Abstract
The integration of microfabrication technologies with advanced biomaterials has led to the development of powerful tools to control the cellular microenvironment and the microarchitecture of engineered tissue constructs. Here, we review this area with a focus on the work accomplished in our laboratory. In particular, we discuss techniques to develop hydrogel microstructures for controlling cell aggregate formation to regulate stem cell behavior as well as a bottom-up and a top-down microengineering approach to creating biomimic tissue-like structures.
Introduction
At the interface of micro- and nanofabrication and experimental biology lies enormous potential to address important problems in biology and medicine. This is because biological systems are highly complex and cannot be easily understood without tools that can study such complexity at length scales that are relevant to biological systems. This complexity extends from developmental processes, where a single cell undergoes many rounds of division and proliferation to become a fully developed organism, to the maintenance and regeneration of adult tissues.
Cellular processes are controlled by the genetic factors in each cell, which are not only intrinsically regulated but are also controlled by the local cellular microenvironment. Thus, the local chemical, biological, and mechanical environments can provide a coordinate set of regulatory cues to control cell behavior. For example, in the early stages of development, embryonic cells in the inner cell mass communicate with one another through paracrine and autocrine signaling and cell-cell contacts that influences organogenesis. 1 Cellular communication and signaling occurs through a number of different mechanisms, including direct cell-cell contact, soluble factors, and cell-matrix interactions. Soluble factors and signaling molecules provide different cues depending on the identity, concentration, and context. Furthermore, mechanical forces imparted by the surrounding extracellular matrix (ECM) can signal cells to specific fate decisions.
The complexity of the interactions between cells and the microenvironment is not only found during development and organogenesis but also found in mature organisms. For example, blood cells are continually replenished throughout an organism's lifetime. It is known that hematopoietic stem cells, which reside in the bone marrow, interact closely with the surrounding endothelial cells, osteoblasts, and fibroblasts that regulates their self-renewal and differentiation toward different blood cell fates.2,3
Our research group, along with others, is interested in developing technologies at the interface between micro- and nanoengineering and materials science for engineering controlled microenvironments that can aid in understanding the interactions between cells and their microenvironment. The goal is to overcome a major experimental challenge in experimental biology, which is to recreate the in vivo cell-microenvironment interactions in vitro.4,5 In a tissue culture Petri dish, cells interact with a two-dimensional plastic surface that is drastically different from the environment that is found in vivo. A salient example of the deleterious effects of disrupting the natural cellular microenvironment is the loss of liver hepatocyte function on culture outside the body. In the liver, hepatocytes reside in controlled tissue units called liver lobules, which have a high degree of control and complexity. 6 Liver lobules are made from organized hepatocytes that are assembled in hexagonal structures that are highly vascularized and are in intimate contact with the surrounding endothelial cells. The inability to recreate this complex microarchitecture in vitro may be a key reason that the significant regeneration capacity of the liver and its diverse metabolic functions have not been recreated in vitro.
Our research group aims to develop and use technologies to recreate cell-microenvironment interactions to produce in vitro culture conditions that can be used for understanding cell biology or to generate three-dimensional (3D) tissue constructs for cell-based therapies. Furthermore, engineered tissues can be used for screening drugs and to investigate the underlying mechanisms of disease. 7 These modulations of the cellular microenvironment will be made by using microfabrication to sculpt and assemble advanced materials to control the interactions of cells with the microenvironment. 8 For example, through the use of microfluidics, it is possible to control the temporal and contextual presentation of soluble factors to induce cellular events; 9 patterned surfaces can be used to generate spatially controllable cocultures to control cell-cell interactions;10–12 and, cell-laden hydrogels can be created to investigate cell behavior in 3D.13,14 Finally, it is possible to perturb mechanical properties of materials and local sheer stresses induced by fluids to modify cell responses. 15
During the past several decades many tools required to investigate biology at the micro- and nanometer length scales have been developed. 8 There is still much work to be accomplished, but great strides have been made in making micro- and nanofabricated systems much more accessible to common laboratory use. 16 For example, with rapid prototyping or soft lithography, micro- and nanopatterned silicon wafers, or other templates, can be quickly and easily replicated with an elastomeric copolymer such as polydimethylsiloxane (PDMS). 16 PDMS replicas can be used as microfluidic channels, for molding of biomaterials or as stamps to pattern surfaces. The power of these techniques is that they can be used to control the architecture of materials at length scales much smaller (<100 nm) or much larger (>1 mm) than a typical cell.
