Abstract
By studying a series of soybean oil-based polymers, using low-resolution nuclear magnetic resonance (NMR) spectroscopy, an easy method to estimate molecular weight (MW) was developed. The relationship between a polymer's MW and the instrument's response can be correlated in a linear relationship. Correlation constants (R 2 ) of 0.988, when data were taken at −10°C, and 0.999, when the study was done at 0 °C, were observed. Overall, this method is simple and with instrument modification, could be applied to other types of polymers as well.
Introduction
As part of the growing green chemistry 1 3 movement, the use of polymers made from natural materials in products is an important goal 4 6 as we move away from using our remaining petroleum resources. The use of polymers based on chemically modified soybean oil has attracted a lot of interest in recent years where products, such as inks, 7 9 composites, 10 14 surfactants, and lubricants 15 –19 have generated considerable results. In its simplest form, thermal vegetable oil polymerization, also termed heat bodying, can be accomplished by heating the oil under a nitrogen atmosphere. In oil with a significant amount of double bonds, such as soybean oil, the Diels—Alder (Scheme 1) reaction occurs and gives polymeric oil with different properties than the starting material.
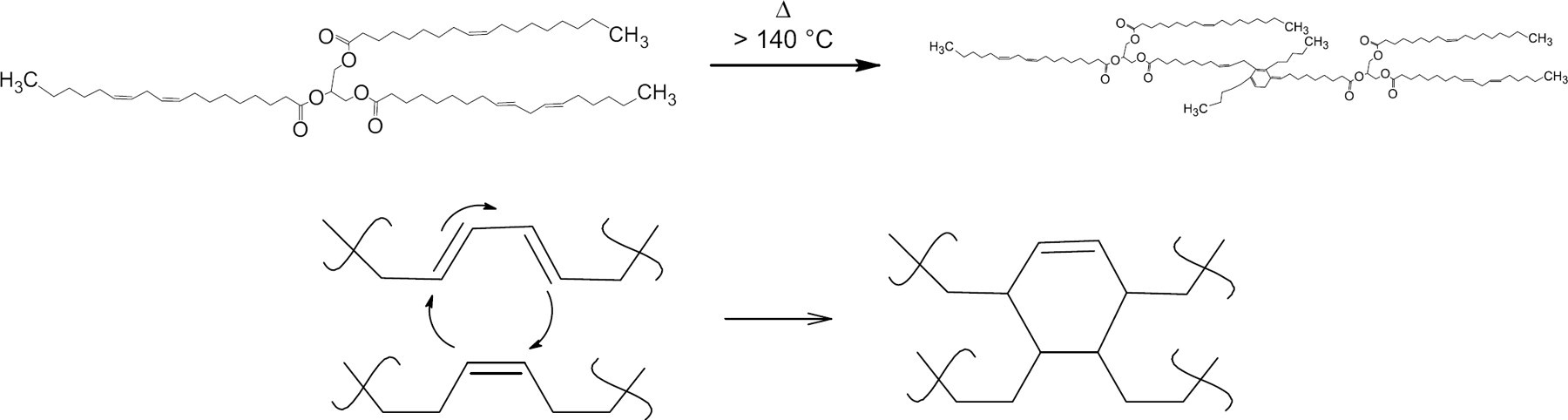
The Diels—Alder polymerization reaction, which occurs during heat modification of soybean oil (top). The reaction in more detail (bottom). The MW of the oil increases from ∼ 1 kDa up to greater than 16 kDa.
One of the keys to getting a polymer with desired performance properties is in the control of its MW. 20 The current state-of-the-art technology for this determination is gel permeation chromatography, which suffers from two drawbacks. First, it is not direct. The interaction which is actually measured is that of the sample with a column gel, which is related to MW. Second, each sample run can take up to half an hour and the instrument expense can be considerable; both the instrument and column costs, as well as the solvent that is consumed with every run.
