Abstract
This JALA feature story reports the results of two surveys conducted by the Association for Laboratory Automation (ALA) to evaluate the state of industrial laboratory automation. The purpose was to gain more insight into the results of the 2006 ALA survey by posing more detailed questions based on the 2006 responses. The 2008 survey polled 43 carefully selected ALA members whose organizations use laboratory automation. These organizations represented the biotech, pharmaceutical, agricultural science, consumer goods, materials and formulation, research institute, and government business sectors. The 2008 survey also polled 12 leading laboratory automation technology providers. Questions were designed to compare the trends, needs, and priorities of the user and provider communities.
Introduction
The 2006 Association for Laboratory Automation (ALA) Survey 1 on Industrial Automation resulted in several observations. One such observation was that laboratory automation technology had matured to the point that the factors limiting the use of such automation had shifted away from the long-standing “nuts and bolts” issues of reliability and throughput, and toward the challenges of doing science with automated tools, such as the difficulty of validating new automated processes. This 2008 survey seeks to probe more deeply into such observations and conclusions from the previous survey.
The 2008 survey also seeks to compare and contrast the opinions and priorities of the technology user and provider communities by posing complementary questions to each group. To offer supplemental analysis of the survey data, we include commentary from Dr. Hansjörg Haas, director of marketing and integrated systems, laboratory automation at Thermo Fisher Scientific, and Dr. Peter Grandsard, executive director of research at Amgen, and long-time leader of Amgen's Research and Automation Technologies group. Their comments are designated as
Automation System Use and Needs
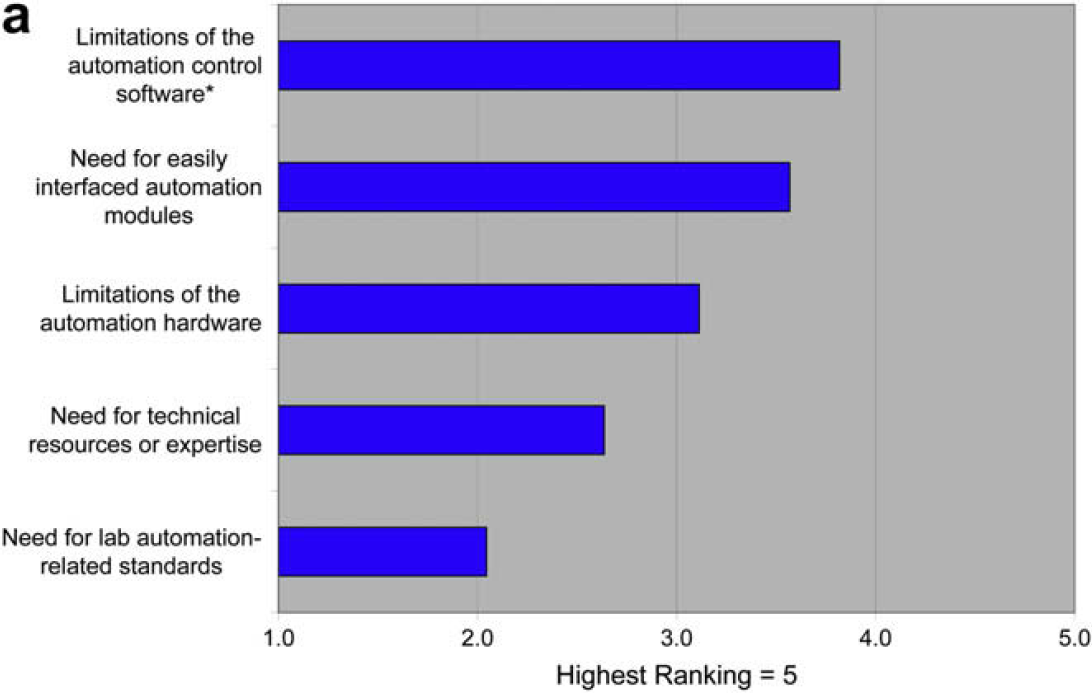
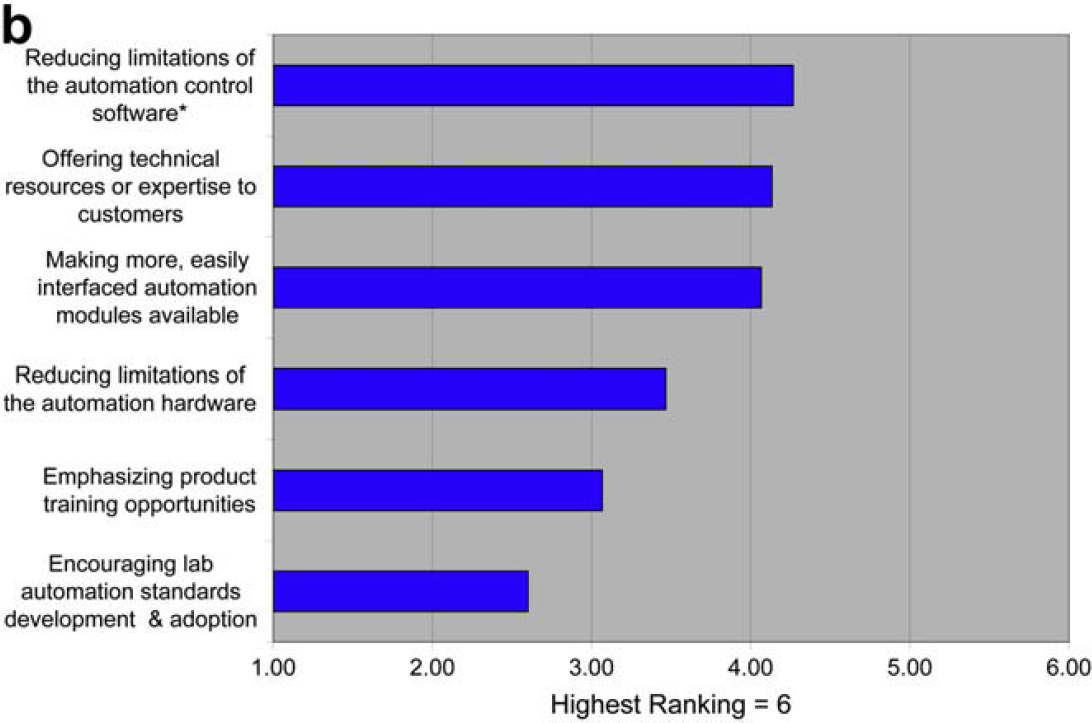
Questions 1.1–1.3 are focused on in-depth detail about the factors limiting the use of laboratory automation that were identified in the 2006 survey. The graphs represent respondent ranking of responses. We also indicate (*) the response that received the most “top” rankings. This response represents the choice most people would make if they could make only one choice versus the ranking of all responses, which represents the choices that would be made if multiple options are available. Some small variation between the two modes of analysis is normal.
Validation of New Processes on Automated Systems
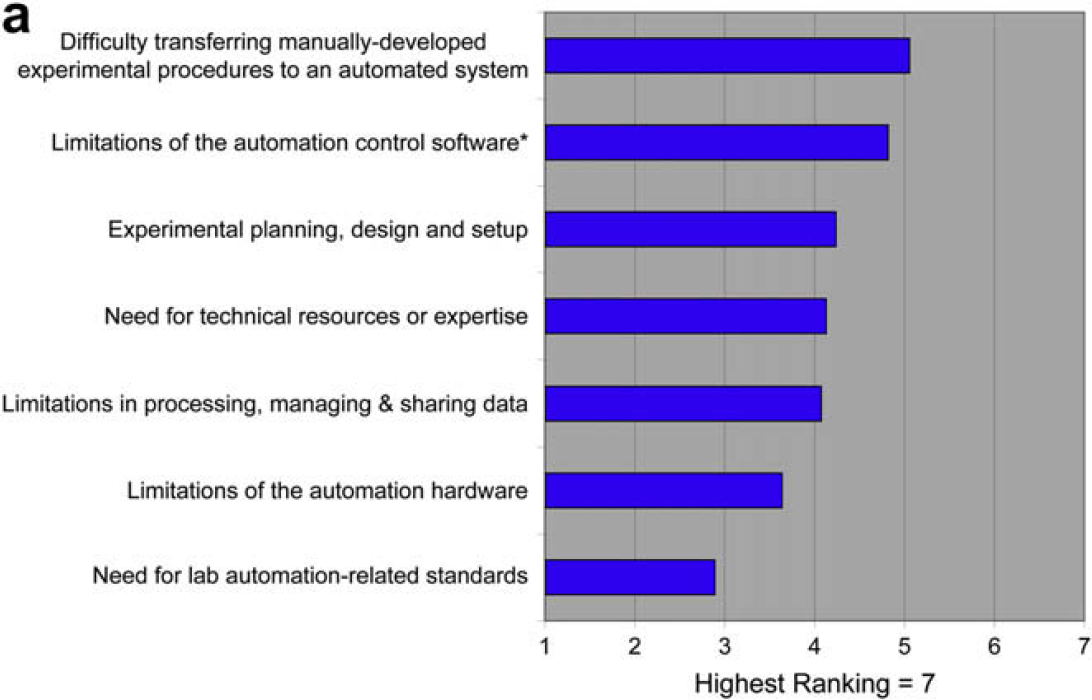
When asked what could be done to improve the situation, users offer the following (unedited) responses:
Lab automation standards.
More training for automation implementers.
More focus on automation issues when developing manual procedures.
To improve the flexibility of the software full control of tool placing and timing needs to relinquish to the user especially for importing data or integration with third party equipment. The variables used to control the automated tools should be able to systematically and independently controlled to allow the optimization of the whole process to become possible.
Connect Electronic Laboratory Notebook (ELN) with software… or change the culture to design manual experiments with automation implementation in mind.
Clear understanding of needs and route for fulfillment.
Clearer technical documents + more intuitive/user-friendly software (=quality control (QC) of the product should involve users, not only developers).
Reduce the number of manually developed experimental procedures.
Develop standards for lab automation … both in labware (plates/tubes/reaction vessels) and control software. End users of lab automation tools are required to become experts in multiple software platforms. The transferring of methods becomes very tedious.
Closer collaboration between assay development and HTS, plus better up-front education on the HTS process for assay developers.
Educate scientists about what works as an automated method and what does not.
Better experimental design.
I think it is too early for specific standards, but a general body of knowledge about system suitability needs to be created and documented.
Need to do better in getting vendor experts talking to internal IT.
Configuration and validation of pipetting heights and speeds. Rapid validation of proper volume dispensing.
Better planning, more prework, and up-front validation.
Try to avoid to literally copy the manual process and automate it. It should be an opportunity to rethink the process and choose the corresponding hardware and software to translate the process.
These improve as more processes are automated!
