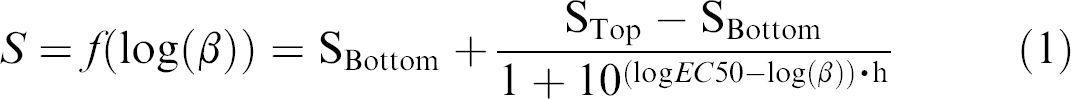Abstract
Robotic liquid-handling systems can be equipped with disposable pipetting tips or fixed reusable pipetting tips. The use of disposable tips is perceived as the best option to avoid carry over (CO) of analyte from sample to sample. We recently developed standardized CO test procedures that allow precise and reproducible quantification of CO for fixed reusable tips. We used these test procedures to reduce CO of the analytes fluorescein, IgG, and hepatitis B surface antigen (HBsAg) to minimal levels. Variations of washing intensity, using water as a washing solution, did not lower CO below acceptable target levels. These target levels would preclude a false-positive detection of IgG and HBsAg in human serum when a negative sample is measured subsequent to a sample with a high analyte concentration. We therefore integrated a decontamination step into the washing procedure. Screening of 12 decontamination solutions showed that sodium hypochlorite solution was very efficient in reducing CO. Optimization led to a final washing routine in which tips are exposed for 0.2 s to 0.17 M NaOCI and subsequently rinsed with 2 mL of water to remove any remaining decontaminant solution. The washing procedure only takes 15 s and is thus suitable for high-throughput applications. The procedure was able to lower CO of IgG and HBsAg in human sera below relevant levels. The decontamination step with hypochlorite can easily be integrated into different liquid-handling systems and is likely to be effective against CO of most proteins and peptides.
Introduction
Robotic liquid-handling systems have become standard for clinical in-vitro diagnostic analyzers and in pharmaceutical, biotechnological, forensic, environmental, and further research laboratories. Two options for pipetting tips are available for most liquid-handling systems. Pipetting instruments can be equipped with either fixed reusable tips, usually made out of stainless steel or alternatively with adapters allowing the use of disposable plastic tips. Disposable tips for robotic use require conductive materials to allow capacitive liquid-level detection and must fulfill stringent tolerance criteria to allow consistent tip pick-up. The use of disposable tips can thus substantially increase assay costs, as assays usually require a large number of pipetting steps. 1 In addition, assay throughput can be limited, as additional space on the worktable must be allocated for tip racks. In the fixed tip set-up, a washing station has to be integrated to allow the tips to be washed between pipetting steps. Carry over (CO), which is defined as the process by which samples or reagents are carried into a reaction mixture to which they do not belong, 2 is perceived as a major problem for fixed tip set-ups, and therefore the use of disposable tips is often preferred. 3 Sample-to-sample CO (also termed sample cross-contamination) can, for example, occur when a sample containing a high analyte concentration precedes one with a low (or zero) analyte concentration.
Accordingly, liquid-handling system users have to carefully optimize the pipetting and washing procedures for each analyte to ensure that CO does not significantly affect results. The absence of clinically significant CO in commercial in-vitro diagnostic analyzers that use fixed reusable tips has been reported. 4 11 However, in these publications, pipetting and washing procedures are not described in detail. The elimination of CO of nucleic acids is particularly challenging because of highly sensitive amplification methods. 12 Although extremely low levels of CO can lead to false-positive results, fixed tips can be used for such analyses. 1 13 14 Frégeau et al. 1 13 showed that careful optimization of washing procedures and integration of sodium hypochlorite decontamination steps allow the use of fixed tips during automated processing of forensic samples for DNA profiling.
Liquid-handling systems with reusable tips have found widespread use; nonetheless, we are not aware of published studies that have systematically investigated CO for analytes other than nucleic acids. Briem et al. 15 reported that CO of two organic molecules of about 400 Da molecular mass in plasma was eliminated by thorough washing of the tips in DMSO; however, no details were given.
Together with a consortium of three liquid-handling system suppliers, we have recently established and validated test procedures for the quantification of CO of three model substances; the small organic molecule fluorescein; the protein IgG; and, as a model for a lipid—protein complex, the hepatitis B surface antigen (HBsAg) particles. 16 In these test procedures, tips were first exposed to a solution with high analyte levels. After a washing step, a recipient buffer solution was aspirated with the washed tips, and the CO was determined by measuring the analyte concentration in the recipient buffer solution. Standardization of all parameters allowed the reproducible quantification of CO. 16 For this article, we used these test procedures for evaluating the effectiveness of different washing procedures, tip coatings, and agents to lower CO below relevant levels.
Materials and methods
Liquid-Handling System
CO was measured on a commercial liquid-handling system (Freedom EVO; Tecan, Mannedorf, Switzerland) equipped with eight pipetting channels, a membrane pump, and a wash station (Low Volume wash station, Tecan). This wash station is divided in three parts. A rectangular trough with an outlet is used as the waste position. The other two parts are both composed of eight wash cavities, each with a 4-mm diameter. In one part, the eight wash cavities have a depth of 4.0 cm (shallow reservoir); in the other part, the eight wash cavities are 8.5-cm deep (deep reservoir). In this deep reservoir, an inlet is integrated in the bottom of the wash cavity and allows a flow of system liquid around the tip immersed in the wash cavity. In addition, system fluid can be expelled through the tips at high speed to wash the inside of the tip, while creating turbulence in the wash cavity to facilitate the removal of aggregate material and liquid adhered to the outside of the tip. 1 Accordingly, three variants for the washing of the tips in the wash cavity are possible (1) through the inside of the tip alone, (2) through the bottom inlet alone, thus washing only the outside of the tip, or (3) both together. Only the waste position and the wash cavities in the deep reservoir were used in this study. Four channels were equipped with fixed multiuse tips, and the remaining four channels were equipped with adapters for disposable tips, which were used for pipetting of calibration samples, reagents, and diluents.
Fixed tips for different liquid-handling systems have different geometries. Therefore, special tips with simplified tube geometry (length: 160 mm; outer diameter: 2 mm; and inner diameter: 0.8 mm) were used for optimization studies to allow transferability of methods to the different liquid-handling platforms and comparability of results. In the following, these tips are described with the term “tube geometry.”
Measurements of CO of analytes in serum were performed with standard multiuse tips from Tecan or tips with the same geometry as the standard Tecan fixed tips. These tips are 155-mm long with 2-mm outer diameter and 1.5-mm inner diameter. The last 12 mm of their length is narrower, with an inner diameter of 0.5 mm. In the following, these tips are described with the term “standard Tecan geometry.” The outside of the Tecan stainless steel standard tips is coated with a polytetrafluorethylene (PTFE)-based fluorpolymer; the inside is not coated.
The effects of different coatings were evaluated with SS tips (both tube and standard Tecan geometry), which were either left uncoated (uncoated stainless steel, USS) or coated inside and outside with FP1, sodium silicate glass (SSG) or a silicone resin coating (methyl-modified silicone resin [MSR]). FP1 is a PTFE-based pigmented fluorpolymer coating with antiadhesive properties, hydro-/oleophobicity, high hardness, and chemical stability, in particular, against some critical organic solvents like DMSO and acetonitrile. SSG is a sol—gel derived, low temperature curing, pure sodium silicate glass coating. The MSR has antiadhesive properties. All coatings are products of Surface Contacts GmbH, Saarbrücken, Germany.
The liquid-handling systems from Tecan, Hamilton, and Sias use demineralized or reversed osmosis water as washing solution. In the Freedom EVO liquid-handling system, demineralized water is used as the system liquid for pipetting and as washing solution.
