Abstract
This article reports the results of a survey examining the practice of laboratory automation in industrial laboratories. This survey polled 400 ALA industrially employed members and received 72 responses representing the R&D components of 37 different companies in Pharmaceutical, Biotech, and Agriculture Science sectors. The survey consisted of 14 questions. For purposes of this report, the survey results are broken into three sections: (1) How laboratory automation gets done; (2) Why automate—success and return on automation; and (3) Technical aspects of automation. Some of these same questions have been part of surveys conducted in the past among attendees of LabAutomation Conference short courses, and that historical data will be compared. (JALA 2007;12:239–46)
Introduction
There are few benchmarks available about why or how laboratory automation is implemented, or what the impact of such is. This survey is an attempt to explore those questions and begin a base of information from which we may be able to track trends and directions in coming years.
The companies represented by the 72 survey respondents are:
3M Pharmaceuticals
Allergan Inc.
Ambit Biosciences
Amgen Inc.
Amylin Pharmaceuticals
Astra Zeneca
Biobide
Boehringer Ingleheim
Bristol-Myers Squibb
Celgene Corporation
Centocor R&D, Inc.
Codexis
deCODE genetics
Dow Chemical
Egea Biosciences Llc
Elan Corporation
Eli Lilly
Genmab B.V.
Genzyme
Glaxo Smithkline
Integrated DNA Technologies, Inc.
Merck
Monsanto
Neurogen Corporation
NNE
Novartis
Novonordisk
Novozymes
OSI Pharmaceuticals
Pall Life Sciences
Pfizer
Pharmacopeia Drug Discovery
Pioneer HiBred
Sanofi-Aventis
Schering Plough
Transnetyx, Inc.
XDx
Survey responses were not linked to a respondents name or company, but were logged with an anonymous numerical identifier. The answers of a given anonymous respondent to various questions can be compared and contrasted. Select questions have been part of surveys given to short course attendees at past LabAutomation Conferences. The size and diversity of those respondent groups were similar. This data is presented for historical comparison.
The author has added commentary to supplement the survey data. The opinions expressed are his own, based on 25 years in the industry. Dr. John Houston, VP of Applied Biotechnologies and Discovery Biology for Bristol-Myers Squibb, Wallingford, CT, was asked for his perspective on the questions that are of a more strategic nature. His comments (in italics) will be found throughout the survey discussion and are meant to lend the perspective of someone in an executive management position, which may be somewhat different from the average survey respondent.
Section 1: How Laboratory Automation Gets Done?
Question 1.1: Describe the Percentage of Automation Sourcing in Your Laboratory/Organization from Each of the Listed Sources.

The data indicate that the usage of off-the-shelf automation has been relatively constant over since 1996, and therefore about half of the lab automation purchased continues to need some level of customization. How that customization is achieved is the changing factor. The amount of automation developed in-house has declined, whereas that developed/customized via a third party integrator has increased since 1998. This is a natural evolutionary trend for any technology, not just lab automation. Early in any technology cycle, development and customization work is done by the “gurus,” the tinkerers, and early adopters of a given technology. Eventually, sensing a business opportunity, the commercial marketplace responds by offering similar services and tools—often with better quality and at lower cost—so the need for “in-house” guru's to tinker decreases. It is also worth noting that customization via the original equipment vendor has stayed fairly constant, and is the largest single source of customization. Although many vendors ideally would like to simply sell “boxes,” the fact remains that the lab automation business is still very “custom driven.” Despite the fact that today's off-the-shelf lab automation is increasingly sophisticated and feature rich, no lab is exactly the same, and customization is still in demand.
Question 1.2: Does Your Company/Organization Employ Dedicated Internal Staff Serving as Automation Resources?

There is a trend toward more organizations employing in-house expertise, which is interesting in light of the trend toward less in-house customization. Even though more work is being “farmed out,” internal expertise is necessary. These internal experts may be spending more of their time planning and managing outsourced projects than in the past. Coordinating a complex project with one or more outside resources can be a very time-consuming task. These results may also indicate that the customization work that remains to be done in-house is the “high-hanging fruit,” that is, difficult tasks requiring more resources, not less. Or, the results may simply reflect that the growing total amount of automation in labs requires more staff devoted to programming, reconfiguring, tweaking, and maintaining existing automated tools, but not actually creating new automated systems.