In this article, we describe our laboratory's work in merging biomaterials and advanced fabrication techniques to control the cell microenvironment and engineer tissues with controlled microarchitecture. The common element of the various projects is that they use biomaterial microstructures to engineer cell aggregates and generate tissue constructs.17,18 We describe two different ways to engineer tissue-like assemblies with controlled microarchitectures by using either a top-down 19 or bottom-up approach. 20 With a bottom-up or “Lego-like” approach, we create small microfabricated tissue units that assemble into larger tissuelike constructs with controlled microarchitectures. With a top-down engineering approach, we sculpt biomaterials into micro- and nanoscale structures that mimic tissue constructs. In each of our approaches, we use microfabrication techniques to mold and control the size, shape, and microscale features of biocompatible hydrogels. We use photo-and soft lithography as well as micromolding techniques as they are simple methods that can be easily made compatible with a variety of different materials.
Hydrogel Microstructures for Stem Cell Bioengineering
Polymers such as poly(ethylene glycol) (PEG) are so hydrophilic that water complexes with their polymer chains to prevent subsequent protein adsorption or cell adhesion. We have been interested in exploiting this phenomenon to create microscale structures with controllable surface properties. Figure 1 shows a number of different microscale hydrogel structures made by micromolding of photocrosslinkable PEG hydrogel microstructures. Scanning electron micrographs of (A) PEG-bottomed microwells and (B) microwells exposed to the underlying substrate. Cell docking in various microstructures, including (C) round 100-μm diameter microwells, (D) grooves 100 μm in width, and (E) 200-μm square microwells. Images reproduced with permission.
21
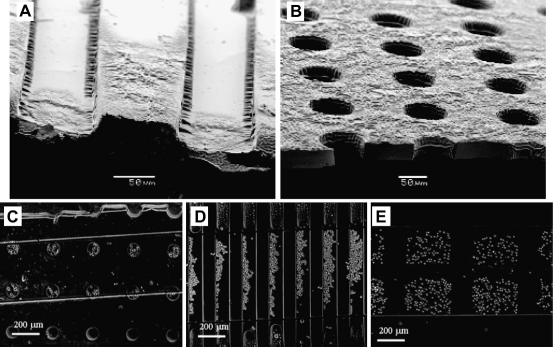
It is important to note that fluid elements around microscale structures behave differently from those in macroscale systems. For example, mathematical modeling of arrays of microwells under fluid flow reveals that as the depth of the microwells increase or the diameter of the wells decrease, the regions in the wells become protected from shear stresses. 22 Thus, cells in deeper- or-smaller diameter microwells that are exposed to less shear stress, are not washed away under flow, and can be stably integrated into the device. This system of arrayed microwells made from PEG hydrogel can be used to capture cells and other particles of interest in a rapid and controllable manner.
The flow patterns inside the structures can also be used to control cell location. For example, we have used mathematical simulations to show that when fluid flows over arrays of microwells that are less than 50 μm in diameter, fluid recirculated in the structures, whereas in larger structures, fluid penetrated the structures. These results were also confirmed experimentally. In the 75-μm grooves, there was no recirculating flow or back flow, and cells were somewhat evenly distributed across the width of the grooves. However, in the 50-μm grooves, recirculating flows aligned cells along the upstream side of the grooves. 22
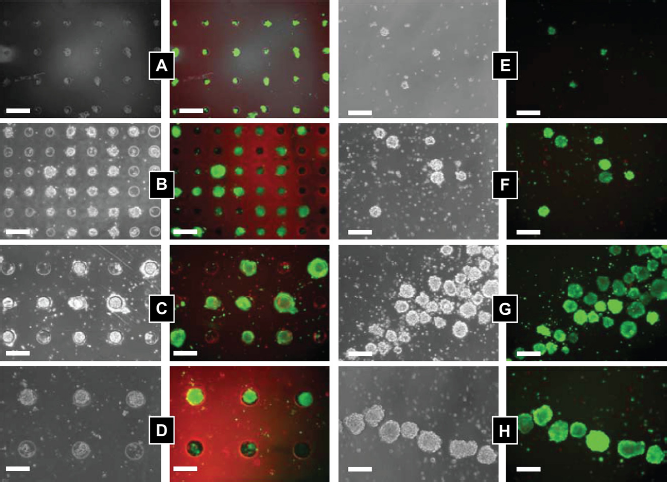
Arrayed microwells can be used as a platform to ask questions in stem cell biology and to direct cell fates for therapeutic applications. Specifically, the microwell platform is well suited to control the size of stem cell aggregates.17,23 In these aggregates, embryonic stem (ES) cells begin to initiate differentiation pathways that more closely mimic the developing embryo. In a typical ES cell aggregate experiment, ES cells are plated in a nonadhesive dish, and cells cluster together into aggregates of varying sizes and begin to differentiate. PEG microwells can be used to control this process and form cell aggregates in a simple manner. Cells seeded inside the microwells are in low shear stress regions and remain in the microwells under flow. In this way, we can control the formation and size of cell aggregates and produce homogeneous populations of microsphere cell aggregates. Figure 2 shows examples of controlled cell-cell interactions and the culturing of populations of cell aggregates, embryoid bodies (EBs), with homogeneous sizes. Figure 2A-D shows EBs of varying sizes cultured in microwells and Figure 2E-H shows EBs after retrieval from the microwells after 10 days of culture.
