Abstract
A totally integrated serial dilution assay plate preparation system that fully uses the high precision nanoliter dispensing capabilities of acoustic liquid handlers has been developed and implemented. The application uses a hybrid of a serial dilution method and a direct dilution method, achieving a wide concentration range for the dilution series, while avoiding additive errors inherent to traditional serial dilution methods. The method allows assay miniaturization, which greatly reduces reagent and consumable costs to the customers. The system is in production at AstraZeneca and has generated high-quality assay ready plates for high-throughput screening and secondary screening since 2005. Further development in recent years has expanded the flexibility of the assay ready plate creation process to meet varied screening requirements.
We will discuss the requirements for assay ready plates for concentration response testing and describe the novel plate creation method in detail with the rigorous validation procedures. Along with method validation data, some real-life screening results will be presented to compare an experiment conducted on compounds prepared using the novel hybrid method and those prepared using a more traditional serial dilution method, which endorses the application of the novel method.
Keywords
Introduction
In early drug discovery, the potency of compounds on the disease target is generally deduced by experimentally evaluating the effect of the compound on the target across multiple concentrations of the compound. This generates EC50 or IC50, which is the concentration producing 50% effect or inhibition. To achieve this result, it is necessary to dilute the compound in an organic solvent such as di-methyl sulfoxide (DMSO) at the highest required concentration, and then prepare a series of diluted solutions in the required range of concentrations. Depending on the concentration range desired, and the planned tests, the number of dilution points and the fold difference between points will be decided. Typically, any number of points from 5 to 12, and factors of 2-, 3-fold, 1/2 log, or full log are considered.
Traditionally, serial dilution of compounds has been carried out using either manual pipettes or automated liquid handlers with pipette tips and syringes. These methods would typically use a column (or row)-wise dilution on a microtiter plate. Typically, an aliquot of the compounds in DMSO at the highest concentration would be transferred from the first column to the second column filled with DMSO to prepare the second dilution point at the required second concentration. The solution would be mixed and then another aliquot would be transferred to the third column to prepare the third dilution point. This would be repeated until the full dilution series is achieved. Generally, a set of 8 compounds could be diluted in this manner at the same time using an 8-channel pipette or an automated liquid handler with 8 channels for a 96-well plate, or 16 compounds at the same time using a 16-channel pipette on a 384-well plate. When using this method of dilution, it is possible to accumulate errors that are associated with each transfer and dilution step, at each dilution point. Once the large-volume intermediate plate is prepared in the above manner, the assay ready plate will be prepared by transferring the required compound volume to an assay plate, producing the low volume serially diluted assay ready plate. The remainder of the solution in the intermediate plate at this point is considered as waste, because it will not be used for any testing purposes.
In contrast, direct dilution in making an assay ready plate is where each point of the concentration series on the final assay plate is prepared from the dilution of compound from the same compound stock solution and DMSO. This method would not result in such additive errors as experienced in a traditional serial dilution method, 1 and would not require a second transfer step from a large volume serially diluted intermediate plate to prepare a low volume assay ready plate. However, the issue would be that none of the liquid-handling instruments currently on the market are capable of achieving a wide range of concentrations typically called for a concentration response test at low volumes, using a direct dilution method. To achieve a serial dilution prepared from direct dilution, a liquid handler that has a wide range of dispensing capability with low resolution is best used. 2 The Labcyte (Sunnyvale, CA) Echo, is one of the most versatile liquid-handling instruments that is currently available in the market for low volume dispenses, and uses focused acoustic energy to transfer compounds in a noncontact dispense mode. 3 The Echo resolution is a very low 2.5 nL, whereas the achievable volume range is 2.5 nL to 10 μL. 4 There are a number of liquid-handling devices currently in the market that achieve similar high resolution and dispense volume range, and use different technologies. Typical requirements from the high-throughput screening (HTS) groups are for nanoliter volumes and for the application under discussion in this paper, an Echo555 unit was used as the primary liquid handler to prepare the final serially diluted assay ready plates.
As an example, consider a typical requirement of an assay ready plate for a concentration response test, which could be a seven-point serial dilution of 1/2 log dilution factor, starting at 10-mM concentration at the top dose with 250-nL volume across the plate. If an Echo liquid handler was used to prepare this serially diluted assay ready plate using a direct dilution method, it is only possible to achieve five out of the seven required points because the sixth point would require a volume smaller than the lowest volume the Echo can deliver (table 1). Other liquid handlers that are less versatile than the Echo can achieve even less range, unless an intermediate serial dilution plate of large volume is prepared, which is undesirable due to the unnecessary wastage of compound stock solutions.
Direct dilution from one compound source well to the assay ready plate serially diluted well

The work presented in this article describes a novel method developed that is a hybrid between a direct dilution and a traditional serial dilution, and prepares the serial dilution curves in just two steps, using only one intermediate plate, which is to be used as the source plate for the Echo555 to prepare the final assay ready plate. The original work on the development of this method was first presented by Cesarek and Nie in 2005 5 as an industry first, and this current paper describes the method in full application with extended validation data, while discussing the benefits to drug discovery. One recent publication describes the use of a similar methodology to create serially diluted plates using the acoustic droplet ejection (ADE) capability of the Echo instrument, achieving a large concentration span at low volume, by creating a 12-point 1/2 log serial dilution of 100 nL. 6 Four transfer and/or dilution steps and a number of source and intermediate plates are used in this method. It is suitable for liquid inventories that are in plates rather than in individual microtubes because if the source plate was to be prepared from an inventory of microtubes, that would require another additional step, however it will be also necessary to then use a compound storage plate and volume that is compatible with the Echo. The hybrid method described in this paper still manages to achieve a large concentration range in only two steps and avoid such additive errors as seen in the traditional methods. A Velocity11 (Santa Clara, CA) Vprep instrument is used for the first step to create a three-point serial dilution intermediate plate. A Labcyte Echo acoustic liquid dispenser is used for the direct dilution portion.
The use of a noncontact liquid dispenser such as the Echo also gives additional benefits over other automated liquid-handling systems that use pipette tips, due to decreased or no cost of wash reagents or disposable tips. Furthermore, due to the ability to transfer nanoliter amounts with high precision there is a large advantage in the fact that compound wastage can also be minimized. Nanoliter dispensing capability allows assay miniaturization resulting in cost reductions to the customers, as the samples can be prepared in less number of steps using less reagents and consumables. Because the final test plate is prepared directly at the Echo, it also removes further potential error that would be added at a transfer step from a less accurate instrument. The fully automated assay ready plate preparation system consisting of source and destination plate stackers, centrifuge, heat sealer, de-sealer, barcode label print and apply unit, and the Echo liquid handler, has been integrated by the AstraZeneca Compound Management Group in Wilmington. This fully automated system allows the Compound Management Group to operate in a highly efficient manner and achieve high throughput. Each serially diluted assay ready plate will take 4–9 min to prepare, depending on the dilution schema. Over 300 plates can be prepared on a daily basis.