It should be noted that this question only evaluates the “presence” of internal resources, not the number of internal resources, which is very difficult to quantify in a survey. Also, this survey did not reach companies without ALA members—companies with potentially a lesser commitment to automation technology. Still, it is apparent that the industries represented in this survey are technology driven, and as such they continue to use resources to manage that technology drive.
Question 1.3: Describe the Ways Your Organization Provides On-Going Support for Laboratory Automation.

Outsourcing is the single largest source of support, but over half of the total support is still provided by internal resources. This may be a reflection of the highly customized nature of the final installations of much lab automation.
Question 1.4: Has Your Company/Organization Standardized on Automation Vendors to Simplify On-Going Support?

Given the highly independent nature of laboratory scientists, it is surprising that the “Yes” number is substantial. Although IT groups have long been successful at controlling what office computer scientists use, the same scientists usually bristle at restrictions on what goes into their laboratory. Internal automation development resources can easily become bogged down with on-going support responsibilities, so perhaps these numbers reflect an attempt by such resources to simplify their support role and/or make outsourcing of support more effective? Certainly, outsourcing support is easier if an organization owns a “critical mass” of equipment from a given vendor.
At Bristol-Myers Squibb, Dr. Houstons’ team has become more involved in helping to manage capital budgets in order to guide automation and technology purchases and they are involved in this dialog much earlier than in the past. Their goal is to help scientists see their equipment wants and needs in the context of the broad range of equipment offerings, and to encourage them to purchase equipment that has a proven track record within the organization. They stop short of actually vetoing the purchase of equipment from “non-approved” vendors, but find that most scientists are quite reasonable when given a comprehensive set of information to guide their decision making.
Section 2: why automate? success and return on automation
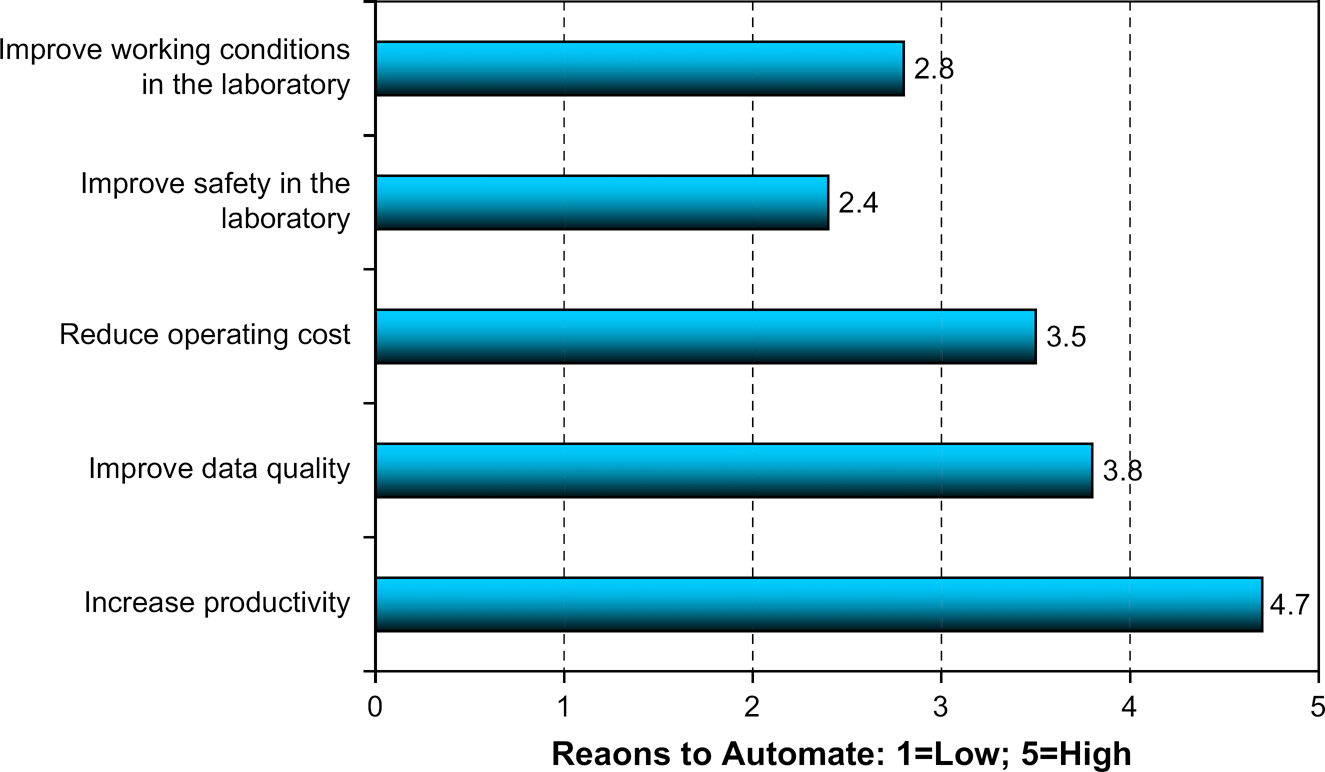
Question 2.1: Rate These Reasons Why Your Organization Uses Automation, Five Being the Highest Rating and One Being the Lowest Rating.