Companies should do their homework.
No easy amelioration for this other than moving automation higher in priority so that more resources are directed toward it.
Nothing—it is planning and resource allocation and expertise of the staff to implement.
In my opinion, difficulty with transference of manual processes to an automated platform is inextricably linked to software inflexibility for both control of instrumentation and definition of experimental design. The opportunity for improvement is to focus on the development of flexible design and execution software.
Limitations in processing and managing data and the actual regulatory validation take too much time at my company. The same data related limitations in support of strict scientific validation are not as restrictive as limitations of automation hardware.
More lab automation papers.
More opportunities for training of qualified individuals.
The lab typically dose not have the knowledge to help design and plan proper experiments.
Better accessible software.
Improve the communication between lab and automation.
The most limiting factor at our firm is the interaction between the automation group and the group that developed the manual assay. Typically, our resources are stretched very thin and it is difficult to get time from people to dedicate to automation. This is starting to change, however, with a mandate from upper management for increased efficiency through automation.
Industry wide effort to standardized software development for life sciences automation.
More time in the development cycle and beta testing.
Establish a standard group (ALA or Society for Biomolecular Sciences (SBS) or together) and start working on standards.
Improve control software—get hardware developers early on involved with users and feedback. Push developers/vendors to establish networks to share programming examples'/methods' patches.
Make sure controlling software is as bug-free as possible before release.
More education that leads to people with automation knowledge.
Developing New Assays/Procedures Directly on Automated Systems
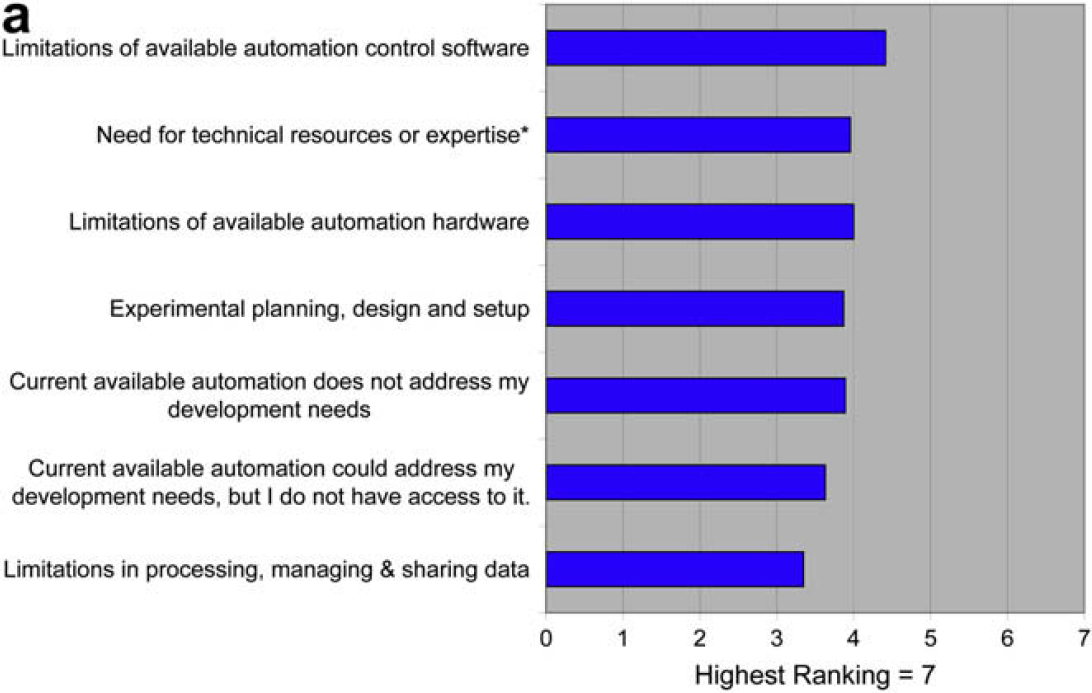
When asked what could be done to improve the situation, users offer the following (unedited) responses:
Loaner equipment.
Develop expertise.
No idea.
Automation software to be compatible with DoE software to optimize and develop quickly new processes and assays.
N/A.
Larger budget.
Same as previous.
Increase level of automation.
Data crushing/mining using multiple software platforms becomes extremely tedious for the end operator.
More education at the level of assay development.
Expand commercially available control software options.
Better automation.
It is very difficult creating automation flexible enough for assay development.
Better use automation for assay development.
To absolutely get the commitment from management and end users before to get money (central nerve of all wars…).
Not enough systems available in labs so more capital expenditure.
Companies should do their homework.
Need more automation to meet typical analytical measurements needs—ALA is too focused on drug discovery and DNA research.
Once available the resources if trained can deal with the hardware and software issues.
Same answer as for previous question.
Partnerships with external instrumentation/system development labs, internal focus.
Further development in the use of automation for cell culture and cell-based assays.
Same as question #1.
Depending on the method requirements we may sometimes need to purchase new peripherals for our existing hardware, or completely new systems. We try to be creative, although and have come up with some stop-gap solutions, which work surprisingly well.
Improving problem definition.
User-friendly software—incorporation of design of experiment modules in liquid handlers.
Making sure automation control software is as bug-free as possible.
Same as question before.
Actually more time is needed.
Labs should do more to foster communication between automation specialists and scientist.
Improved labware teaching implementation and integration of smaller devices from third party.
Again, we have a low pool of technical expertise, and had a great deal of difficulty in hiring someone with sufficient experience to help grow our knowledge base.
Corporate budget increase.
Widespread adoption of control and data exchange standards.
Every device has its own graphical user interface (GUI) or control software architecture that needs to be learned individually. It would be interesting to have automation control software working group put together that lists strengths and weaknesses across various platforms and then makes recommendations.
More training of more individuals.
Improve user expertise and partnership with automation experts.
Push management to allow for more funding to support automation projects.
Again, a commitment by vendor to address important but still underserved segments.
Again resources…
Acquire additional automation.
Know who your customers are and their requirements, so the experimental planning and design time can be minimized and efficient.
New assays are the bane of every project as they require de novo construction of routines, assays, and training. It would be interesting to have more standalone equipment that encompass flexibility and then coordinate these machines in a workcell or integrated piece more easily.
An assay developer familiar with the science of the assay and familiar with automation.
Improved acceptance of DoE and QbD approaches to these efforts.
Resources.
Reconfiguring Automated Systems for New Assays/Procedures
When asked what could be done to improve the situation, users offer the following (unedited) responses:
Plug-and-play interoperability of different vendor equipment.
Build awareness of training opportunities.
More and intelligent control software.
The ease of integration and control of third party equipment and sensors, for example having software that could easily interface with flexible control software such as Lab View.
Use automation with similar control software.
Greater flexibility of scheduling software.
Same as previous.
Improve/enforce standards.
Again, end users of lab automation tools are required to become experts in multiple software platforms. The transferring of methods become very tedious.
Reconfigure requires expertise not available in-house.
Plug-and-play standards adopted by all vendors.
Better hardware.
Create a knowledge base around creating automation-friendly processes. For example, if you modify your manual method to more closely match the automated method, incorporation becomes much easier.
Truly flexible, yet simple to operate control software.
Flexibility of the existing developed HW/SW is the key: modular and exportable.
Manufacturer collaboration—open architecture for software/firmware.
Software concepts still not adequate, interoperability limited, each company following own approaches.
More technically trained resources.
Again, software flexibility is the answer, at least in Pharma Dev.
Collaborative effort to improve protocol engines of control software and their flexibilities. Collaborations with bench scientists and external software providers.
Limited by lack of interfaced modules.
There needs to be highly skilled programmers on staff to make the automation perform as desired.
We do not usually have too many problems with this. It is important to harmonize our different assays as much as possible with regard to set up.
More flexibility in software and hardware.
Train staff as best as possible—High performance system = software + hardware + people.
See last question.
Cheaper cost so I can buy all the hardware I need.
Companies should strive to create modular of their hardware and especially the software that controls them, so that new processes can be created quickly by using modules already optimized.
Software improvements.
We have found that we are attempting to use our system at the outer most limits of its capabilities.
Vendors need to recognize the flexibility demands of industry.
Automation standards development.
Ideally, running a new assay should simply require writing a new method. This works as long as systems are designed with enough capacity in process zones or if they have modularity that allows quick hardware changes that are automatically recognized by the scheduling software.
More intervendor cooperation (not just in the form of documents/& standards).
As in 1 and 2.
Improved training.
Resources in nonpharma areas.
Resources.
Ability to bring new modules on-line quickly and easily.
Have the experts readily available and who are familiar with your process.
Reconfiguration of hardware requires lots of time and would need a consulting firm or in-house mechanical and software group for the rearrangements. For software, it is possible to rewrite software for the same system in-house usually. Most times getting new hardware and software is the only way to perform brand new tasks.
Reconfiguration is often due to a labware change. Control software deliberately makes this difficult to protect the hardware.
The software isn't that difficult any more.
Open source, better documentation, and universal language.
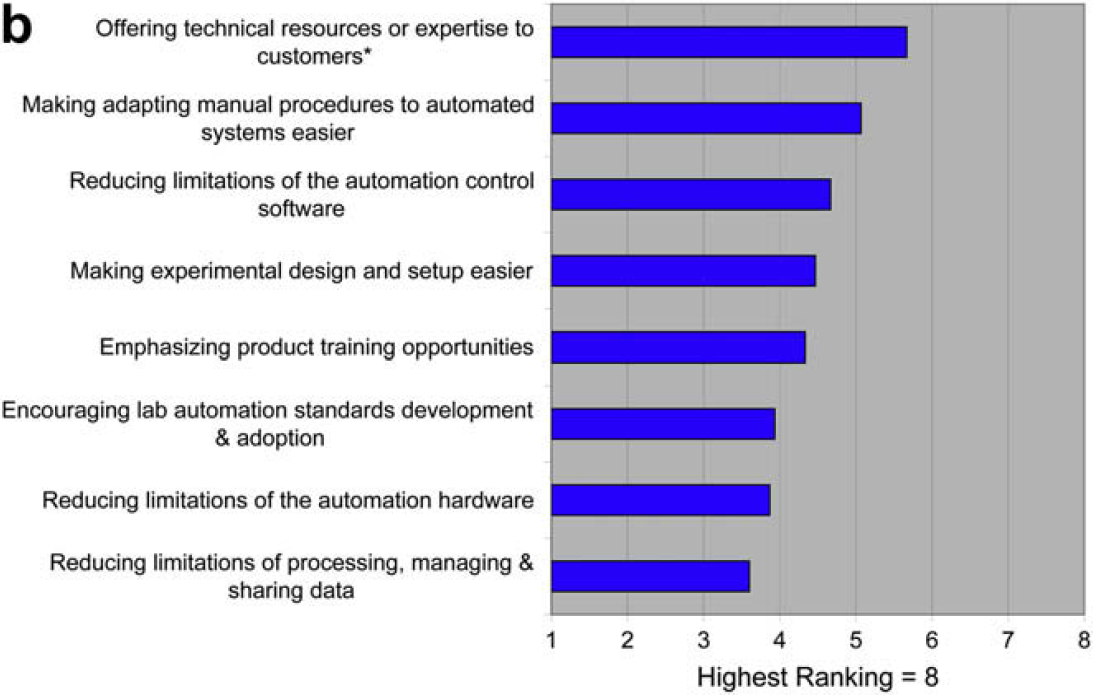
One goal of this survey was to cast further detailed light on the results of the 2006 survey. That previous survey indicated that the use of laboratory automation was limited by developing and validating new automated procedures and reconfiguring for those procedures. This 2008 survey shows that these limitations are the result of the difficulty of translating manual procedures to automated procedures and the limitations of automation control software. This survey also indicates that the technology providers understand these issues, and place them at the top of their priority lists.