Materials and Methods
To describe the method, an example of creating an 11-point 1/2 log serial dilution on a 384-well plate is used. It should be noted that the hybrid method can be and is currently used to create a wide variety of dilution schemes, ranging between 96- and 384-well plates of volume across the plate from 100 nL to 2 μL, with 1/2 log, three-fold, two-fold dilution factors, and 5-, 7-, 10-, and 11-point dilution series. The schema used for the example has serial dilution curves for 16 unique compounds on the plate, with each compound having duplicate series. Therefore, the number of serial dilution curves on one plate is 32. It is required to prepare the serial dilution of the compounds on the 384-well plate where columns 1 and 13 have the highest concentration at 10 mM and columns 11 and 23 have the lowest concentration at 0.0001 mM. The volume across the plate needs to be 1 μL. This plate will be referred to as the assay ready test plate.
The compound stock samples in DMSO at 10 mM are stored in Abgene (Epsom, UK) 2D barcoded vials. The samples are picked at the AstraZeneca automated compound management facility (ACMF) from a tube-based liquid sample store into a 96-well transporter rack. Column 1 (wells A01-H01) and column 7 (wells A07-H07) of the rack are populated with eight Abgene vials per column that hold the compounds of interest that are to be diluted.
The creation of the serially diluted test plate is done in two main steps. The first will create an intermediate plate with a three-point dilution series from stock sample obtained from the Abgene stock tubes. The second step will create the assay ready test plate using direct dilution from the solutions in the intermediate plate. Figure 1 describes the two-step method visually.
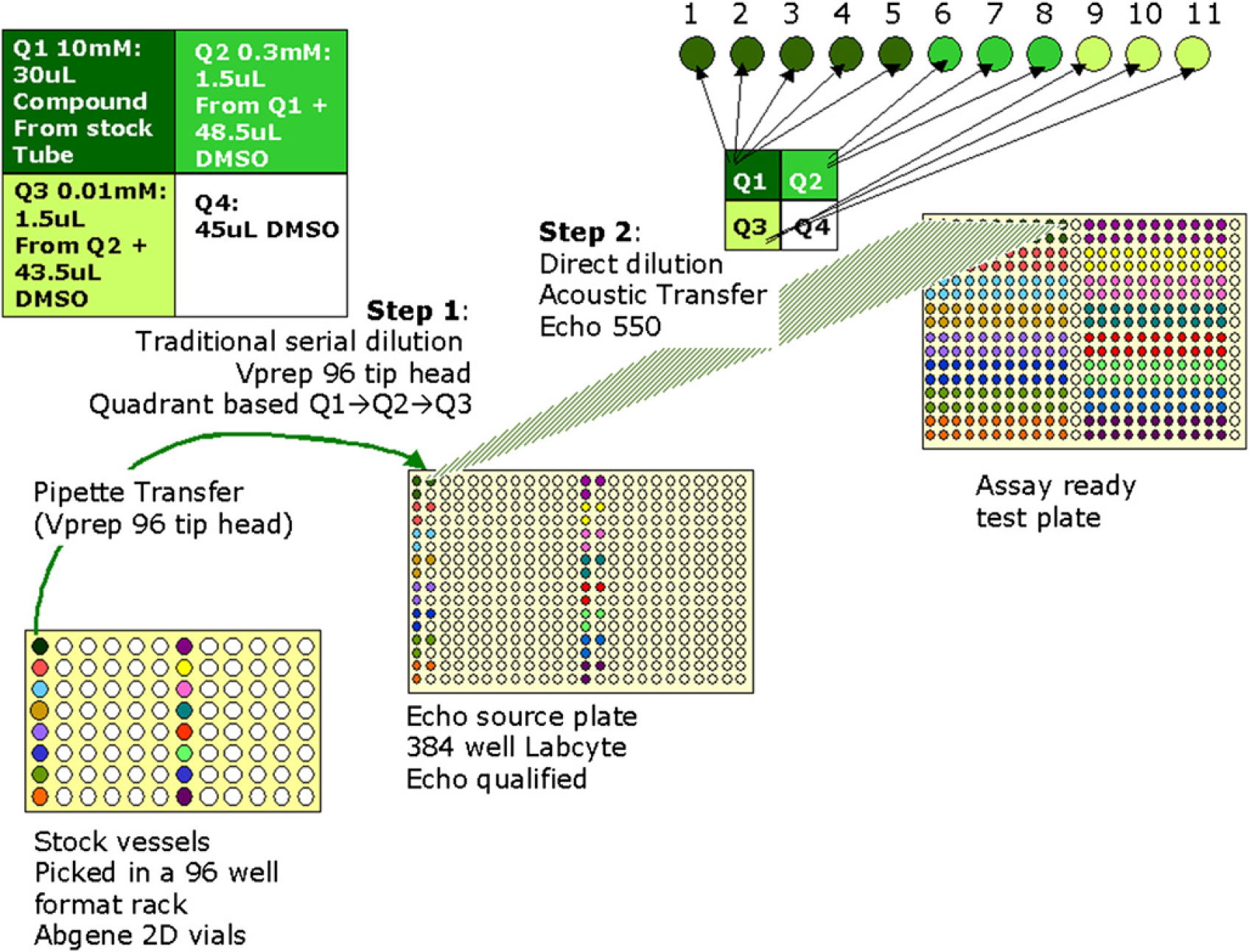
Visualization of the full two-step method to produce the 384-well assay ready plate with an 11-point serial dilution series in duplicate.