Throughout our time of asking this question in surveys, the improvement of productivity has always ranked clearly as the #1 reason industrial R&D laboratories automate. This is no surprise. The drive to increase productivity cuts across most industries, but in decades past was mostly focused on the manufacturing or operations components. Only in the last decade or so has productivity improvement become a strong focus for the industrial R&D component, largely because many R&D components have not produced new products at the desired pace. Our survey respondents do not appear to equate increasing productivity with reducing operating cost. One can surmise that generating more new products faster is more important than simply cutting costs. This is characteristic of technology-driven industries, that is, those industries that seek to reinvigorate themselves via technology. The spate of pharma spending on HTS technology in the past decade reconfirms that this mindset exists. Such a mindset is not necessarily bad or unproductive unless taken to an extreme of blindly throwing money at technology in the hope that at least some of the blind spending will pay off or in the fear that the competition will spend more.
We were curious if a senior Pharma manager would share the same perspective as the survey respondents. Dr. Houston commented that that his organization has certainly used automation to improve productivity via increasing capacity, improving timelines, reducing costs via miniaturization. However, the biggest strategic driver has been using automation and the accompanying process changes to drive change in the organization. They find that the discussion of how to use automation in the lab or how to do a process differently can be a polarizing moment. There can be initial resistance to change, but through these discussions scientists will often discover that there are new and better ways to their work. Automation can be leveraged to change thinking and to change cultural aspects of how teams work, which leads to an overall optimization of drug discovery.
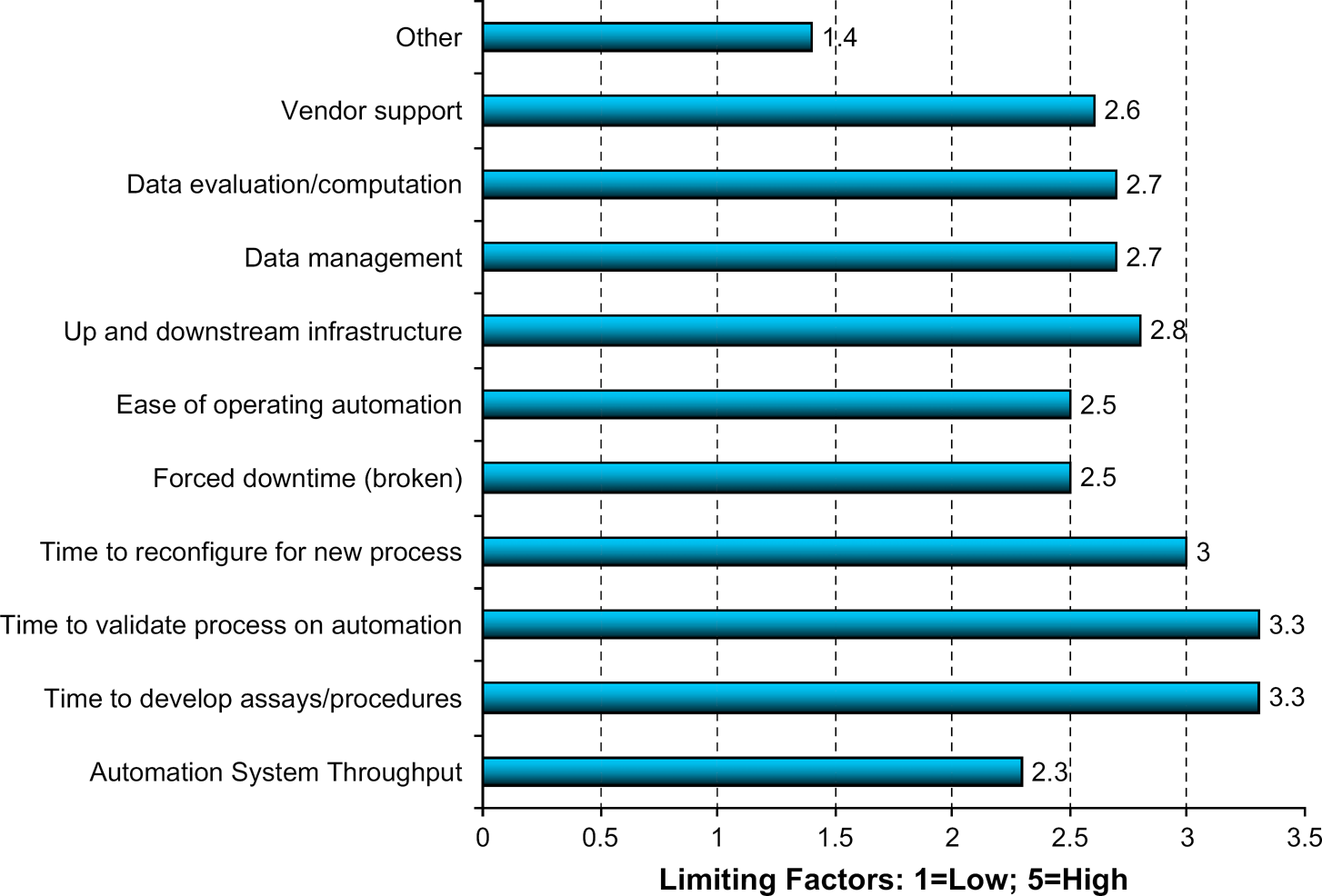
Question 2.2: Rate the Factors Most Limiting to Your Laboratory's Use of Automation, Five Being the Most Limiting and One Being the Least Limiting.