CO Measurement Procedure
CO was provoked using a three-step procedure, as recently described. 16 This procedure includes (1) aspiration and dispense of a solution with a high analyte concentration, (2) a washing step, and (3) aspiration and dispense of a buffer solution in which the concentration of analyte is subsequently quantified. Tips with tube geometry were immersed in the analyte stock solution (140 μL per well of a 96-well plate; see the Results section for the specific stock concentrations used) with a tip end position 0.75 mm above the bottom of the plate, resulting in an immersion depth of 3.5 mm. Analyte stock solution (70 μL) was aspirated at an aspiration speed of 150 μL/s, which resulted in an aspirated column height of 139.3 mm. The analyte solution was kept in the tips for 5 s and was then dispensed back into the same well with a dispension speed of 600 μL/s. Tip position remained at 0.75 mm above the bottom of the plate for another 5 s before the tips were retracted from the solution without a recurrent aspiration. For measurements with tips with the standard Tecan geometry, volumes were adapted: tips were immersed into 290 μL of analyte stock solution resulting in an immersion depth of 7.4 mm, and 190 μL of analyte solution was aspirated resulting in an aspirated column height of 118 mm.
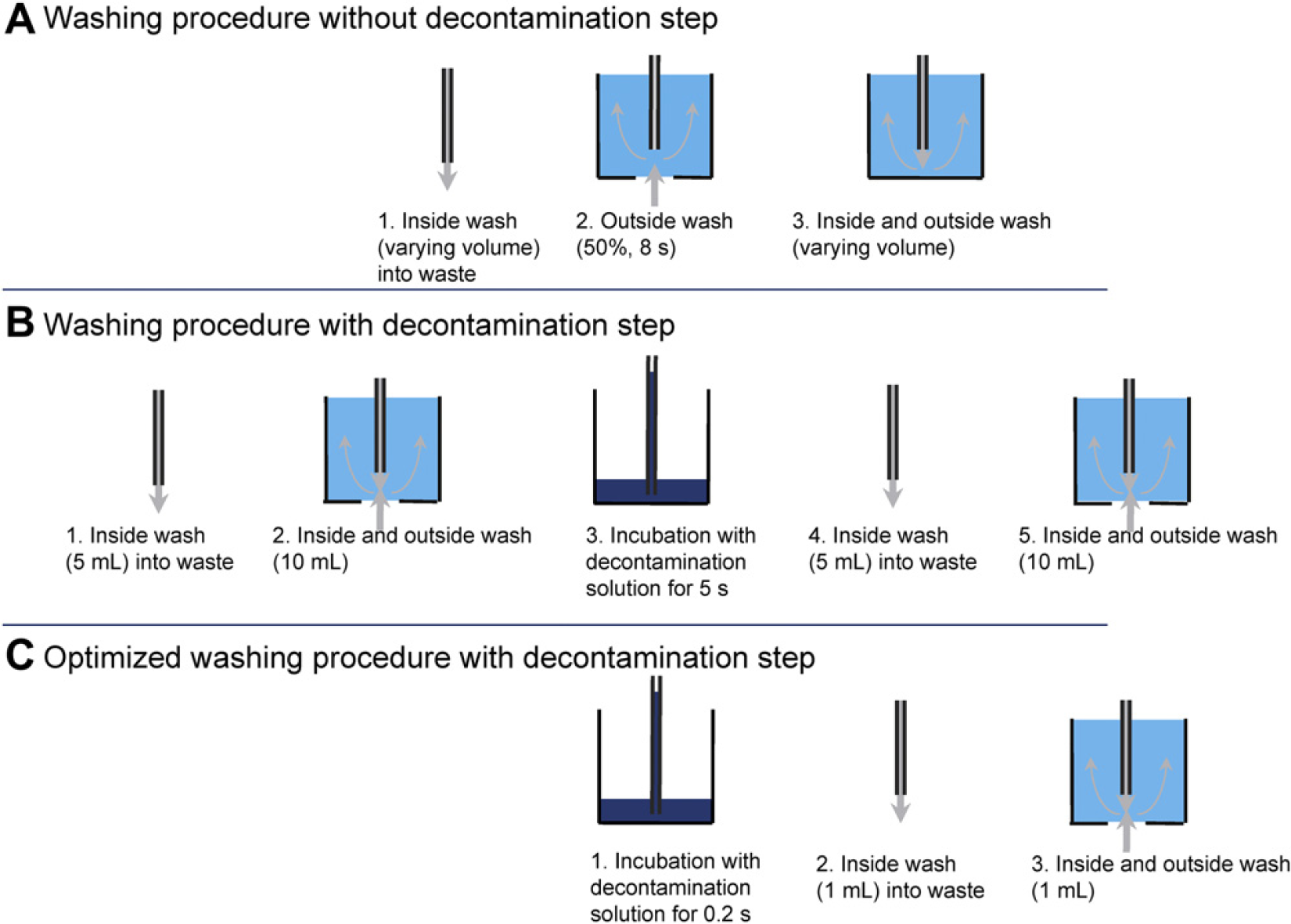
For measurements of CO of IgG and HBsAg in human serum without a decontamination step, the tips were washed using a three-step procedure: First, the insides of tips were rinsed with 13 mL of wash solution with the tip positioned above the waste position (speed 0.9 mL/s). In the second washing step, the outer surfaces of the tips were washed in the wash cavities using the membrane pump (8 s, intensity 50% corresponding to 150 mL). In the third washing step, the tips were washed inside and outside by pumping 13 mL of washing solution through the tip with the tip positioned in the washing cavity. Five CO measurements were subsequently performed, with decreasing washing solution volumes from 26 mL (13 mL into waste and 13 mL in wash station) to 11, 6, 3.5, and 2 mL.
For optimization of the washing procedure, a decontamination step was integrated. Tips were first rinsed with 5 mL of washing solution with the tip positioned in the waste trough (membrane pump; speed 0.9 mL/s) followed by a washing step with 10 mL of washing solution in the wash cavities. In this step, the washing solution was pumped simultaneously through the tips and from the bottom inlet of the wash cavity. Thereafter, the tips were moved to a 96-well plate that contained 300-μL decontamination solution per well. The decontamination solution (71 μL) was aspirated with tips with tube geometry, kept in the tip for 5 s and then dispensed into the same well. The decontamination solution (200 μL) was aspirated for tips with standard Tecan geometry. The decontamination solution in each well was only used once. Finally, the tips were washed again in the washing station by pumping 5 mL of washing solution into the waste and performing a washing step with 10 mL of washing solution in the wash cavities. Four different volumes were tested for the washing step that was performed before the decontamination step: 15 mL (5 into waste plus 10 in wash cavities), 1.5 mL (0.5 plus 1), 0.16 mL (0.08 plus 0.08), and 0 mL. The washing step after the decontamination step was varied using four different volumes of solution: 15 mL (5 plus 10), 4 mL (2 plus 2), 2 mL (1 plus 1), and 0 mL. The incubation time for the decontamination solution was varied between 5, 1, and 0.2 s. Two subsequent aspirations for 0.2 s were also tested.
After the washing procedure, tips were positioned in a second 96-well plate that contained 150 μL of buffer per well (0.75 mm above bottom of the plate). The buffer (71 μL) was aspirated with tube geometry tips, kept in the tips for 5 s and was then dispensed into the same well. The tip position remained at 0.75 mm above bottom of the plate for another 5 s before the tips were withdrawn from the solution without a recurrent aspiration. Tips with standard Tecan geometry were immersed into 300 μL of buffer, and 200 μL of buffer was aspirated.