Step 1: Preparation of the Intermediate Echo Source Plate at the Velocity11 Vprep
In the first step, a Velocity11 Vprep automated liquid handler with a fixed tip 96-well pipette head is used to transfer the stock samples from the Abgene vials to a Labcyte P-05525 plate, creating a three-point serial dilution in a quadrant wise manner (Fig. 1 Step 1). This intermediate plate will be referred to as the Echo source plate. Initially, the Vprep first transfers 30 μL, 10-mM stock from the Abgene tube into quadrant 1 of the 384-well Echo source plate. This will result in 30 μL at 10 mM of the 16 compounds that were in the transporter rack columns 1 and 7 now available in the odd rows of the columns 1 and 13 of the Echo source plate (wells A01, C01, E01, G01, I01, K01, M01, O01, A13, C13, E13, G13, I13, K13, M13, and O13 in quadrant 1). The second step will perform a 33-fold dilution from quadrant 1 to quadrant 2, by adding 1.5 μL from the quadrant 1 to 48.5 μL of DMSO in the quadrant 2 well. This step also includes a mixing of five cycles. This will result in the wells A02, C02, E02, G02, I02, K02, M02, O02, A14, C14, E14, G14, I14, K14, M14, and O14 in quadrant 2 of the Echo source plate having 50 μL at 0.3 mM of the 16 different compounds. Then, the Vprep will perform a 30-fold dilution from quadrant 2 to quadrant 3, by adding 1.5 μL from quadrant 2 to 43.5-μL DMSO in quadrant 3. This will result in the wells B01, D01, F01, H01, J01, L01, N01, P01, B13, D13, F13, H13, J13, L13, N13, and P13 in quadrant 3 of the Echo source plate having 45 μL at 0.01 mM of the 16 compounds. Quadrant 4 will be filled with 45-μL pure DMSO. This will result in the wells B02, D02, F02, H02, J02, L02, N02, P02, B14, D14, F14, H14, J14, L14, N14, and P14 in quadrant 4 of the Echo source plate having 45 μL of DMSO. The addition of DMSO to quadrants 2, 3, and 4 is carried out by the Vprep itself from a DMSO reservoir of 96-well format.
Step 2: Preparation of the Assay Ready Test Plate at the Echo555
The second step to create the Assay ready test plate is carried out at a different automated system where the liquid handler involved is a Labcyte Echo555 unit. The intermediate plate with the three-point serial dilution prepared at the Vprep is used as the source plate to the Echo. An Echo protocol is run to prepare the assay ready test plate in a direct dilution method according to the volumes given in table 2. Figure 1 Step 2 visually describes the transfer from the source wells to the final assay ready plate wells.
Dilution volumes from the three source wells of the intermediate plate and di-methyl sulfoxide (DMSO) well, to prepare the serially diluted assay ready plate wells

The Echo protocol defines how much liquid is to be transferred from which source well to which destination well. In current practice at the AstraZeneca compound management group in Wilmington, the lowest amount transferred by the Echo is 10 nL, although the Echo is capable of transferring 2.5 nL. Figure 1 show that the serial dilution points 1–5 are prepared by a direct dilution of the compound at 10 mM in the source well #1, which is the highest concentration well of the three-point dilution series in the source plate. The DMSO used for the dilution is taken from the quadrant 4 wells of the source plate, which had 45-μL pure DMSO transferred at the Vprep. The serial dilution points 6–8 are prepared by the direct dilution of the compound at 0.3 mM in the source well #2 and DMSO. The serial dilution points 9–11 are similarly prepared from a direct dilution between the compound at 0.01 mM in source well #3 and DMSO.
This achieves the desired concentration and volumes on the test plate. For the example being discussed, the second duplicate series of the dilution curve is dispensed in the same manner from the same source wells to the row below.
Results
The Vprep and Echo dispense protocols are validated using Fluorescein solution and reading the Fluorescence intensity on a Tecan (Männedorf, Switzerland) SpectraFluor reader. Fluorescein is typically used at AstraZeneca and industry wide for instrument quality control and method validation experiments. 7 The rigorous validation procedure established requires three initial validation tests.
Method Validation Test 1
First, it was necessary to validate the Echo protocol, to show that the wells prepared by the protocol produced the expected dilution effect. All three wells of the source plate were filled with Fluorescein in 100% DMSO at 24 μM, rather than creating a three-point dilution as described in the previous section, due to the limitation of the instrument detection range. The Echo protocol was run to prepare the serially diluted output plate according to the schema to produce an 11-point 1/2 log serial dilution with 1000 nL across the plate on a 384-well plate; 79 μL of 1 x phosphate buffered saline (PBS) buffer was added to each well using a Thermo Scientific-Matrix (Hudson, NH) WellMate bulk dispenser and centrifuged for 1 min at 1000 RPM before reading on the SpectraFluor reader.
The fluorescence intensity from each test plate well produced by the three source wells was plotted as separate series against the concentration at each well (Fig. 2). The linear response on the log scale indicates that the dilution carried out by the Echo is in agreement with the expected dilution factor.
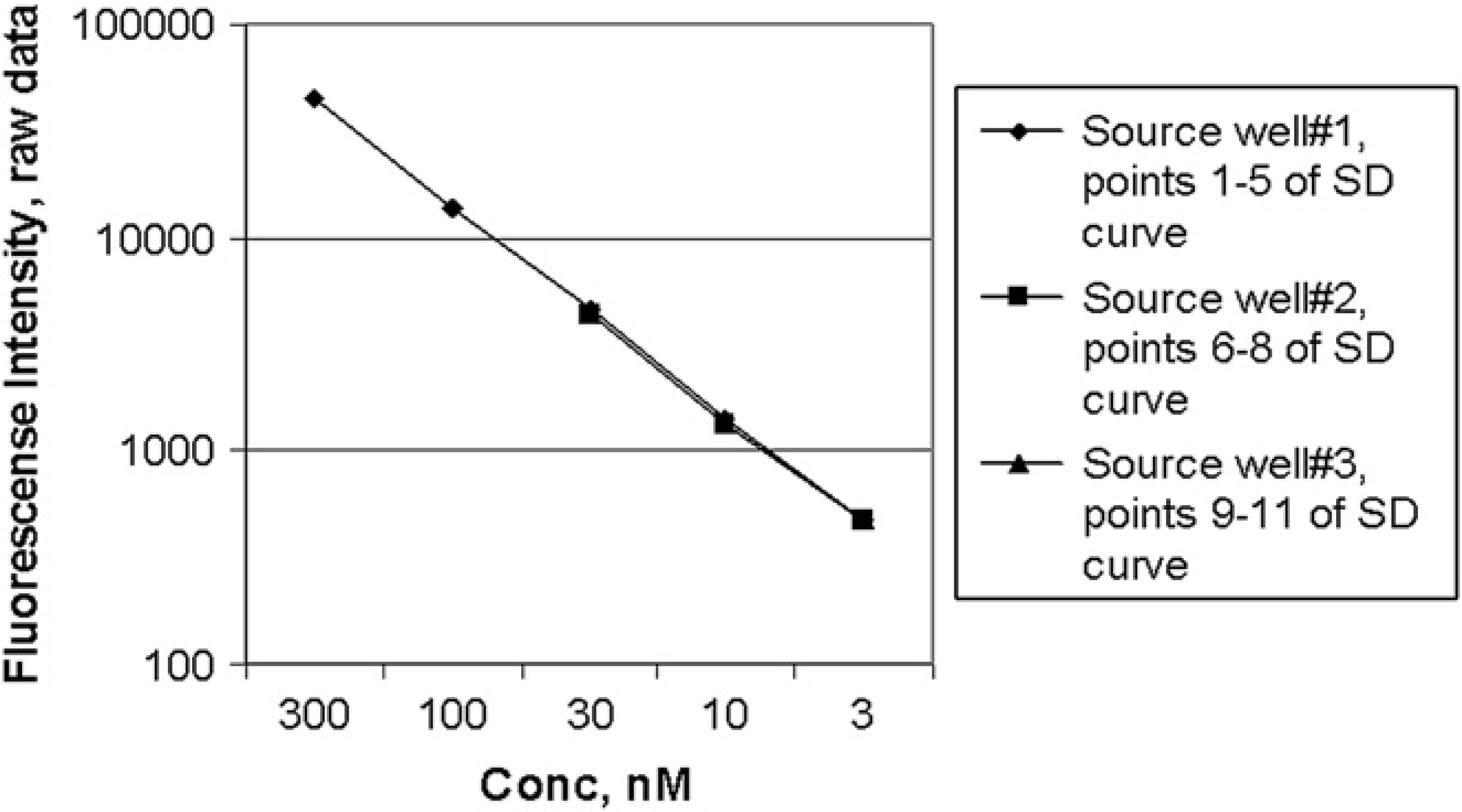
Eleven-point 1/2 log serial dilution, 1000-nL/well Echo method validation with the same Fluorescein concentration in all three source wells. Each point is the average of 32 wells, of a 384-well plate with 32 dilution series.