The interesting feature about these responses is that the top three factors limiting the use of automation are the result of using automation to do science. Creating new science, that is, assays or procedures, takes time—lots of it. Reconfiguring automation for those new procedures and then validating those procedures on automated systems also takes lots of time. Notably, one of the factors we hear a great deal about—throughput—gets the lowest rating. Forced downtime, that is, when a system is broken, is rated next lowest, together with ease of system operation. The point is that doing R&D science is hard. It always has been and always will be. Once the science is developed and developed well, the machines repeat it reliably enough and fast enough. The “gap” to be filled involves better ways to develop the science and get it running on the automated systems. The technology-driven approach to filling this gap is to develop better tools to assist in developing assays/procedures and the subsequent automated reconfiguration and validation. The science-driven approach is to find ways to simplify the science. It is always easier to validate and implement a simple automated procedure versus a complex one. The solution undoubtedly involves both approaches.
Question 2.3: My Company/Organization's Senior Management Feels its Investment in Laboratory Automation Has

It is a positive sign that over half of the respondents feel that lab automation has delivered the expected result in their organization. The sizable contingent indicating “mixed results” is not surprising because implementations of technology do not all work out well, despite best intentions or expertise. This survey does not quantify whether the “mixed results” leaned toward good or bad, nor does it attempt to gauge whether the “expected benefits” were reasonable or not. Only one person indicated that automation did not work out in their organization. That same person also indicated their organization use 30% off-the-shelf automation versus the survey average of 47% (Question 1.1). Certainly, using less off-the-shelf tools will lower the odds of success.