ES cells seeded in microwells of varying size: (A) 40 μm; (B) 75 μm; (C) 100 μm; and (D) 150 μm. Fluorescent images show cell stained with calcein AM (live cells) and ethidium homodimer (dead cells) after 10 days of culturing. (E-H) Harvested cell aggregates after 10 days of culture. Images reproduced with permission.
23
The number of cells that form an EB can be used to direct stem cell differentiation. If one can control the number of cells per EB, then comparisons of small cell number and large cell number EBs are possible. In different sized EBs, individual cells will experience a different microenvironment and will be directed toward different fates. Figure 3 shows the outcomes of experiments with EBs of different sizes that were investigated with respect to the size-dependent expression of cardiac and endothelial cell differentiation.
24
In this example, we examined the differentiation of EBs of three different sizes by using two different experimental protocols. In both cases, ES cells were cultured in microwells of different sizes for 5 days. In one protocol, we continued culturing the EBs in microwells for an additional 10 days. Alternatively, we retrieved the EBs and replated them on Matrigel for an additional 10 days. In both cases, we analyze the outcomes with respect to gene expression and protein markers of cardiac and endothelial cell differentiation. In particular, we found that a higher fraction of larger EBs were spontaneously beating, a characteristic of differentiation into cardiac cells. In addition, the cardiac marker sarcomeric alpha actinin (SαA) was highly expressed in the beating cells. Large EBs (450 μm in diameter) cultured in the microwells showed strong SαA staining and beat spontaneously. In the smaller EBs (150 mm in diameter) that were cultured throughout the experiment in microwells, we saw minimal expression of SαM and a significant reduction in spontaneous beating. Larger EBs also had more cells with typical cardiac cell morphologies, when replated after 5 days of microwell culturing and when culturing in microwells for the course of the experiment.
Microwell-mediated control of EB size and cardiac and endothelial cell differentiation. (A) Immunocytochemical characterization of Wnt5a-siRNA transfected (green) EBs shows that transfected siRNA is delivered (Left). EBs are analyzed for the presence of endothelial cell marker CD31 (red) and cardiogenic marker SαA (green). (B) Characterization of EB sprouting and beating frequency. (C) Gene expression of endothelial cell and cardiogenic differentiation marker from ES cells cultured in 150-μm diameter microwells. (D) Characterization of ES cell aggregates with the addition of recombinant mouse WNT5a in 450-μm diameter microwells (scale bar, 100 μm). (E and F) Analysis of cardiogenic and endothelial cell differentiation by EB beating, vessel sprouting frequency, and gene expression. (n = 3, *indicates P < 0.05 compared with controls). Images reproduced with permission.