Method Validation Test 2
To assess the repeatability of the Echo protocol, three serially diluted output plates were prepared from the same source plate, and their fluorescence response was compared (Fig. 3). The source plate was prepared to have the three-point dilution as discussed in the Materials and Methods section. The response obtained from the three plate copies show excellent repeatability. Only the first seven points of the 11-point curve are displayed, as the fluorescence intensity obtained from the last four points do not fall within the detector linear range. The linearity of the dilution series is validated in the previous test, as shown in Figure 2.
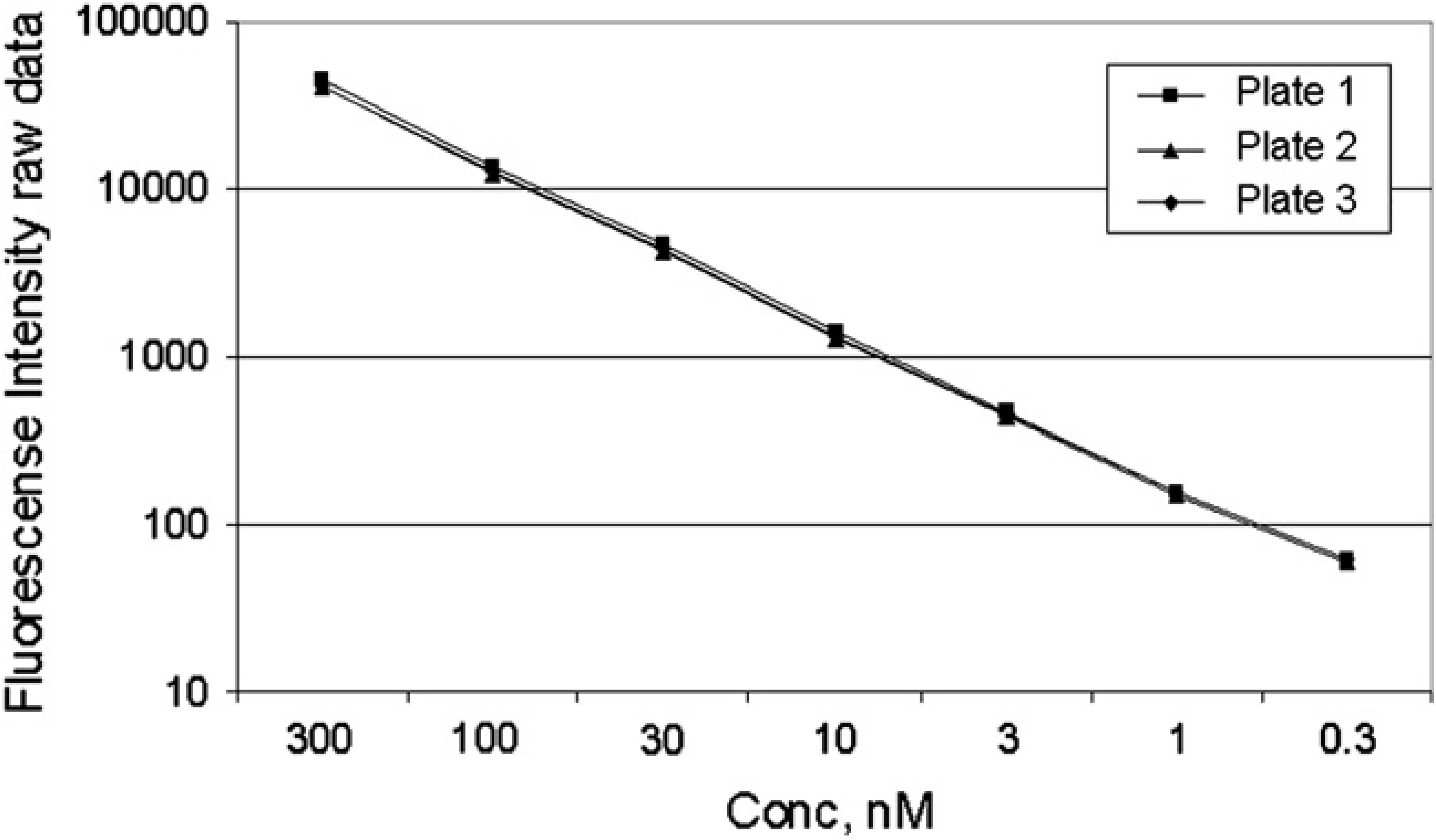
Eleven-point 1/2 log serial dilution, 1000-nL/well Echo method validation with Fluorescein. Fluorescence intensity plotted for three separate plates prepared from the same source plate by running the same Echo protocol. Only first seven points are displayed due to last four points being below the detector linear range. Each point is the average of 32 wells, of the three 384-well plates with 32 dilution series each.