A potential direct method for MW determination is nuclear magnetic resonance (NMR) spectroscopy. The literature is full of recent methods 21 where high-resolution NMR techniques have been developed for determination of the MW of glassy polymers, poly-saccharides, 22 fluoropolyethers, 23 hydrogels, 24 25 and other complex hydrocarbon systems. 26 28
Although these methods are of considerable interest, they require an even more expensive instrument than gel permeation chromatography. Their superconducting magnets require cryogenic cooling, and considerable upkeep. However, low-resolution NMR spectroscopy is an alternative without the high-cost or high-maintenance requirements. Low-resolution NMR spectrometers (Figure 1) have been available for many years, and come in a variety of field strengths. There is even an automated system available. 29 Low-resolution NMR has been used specifically to look at the solid fat content of materials since 1960s. 30 33 Pulsed NMR brought the method into the mainstream, 34 35 eventually leading to the acceptance of the American Oil Chemist's Official Method Cd 16–81, in 1983. Now, these instruments are common and are used not only for solid fat content, but also for emulsion analysis of complex mixtures, including ice cream. 36

The low-resolution NMR instrument used in this study.
The instruments work by subjecting the sample to a radio-frequency pulse in the presence of a magnetic field. Because hydrogens in the solid state will return to equilibrium much faster than those in the liquid state, the instrument can give a direct measurement of the amount of hydrogens in each state. A working hypothesis is that soybean oil-based polymeric material, due to its increased MW, will display the same phenomenon that will be detectable by a low-resolution NMR instrument. Furthermore, because this response will be greater in polymers of greater MW, a convenient method for determination of MW is possible.
Experimental
Instrumentation
Low-resolution NMR spectroscopy was performed on a Bruker Minispec MQ20 (Bruker, Ontario, CA) spectrometer (Figure 1). It was set up to run at its normal field strength of 0.47 Tesla corresponding to a proton Larmor frequency of 20 MHz. Glass NMR tubes (180 mm x 10 mm, 0.6 mm thickness) were used. Data were acquired using the Minispec Software version 1.81 running on a Dell Optiplex GX110 with a Microsoft Windows 98 operating system. A Lauda E 100 Ecoline (Lauda, Germany) circulating heater was used to heat samples above room temperature (Figure 2) and a Neslab RTE 17 refrigerated bath (Thermo Electron Corporation, Waltham, MA) circulating 2-propanol (Certified ACS; Fisher Scientific, Fairlawn NJ) was used for temperatures below room temperature.

The sample tubes used in this study in a temperature control unit (The pictured unit is used for temperatures above room temperature. The bath used for temperatures below room temperature contained a refrigeration unit.)
Materials
Soybean oil (RBD grade; KIC Chemical, New Paltz, NY) was used as received. Polymerized soybean oil was made and characterized by literature methods. 9,37 The polymerization involves heating the soybean oil under a nitrogen atmosphere to 330 °C. The extent of reaction was determined by measuring the viscosity. The MW of the samples was determined by gel permeation chromatography as described elsewhere. 16
Testing
The samples were run using a modified version of AOCS Official Method Cd 16b-93. It is a direct method, which compares the intensity of the free decay signals. Because the signal for the hydrogens in the solid state decays much faster than that of hydrogens in a liquid, this information can tell the percentage of the hydrogen spin density that is in each physical state. We modified the method to incorporate the coldest temperatures necessary in our experiment, but otherwise used the method as written. Typically, each sample was placed in a glass vial. The vial is heated with a heat gun to warm the sample. The vial was then put on a vortex-type blender and mixed for several seconds to be sure the samples were well blended. Next, 1.7–2.0 g of blended sample was poured into a glass NMR tube and capped. The sample was heated to 100 °C for 10 min, cooled to 70 °C for 30 min, cooled to 26.7 °C for 15 min, cooled to −11 °C for 15 min, heated to 26.7 °C for 30 min, and finally cooled to −11 °C for 45 min. NMR was then performed at each of the experimental temperatures in order of coldest to hottest. Although the temperature pretreatment would probably not be necessary for our samples, it was performed for consistency.
Results and Discussion
Temperature
Soybean oil, and a selection of soybean oil-based polymers, all with known MW, were studied by low-resolution NMR spectroscopy at a variety of temperatures. The data (Figure 3) show two important factors. First, the larger polymer had a much higher response, supporting our hypothesis. Second, the overall response, as well as the difference between the samples, was much greater at lower temperatures. This shows that the method will be the most effective at these colder temperatures, where 0 °C and −10 °C were used for MW elucidation.