The need for additional lab automation standards appears not to be limiting to users or a top priority for providers.
Transferring or Validating Assays
The following two questions were asked to gain further insight into how users typically transfer and validate assays on an automated platform.
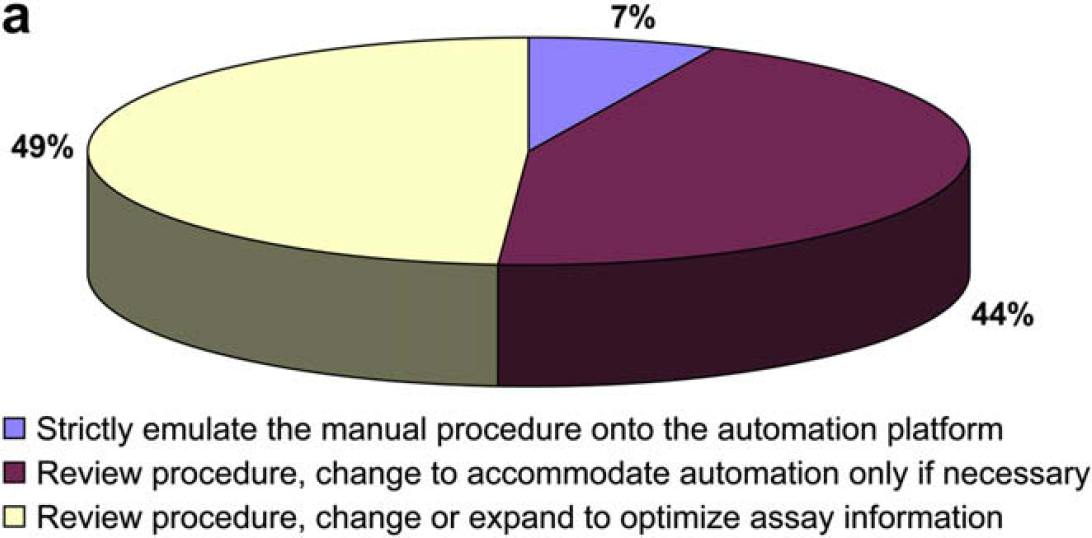
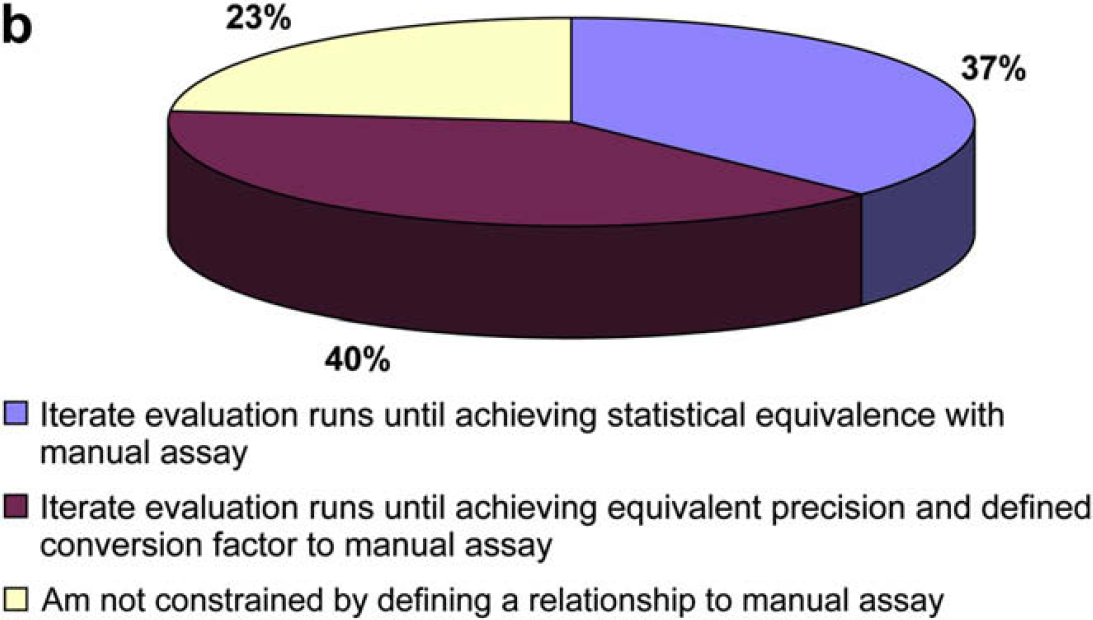
It would seem that the respondents do not feel at all constrained to strictly emulate a manual procedure on an automated system, but they do feel constrained to demonstrate a defined statistical relationship between manual and automated methods.
Automation Trends
The following questions focus on identifying general trends in laboratory automation and technology.
Characterize the State of Laboratory Automation in the Following Disciplines as Embryonic, Developing, or Mature