Method Validation Test 3
The two previous tests demonstrated that the Echo protocol produces the 11 points precisely and is repeatable. The next test was to validate the accuracy of the full procedure, including the three-point source plate preparation at the Vprep. The validation test compared the Fluorescence response of assay ready test plates prepared from three different source plates that had been created using different methods. All test plates were prepared to give the same serial dilution schema of an 11-point, 1/2 log dilution factor, with the top concentration at 300 nM of Fluorescein solution.
Two assay ready plates were prepared by running the Echo protocol for the 11-point, 1/2 log dilution for 1 μL across the plate volume. One of these was dispensed from a three-point source plate that was prepared on the Vprep using the Vprep protocol. The other was dispensed from a three-point source plate prepared manually; 79-mL PBS buffer was added to all wells of these plates to bring the top concentration to 300 nM. A third plate was prepared manually using a traditional serial dilution method to achieve an 11-point, 1/2 log dilution schema with 300 nM as the top concentration. All three plates were read on the Tecan Spectra-Fluor and the intensity response data was plotted against each other for comparison (Fig. 4). Once again only the first seven points are displayed, as the lower four points of the dilution series do not fall within the reader linear detection range. The results show that the three assay plates prepared from the three different source plates give close readings, showing comparable behavior.
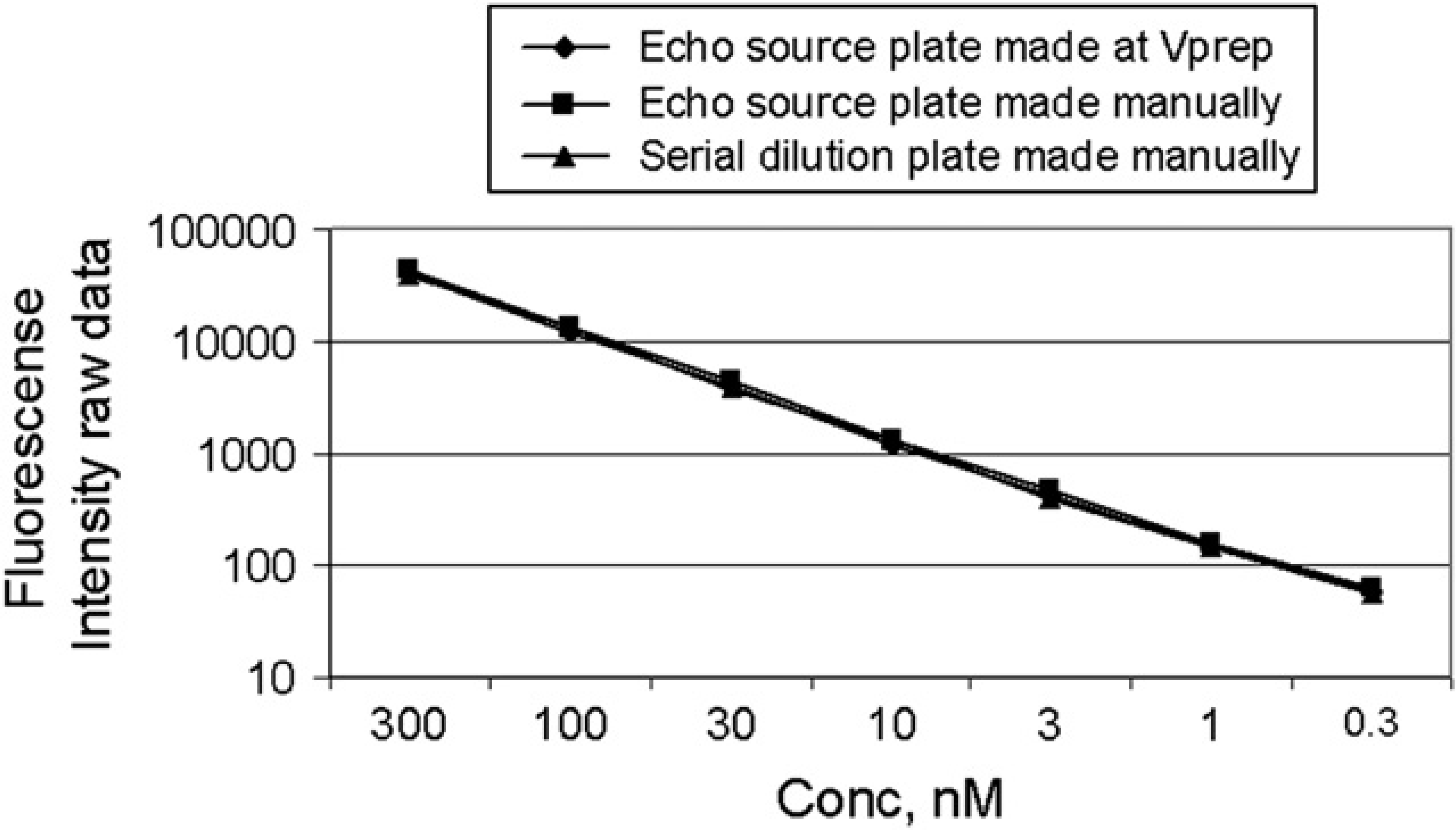
Eleven-point 1/2 log serial dilution, 1000-nL/well Echo550 method validation with Fluorescein. Fluorescein intensity plotted for plates prepared from three different source plates that are prepared using different methods. Only first seven points are displayed due to last four points being below the detector linear range. Each point for the manually prepared serial dilution plate is the average of 32 wells of a 384-well plate with 32 dilution series. Each point for the other two data series is the average of 96 wells, from three 384-well plates each with 32 dilution series each.
The three tests conducted as above give us the confidence to use the developed Echo protocol in a production setting. Each new schema that is developed is put through these rigorous validation steps, to ensure that it is ready for production use. Once thus validated, the new schema is available for customers for requesting compounds.
Method Validation Test 4 (Biology Validation)
Customers are also offered the option to conduct a biology validation of a newly created dilution schema if desired. Although some customers opt to pass on this extra validation test and use the new schema in a production setting immediately, some customers prefer to have the additional biology validation. This usually consists of conducting a concentration response test on the assay of interest from compounds prepared using the new Echo protocol. The same compounds are also prepared by the customers using their standard lab methods, and run on the assay along with the compounds prepared from the Echo. The biological results are compared. Often, customers use reference compounds that are known to be active in the assay of interest, and have published data in literature. Figure 5 shows an example of such a biology validation conducted using an 11-point, 1/2 log serial dilution protocol, where the results determined from a secondary screening assay were compared with published Ki values for the histamine reference compounds used for the test. The Ki values determined from the in-house prepared assay ready plates are similar to the published data found in literature, thus giving confidence to use the developed schema in a production mode.