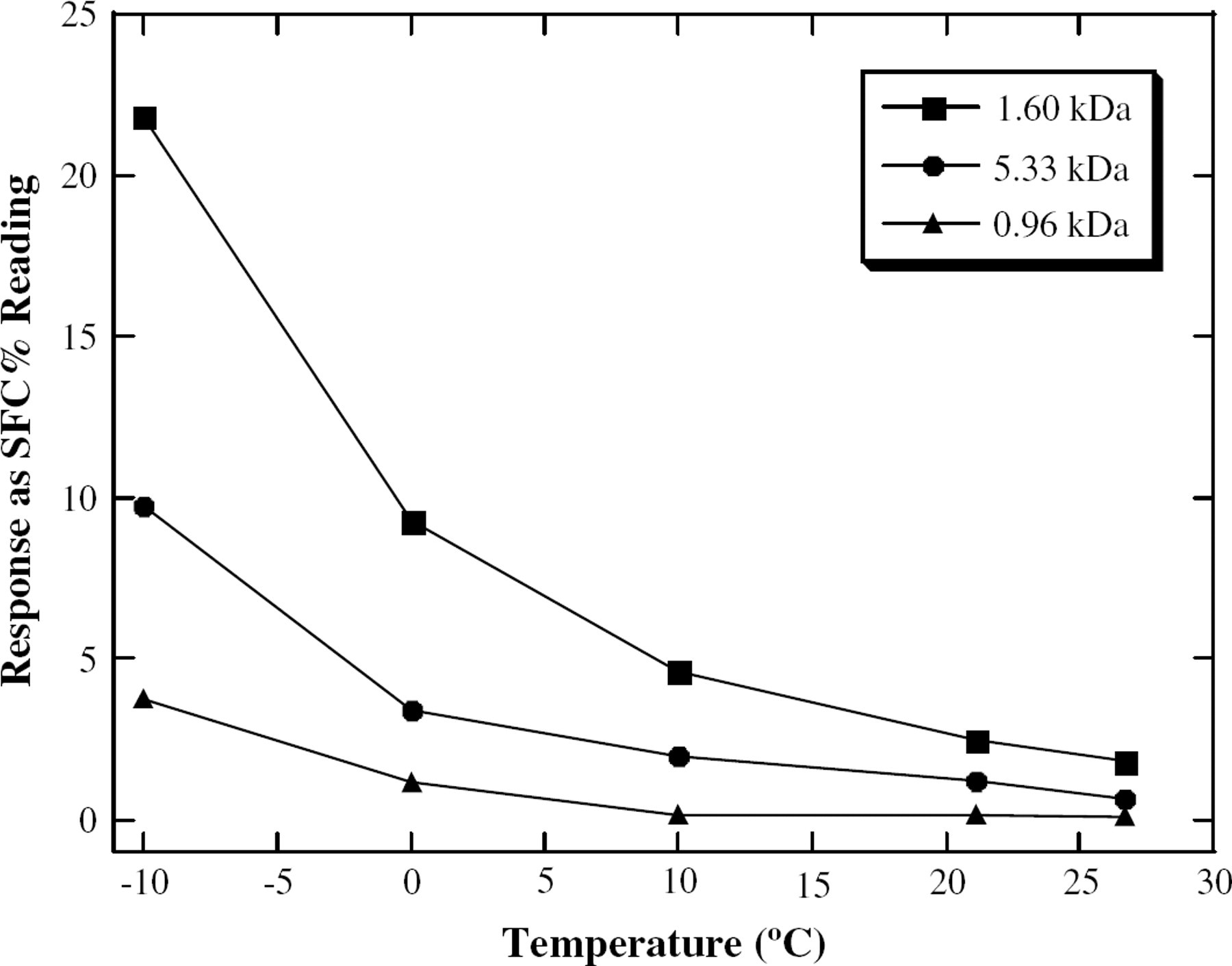
A plot of the observed response of the low-resolution NMR (read as %SFC) as a function of temperature.