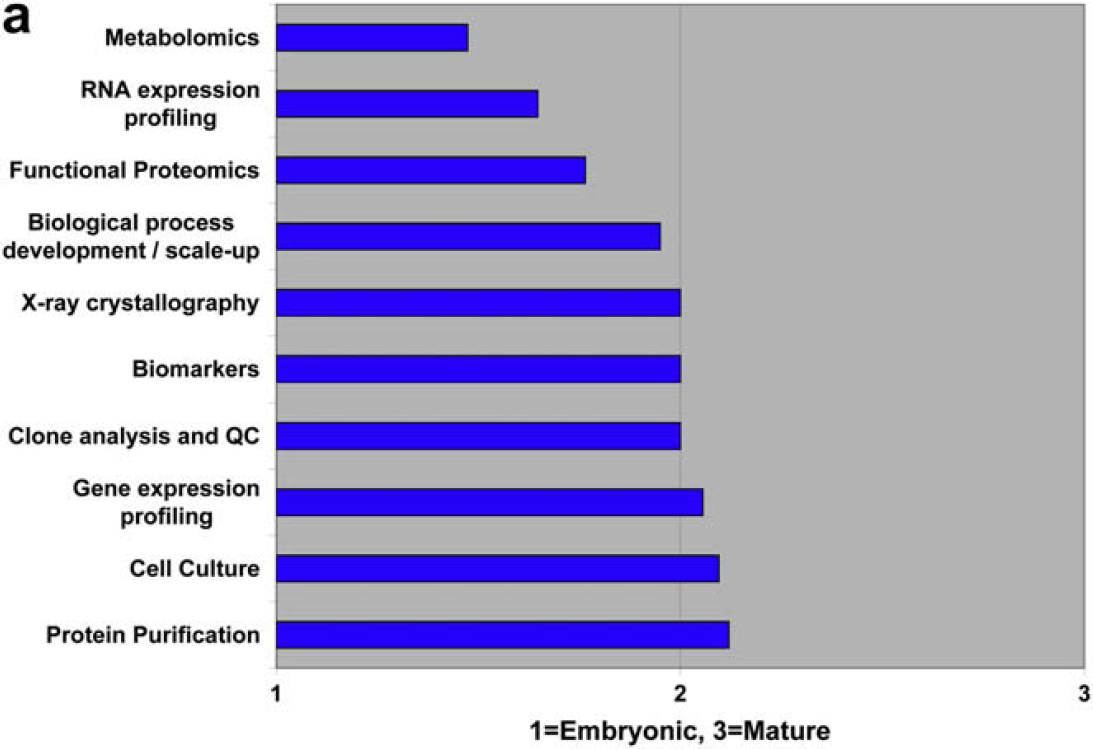
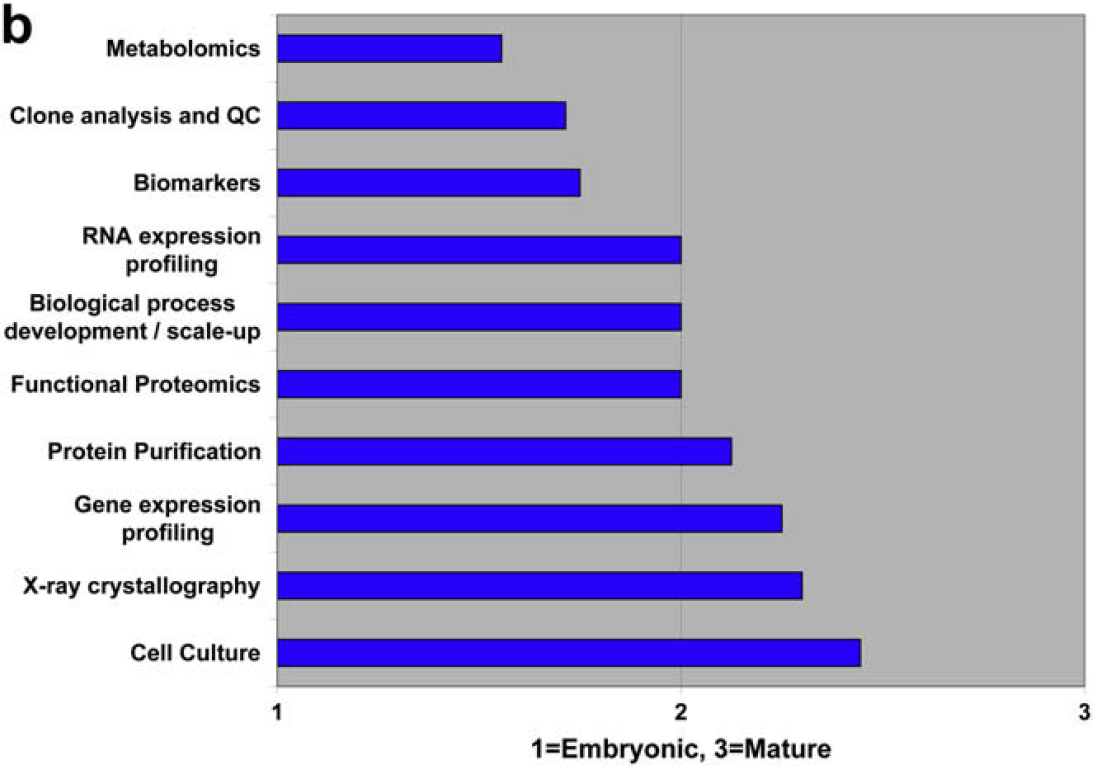
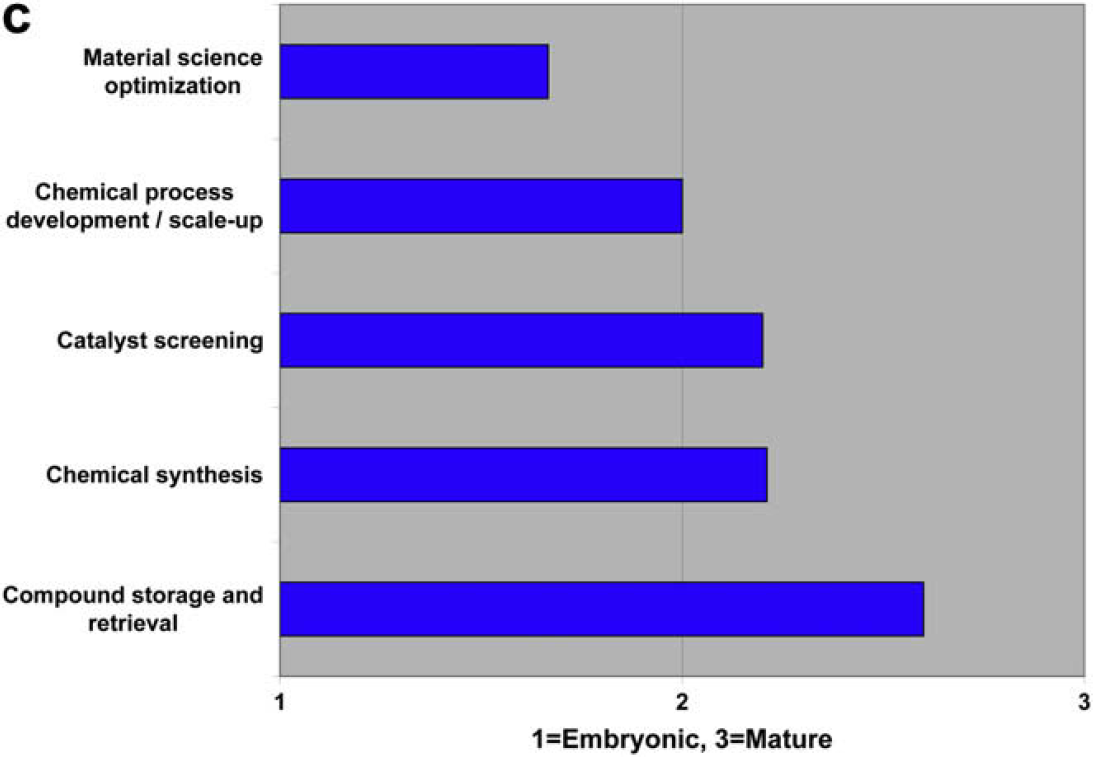
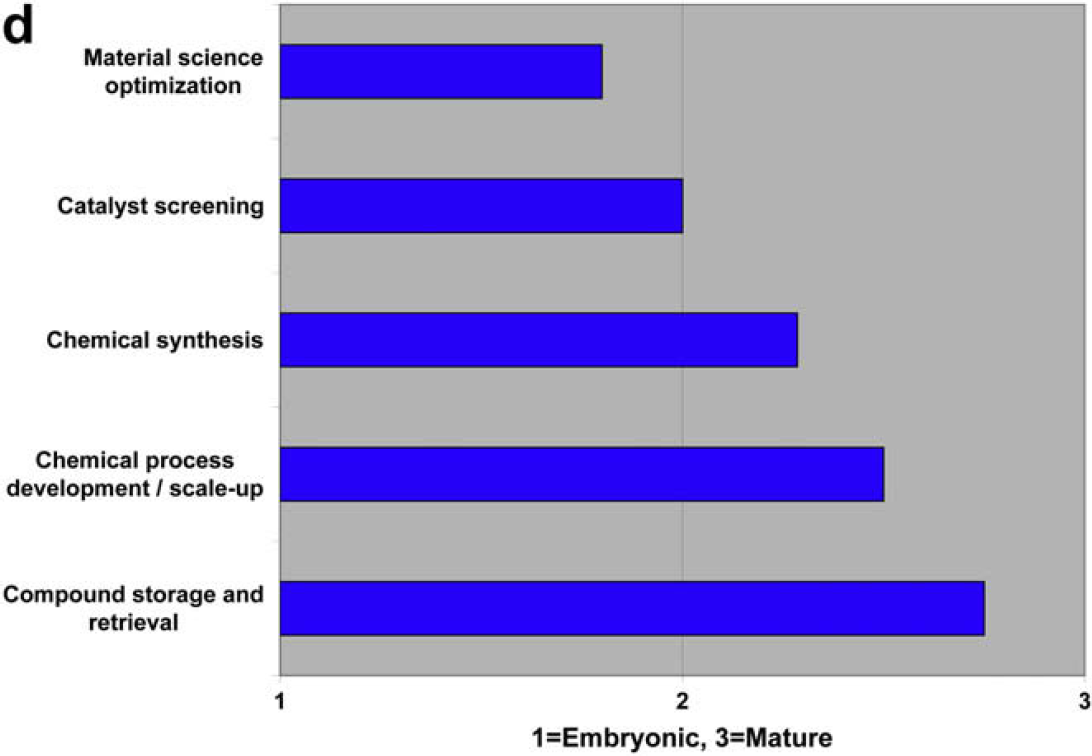
Both communities agree that compound storage and retrieval are automation mature, and that material science optimization is embryonic. There is disagreement regarding chemical process development and scale-up.
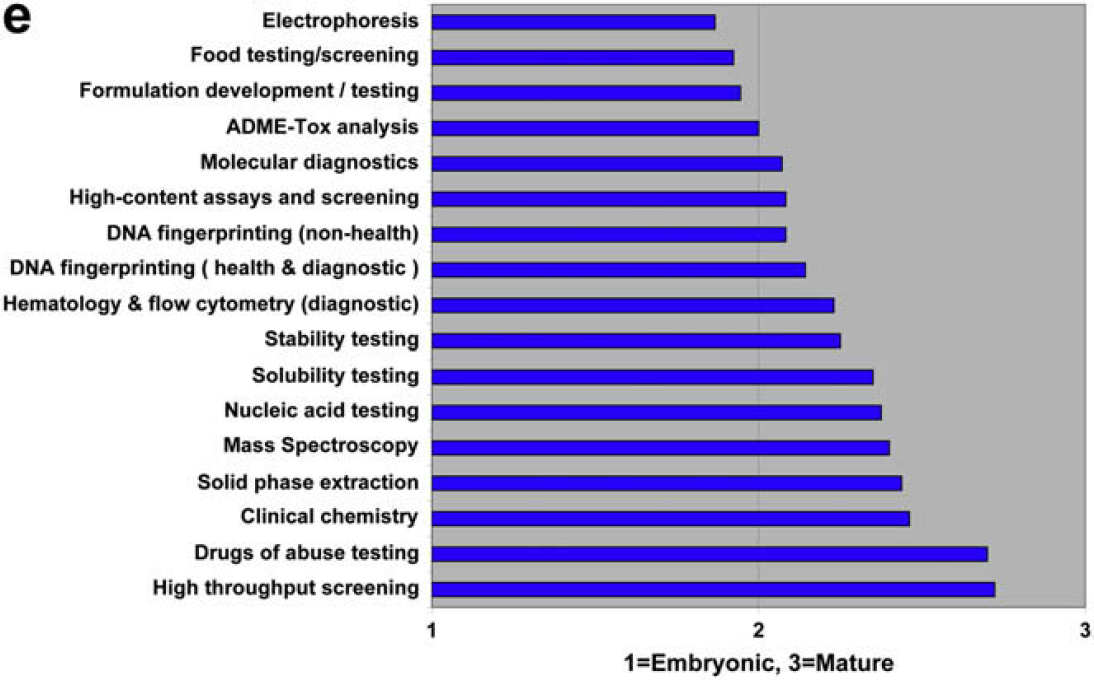
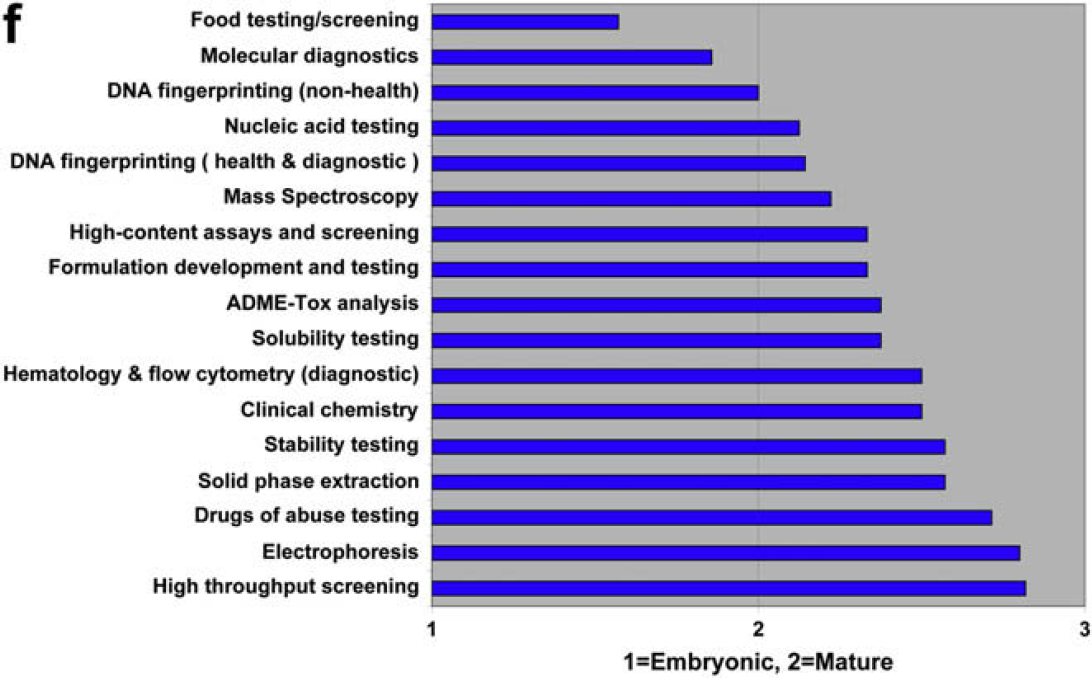
Both communities agree that HTS and drugs of abuse testing are automation mature. They also agree that food testing/screening and molecular diagnostics are among the most embryonic. A striking difference between users and providers are the rankings for electrophoresis. Users rank it as the most embryonic discipline, whereas providers rank it as the second most mature!