Does the presence of internal automation resources (Question 1.2) impact the degree of success with automation implementation? Of the organizations with internal resources, 61% reported “succeeded,” 37% reported “mixed results,” and 2% “not delivered.” Of the organizations without internal resources, 39% reported “succeeded” and 61% reported “mixed.”
Dr. Houston indicates that his organization has definitely achieved the expected success from their automation investments, but only after spending the past several years focused on integrating technology platforms they have come to know and trust and plugging gaps in the processes. They have seen significant improvements in their companies ability to move projects along through the drug discovery process, predict where bottlenecks may appear, and deploy resources and technology to deal with those appearing or potential bottleneck areas.
He feels that in many cases less-than-expected results from lab automation stem from an incomplete understanding of what the success factors really are. For instance, their compound screening program initially defined success in terms of the number of compounds that could be screened per day, but eventually came to realize that producing reliable, reproducible screening results was far more important than raw throughput. The shift from emphasizing speed to emphasizing quality was important key to their success.
Dr. Houston feels that there is simply no way they could have achieved this level of success without their internal automation group, an experienced core group of people with the background to ask good questions. The success of that group has led to their becoming involved in other parts of the drug discovery process beyond in-vivo assays.
Question 2.4: In the Future My Company/Organization's Senior Management Will

Despite the response to the previous question (indicating that over half felt lab automation had delivered the expected results), the response to this question indicates that the majority feels their management will still evaluate lab automation opportunities on a case-by-case basis. One might think that there would be some correlation between the degree of success reported in the previous question and the prediction of future support in this question, but here appears to be little if any correlation. Many who reported mixed results predict broad support and many who reported success predict case-by-case evaluation. The person who reported “not delivered expected benefits” predicts a case-by-case evaluation by management. The person who predicts “no continued support” reported mixed results in the previous question.
Question 2.5: My Company/Organization Values Laboratory Technology and Automation As

A very strong majority reports lab automation and technology to be strategically important to their organization. Keep in mind that the respondents are ALA members, that is, practitioners of lab automation. We do not expect them to indicate that their work is irrelevant! In general, this response indicates once again that the industries represented by ALA members are technology driven. They have and will continue to use technology in their attempt to gain competitive advantage.
Automation is definitely seen as having strategic impact at Bristol-Myers Squibb, much bigger than just automating a specific task, says Dr. Houston. He is not sure that all organizations see automation and technology this way, and in fact feels that were the above question posed to many companies executive level decision makers, the “strategic impact” response might well be lower. A good track record of judicious use of money and resources to successfully implement technology projects has developed credibility for his organization that has led to a broadening of their mission to other parts of the drug discovery process.
Section 3: Technical Aspects of Laboratory Automation
Question 3.1: My Laboratory Prefers Automation Solutions Using

Note that this question focuses on “my laboratory,” not “my organization,” given that various laboratories within an organization have different missions, workloads, and budgets. A strong majority favors a mix of workstation and larger, integrated automated systems. Very few favor only large, integrated systems and a significant number favor only workstations.
Question 3.2: What is the Technology Lifecycle in Your Functional Area (Time from Which Technology Appears to When it is Superceded, Changed, or Irrelevant)?

Question 3.3: What is Your Preferred Entry Point to New Technology?

It's appropriate to discuss the response to Questions 3.2 and 3.3 together. A very strong majority favors a “leading edge” entry point to new technology—a point which is ahead of knowing whether a given technology is the “right solution.” The largest group of respondents indicated that they experience a 5-year lifecycle for technology, with 3 and 4 years also getting a significant response. Taken together, these responses show that risk taking with technology is common, but also that such technology is going to be used for 3–5 years. This highlights the importance of making good choices when taking a risk on leading edge technology. Or perhaps even more importantly, it highlights the impact of a bad choice of technology. Leading edge technologies, by definition, require significant internal energy and resources to implement and maintain initially. Deciding to abandon a poor choice of leading edge technology can also take quite a bit of time, as no one likes to admit having made a mistake. In the end, technology misadventures can actually set an organization further behind than they would have been had waited for the technology picture to reach the more defined “state of the art” stage.
Does the presence of internal technology resources (Question 1.2) affect an organizations technology entry point? By cross-referencing the responses to Questions 1.2 and 3.3, we get the results in Table 3.3a.