Linear Relationship
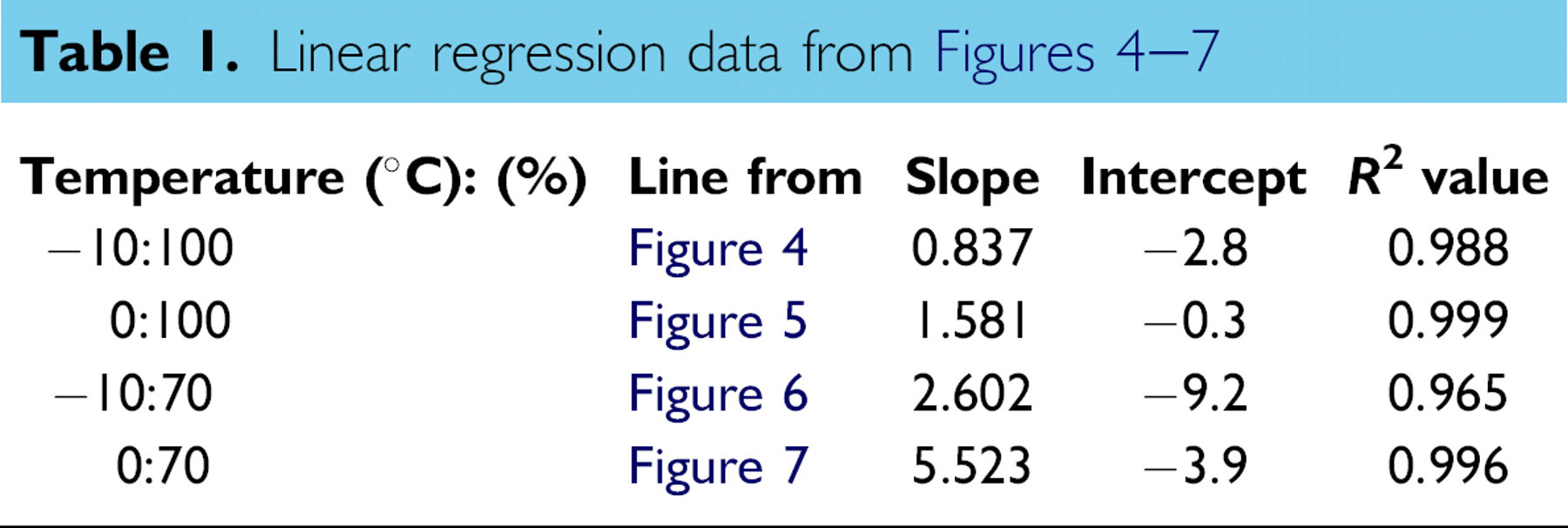
A relationship between the observed NMR response and the known MW of the samples was plotted. Linear trends were observed in both the −10 °C (Figure 4) and 0 °C (Figure 5) data. Linear regression analysis was performed on the samples, and the resultant fits (Table 1) give slopes between ∼0.8 and ∼1.5, ideal for the determination of a variety of polymer MWs.
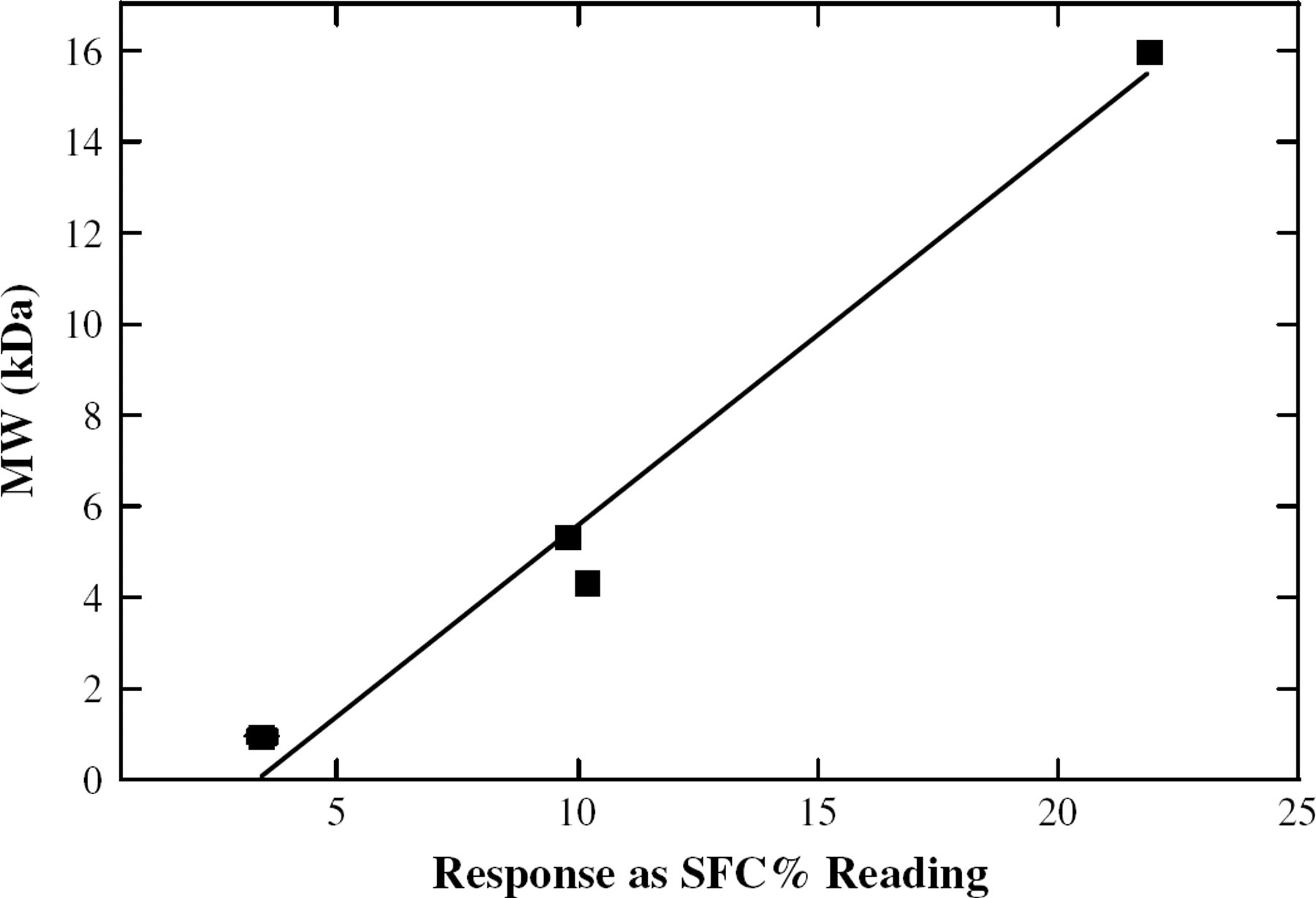
A linear trendline for the response of the NMR vs the MW of the polymerized soybean oil samples, at −10 °C.
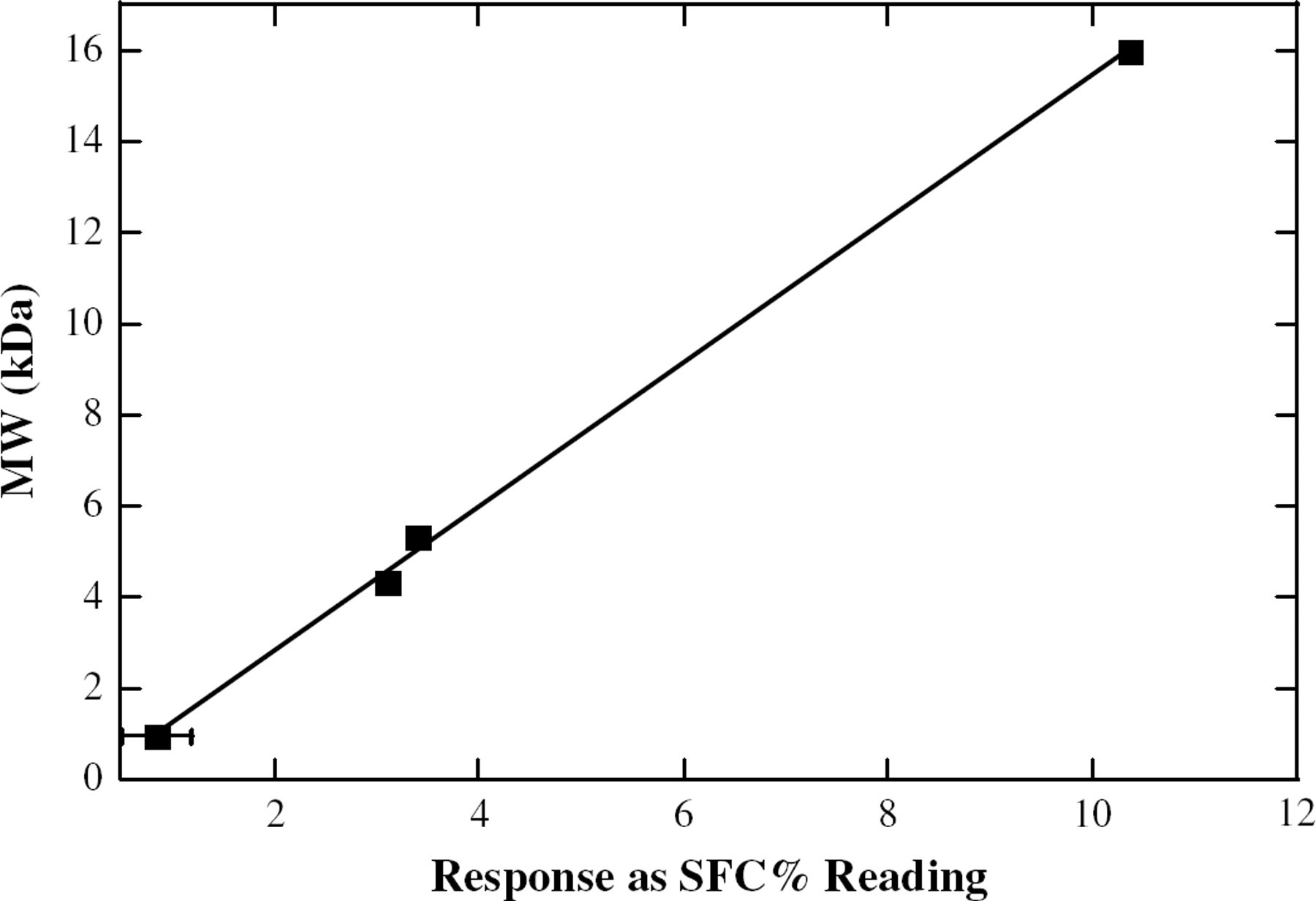
A linear trendline for the response of the NMR vs the MW of the polymerized soybean oil samples, at 0 °C.
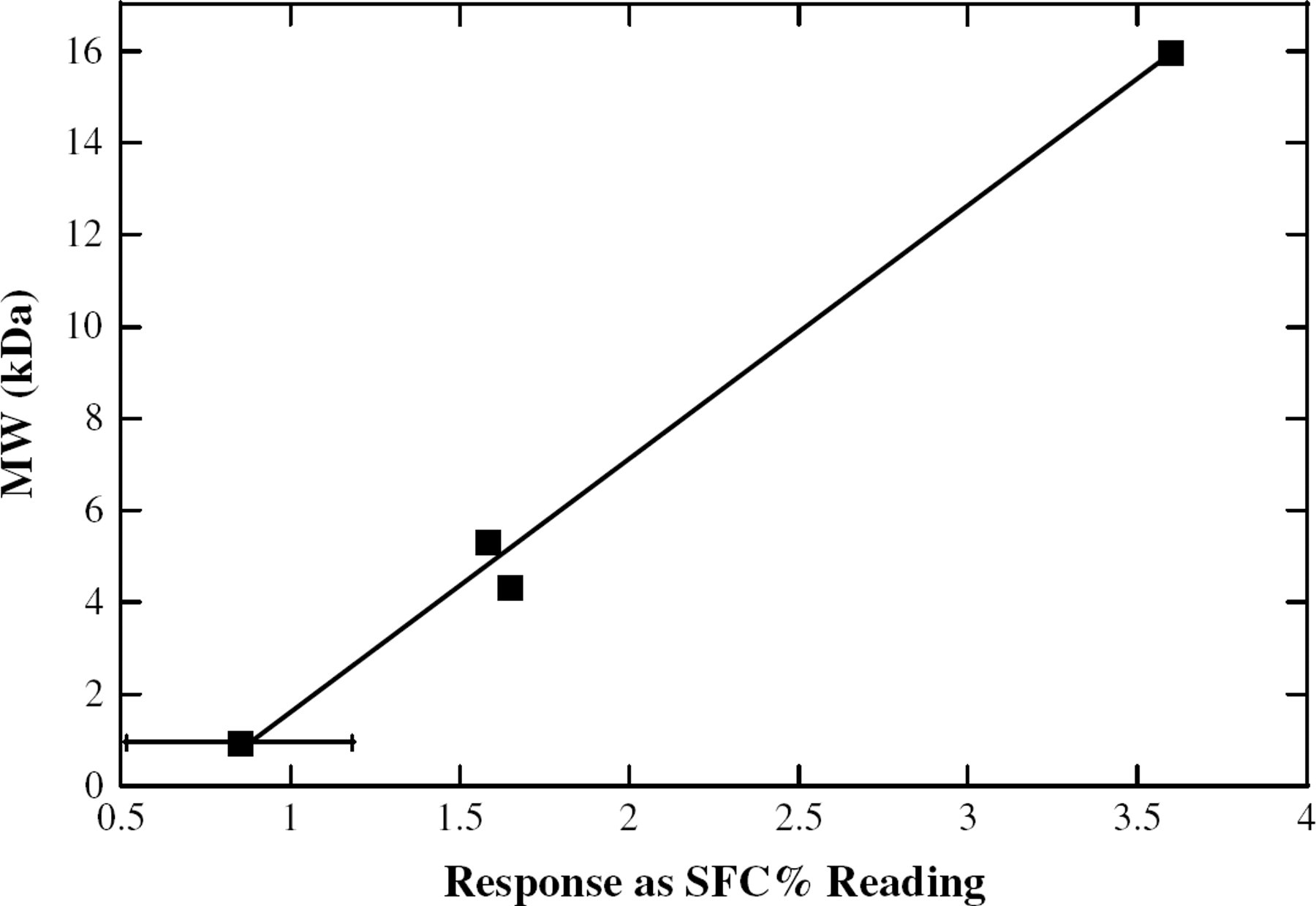
A linear trendline for the response of the NMR vs the MW of a solution of 70 wt.% polymerized soybean oil and 30 wt.% normal soybean oil, at −10 °C.