For the decontamination step, the following decontamination solutions were used: 1 M sodium hydroxide (Riedel-de Haën, Seelze, Germany), 1 M sulfuric acid (Fluka), 70% ethanol (Fluka), 8 M urea (Merck), water, phosphate-buffered saline (PBS) with 0.05-1% Triton X-100 (Sigma-Aldrich), PBS with 1% Tween 20 (Sigma-Aldrich) and 0.17 to 1 M sodium hypochlorite (Riedel-de Haën; diluted from a solution with 2 M active chlorine [15.2%]). The concentration of active chlorine was determined by iodometric titration. Briefly, potassium iodide is oxidized by sodium hypochlorite to iodine, which is back titrated with a standardized sodium thiosulfate solution using starch as indicator. The active chlorine content of the hypochlorite solution can be calculated from the amount of thiosulfate needed for back titration.
Quantification of analytes was performed similarly as described previously. 16 Fluorescein was quantified by fluorescence spectroscopy. Human serum was inactivated at 60 °C for 30 min. HBsAg (Genovac, Freiburg, Germany) in human sera was measured by enzyme-linked immunosorbent assay (ELISA) using the Enzygnost HBsAg 5.0 kit (Dade Behring, Deerfield, IL), which was adapted for quantification of HBsAg. As human serum contains human IgG with different specificities, we spiked human IgG with murine IgG and used a sensitive murine IgG-specific ELISA 16 for quantification of CO. This mimics the situation when, for example, virus-specific IgG is present in human serum after an infection or after a vaccination. Human and murine IgG have the same composition of two heavy and two light chains, similar amino acid composition, and similar glycosylation.
The lower limit of detection (LLOD) for IgG in serum was 9 pg/mL, and for HBsAg, it was 0.27 ng/mL.
Quantifications
with β being the concentration of the analyte (ng/mL), S the signal (optical density or fluorescence), SBottom the signal at analyte concentration of 0 ng/mL, STop the signal at analyte saturation, EC50 the effective concentration of analyte to reach 50% of the signal, and h the slope of the curve. GraphPad Prism 5.0 was used to calculate the 4PL fits. Sample concentrations were determined from the 4PL fit of the standard curve. To determine the LLOD and the lower limit of quantification (LLOQ), a blank (the respective assay buffer) was measured six times. LLOD was calculated from the average signal of the six measurements plus three standard deviations thereof. LLOQ was defined as five times the LLOD.
As CO values may be very small, pCO was defined as the negative logarithm of CO.
Results
Definition of Acceptable CO
Sample-to-sample CO occurs when a sample with a high analyte concentration is pipetted before a sample with low analyte concentration, and analyte is transferred from the high to the low concentration sample. This can lead to a false-positive result or an overestimation of analyte concentration in the recipient sample. The magnitude of CO depends on the concentration of analyte in the high sample. The definition of acceptable CO has to be performed on a case-by-case basis for each assay. For example, for most clinical in-vitro diagnostic assays, a concentration threshold is defined, and above that threshold, a sample is judged to be positive. Obviously, CO from a sample with a very high analyte concentration should not increase the concentration in the recipient sample above this threshold.