Correlation of internal automation resources to technology entry point
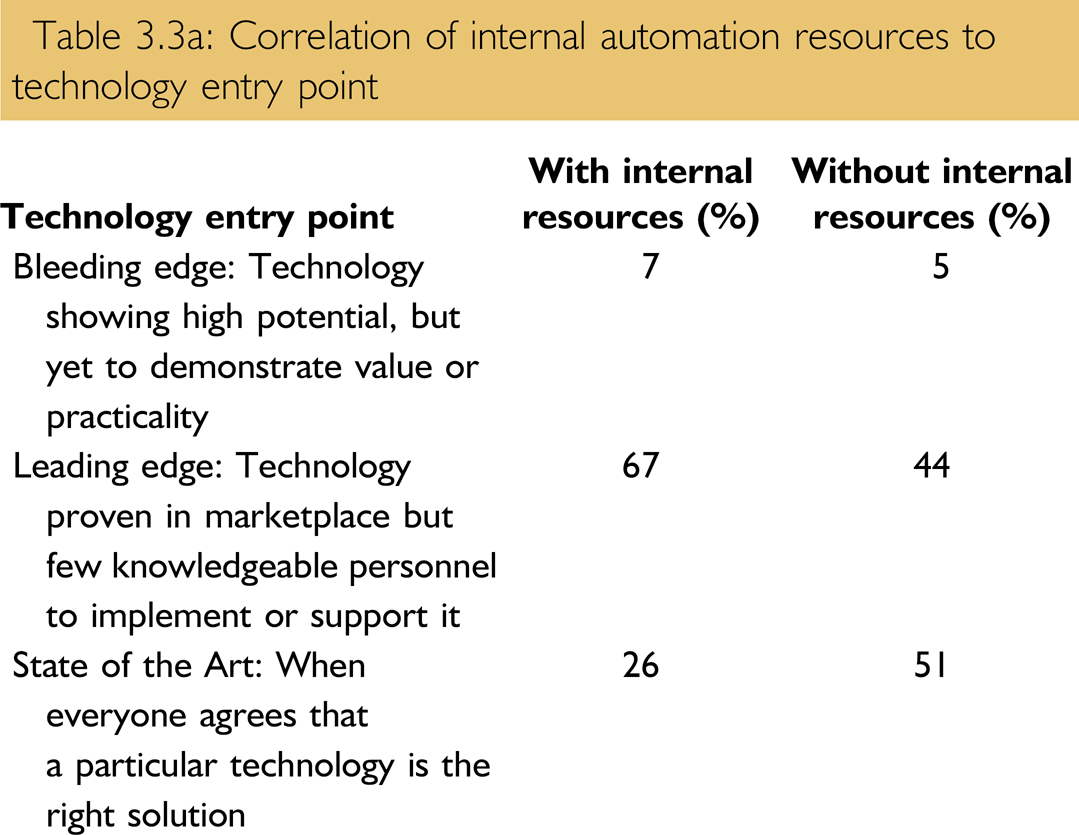
It would appear the answer is “yes.” Those organizations with internal automation resources are measurably more aggressive in their technology entry point.
According to Dr. Houston, his organization has had debates about the preferred entry point to technology. They have come to favor the leading edge entry point and have focused their efforts around “robust” technologies, which he feels has been key to their success. They see a technology lifecycle of around 3 years, at which time a technology needs to be changed or at least upgraded. He feels that the lifecycle period at BMS may be shortened by the presence of internal automation resources to evaluate or initiate upgrade projects.