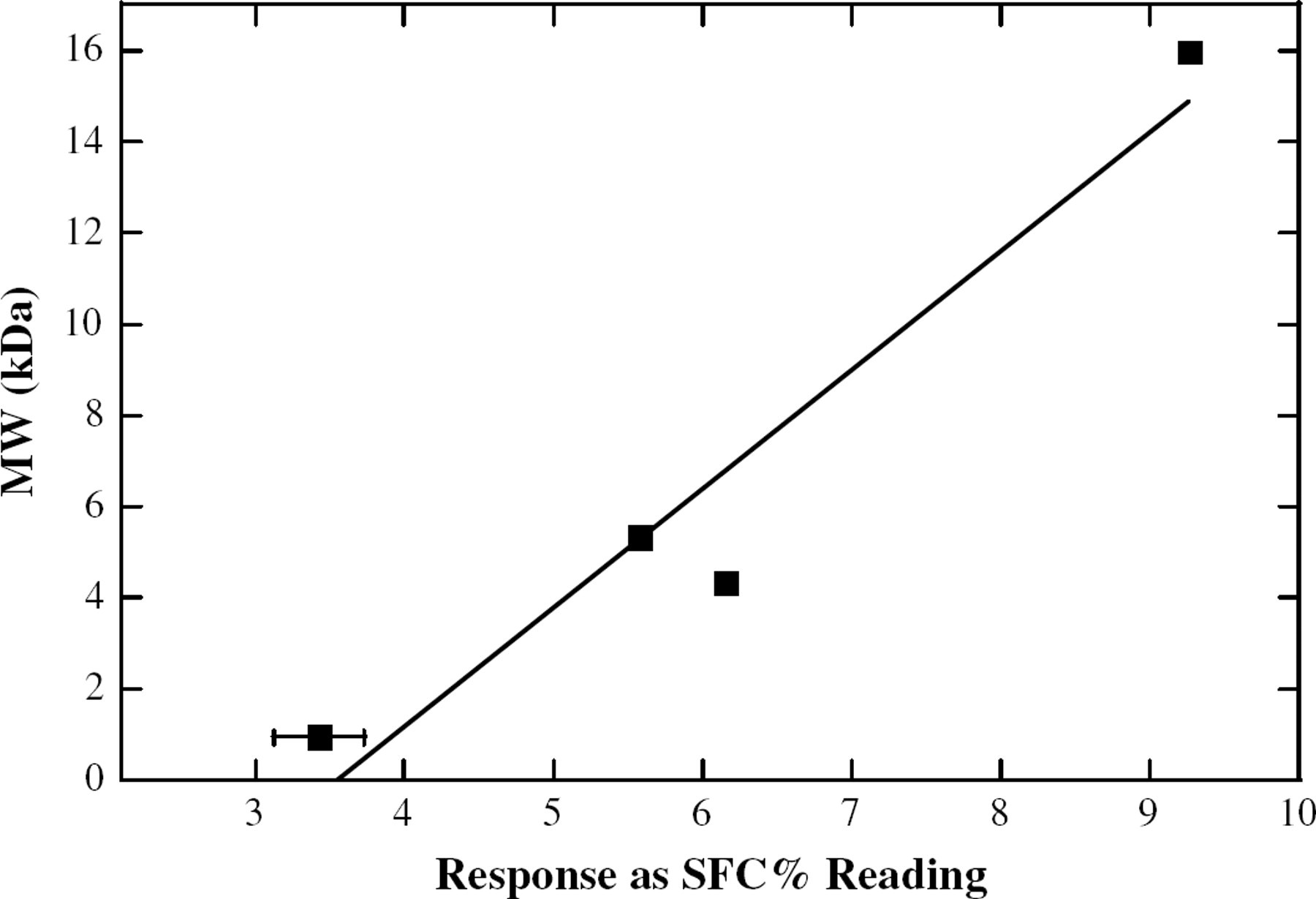
A linear trendline for the response of the NMR vs the MW of a solution of 70 wt.% polymerized soybean oil and 30 wt.% normal soybean oil, at −0 °C.
Concentration
One potential drawback of this method is that at higher MW, the soybean oil-based polymers become too viscous for convenient handling. We overcame this problem by testing the samples after dilution in ordinary soybean oil. Although other solvents could be considered, we chose ordinary soybean oil for several reasons. First, it was effective in aiding the sample handling. Next, because it is also one of the components in the sample, its NMR signal was easily within the operating range on the instrument. Finally, because it is a food grade solvent, it minimizes the potential impact of contamination, a large concern in some laboratories. At the concentrations above 30 wt.