HBsAg is determined in human sera for diagnosis of a potential hepatitis B virus infection. The lower limit for a positive diagnosis is 0.3 ng/mL HBsAg when using the Enzygnost HBsAg 5.0 kit. 17 Serum samples from patients who are infected with hepatitis B virus can contain up to 510 ng/mL HBsAg. 17 18 Accordingly, an HBsAg-free sample would become rated as positive if 0.3 ng/mL HBsAg or more was transferred from a sample with a high HBsAg concentration. This corresponds to a CO of 1/1700. Including a safety factor of 100, the target CO should be a maximum of 1/170,000, which corresponds to a target pCO value of 5.2. The safety factor of 100 was arbitrarily chosen to guarantee a sufficient safety margin for the avoidance of detecting false-positive samples.
IgG concentrations are important for a number of clinical indications. Prophylactic vaccinations against bacterial and viral infections, such as diphtheria or polio, induce antigen-specific antibodies. The concentration of these antibodies in serum is predictive for protection against a potential infection. Antigen-specific antibody concentrations after vaccination can be as high as 200 μg/mL while the LLOD of a typical antibody determination is about 10 ng/mL (P. Maurer, Cytos Biotechnology AG, personal communication). Assuming a safety factor of 100, a maximum target CO value is 1/2,000,000, which corresponds to a target pCO of 6.3.
In our CO test procedure, we use an analyte-free sample as the recipient sample. If such a sample is not available, a sample with an analyte concentration close to the LLOD of the assay should be used as the recipient sample.
CO of IgG in Human Serum without a Decontamination Step
CO was provoked using a standardized three-step procedure as described previously. 16 Firstly, tips were exposed to highly concentrated IgG stock solution in human serum to allow adsorption of IgG to the tips. Thereafter, the tips were washed using a defined washing procedure with water as the washing solution. Finally, the tips were exposed to an analyte-free solution (human serum) to allow CO of the remaining analyte to occur.
We first tested the effect of the IgG stock concentration on CO. After provoking CO by aspiration of a 400 μg/mL IgG stock solution, an IgG concentration of 10 ng/mL was detected in the recipient solution (Fig. 1A). This corresponds to a pCO value of 4.6. When 100-fold and 10-fold diluted IgG stock solutions were used, the concentrations in the recipient solutions were also reduced 100-fold and 10-fold, respectively. Because the pCO value is defined as the ratio of analyte concentration in recipient and stock solutions, pCO values for the 100-fold and 10-fold diluted stock solutions were also about 4.6 (Fig. 1B).
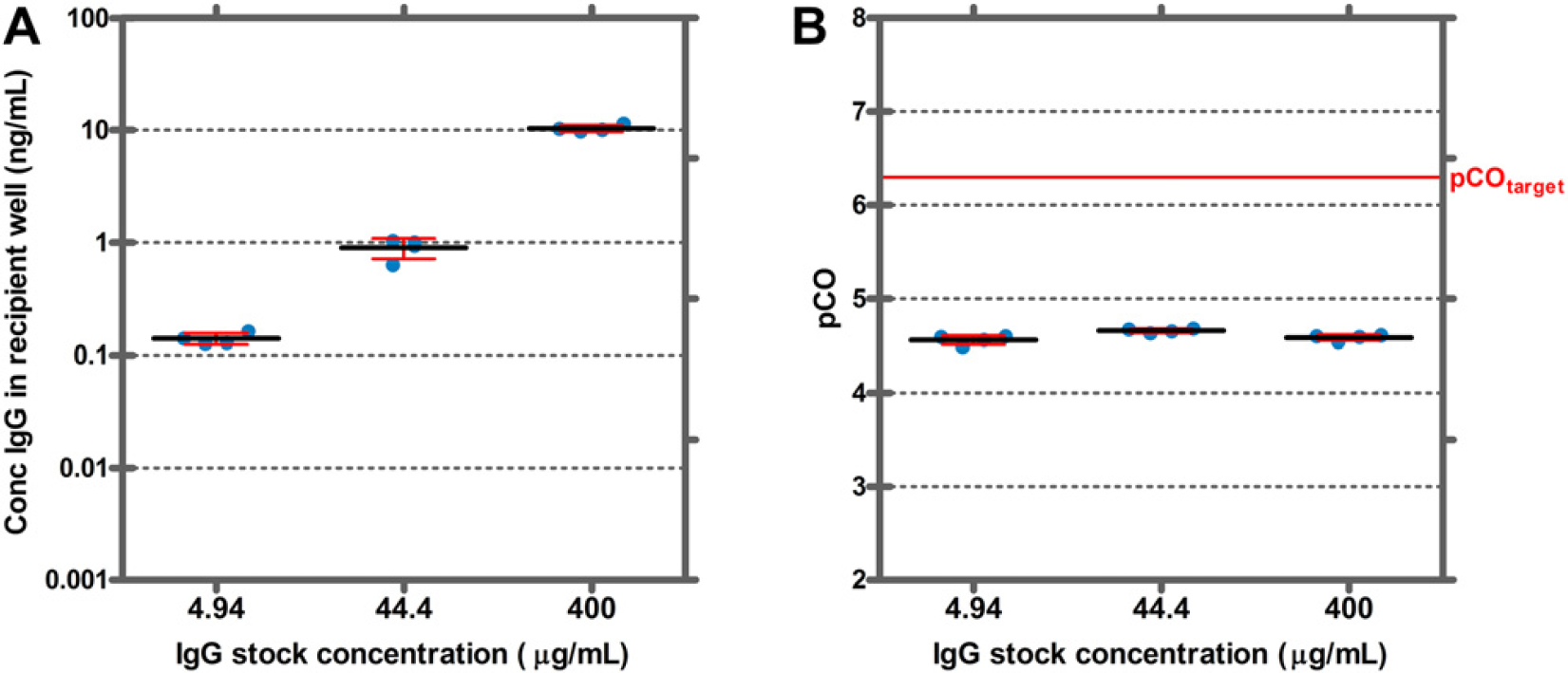
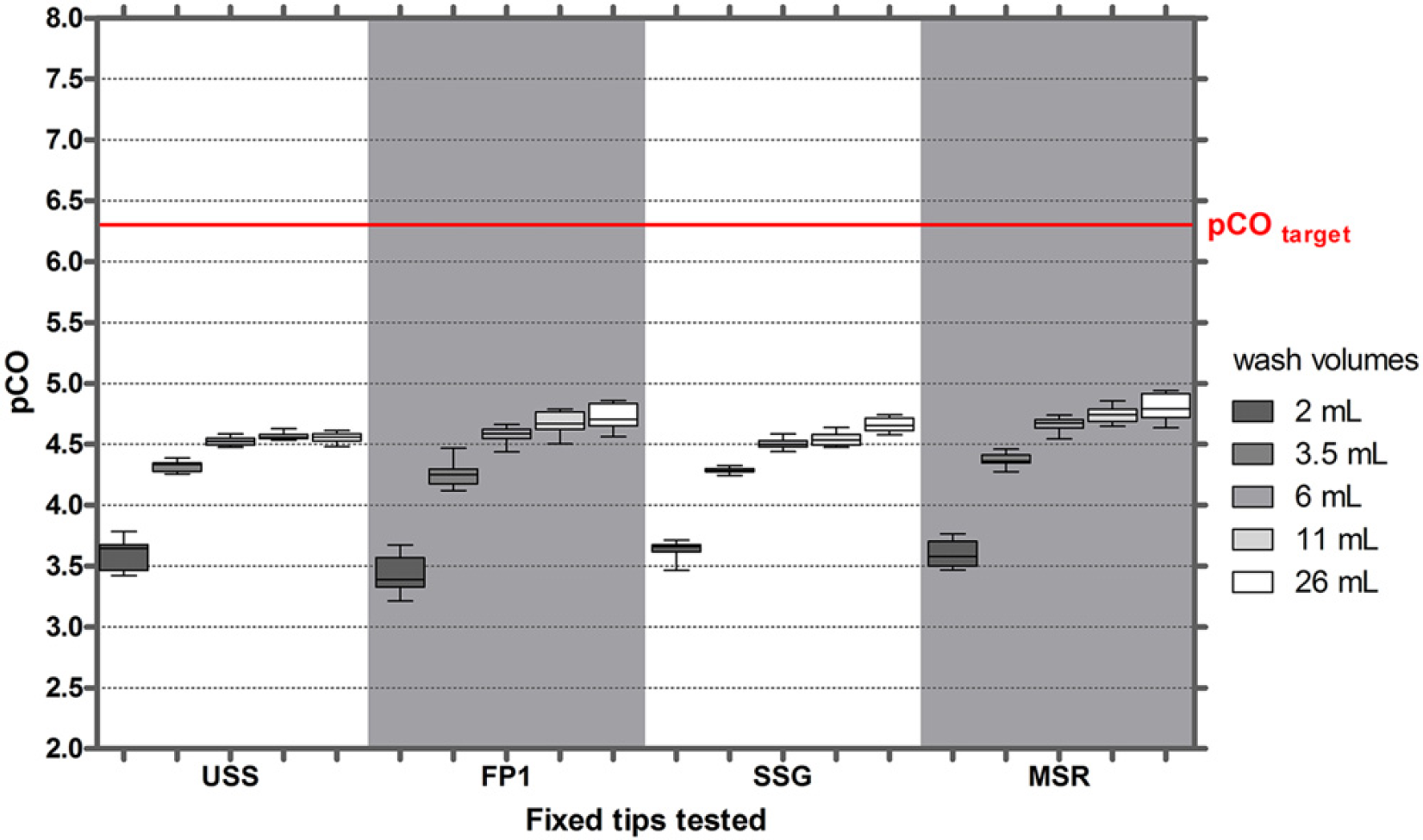
To investigate the effect of washing intensity on CO of IgG in serum, five different washing solution volumes were analyzed within one CO measurement run. In addition, we tested USS tips and stainless steel tips coated with FP1, SSG and MSR. Figures 2 shows that the target pCO value of 6.3 for IgG in serum was not reached even when the tips were washed with large volumes of water. At the lowest washing intensity, pCO values were about 3.5 and increased to about 4.7 at higher washing intensities. No significant differences were observed for uncoated tips and the three types of coated tips.
CO of HBsAg in Human Serum without a Decontamination Step
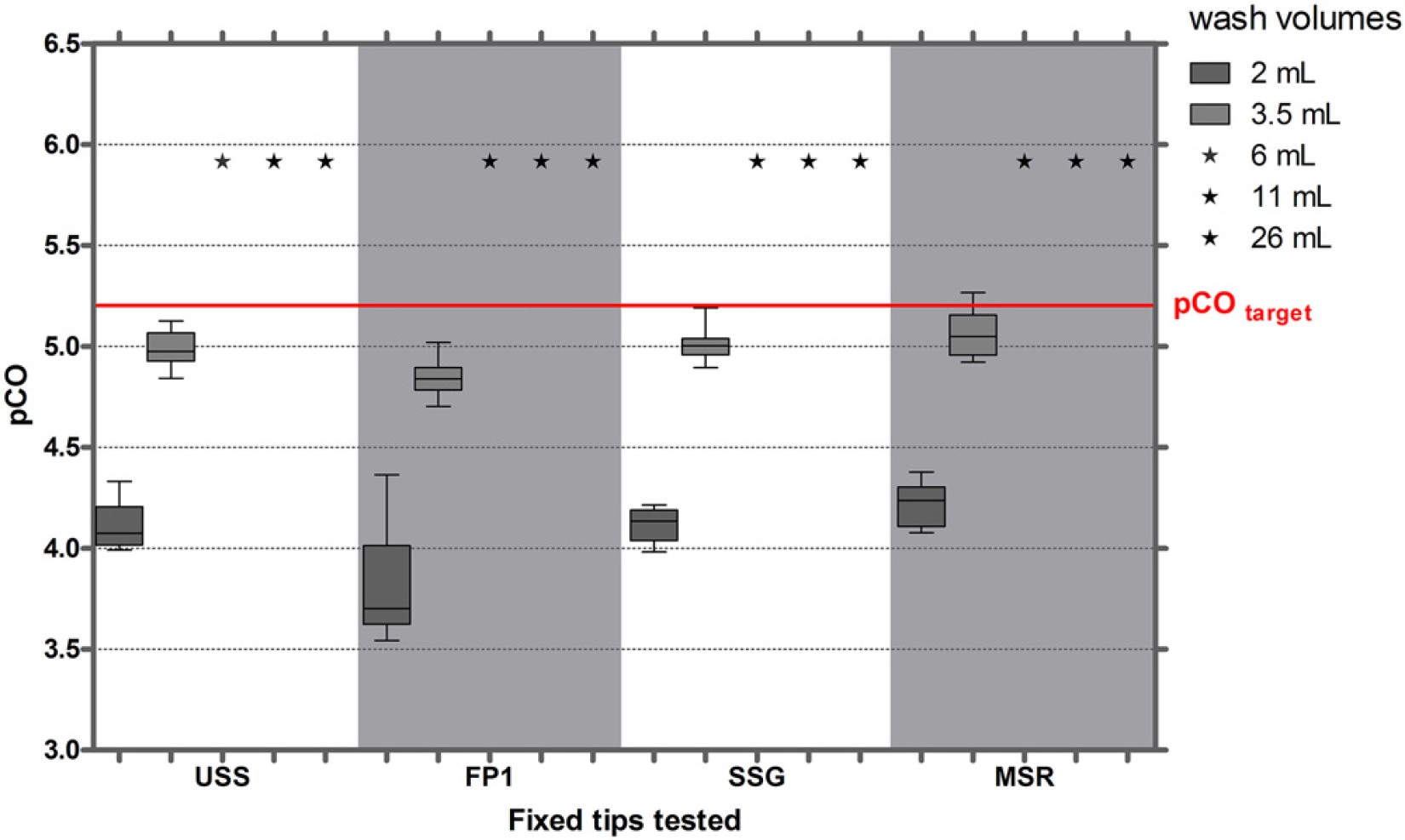
The CO of HBsAg in serum was analyzed using the same procedure as used for IgG. With a washing volume of 2 mL, pCO values of approximately 4 were detected, which were clearly below the target pCO values of 5.2. Increasing the washing volume to 6 mL or more, lowered CO to below the target values (Fig. 3).