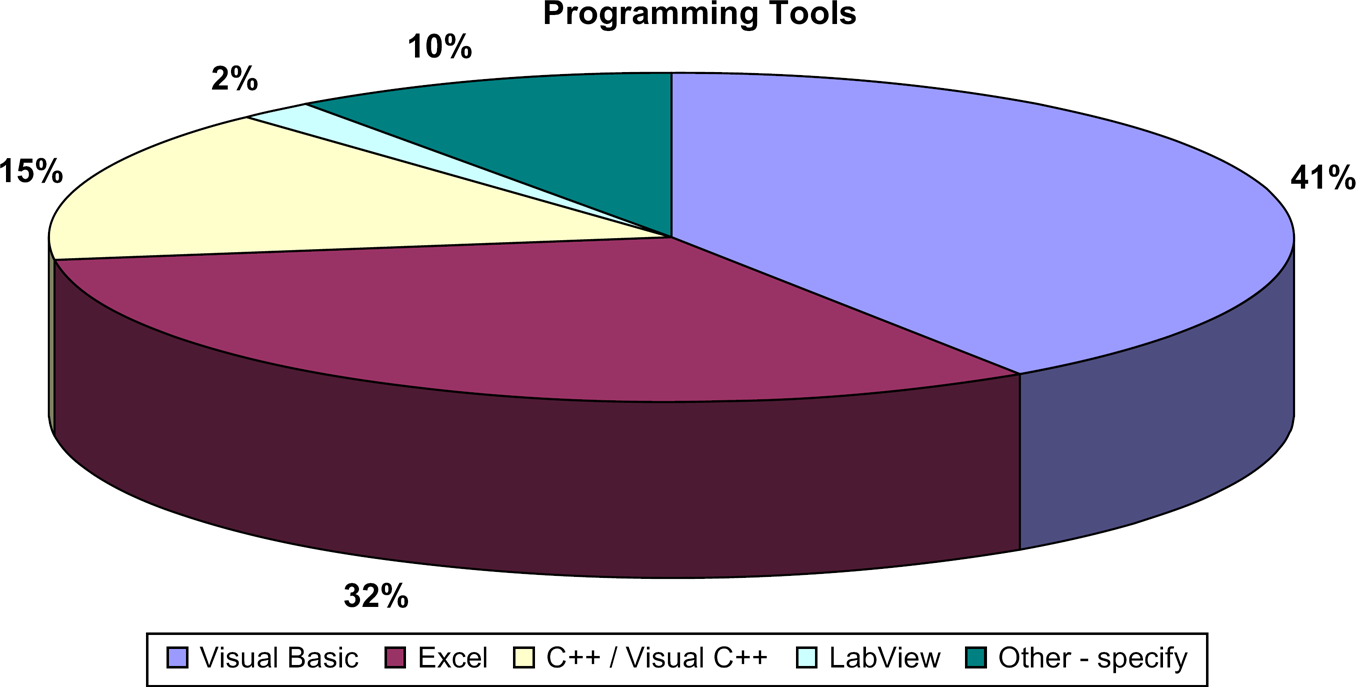
Question 3.4: Describe the Custom Programming Tools You Use in Your Laboratory by Selecting the Appropriate Proportion for Each Tool Below.
It is striking that VB and Excel are so strongly popular among the lab automation community, whereas in the broader world of programming VB ranks behind Java and C++, and Excel is not even on the radar screen. 1 Clearly, the laboratory automation community prefers (and presumably finds adequate) simpler tools and those that integrate well into the work done by scientists, that is, Excel.