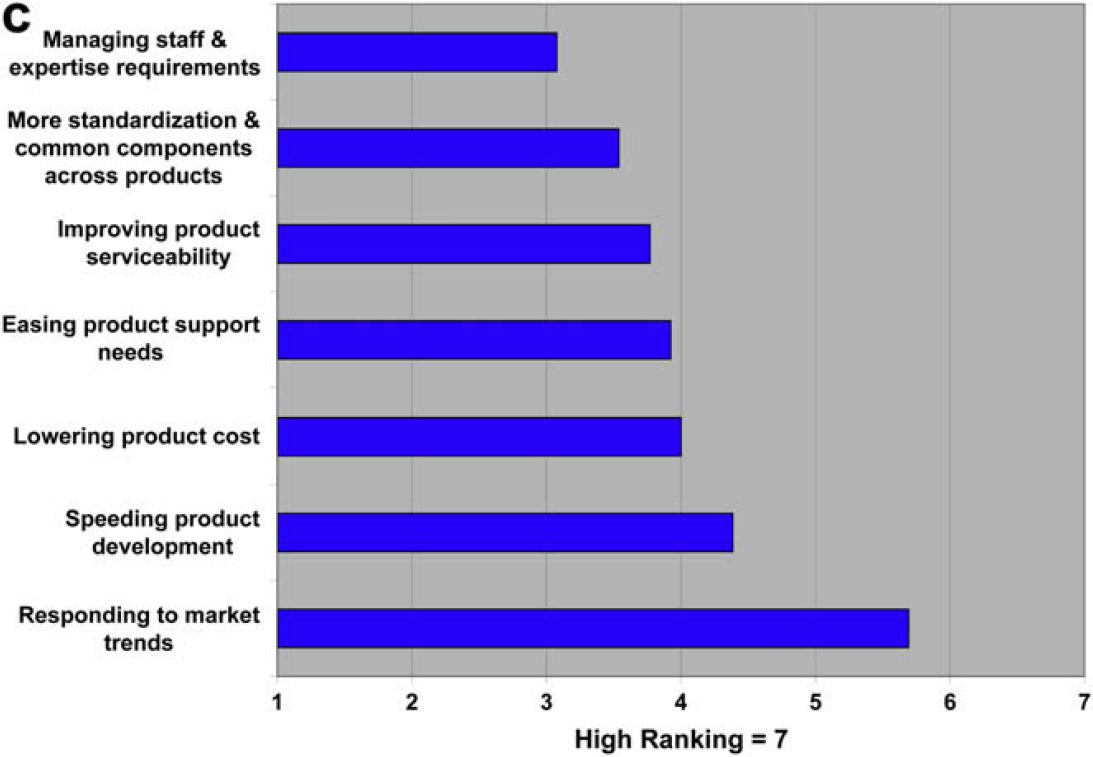
Automation Focus
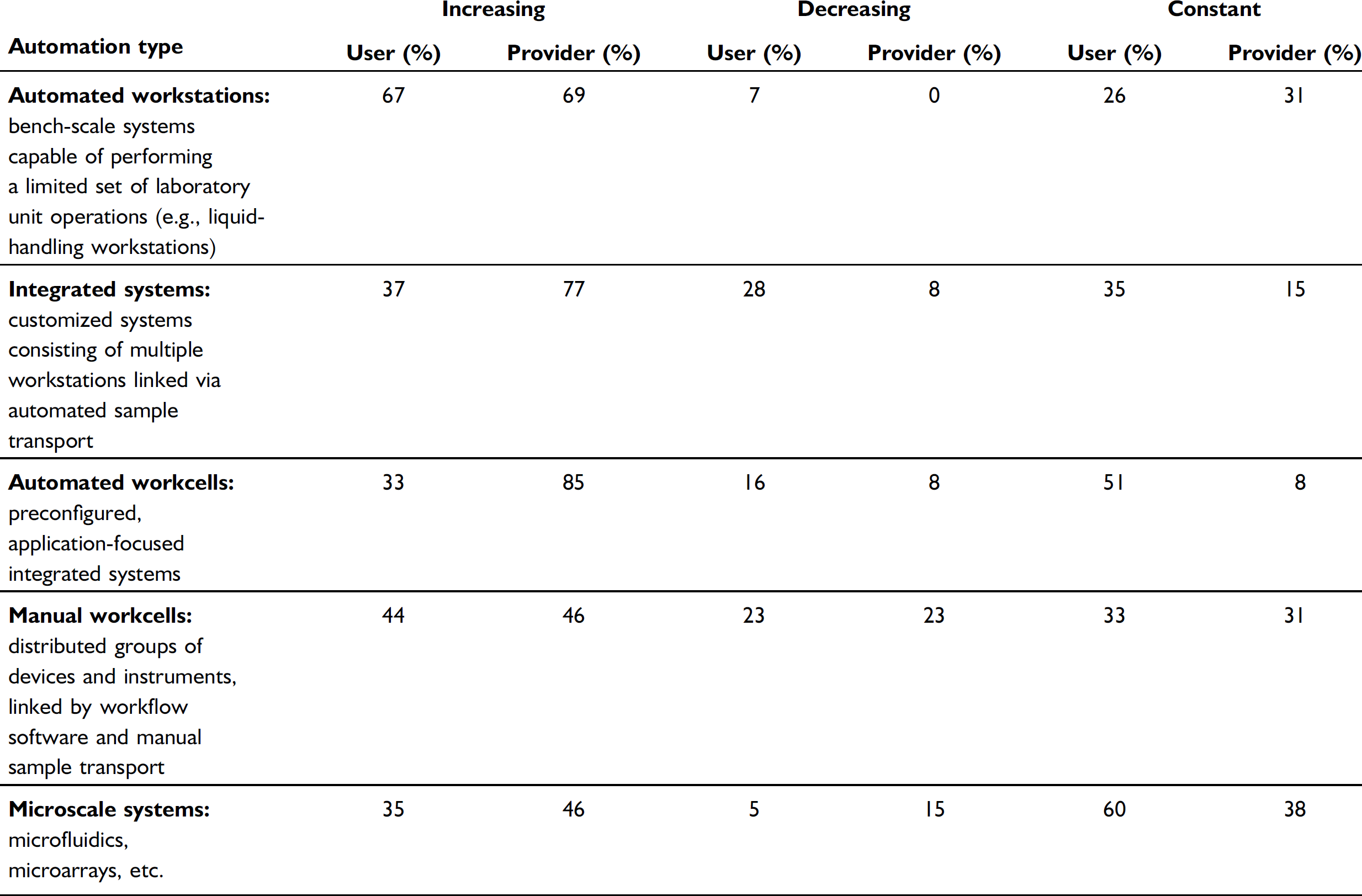
The only type of automation that users report they are increasingly focused on is automated workstations. Otherwise, their focus on other types of automation is neither strongly increasing nor decreasing. Providers, on the other hand, report that their focus on multiple types of automation is increasing. Specifically, 77% of the providers report their focus on customized integrated systems is increasing, but only 37% of the users say the same; 85% of the providers say they are increasing their focus on automated workcells, but only 33% of the users report the same. The providers report that 69% are increasing their focus on automated workstations, which is very similar to users.
We are definitely increasing our focus on miniaturizing our processes. We were involved early in exploring lab-on-a-chip technology (Caliper) for high-throughput screening and have several miniaturization projects underway right now in other areas. It's interesting to note, however, that miniaturization is relative. In the process development world, miniaturization means scaling down from liter scale or scaling process time from hours to minutes. Miniaturization does not always mean micrometer dimensions. It's all about trying to do better and more efficient science.