Development of a Washing Procedure with an Integrated Decontamination Step
The use of water as a washing solution was not sufficient for reaching the target pCO values for IgG; we tried to improve the washing procedure by integrating a decontamination step. To this end, we screened 12 decontamination solutions for their ability to lower CO of IgG. Solutions with high and low pH values (1 M NaOH and 1 M H2SO4), detergents (0.05% Triton X-100 in PBS, 0.1% Triton X-100 in PBS, 0.5% Triton X-100 in PBS, 1% Triton X-100 in PBS, and 1% Tween 20 in PBS), an agent which solubilizes protein aggregates (8 M urea), a solvent (70% ethanol), an oxidizing agent (1 M NaOCl), water, and PBS as controls.
In the initial screenings, the tips were exposed to solutions with high analyte concentrations by aspirating 70 μL. In the subsequent washing procedure, the tips were first washed with 15-mL water. Then 71 μL of decontamination solution was aspirated, kept in the tip for 5 s, and dispensed. The additional volume of 1 μL, which led to a 2 mm higher liquid level in the tips, ensured that the liquid-level of the decontamination solution was higher than the liquid level of the analyte stock solution. Thereafter, the tips were washed again in the washing station with 15-mL water to remove any remaining decontamination solution. All washing steps were performed at 0.9 mL/s. CO of the remaining analyte into the recipient plate containing analyte-free buffer was then performed, as described in Material and Methods.
The CO values for IgG for the 12 decontamination solutions and four tip coatings are shown in Figure 4. CO values varied by a factor of approximately 10,000. With USS tips and water as a decontamination solution, a pCO value of about 3 was determined, while a 1 M NaOCl solution reduced CO to a pCO value of 6.6. The 1 M NaOCl solution resulted in the lowest CO for all coatings, whereas the other decontamination solutions produced differing results, depending on the coating used.