24
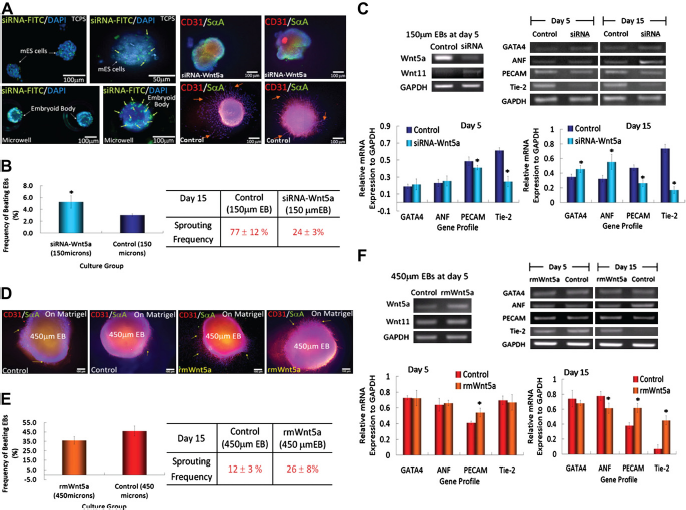
Analysis of vascular differentiation showed an opposite response with smaller EBs giving rise to more endothelial-like cells. In these experiments, a higher degree of expression of the endothelial cell marker CD31 was observed in smaller EBs compared with larger EBs. Also, CD31+ cells formed capillary-like structures. Additionally, smaller EBs that were replated on Matrigel started to form sprouts that stained positive for CD31 at a higher frequency and length than those observed in larger EBs, further suggesting that there was more endothelial cell differentiation in smaller EBs than in larger ones.
The microfabricated well arrays can also be used to generate homogeneous cultures for studying the molecular and biochemical mechanisms that regulate the observed size-dependent outcomes. In further analysis of the EBs of different sizes, we did not see significant differences in expression of a numberof ECM components including fibronectin, collagen IV, and laminin a and b. We also analyzed the expression of Wnts, a family of signaling molecules that control a number of events during embryonic development. Although we did not observe differences in the expression of canonical Wnt family members, beta-catenin or Wnt2, we observed differences in expression of two member of the noncanonical Wnt family molecules. In particular, it was observed that Wnt5a was highly expressed in smaller EBs, whereas Wnt11 was more prominently expressed in larger EBs. The size-dependent Wnt5a expression and the effect oncardiac and vascular differentiation were confirmed by si
These size-controlled EB experiments demonstrate the utility of microwell-based cell culture in answering interesting questions in stem cell biology; however, there are still many interesting aspects of the microwell culturing that have yet to be explored. For example, we have yet to investigate the effects of size restriction on stem cell proliferation and differentiation. Furthermore, the mechanisms that direct stem cell differentiation in these systems are yet to be fully elucidated.
Microwell technologies have many advantages over tradition cell culturing methods, such as the ability to control aggregate size, and the ability to protect cells from external shear stress. It is also possible to control microenvironmental factors including cell-cell and cell-matrix interactions with engineered hydrogel microwells. However, there are some potential shortcomings including an inability to individually address single microwells and an inability to dynamically control aggregate size as aggregates expand with cell proliferation. Additionally, aggregate size is controlled by restricting lateral growth, and during long-term culturing cell aggregates can proliferate outside of the bounds of the microwell structures. Retrieval of aggregates from the microwell structures has also proven challenging, and great care is needed to retrieve high yields of intact aggregates. With new microscale technologies and hydrogel microstructures, we are beginning to address some of these shortcomings.
Bottom-up Assembly of Microgels for Tissue Engineering
Natural tissues are made from highly complex tissue microarchitectures. A major challenge in tissue engineering is to recreate tissue constructs with appropriate microarchitectures and functions. To this end, we have explored two different microengineering approaches: (1) the bottom-up assembly of microgels 19 and (2) the top-down engineering of microscale biomaterials. 20 The latter is described in detail in the next section. The former, bottom-up assembly, is a bioinspired approach to the assembly of complex tissue microarchitectures using microgel building blocks.
In nature, many tissues are made up of assemblies of small tissue modules (i.e., repeating functional units). For example, muscles are made from bundles of myofibers; the liver is composed of lobules; the kidney is made up of nephrons; and the pancreas contains islets. These functional tissue units are well vascularized and are made of patterned assemblies of the units. In our laboratory, we are using a bottom-up approach to tissue engineering to mimic this concept. This is done by using microfabrication techniques to engineer microscale tissue units and then assembling the resulting structures to generate tissue-like complexity.
Hydrogels are attractive materials for making microengi-neered tissue building blocks because of their hydrated nature and biomimetic mechanical properties.25,26 We have previously used a number of methods to engineer cell-laden hydrogels of controlled shapes and sizes. For example, photocrosslinkable hyaluronic acid-based hydrogels were micromolded into controlled shapes (Fig. 4).