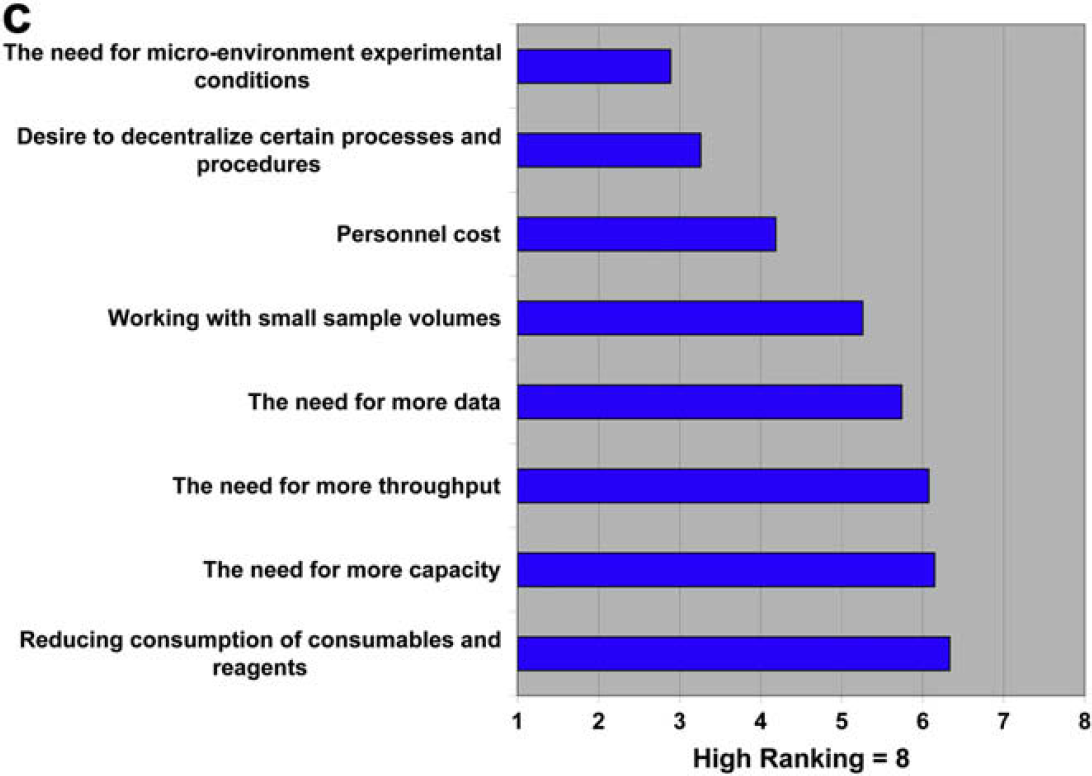
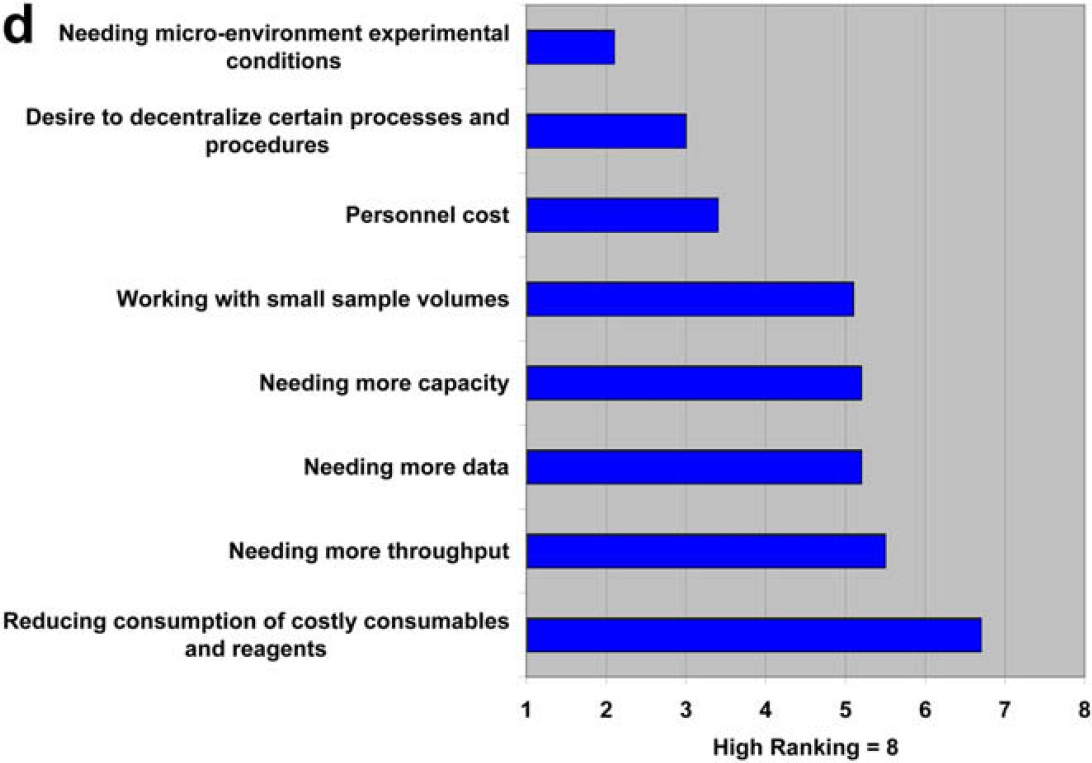
Both communities are in agreement about the trend toward miniaturization, but as was seen in question 2.2, the technology providers seem to be more aggressive in pushing the technology than the users are in adopting it.
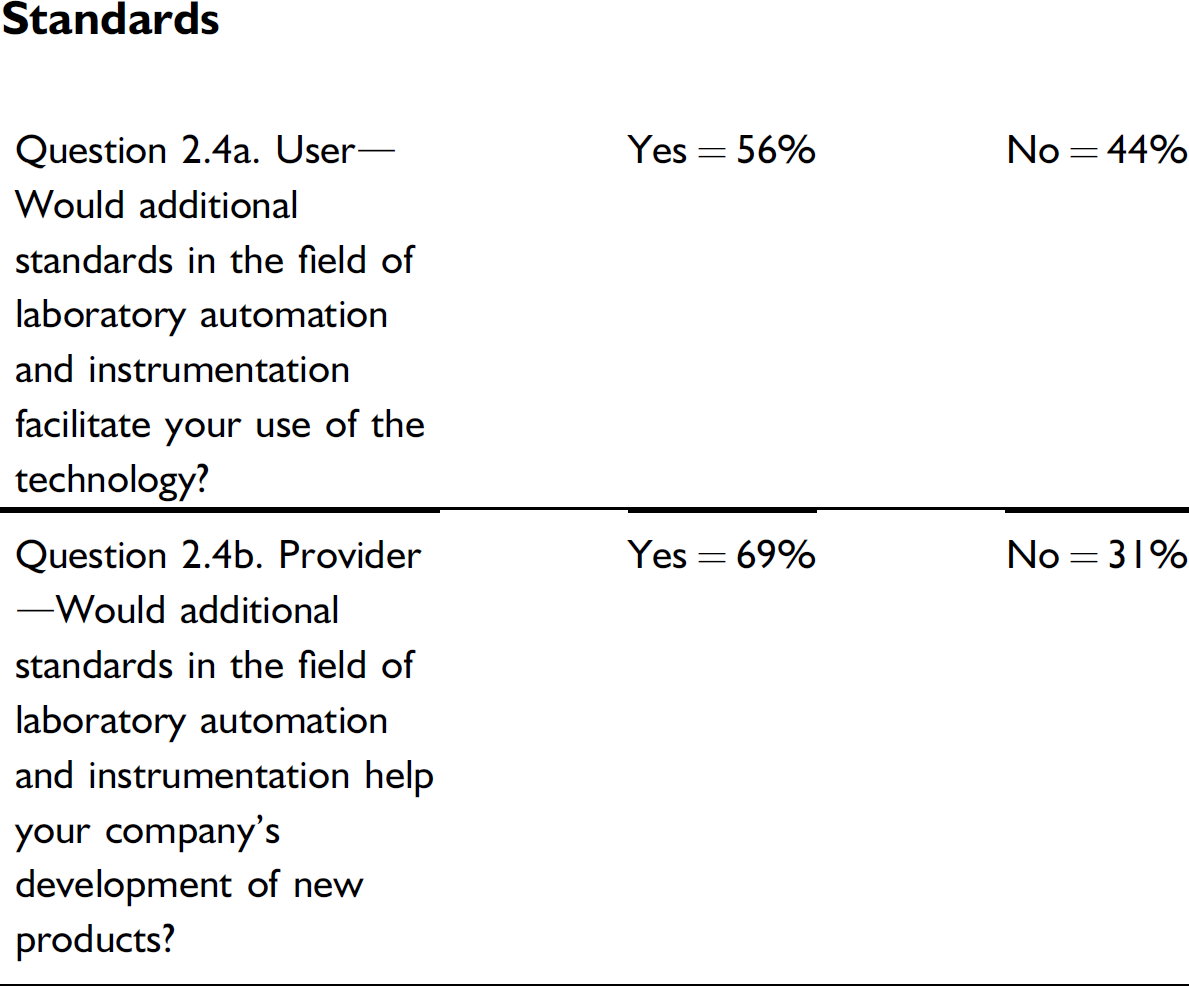
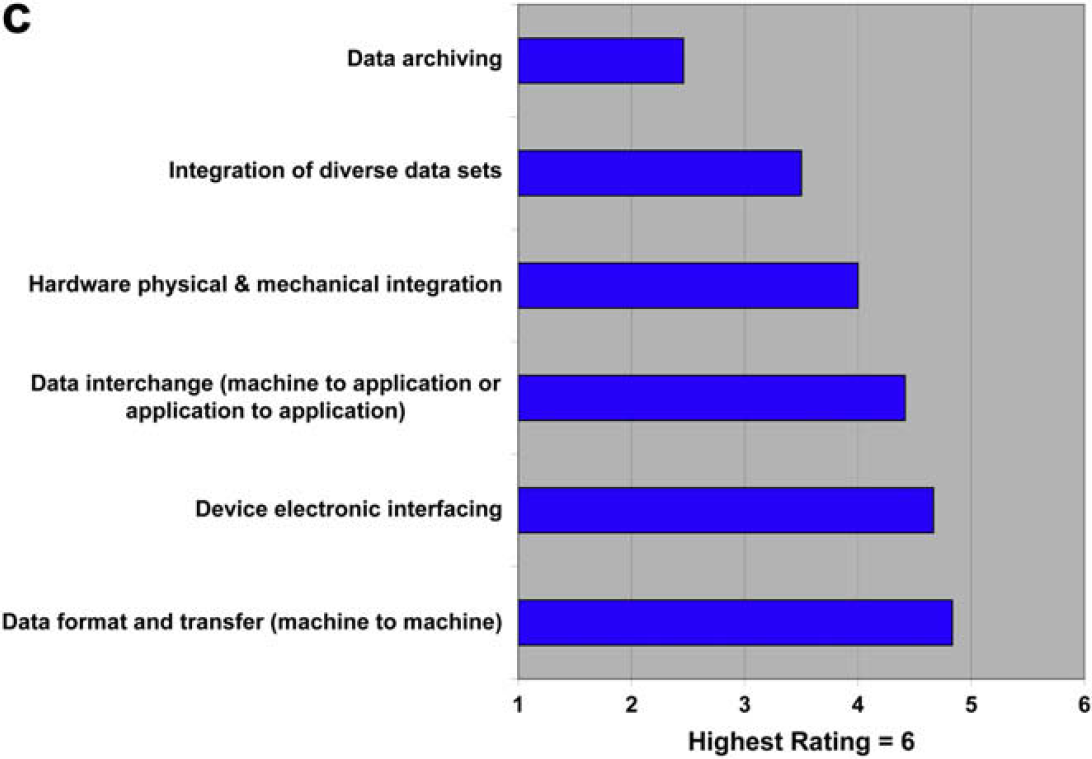
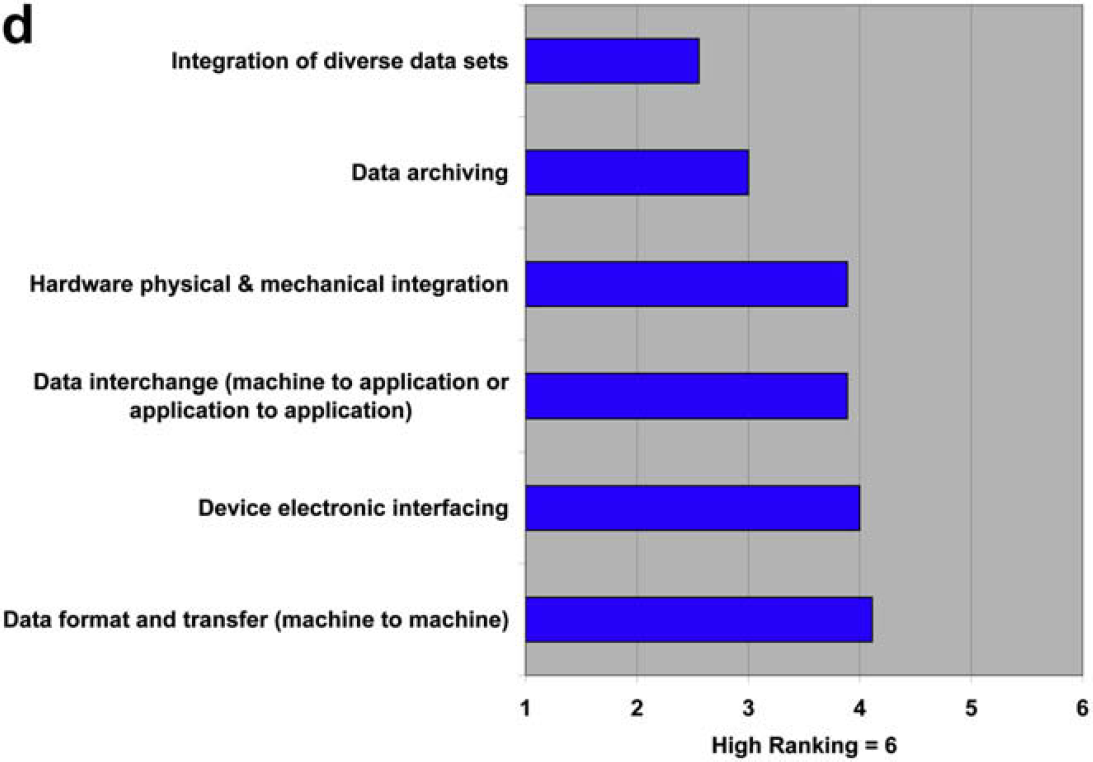
The results of this question are interesting when compared with the responses regarding standards in questions 1.1–1.3, where the need for new standards ranked at or near the bottom of limiting factors. In question 2.4, both users and providers indicate additional standards would be helpful, with the providers being measurably more positive than users. Both parties agree almost completely in ranking the type of additional standards that would be most helpful. So, are standards something that would be “nice to have,” but never rise to high priority?