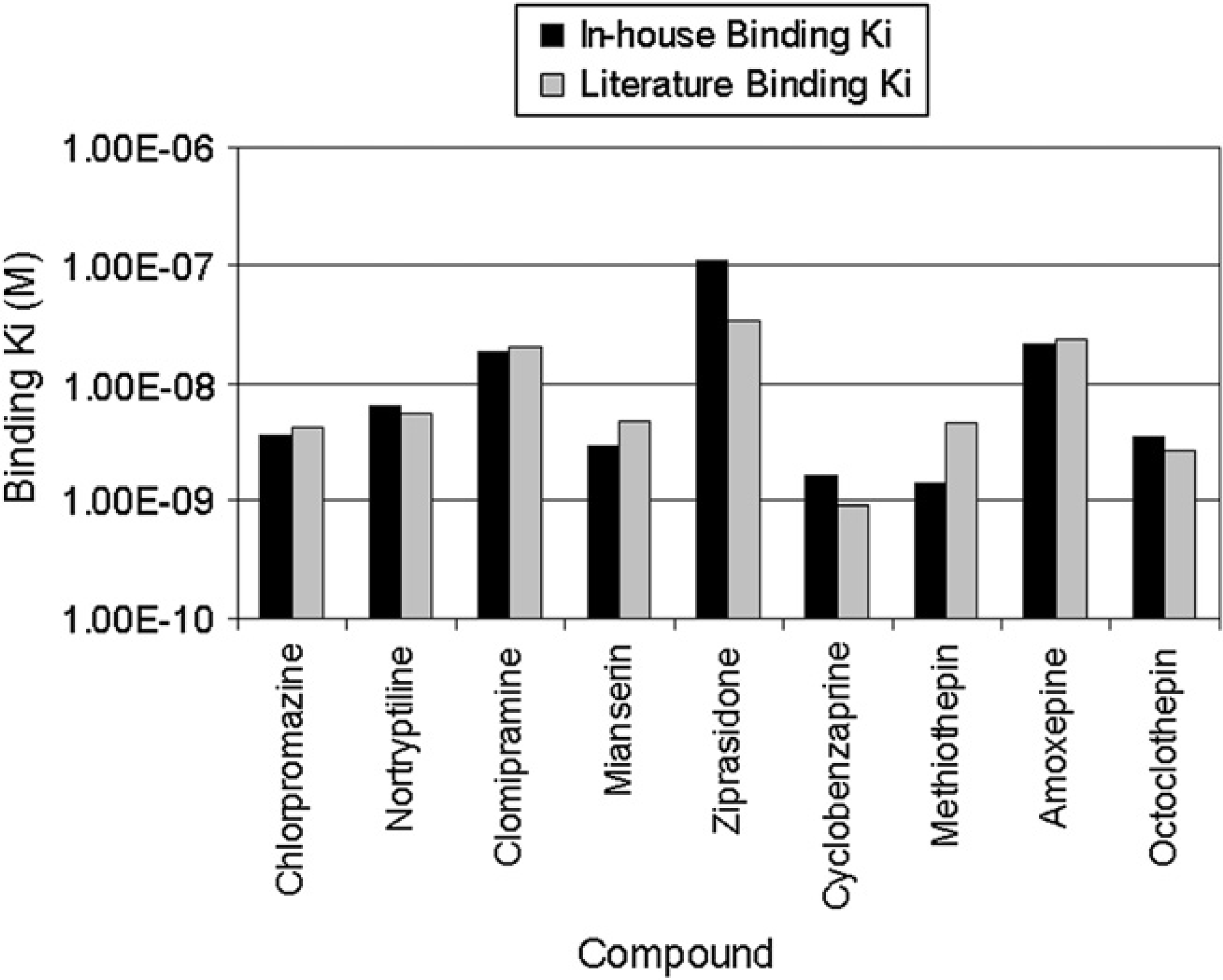
Binding Ki determined for known Histamine reference compounds prepared using novel method compared with literature data.
Experimental Example for Discussion
A further experiment was carried out to compare the biological activity of 31 compounds that were active in an HTS campaign. The results presented show the comparison of IC50 values obtained from an experiment where the serially diluted compounds were prepared using the hybrid method on an Echo as explained above, and a traditional serial dilution method on a Thermo Scientific-Matrix (Hudson, NH) PlateMate Plus liquid handler. The source compound solution for both plates was acquired from the same original stock tube. Two copies of the output test plate were prepared for each method and run on an HTS concentration response assay. The dilution series used is a seven-point dilution with the highest concentration at 10 mM. The assay ready plate was prepared with 200-nL volume across the plate.
The procedure to prepare the assay ready plate at the Plate-Mate Plus was achieved in four steps: 1) prepare an intermediate plate from stock solution tubes at the Vprep to give 36 μL of compound at the highest concentration in the first column; 2) add 24 μL of pure DMSO to second to seventh columns using a WellMate bulk dispenser; 3) run a serial dilution protocol at the PlateMate Plus to transfer 12 μL from the first column to the second column, mix five times, transfer 12 μL from second column to the third column, mix, and so on. 4) Once the intermediate plate was serially diluted, a low volume assay ready plate was made at the Echo555 by transferring 200 nL, as opposed to transferring at the PlateMate Plus unit for which the lowest transferable volume is 500 nL.
The assay ready plates thus prepared using the two methods were run on an ion channel target using ion flux assay to determine the IC50 in duplicate. Samples were read on an atomic absorption spectrometer (AAS) from Aurora Biomed (Vancouver, BC). Compounds diluted in RbCl buffer were added to the cells expressing the ion channel and incubated for 2 h at 37 °C. After incubation, cells were washed to remove extracellular Rb. Cells were then lysed with Triton-X100 and read on AAS.
The IC50 values were calculated by nonlinear curve fitting through seven points using the following equation:
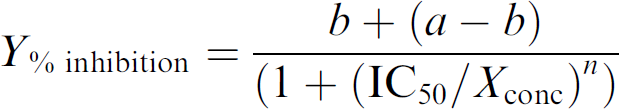
where a = top, b = bottom, n = slope.
Figure 6 shows the average IC50 values for the 31 compounds obtained from the duplicates for the two different methods and their associated errors. It is observed that the variability is much higher from the PlateMate produced test plates, when compared with those from the Echo produced plates. The IC50 values of the Echo produced plates were much tighter, and also generally lower than the PlateMate produced plates.