“Other” results include:
Python; SAS; Java IDEs—Netbeans; Jbuilder; Matlab; C#; JSP; Excel; Java; .NET; C#; Java; System depending software; WinBatch; proprietary tools, and so on. The tools themselves are determined by many factors unique to each project. Updating or customizing an existing system—use the tools that are already in place for that system, Brand new system—use tools that allow extensibility in the future and allow for quick development now, … The environment in which these tools are developed is moving to MS .Net.; ActivityBase; Pipeline Pilot; Visual Studio. NET; SYMYX RAS; Hamilton Vector & HSL; Easylab; Numerous other packages for one or two systems; Database tools; Visual Studio. NET/IIS/C#/ASP.NET
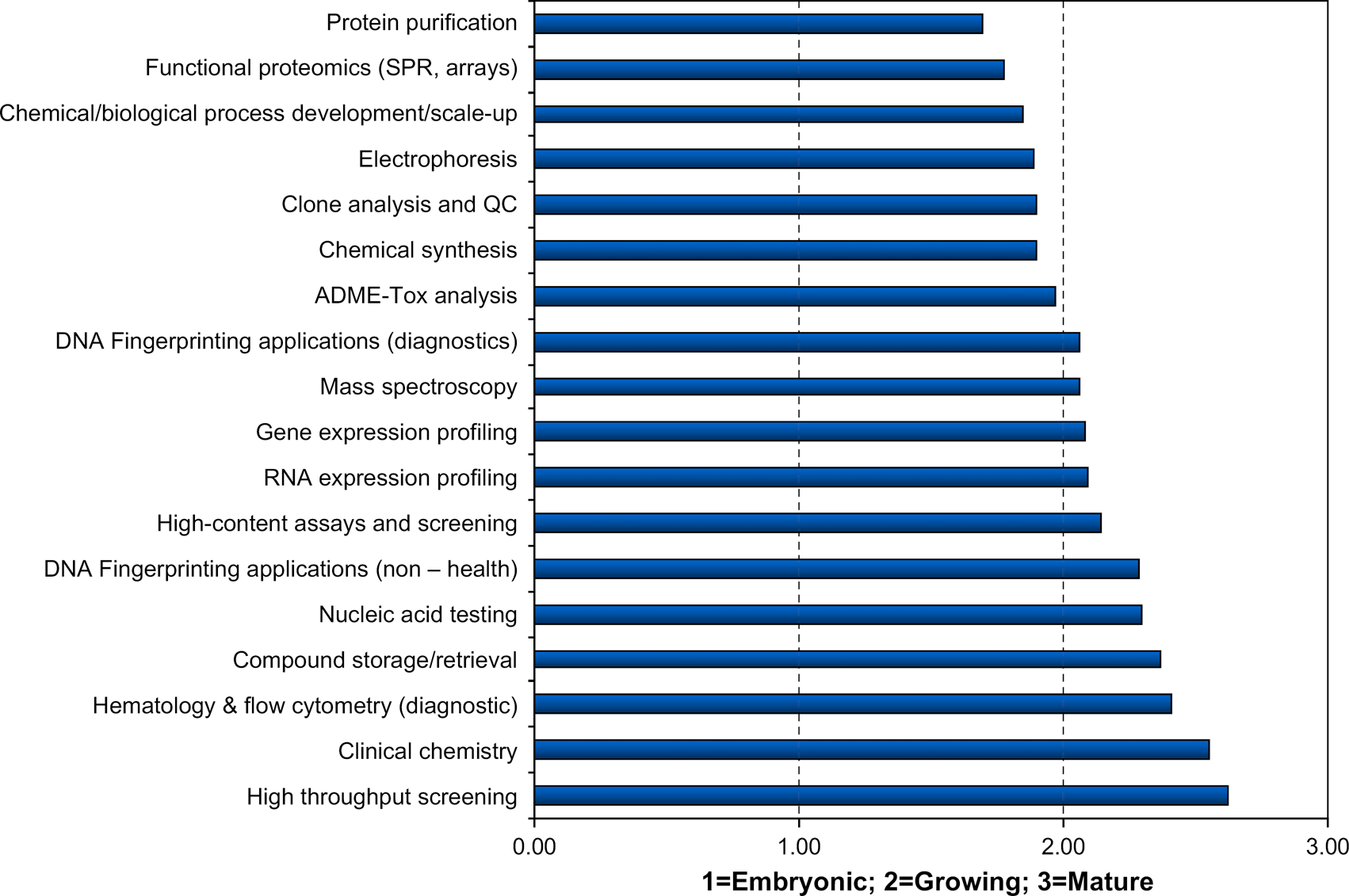
Question 3.5: Characterize the State of Automation & Technology Development in these Fields.
The field that was arguably the “hottest” of the last decade, HTS, is considered now to have the most “mature” state of automation and technology development. This does not necessarily mean that the science of HTS is mature. For instance, high-content assays and screening, a field related to HTS, is considered less mature. The automation in fields related to proteins is characterized as least mature. Chemical/biological process development, a field historically on the periphery of the traditional laboratory automation focus is also considered less mature.
Footnotes
Acknowledgments
The author would like to thank all the survey respondents for taking the time to complete the survey. Thanks also to the ALA management, specifically Executive Director Greg Dummer for supporting this effort and finally Todd Newell, the ALA's web guru at Potomac Digitec for managing the technical process behind the scene.