20
To overcome the potential limitation of micromolding, which include its batch process,
20
we have used microfluidic systems to create a continuous process to generate controlled shape microgels. In this process, the hydrogel precursor solution containing cells was flowed continuously past a light source to produce individual cell-laden microgels (Fig. 5).
27
A shutter was used to control the exposure of the hydrogel precursor to light, and a mask was used to control the microgel shape. In this way, we and our collaborators produced microgel building blocks in a continuous process.
Light (A) and fluorescent (B) micrographs of micromolded cell-laden methacrylated hyaluronic acid (MeHA) hydrogels. The MeHA is crosslinked by exposure to light. The fluorescent micrograph shows cell viability after UV light exposure. Live cells are green (calecin AM) and dead cells are red (ethidium homodimer). Images reproduced with permission.
33
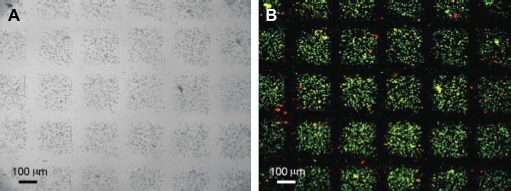
Cell-laden microgels produce by stop-flow lithography. (A) Cell-laden microgel collected at the device outlet reservoir (100-μm scale bar). (B-D) Cell-laden microgels of different shapes (20-μm scale bar). Images reproduced with permission.
27

We have also developed methods of inducing the assembly of microgels into tissue-like structures. The self-assembly of microgels differs substantially from molecular self-assembly in that the forces at work in molecular assembly differ from those that drive the assembly of microscale hydrogels. Thus, a key aspect that must be considered for microgel assembly is the type of forces that will be used to drive the assembly process.
One of our first ideas was to direct the assembly process by exploiting the tendency of hydrophobic and hydrophilic substances to minimize the interaction with each other. Specifically, we used the difference in chemical properties at a hydrophobic-hydrophilic liquid interface and at an air-liquid interface to drive the assembly process.19,28 Following on the work of others,29,30 we have used the forces at these interfaces to drive the assembly of microgel building blocks as the energy of the system is minimized.
Figure 6 shows two different examples of self-assembled microgel structures. In one case (Fig. 6A), two differently shaped microgel building blocks are assembled using an oil-water interface.
19
In the simple case of oil in water, the solutions minimize the surface interactions and droplets of water form in oil (or oil droplets in water). With microscale hydrogels in a bulk oil phase, a similar process occurs: the hydrogels are driven together as the surface interactions are minimized. Controlling the microgel shape leads to control of the assembly process. With appropriately shaped microgels, it is possible to create “lock-and-key” assemblies, where the shape of different microgels is matched to create mesoscale hydrogels with controlled microarchitectures. The examples shown in Figure 6 were made by using an oil-water interface, but we have also been able to create similar structures using an air-liquid interface. We have also been able to use this assembly process to create larger structures. By using two interfaces, oil-water and a wetting substrate, we can “wrap” assembled microgels around a wetting substrate to generate 3D structures (Fig. 6B).
Bottom-up assembly of microgels. (A) Cross- and cylindrical-shaped PEG hydrogels assembled in mineral oil.
19
(B) A semispher-ical shell assembled from PEG hydrogel microgels.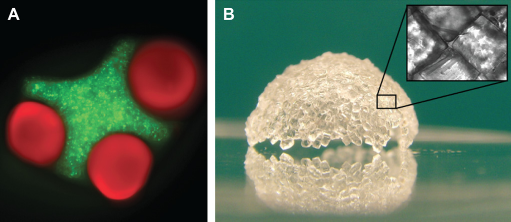
The examples of bottom-up assembly of microgels demonstrate the potential utility of this technique to create complex tissue-like structures; however, there are some potential disadvantages with the process. For example, it is challenging to create structures that are 10s of centimeters in length. Such structures will require strong secondary crosslinking to ensure adequate bonding between microgels, and, if free standing, will require building blocks with mechanical properties sufficient to support the weight of the structure. Additionally, it would be advantageous to develop two-phase liquid systems other than mineral oil and water to drive the assembly process.
Further research is required to overcome these limitations, and we are actively pursuing microscale techniques to advance this bottom-up approach to tissue engineering. We are interested in exploring different aspects of the approach including the ordered and sequential assembly of microgels. We are also interested in exploring new hydrogels for microgel fabrication and developing composite hydrogels for controlling cell-ECM interactions within each microgel building blocks.