Data Sharing
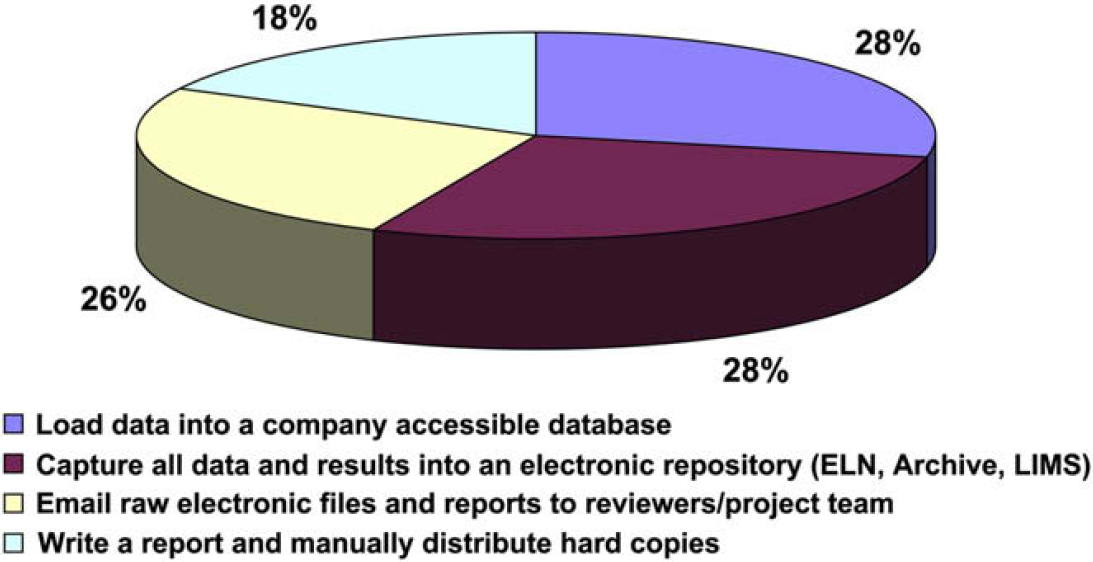
In this supposedly paperless age, nearly a quarter of the respondents report they share data via written hard-copy reports despite the fact that they likely prepare these reports on computers and could use e-mail as the means of sharing such documents.
Electronic Interfaces
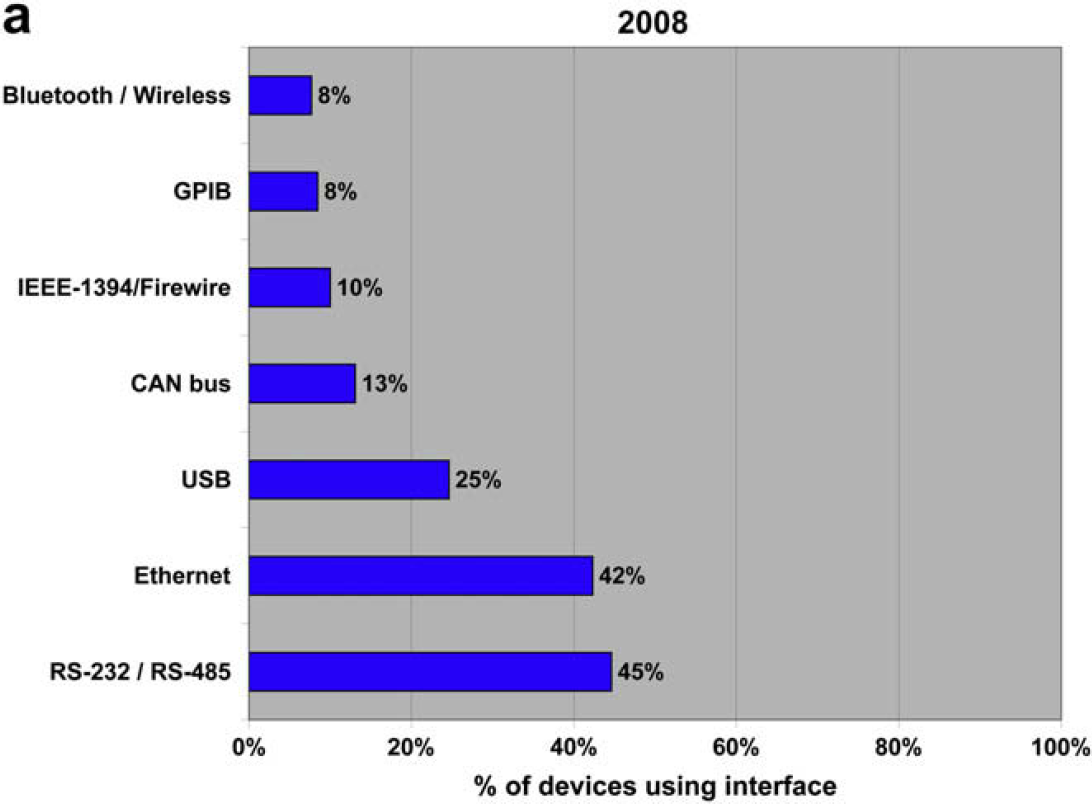
Despite being over 40 years old, the RS232 interface is still the most widespread electronic interface in our industry. It's closely followed by Ethernet, and everyone predicts that it will be overtaken by both Ethernet and USB over the next 5 years, but this has been predicted for a number of years. The fact remains that RS232 is inexpensive and simple, and so is not likely to disappear.
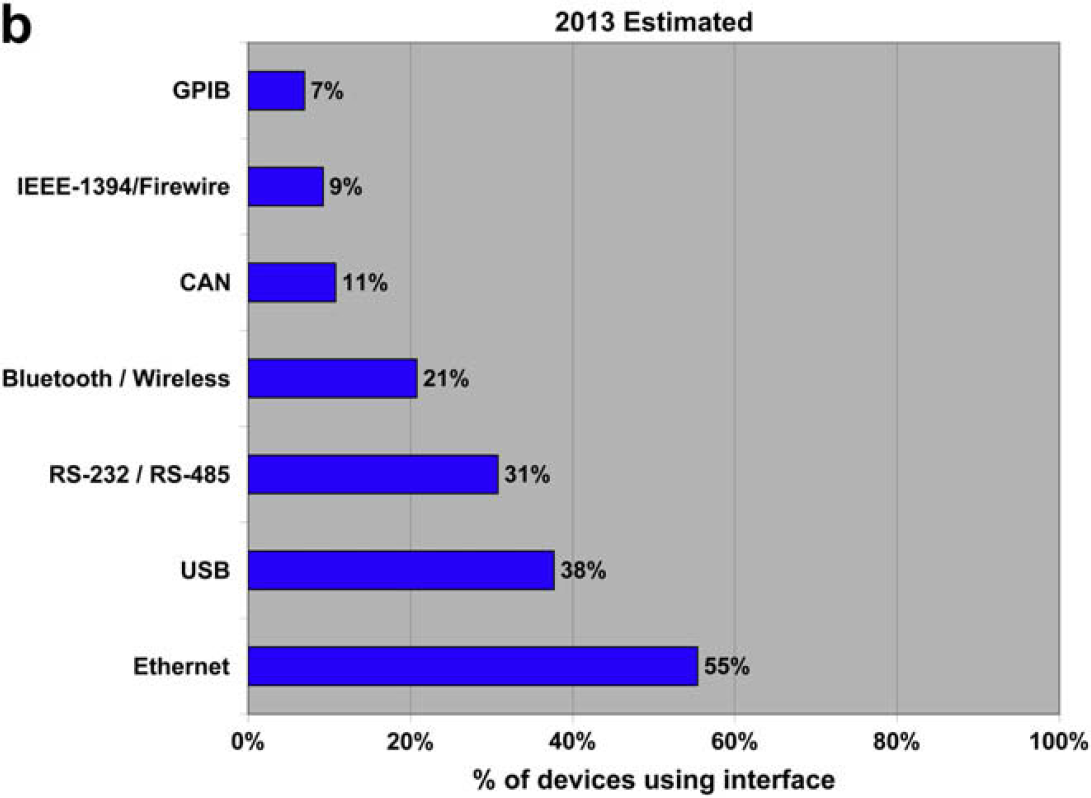
Organizations and Laboratory Automation
The questions in this section focus in-depth on user organization staffing trends for supporting laboratory automation. In the 2006 survey, 75% of users indicated that their organizations used full-time internal staff to serve as laboratory automation resources.