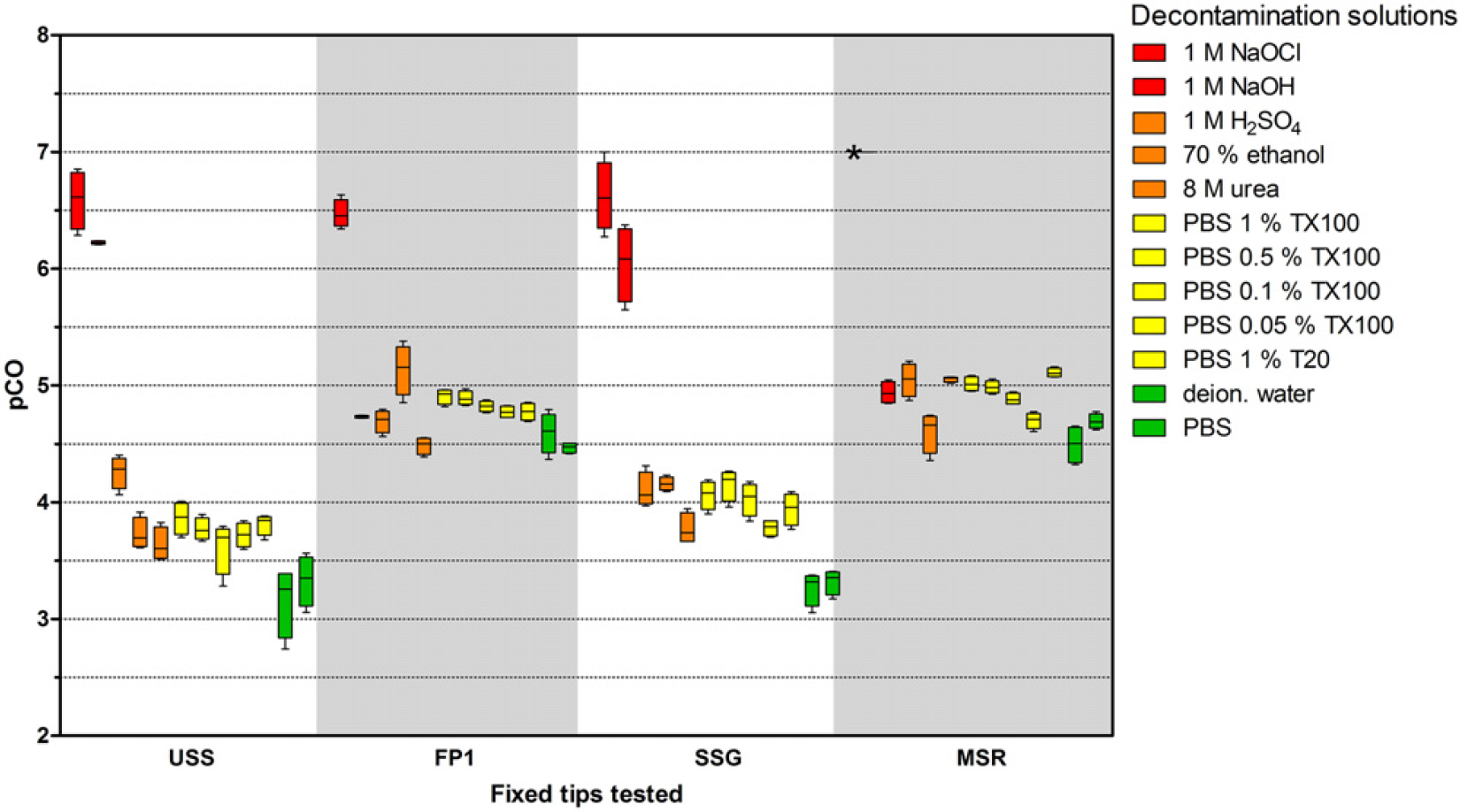
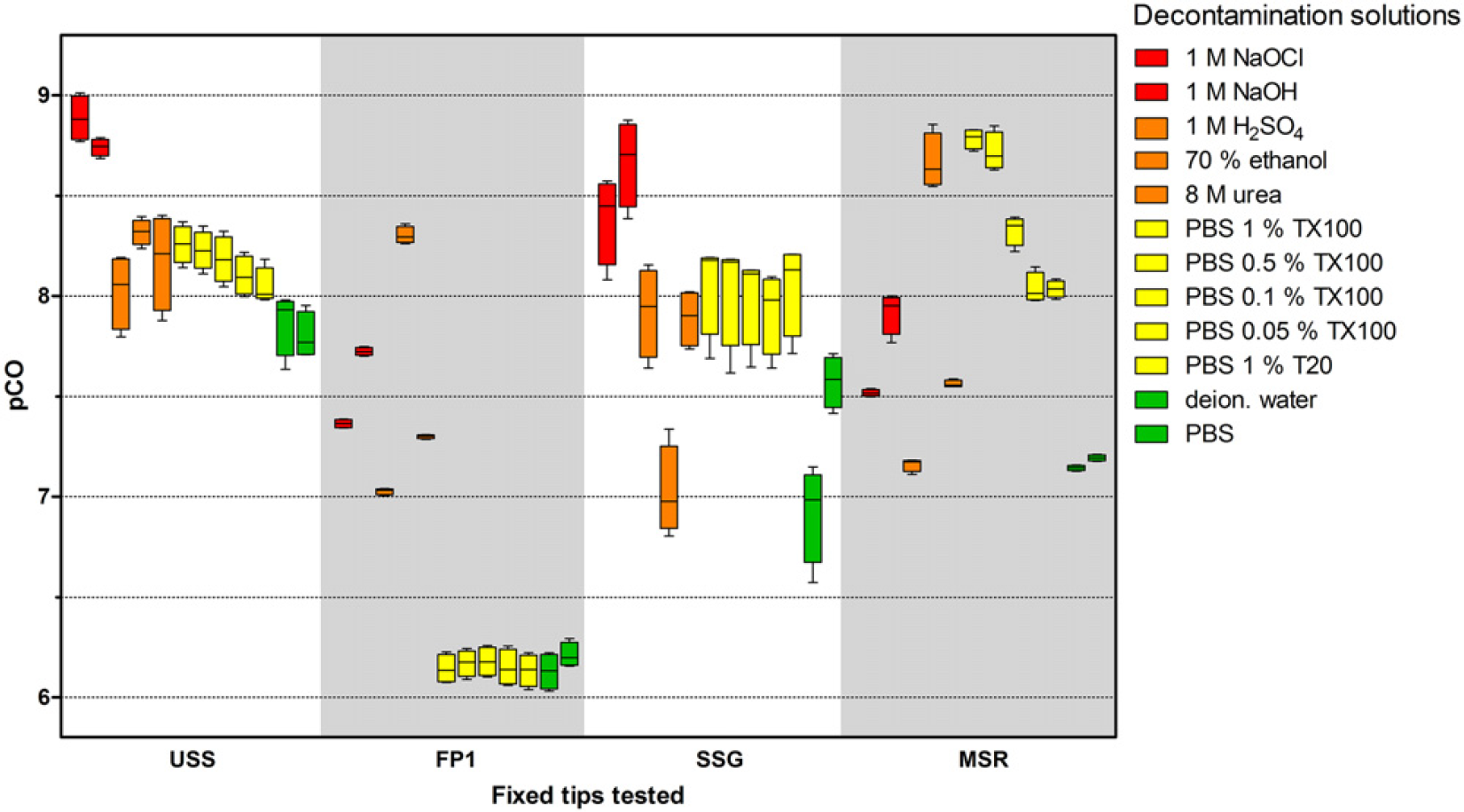
To investigate the effect of the decontamination solutions for a further analyte, the same 48 combinations of tip coatings and decontamination solutions were tested using fluorescein as a model analyte for a small organic molecule (Fig. 5). CO differed by a factor of about 1000-fold. The lowest pCO values were measured using FP1-coated tips with water as the decontamination solution, and the highest values were measured using USS tips with 1 M NaOCl as the decontamination solution. For IgG, as noted before, the 1 M NaOCl was the best decontamination solution not only for uncoated tips but also for the tips with different coatings. In contrast, PBS with 1% Triton X-100 achieved the highest pCO values of fluorescein for the MSR-coated tips, while 70% ethanol was the most efficient for FP1-coated tips, and 1 M NaOH achieved the best results for the SSG-coated tips.
Optimization of the Decontamination Step
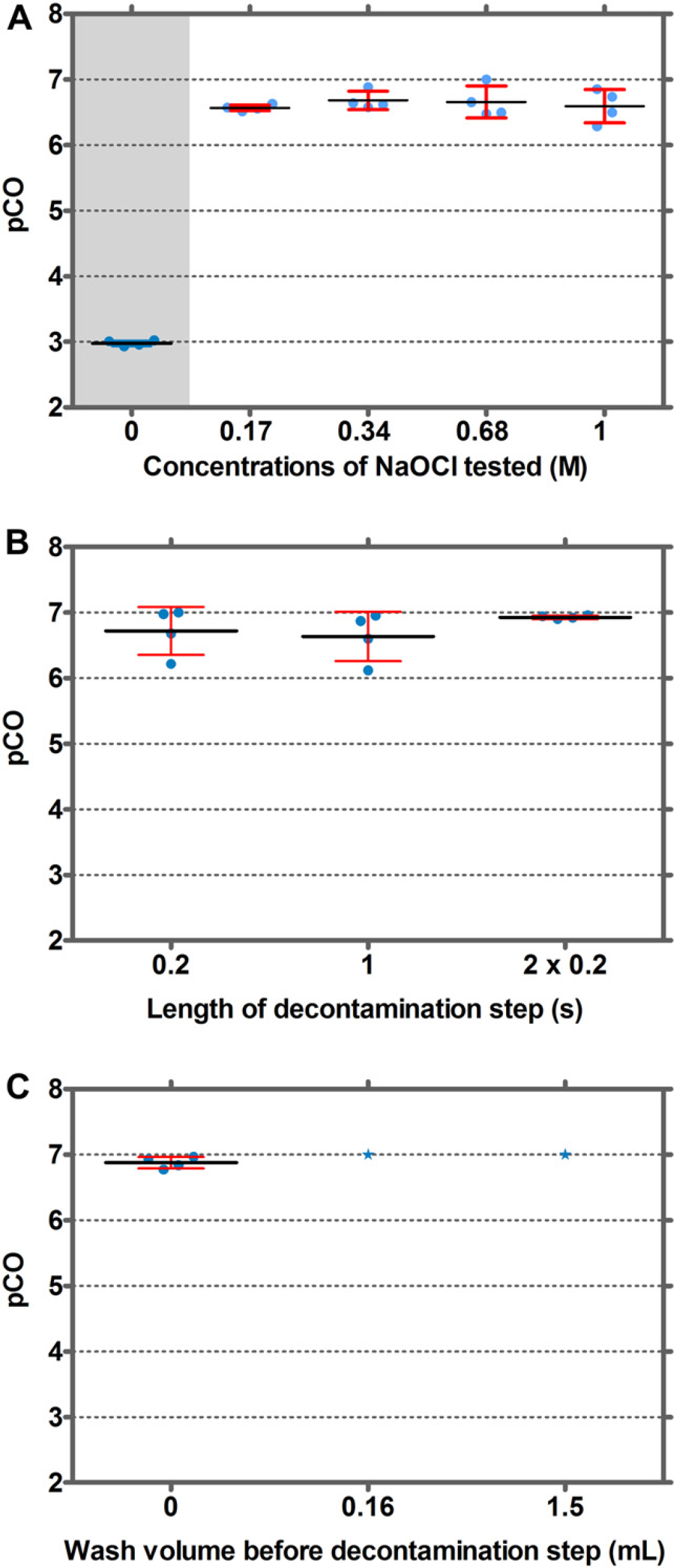
Sodium hypochlorite was a very efficient decontaminant for IgG and fluorescein. However, because it is a strong oxidizing agent, it could potentially cause tip corrosion. Therefore, lower NaOCl concentrations were investigated. NaOCl (0.17 M [1.27% w/v]) proved to be as efficient in suppressing CO as 1 M NaOCl (Fig. 6A). Subsequently, the length of the incubation time, when using NaOCl, was investigated. It was discovered that lowering the incubation time from 5 to 0.2 s did not cause a significant increase in CO (Fig. 6B). A further parameter that was investigated was the need for a washing step with 15-mL water before aspiration of the decontamination solution. It was observed that the volume of washing solution could be reduced to zero without compromising CO (Fig. 6C). For fluorescein, a similar dependence on NaOCl concentration and incubation times was observed (data not shown).