Top-Down Microscale Tissue Engineering
Some of the initial studies in the use of microfabrication techniques for tissue engineering were done in attempts to engineer microfluidic networks to recreate vascularized tissue structures in scaffolds made from materials such as silicon, PDMS, or degradable synthetic polymers.31,32 Although these materials are well suited for microscale fabrication, they are not amenable to cell encapsulation. Recently, we have been creating microfluidic channels with hydrogels made from natural polymers to enable the formation of hydrated vascular networks within which cells can be encapsulated in the bulk phase of the materials.
Figure 7 demonstrates an example of a micromolding approach to produce cell-laden hydrogel microfluidic channels.
33
In this example, hepatocytes were embedded in agarose and molded into a microchannel. Perfusion within the hydrogel could be achieved through diffusion from the channels into the hydrogel walls and was confirmed in cross-sections of agarose microfluidic channels after only a few minutes of flow.
33
In this way, the microfluidic hydrogel channels have the potential to mimic native vasculature, as oxygen and nutrients from liquid in the channels diffuse into the surrounding hydrogel. This effect can be seen in cell-laden agarose hydrogels with engineered microporosity.
34
After 3 days of culture, a ring of viable cells can be seen on around the channel that corresponds to the diffusion pattern of oxygen and nutrients. With these types of micro-engineered structures, one can begin to create tissue-like constructs with encapsulated cells and endothelialized channels. Multiple constructs can be stack to create more complex vasculatures and tissue-like constructs.
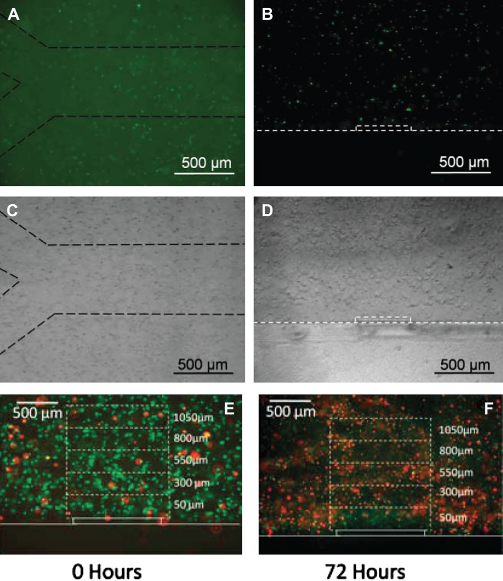
Fluorescent and brightfield images of carboxyfluorescein succinimide ester-stained AML-12 cells encapsulated in a microengineering agarose channel. (A, C) top view, and (B, D) are cross-sectional view of the channel. Dashed lines were added to images to aid in visualization at print resolutions. (E, F) Representative live/dead staining of AML-12 hepatocytes immediately after encapsulation and after 72 h of culturing. Reproduced with permission.
33
The size, structure, and complexity of multilayered constructs are limited by the material properties of the hydrogels used to create the constructs. Although the examples presented here make significant progress in mimicking native vasculature, there are many technological challenges that remain. For example, microscale technologies that can create high densities of microscale channels with complex branching are required. Fabrication of microchannels that extend out in 3D and that have hierarchical structures are also needed. We are actively pursuing new microscale technologies to address these limitations.
Conclusions
We have described three different approaches to merge easily accessible microscale fabrication technologies with hydrogel biomaterials. With hydrogel microstructures, we have been able to systematically manipulate stem cell microenvironments and analyze the outcomes of those manipulations. Furthermore, we have developed a bottom-up approach to create cell-laden hydrogels with controlled microarchitecture. We have also developed a top-down approach to microengineer cell-laden hydrogels that mimic native vasculature. These techniques may be of potential benefit for generating tissues and directing stem cell differentiation for regenerative medicine applications and for generating in vitro models for drug discovery and pathological studies.
Footnotes
Competing Interests Statement: The authors certify that they have no relevant financial interests in this manuscript.
Acknowledgments
This paper was supported by the National Institutes of Health (EB008392; DE019024), National Science Foundation (DMR0847287), the Institute for Soldier Nanotechnology, and the US Army Corps of Engineers. The authors certify that they have no relevant financial interests in this manuscript.