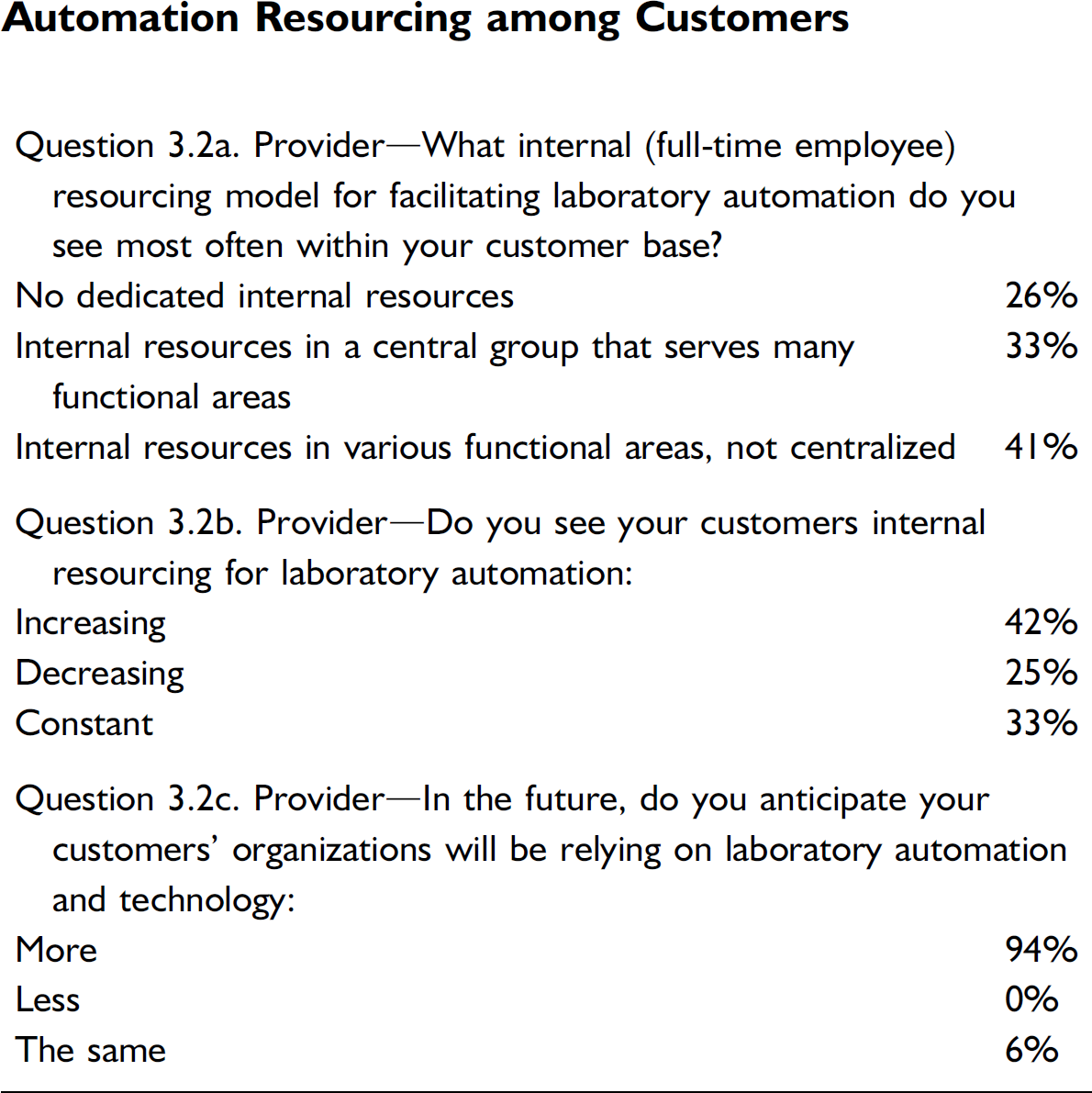
Internal Automation Resourcing

Automation Resourcing among Customers
Future Trends

Conclusions
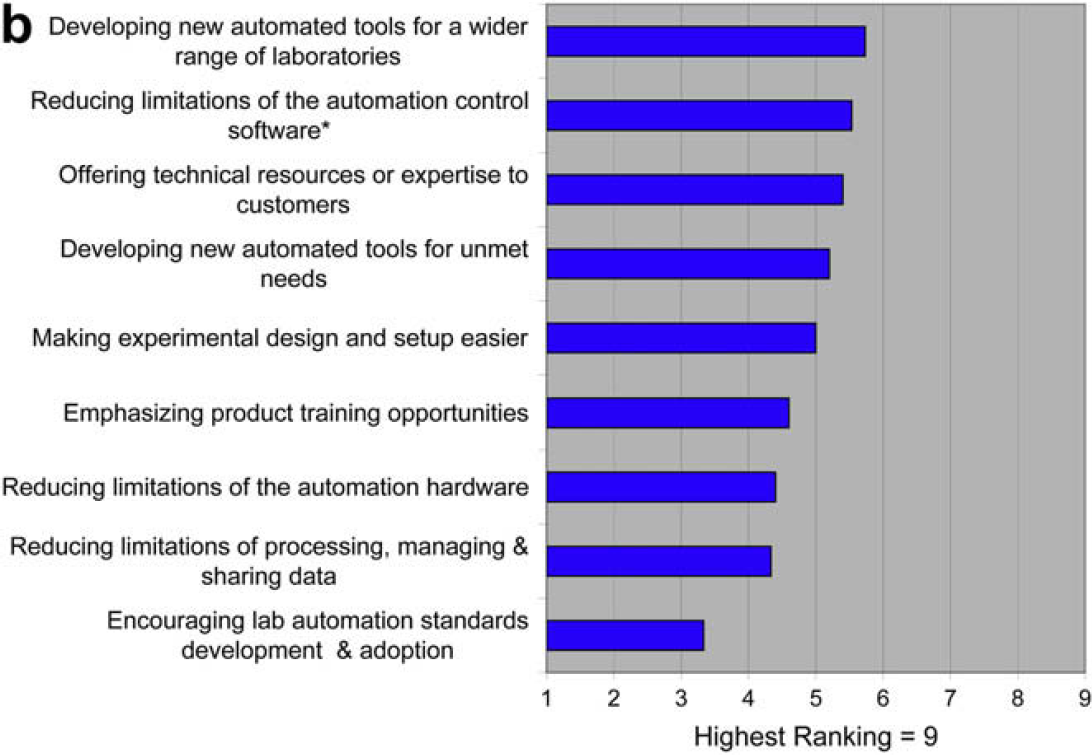
This 2008 ALA survey was meant to be a detailed follow-on to the 2006 ALA survey. In this 2008 survey we find, with little surprise, that the difficulty of translating manual procedures to automated procedures is a major issue. What is enlightening, however, is the opinion that the limitations of automation control software contributes much more to this problem versus the limitations of hardware. This echoes the feedback from the 2006 survey, which indicated that issues of hardware reliability and capability had dropped to the bottom of the list of concerns.
A new insight from this survey is that the technology providers understand these issues and have prioritized them accordingly. In the words of Dr. Hansjorg Haas “I found this LabAutomation 2008 follow up survey particular useful because it provides views of both providers and users and shows their alignment and misalignment (relatively few) on automation need and use, market trends and organizational support structure. Many of the findings confirm perceptions and anecdotal evidence we found in customer communications.”
Both user and vendor communities also identify three embryonic areas of laboratory automation development: metabolomics, material science optimization, and food testing and screening.
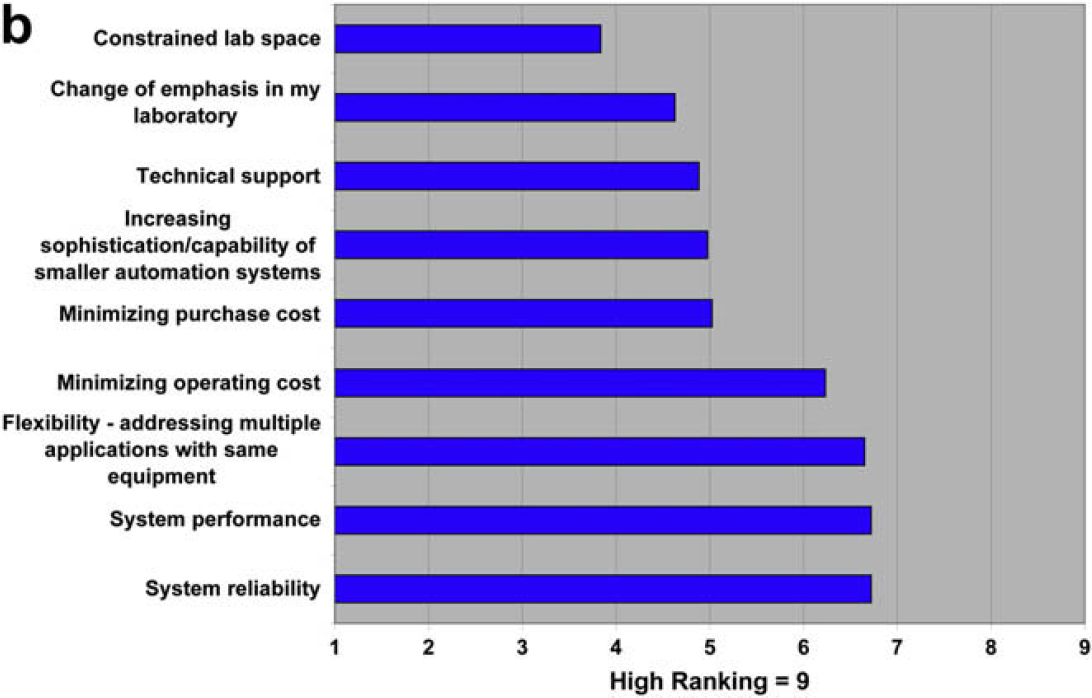
We find that users are increasing their focus on automated workstations, being driven by performance and reliability. This trend is not clearly reflected in the providers focus, so it remains to be seen how this sorts out in terms of future system sales.
Everyone wants to miniaturize the procedures, driven by cost savings. It's a particularly cost-sensitive time in the industry.
The push for additional laboratory automation standards is modest at best. This is interesting given that few standards exist currently. What respondent interest exists is focused on standards for data format and transfer, which happens to be about the only active area of lab automation standards development (analytical data interchange).
According to the survey respondents and our expert commentators, we are a long way from realizing the true paperless laboratory despite many headlines and discussion to the contrary. Don't discard your printers just yet! In that same vein, we continue to hang on to a communications protocol (RS232) that is over 40 years old! Although many aspects of our field change rapidly, obviously some don't.
A strong majority of organizations appear to have some form of internal automation resources. In this survey, we drilled down to find that these resources are roughly split between existing in centralized groups or being scattered throughout functional areas. Virtually all of these resources are involved in both automation development and ongoing support. Almost everyone expects their organizations will be relying more on lab automation in the future to cope with increasing workloads, streamline operations, and add new capabilities.
The final, and perhaps most reliable observation of this survey is that
Footnotes
Acknowledgments
The funding and infrastructure for conducting this survey was provided by the Association for Laboratory Automation.