CO of HBsAg and IgG in Human Serum with an Integrated Decontamination Step
The optimized washing procedure with a decontamination step (Fig. 7) was finally tested with undiluted human serum using standard tips from Tecan.
Without a decontamination step, pCO values for HBsAg were 3.7 when 2-mL water was used for washing and increased to 5.7 when 26-mL water was used (Fig. 8). Decontamination with 0.17 M NaOCl for 0.2 s caused a further increase of the pCO value to more than 5.9. HBsAg concentrations after decontamination were below the LLOD (0.3 ng/mL) (Fig. 8).
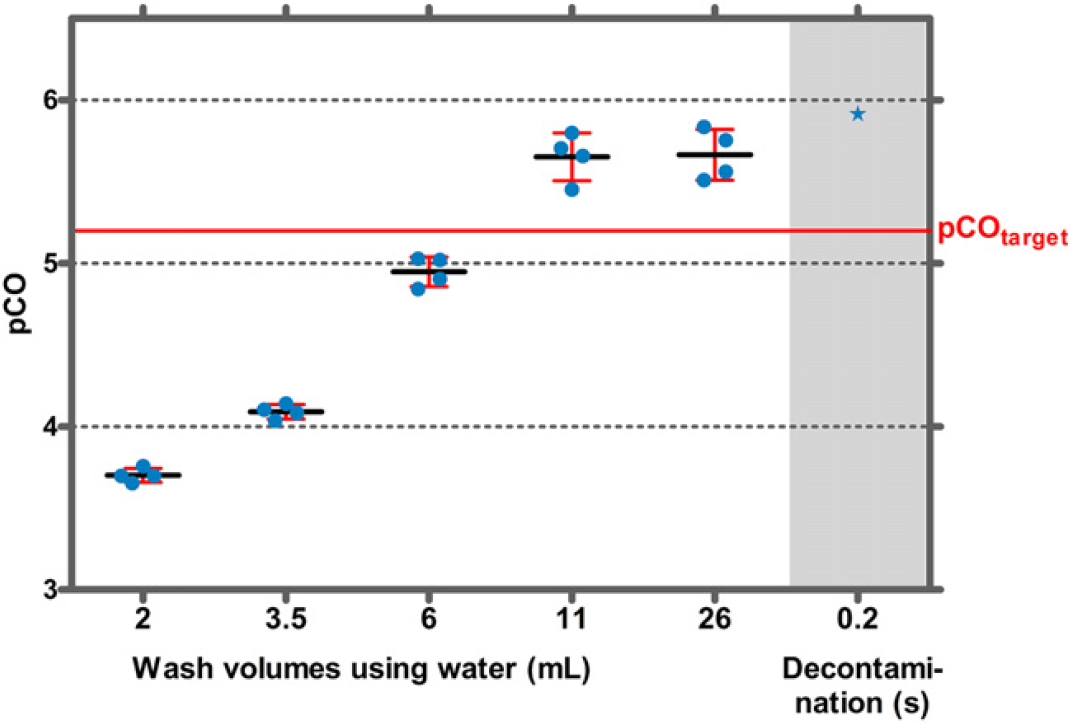
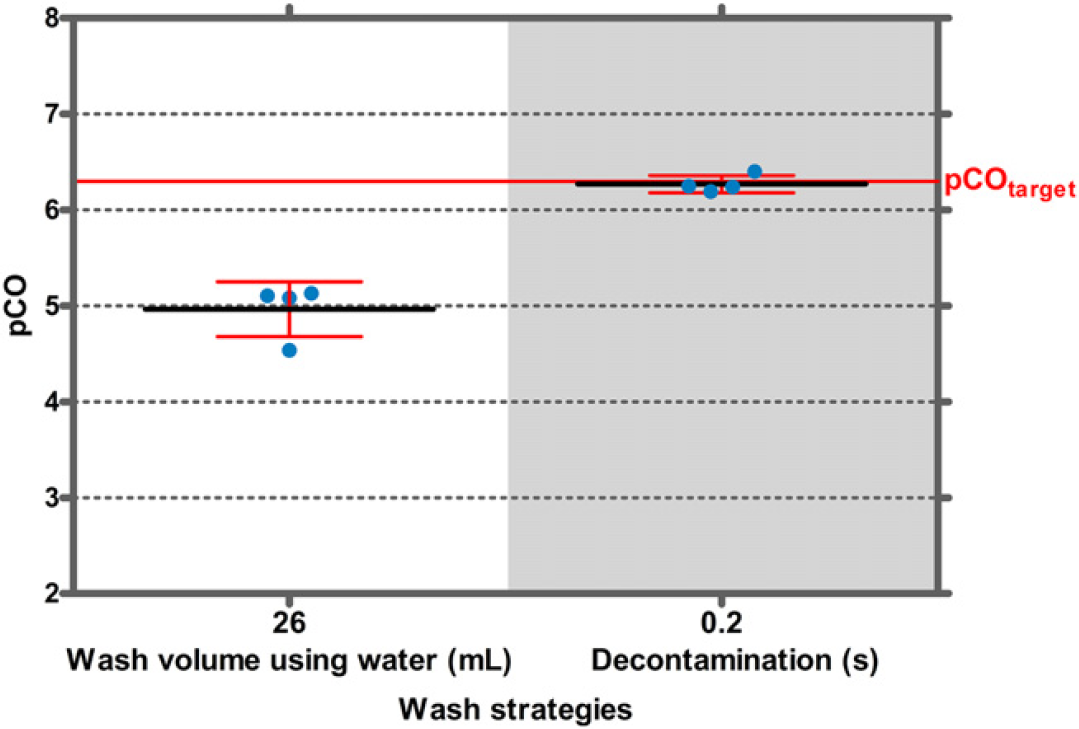
The pCO value for IgG in the absence of a decontamination step was approximately 5 when 26-mL water was used for washing. When decontaminated for 0.2 s with a 0.17 M NaOCl solution, the pCO value was increased to 6.3 and thus reached the target pCO value (Fig. 9).