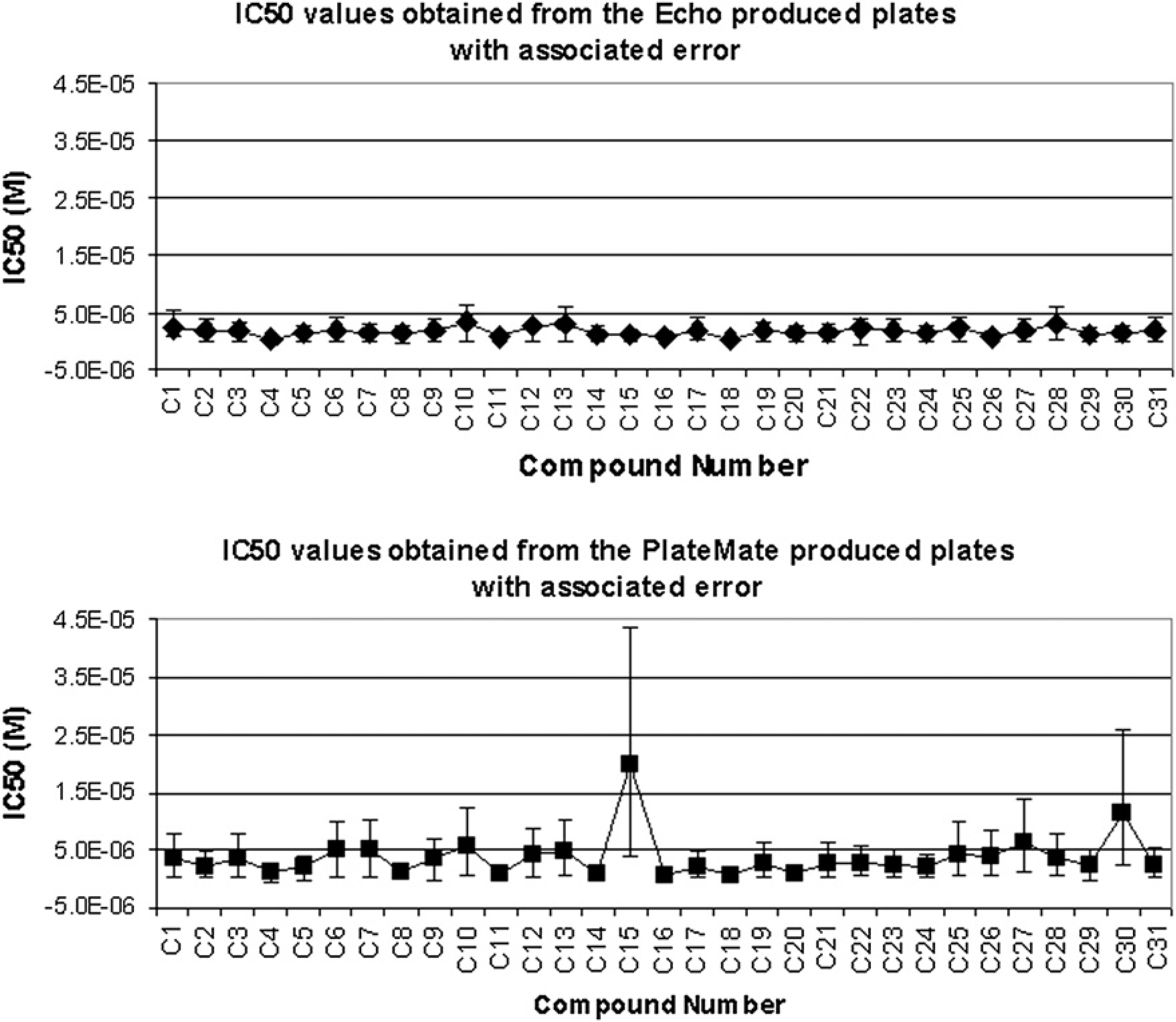
IC50 values obtained from the serially diluted assay ready plates produced by the Echo and the PlateMate Plus, with the associated error.
Discussion
The previous experimental example helps to demonstrate a number of issues that are inherent to the traditional serial dilution methods, which the novel method can help overcome.
Process
The assay ready plates prepared from the traditional method used four process steps, which could easily have been five steps if a liquid handler with a minimum transfer volume more than 200 nL was used for the final replication step. With the novel hybrid method, the assay ready plate was achieved in two steps. This process difference exemplifies an advantage of the novel method over the traditional method, because each extra step adds time, resources, additional reagents, and consumables to the sample preparation. The nanoliter dispensing allows downstream assay miniaturization, thereby greatly reducing experimental costs because less reagents would be used. When using liquid handlers whose lowest transferable volume is in the microliter range, typically it is required to carry out an extra dilution in aqueous buffer before preparing the final assay ready plate. Dilution in aqueous buffer has potential compound precipitation due to solubility issues, which can cause inaccuracies in the concentrations of the dilution series.
Uncertainty
Another concern of the traditional serial dilution methods is the additive dispense errors and potential carryover effects introduced at each dilution point. Because each dilution point is prepared from the previous point, the dispense errors will be added at each dilution point. For an instrument with a 5% associated error, theoretically it is possible that the seventh point concentration of a three-fold seven-point concentration as used in the experiment discussed previously has as much as a 21 % additive error. Unless new pipette tips are used at each dilution point for disposable tip instruments, or a thorough tip wash is carried out for fixed tip instruments, the carryover effect can also add a pronounced error. Also, typically a traditional serial dilution method would require liquid transfer steps from a number of liquid-handling instruments, such as bulk dispensers to add the DMSO for the dilution series and/or low-volume plate-based transfers to make the final assay ready plate. These additional transfer and dilution steps also add to the final error introduced to the dilution series. Due to these reasons, the IC50 values calculated would not be as accurate for the traditional serial dilution series when compared with the series prepared with the novel method. The novel method that uses direct dilution and just one serial dilution step aids to remove some of the uncertainty that can be attributed to the traditional methods. The novel method also provides more consistent data than the traditional method, which showed more variability within the repeated data sets.
To demonstrate the errors associated with the traditional and the hybrid methods, consider an example for a seven-point three-fold dilution series, that prepares a 500 nL serially diluted compound in DMSO plate, which is then diluted 100-fold with buffer for the assay. If such a plate is prepared using the traditional method that uses a PlateMate unit with 8% dispense error, and the hybrid method that uses a Vprep unit with 8% dispense error and an Echo unit with 4% dispense error, the error in concentration at each assay point can be calculated mathematically for both methods. The calculations follow the same process steps as have been discussed previously for both methods in preparing the compounds in DMSO plates, and that 49.5 μL of buffer is added to the wells to bring the compounds to the assay concentration. The example is illustrated in Figure 7. The calculations assumed no additional errors are incurred from the bulk dispensing of the DMSO or the buffer, or from carryover introduced by pipette tips in the traditional method.