%, a linear trend of MW vs response could still be observed. Plots similar to those made using neat polymer (Figures 6 and 7) yield the expected relationship. These linear fits (Table 1) have slopes from 2.6 to 5.5. Although not as ideal as the other data, these slopes should still be adequate for MW determination.
Testing on other Soybean oil-Based Polymers
We decided to test our method on a soybean oil-based polymer made through a slightly different method. The catalyzed polymerization yields a compound with a slightly different structure, but the triacylglyceride structure remains intact. Using the catalyzed method, higher MWs are possible, than with the thermal polymerization. This polymer was analyzed by the NMR method at 0 °C, at both a 100% concentration. The spectrometer gave a response of 15.1. Using the equations (Table 1) derived from Figure 5 we calculated a MW of 23.6 kDa. This is in agreement with the value determined from gel permeation chromatography experiments. An additional experiment was performed using a 70 wt.% concentration. The response, 6.5, gives a MW of 32.0 kDa. This demonstrates that although the method may work for qualitative assessment, better results can be obtained using the 100% concentration.
Limitations
This article focuses on using the instrument without modification. Because the method measures the glyceride hydrogens, it is only applicable to polymers that have this type of structure. Also, the sample must have a viscosity such that it can be put in the NMR tube. However, with instrument modification, these limits could be overcome, and the use of low-resolution NMR may have a future beyond laboratories that already have this type of instrument for other purposes.
Summary
We have developed a simple, convenient method for the determination of the MW of soybean oil-based polymers. This information is important in the formulation of products, such as inks and coatings. Because the method is simple and fast, data taken from it could be used to determine the extent of reaction of various batch processes and give realtime insight into soybean oil reactions. Also, because automation technology for these instruments is already on the market, it opens up further possibilities.
Acknowledgments
The authors thank Drs. Brajendra K. Sharma and Zengshe Liu for supplying the polymers for analysis, Ms. Jennifer R. Wyss and Mr. Eric S. Johnsen for sample preparation, and Mr. Dale W. Ehmke for NMR analysis.