling molecules primarily used as mediators of intercellular communication by immune cells to influence cell proliferation, immune cell activation, inflammation, and physiological changes. IL-4 is released by a subset of CD4 cells and helps stimulate antibody production. Serum cytokine levels, including that of IL-4, are used as a marker for several physiological and pathological conditions. We were able to successfully detect fluorescently labeled IL-4 in serum using NanoProbeArrays at concentrations greater than or equal to 25 ng/mL (Fig. 5). Slightly better linearity was observed over a range of concentrations for pure IL-4 (diluted in PBS) using NanoProbeArray probing versus conventional microarray probing (Fig. 5).
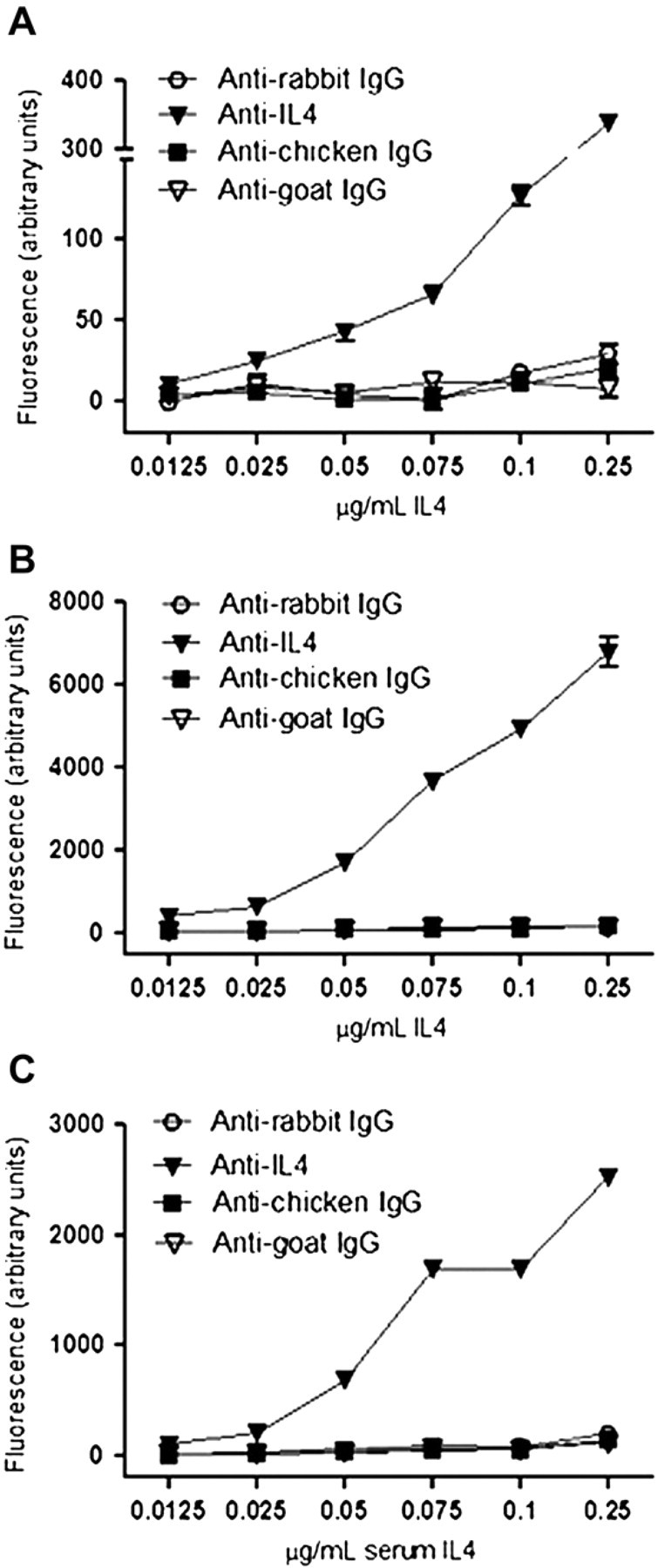
Detection of serum biomarkers using NanoProbeArray. Various concentrations of fluorescently labeled recombinant human IL-4 in PBS was used for conventional microarray (A) and NanoProbeArray analyses (B). The ability to detect biomarkers in complex biological fluids using NanoProbeArray was tested using serum spiked with various concentrations of fluorescently labeled IL-4 (C).
Proteomic Applications of NanoProbeArrays
Proteins lack the ability to be amplified like DNA, prohibiting the use of low-abundance samples for conventional protein-antibody microarray analysis. For example, human body fluids (lymphatic fluid, cerebrospinal fluid, tumor interstitial fluid, tear drop, pin prick, breast nipple discharge, saliva, semen, vaginal secretions, and others), biopsies, and tissue cell aspirates are highly valuable samples but available in limited amounts. Similarly, the small amount of human proteins derived from histopathologically relevant pure-cell populations obtained from laser capture microdissection, flow cytometry-sorted cells, two-dimensional gel fragments, or column-separated protein fractions, is generally a limiting factor for screening antibody microarrays. Model organisms for scientific research, such as the fruit fly (
Using the newly developed NanoProbeArray technology, a microarray consisting of several hundred antibodies can be probed with less than 1 μL of sample. To the best of our knowledge, no other immunoassay currently available is able to use such small sample-probe volumes. For basic and applied research, the same inkjet printer can fabricate and probe the microarray in a high-throughput fashion with no need for additional equipment or reagents. For screening patient samples in a clinical setting, a simple commercial benchtop inkjet printer can be designed with a single inkjet print head to probe antibody arrays with a small amount of patient sample. The benchtop model with temperature and humidity control as well as the capability for either mechanical or optical registration of spots will be necessary for NanoProbeArray analysis. Unlike DNA/RNA microarrays, the binding of each antigen to its corresponding antibody on the microarray requires unique salt and pH conditions. Because the antigen-antibody interactions occur in discrete spots during NanoProbeArray analysis, adjacent spots can be probed in completely different buffers. For the detection of antigens that are low in abundance, depletion of abundant proteins followed by sample concentration can improve sensitivity. Further increases in sensitivity of antigen detection may occur during NanoProbeArray analysis as a result of miniaturization, as explained by the Ekins' theory. 13,14 In addition, the entire printing and probing is automated, saving time and minimizing variations because of user handling. NanoProbeArrays are also compatible with label-free detection methods, such as surface plasmon resonance or electrochemical impedance spectroscopy.
Acknowledgments
The authors thank Jamison Rupnik and Erica Arroyo for technical help with experiments and the Biodesign Institute at Arizona State University for financial support of this work.