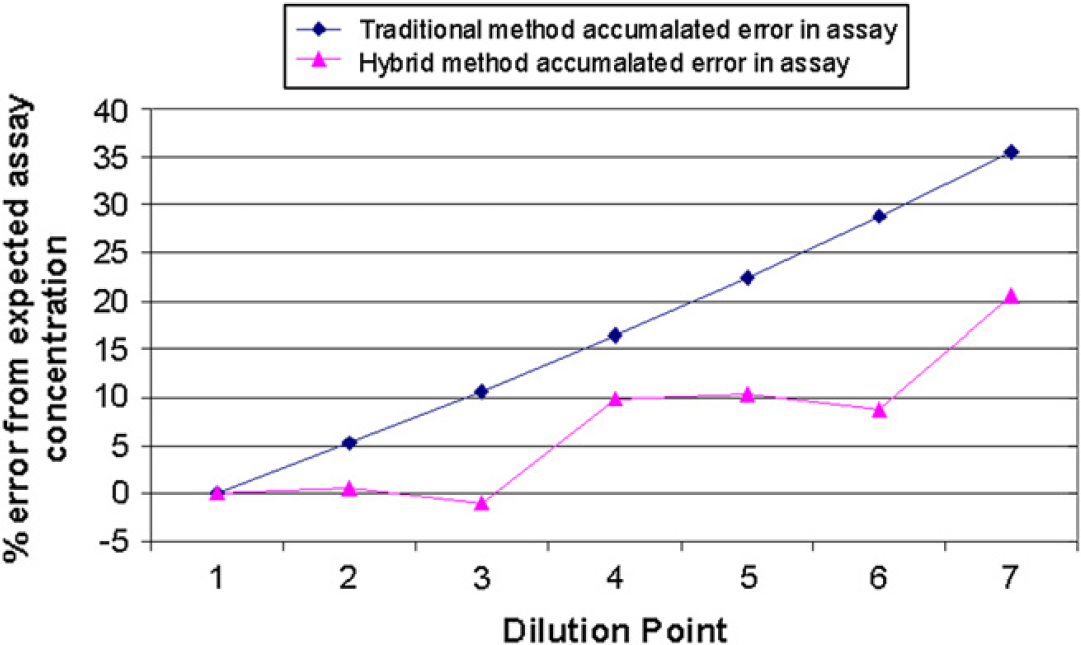
Mathematically calculated accumulated error of concentration in the assay well for a seven-point three-fold serial dilution prepared by the traditional and hybrid methods.
The error in concentration at each point for the traditional method can be up to +35% at the seventh point, whereas for the hybrid method it can be up to +20%. This error is calculated for the instruments overdispensing. Figure 7 illustrates how the error at each point is accumulated for the traditional method, whereas for the hybrid method the error is steady for the dilution points that are prepared from the same source well, and then the error jumps between two different source wells. For example, dilution points 1, 2, and 3 are prepared from the first source well of the Echo source plate and has an average error of 0.16%, and the next three points prepared from the second well of the Echo source plate have an average error of 9.6%. In this example, the ratio of error between the traditional method and the hybrid method can be up to 10-fold for some points.
Compound Usage
Traditional serial dilution methods also typically consume large amount of stock solution and DMSO, due to the dead volume requirements associated with the liquid handlers and tips used in such methods. Because these instruments have a relatively high minimum transferable volume, it is necessary to prepare large-volume serial dilution intermediate plates. In the example discussed previously, 36 μL of the compound stock was consumed to prepare the serially diluted assay ready plates. For each compound, a further 144 μL of DMSO was used for the consecutive dilution points. With the novel hybrid method, 24 μL of compound was used, and a total of 140-μL DMSO was used per compound for the dilutions. Of the 24-μL compound used, only 4 μL is used for the preparation of the assay ready plates, and 20 μL is required due to the dead volume requirements of the Echo liquid handler for the particular source plate type used (Labcyte P-05525).
Cost Benefit
In addition to the savings made on compound usage by the novel method, it also contributes to overall cost savings in other ways. For example, the traditional methods use either disposable tips or a thorough tip wash in DMSO in between the sets of compounds that are being diluted and transferred. However, no such tip wash or disposable tips are needed for the Echo transfers. Because no additional intermediate plates are required, there is also cost savings on limited use of labware. Olechno et al. 6 demonstrate that the cost benefits achieved per assay well can be significant when using ADE for preparing assay ready compound plates. According to their data for a serial dilution application, when compared to traditional dilution methods, the cost incurred per sample with ADE can be between 3 and 10 times less. Labcyte claims that the elimination of intermediate plastic-ware and the savings on DMSO by eliminating tip washes can amount to savings over $1.3 million in 5 years, for a compound library of 2 million and 11 HTS screens per year. 8 Thus, it could be argued that the novel method that used acoustic dispensing helps reduce costs and improve environmental considerations because less wastage is produced in comparison to the traditional methods.
Summary
Acoustic dispensing provides the possibility of an alternative to traditional methods of liquid dispensing. Acoustic instruments such as the Labcyte Echo555 offer high resolution, ultra low-volume liquid dispensing with much improved accuracy and precision over traditional liquid-handling systems. They also offer the opportunity to achieve reasonable direct dilution series that aid to overcome additive errors experienced in traditional serial dilution methods. However, to achieve a dilution series that spans a wide concentration range, it is still necessary to complement the process with a traditional serial dilution step. A hybrid method using direct dilution and traditional serial dilution has been developed and validated for production use. Rigorous method validation carried out using Fluorescein solution and biological experiments has confirmed the acceptance of the novel method. The novel method brings a number of cost and quality improvements over the traditional serial dilution methods, such as higher accuracy; better precision; and less compound, reagent, and consumable use.
Acknowledgments
The authors wish to express their gratitude to Ron Julien and Shephali Trivedi of the CNS/Pain Discovery ADHTS section for generating the biological data for the IC50 comparative experiment, describing the HTS assay protocol and general review of the article. We are also grateful to Qiaoling Jiang of the CNS/Pain Neuroscience Department for her contribution to the Ki determination data.
Competing Interests Statement: The authors certify that they have no relevant financial interests in this manuscript.