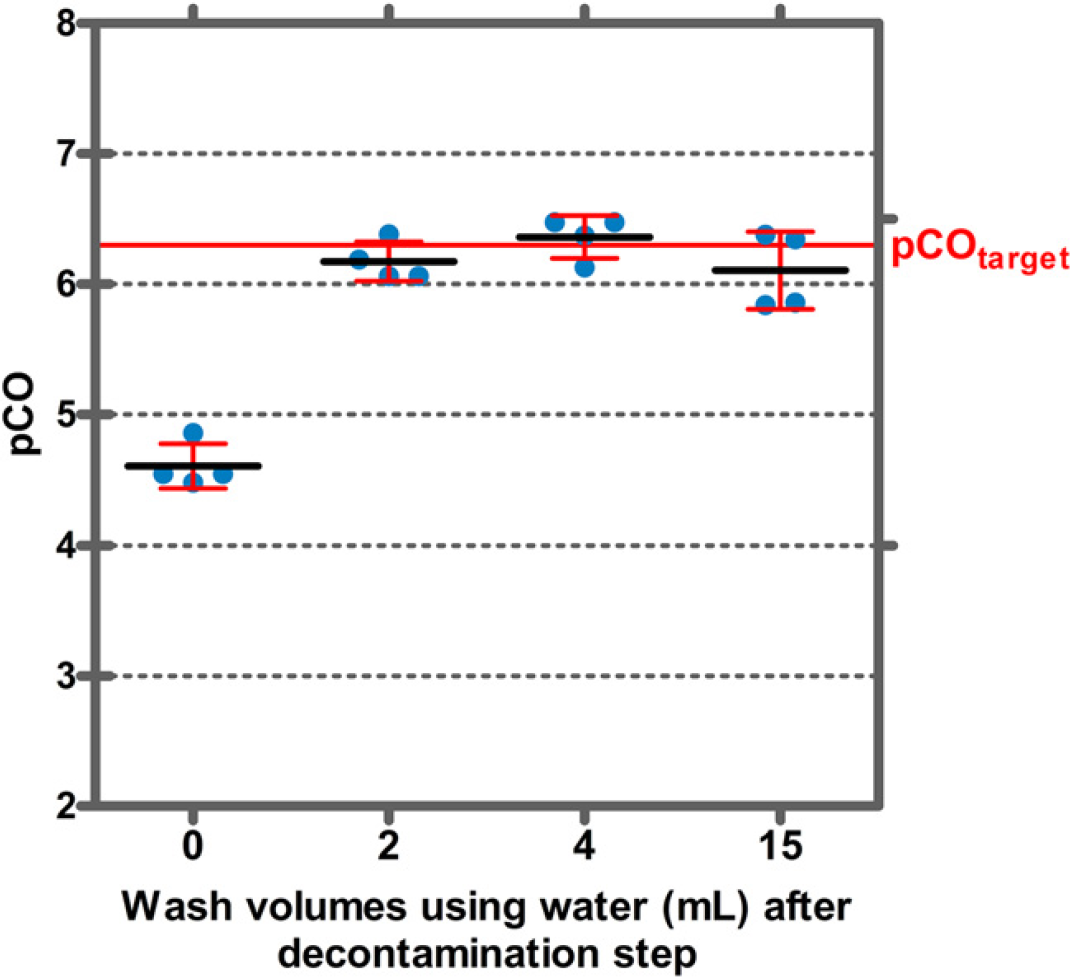
Next, we investigated the volume of water required to remove the remaining NaOCl from the tips. After washing the tips with 2-mL water, no NaOCl could be detected with the iodometric titration (data not shown). This result was confirmed in a CO experiment in which CO of IgG in human serum was measured dependent on the washing solution volume after the decontamination step (Fig. 10). A decontamination step with a subsequent washing step using 2 mL sufficed to reach the target pCO for IgG in human serum. Omitting the washing step led to inferior pCO values.
These results demonstrate that target pCO values for HBsAg and IgG in human serum can be exceeded by incorporating a short decontamination step with NaOCl into the procedure.
Discussion
We have previously reported the development of standardized test procedures for the measurement of CO in liquid-handling systems equipped with fixed reusable tips. 16 These procedures facilitated the reproducible quantification of CO of the small organic molecule fluorescein, the protein IgG, and the lipid-protein complex HBsAg. Using water as a washing solution, significant CO of all three substances was observed. However, neither increasing the washing solution volume nor varying the surface coatings of the tips resulted in a substantial reduction in CO. 16 Here, we confirmed the insufficiency of a standard low-volume washing step with water for both clinically relevant analytes IgG and HBsAg, when they were measured in human sera. Even when using large washing solution volumes, IgG levels were 22 times above a level that would preclude a false-positive measurement of a negative sample, when measured after a sample with a high analyte concentration.
We therefore investigated whether integration of a decontamination step could decrease CO. It was assumed that CO is mainly caused by adsorption of the analytes to the tip surface. Therefore, 12 different decontamination solutions were investigated. These included solutions with high and low pH values, detergents, urea as an agent that solubilizes protein aggregates, the oxidizing agent NaOCl, and the solvent ethanol.
NaOCl was an effective agent in lowering the CO of all three analytes. Optimization runs showed that exposure of the tips to 0.17 M NaOCl for 0.2 s was sufficient to decrease CO below target levels.
Hypochlorite is a strong oxidizing agent. It reacts with side chains of methionine, cysteine, histidine, tryptophan, tyrosine, lysine, arginine, asparagine, and glutamine as well as with the N-terminal amino group. Hypochlorite, at low concentrations, can induce protein cross-linking and aggregation. 19 However, at high concentrations, hypochlorite can attack the amide bond in proteins and cause fragmentation of proteins. Treatment of bovine serum albumin with a 2.25 mM hypochlorite solution caused a rapid and complete loss of the protein band in sodium dodecyl sulfate polyacrylamide gel electrophoresis. 20 Similarly, IgG was reported to fragment at hypochlorite concentrations of 1 mM. 21 We used a 0.17 M sodium hypochlorite solution, and it is thus highly likely that fragmentation of IgG is the reason for the efficient reduction in CO. HBsAg is composed of particles with a diameter of 17-22 nm formed by a lipid bilayer into which the S protein of the hepatitis B virus is integrated. Hypochlorite also reacts with lipids. 22 Accordingly, not only fragmentation of the S protein but also oxidation of lipids in HBsAg could contribute to the decrease in CO on treatment with NaOCl.
It is interesting to note that NaOCl also proved to be the best approach for preventing CO during automated processing of forensic samples for DNA profiling. 1 Because of more stringent requirements for acceptable levels of DNA CO in such samples, the procedure includes decontamination of the tips for 30 s with a 0.26 M NaOCl solution. 1
Our optimizations reduced the time needed for washing to 15 s (0.2-s incubation with the decontamination solution and a subsequent 2-mL washing step using water to remove residual decontamination solution). In our test systems, a tip washing step before the decontamination step could be omitted. If analytes are present in different matrices and buffers, this may not be possible. The greatest portion of the total time is required for moving the tips to their respective positions on the deck. When using disposable tips on the same deck layout, 14 s were needed to remove the used tips and pick up unused tips. Thus, a washing procedure with an integrated decontamination step does not significantly increase assay time.
Conclusions
Integration of a short decontamination step with 0.17 M NaOCl in the washing procedure resulted in a large decrease in CO of IgG and HBsAg in human serum to below target levels, thus ensuring a negative sample could not be judged positive when measured after a sample with a high concentration of IgG and HBsAg. Because NaOCl acts through fragmentation of proteins, it is tempting to speculate that such a decontamination step is applicable for a wide range of protein analytes. The decontamination step could be integrated into our liquid-handling system without hardware adaptations because only the Low Volume wash station was needed. The method is fast and can thus be used in low-, mid-, and high-throughput screenings. Furthermore, the method should be easily transferable to different liquid-handling systems and be testable with any customer-specific assay. Adaptations of washing and decontamination conditions might become necessary for different wash stations and different applications.
Acknowledgments
The project was initiated by Toolpoint for Life Science, Hombrechtikon, Switzerland and was financially supported by the Innovation Promotion Agency CTI of the Federal Office for Professional Education and Technology, Switzerland (CTI 7627.2)
Competing Interests Statements: The authors disclose that C. Zaborosch, R. Weibel, M. Iten, and I. König are employees of the Zurich University of Applied Sciences; R. Beckbissinger and L. Oeltjen are employees of Hamilton Bonaduz; W. Hälg and N. Ingenhoven are employees of Tecan Schweiz; A. Lehnert is an employee of Sias; and T. Benthien is an employee of Surface Contacts.