Abstract
Clinical pathology has a major influence on clinical decisions and the past 60 years have seen an evolution brought about by advances in information technology and automation. The impact of the ever-changing technology in regard to responsibilities and training therefore needs continual appraisal. In this article, the authors have drawn on their experience on automation in clinical chemistry and the experience at Monash Medical Centre in Melbourne, Australia where one of the authors is based. Automation in other industries has also been reviewed, since the reasons to automate and the impact of automation have similarities and these include reduction in errors, increase in productivity, and improvement in safety. Advances in technology in clinical chemistry that have included total laboratory automation call for changes in job responsibilities to include skills in information technology, data management, instrumentation, patient preparation for diagnostic analysis, interpretation of pathology results, dissemination of knowledge and information to patients and other health staff, as well as skills in research.
Research in clinical chemistry should not only emphasize evaluation of performance of automation but also should include pre- and postanalytical phases and training also needs to reflect this.
Introduction
Clinical pathology or laboratory medicine has a great influence on clinical decisions and 60–70% of the most important decisions on admission, discharge, and medication are based on laboratory results. 1 As we learn more about clinical laboratory results and incorporate them in outcome optimization schemes, the laboratory will play a more pivotal role in management of patients and the eventual outcomes. 2 It has been stated that the development of information technology and automation in laboratory medicine has allowed laboratory professionals to keep in pace with the growth in workload. 3 In a paper on “robotics into the millennium,” the various types of automation have been outlined 4 while other authors have classified laboratory automation into total laboratory automation, modular laboratory automation, and workcell/workstation automation. 5,6
This article evaluates the relationship of scientific staff, automation, and expert systems in clinical chemistry with particular reference to the core laboratory and ascertains staff requirements. The changes in work practices due to the introduction of automation and computers in other industries are discussed and similarities with clinical chemistry elucidated as it has been noted that the original total laboratory automation was based on the manufacturing/factory model of production. 7 Others have also written on automation in various industries over the last century and how the patterns of its implementation and effects can be applied to pathology. 8 The goal of a successful automation must be to change the way in which work is done in the laboratory and this involves changing not only the tools and processes, but also the job structure and ultimately the way people think about their work. 7 The progress in automation and convergence of technologies are two key factors, which particularly affect how we think about the future of clinical chemistry. 9 The role of the scientific staff, use of automation and expert systems shall be discussed for a core laboratory focusing on the Monash Medical Centre, Melbourne, Victoria, Australia where one of the authors is based. It is our belief that to consolidate changes that are advocated, 7 it is important to look at skill requirements and training of the operatives in clinical chemistry.
Automation in other Industries
There are similarities in automation in clinical chemistry and other industries. It has been pointed out that under pressure to reduce costs, while maintaining or improving quality, pathology looked to manufacturing models of production for answers and the vision from those observations was of total laboratory automation. 7 However, the same article points out that although many lessons have been, and continue to be, learnt from the manufacturing sector, not all of the concepts and applications are appropriate for the laboratory. For instance, hospitals provide services that to some extent are unpredictable, 24 h daily and cannot increase charges to any significant extent when costs increase because fees are largely dictated by government. 10 The provision of 24-h services in an environment that is unpredictable, coupled with reduced workforce are perhaps the driving forces to automation. In the United Kingdom, the Audit Commission review (1993) however pointed out that pathology staff was slow to change working practices and staff skill mix in response to the changes in technology (Price and Barnes, 1999). 11
In the United States, the growth and profitability of the insurance industry increasingly became dependent on automation and computerization. This involved a decrease in labor-intensive work and an increase in productivity of higher skilled knowledge-intensive occupations. It was concluded that in the insurance industry there was an irreversible and dominant role played by labor-displacing technological change and that other industries including health had adopted labor-displacing technologies. 12 The decrease in the requirements of less technically skilled staff such as office staff and couriers is experienced in pathology as specimens are transported via pneumatic systems, robots, and conveyor belts; downloading of registered requests and reports are computer generated and consolidation undertaken.
In the automobile industry, the driving force by Fiat to introduce automation was to eliminate dangerous and tiring manual operations and to lessen the influence of unions. 13 In clinical chemistry, tiring manual operations include loading and unloading of centrifuges and analyzers as well as making aliquot tubes and dilutions and such phases are amenable to automation. Automation especially of isotopic assays has also improved safety. Automation in the welding division at Fiat decreased direct worker participation and increased indirect workers' participation, that is, inspection and maintenance. The skill profiles of blue-collar workers have been suggested to change as automation increases and that a new job and skill profile has emerged, “the system controller.” 13 Indeed, automation in clinical chemistry has called for increased skills in data management as well as the new field of the automated processing line as opposed to instrument handling as most of the equipment is now highly complex and adjustments to the required performance are carried out by engineers who are employees of the suppliers. At present, most of the equipment is leased and the supplier is also responsible for major maintenance and it is unlikely that automation in pathology is instituted to lessen the influence of unions as noted in Italian automobile industry. 13 The introduction of a fully automated manufacturing system had problems and at the Fiat Cassino plant it was found to be potentially unreliable, fragile, and vulnerable, particularly in management information systems. 13 Clinical chemistry has backup methods, which use less complex instrumentation or are manual, and furthermore in the event of breakdowns work can be sent to other laboratories.
Commitment and the knowledge base of staff are key factors in business success and personnel are the driving force of productivity and quality improvement with support from technology. 13 It has been proposed that the acquisition of equipment used in production is a result of a learning process that is based on internal development, external acquisition, imitation, analogical replication, among others and it has also been noted that investment in equipment is largely non-reversible and represents a long-term commitment to technology. 13 In the author's laboratory, the introduction of an automated line was achieved through learning, selection, imitation, and external acquisition and the irreversible nature of commitment to technology is evidenced by the long-term contract that was entered into. The maintenance of the automated line is the responsibility of a supervisory scientist; a reflection of the experience at Fiat where the position of “systems controller” emerged. There is an on-going examination of this new activity with a possibility of creating a new work position to encompass maintenance and trouble shooting of equipment.
Inadequate response to technological change can endanger viability of an organization and literature categorizes technological change that enhances current skills and knowledge as “competency enhancing” and those that require new technical abilities as “competency destroying,” that is, old competencies are rendered obsolete. “Commitment-maintaining” technology supports employee's job security, whereas “commitment-destroying” technology makes workers redundant. 14 The introduction of an automated line in clinical chemistry with analytical instruments attached at Monash Medical Centre is in keeping with the concept of “commitment maintaining” and “commitment enhancing.” The acquisition of the automated line did not decrease the overall number of staff but there was redeployment and it is the authors' experience that introduction of new equipment often requires maximum numbers of staff for the months that follow and natural attrition or redeployment may be used as the benefits of automation are realized. The new automation may be from a new supplier but the assays/methods in most cases use the same principles hence this can be seen as “commitment enhancing.” The new skills and competencies to learn therefore would consist, for example, of the operation of the new technology, quality assurance, and data handling associated with the new automation.
A workforce is stabilized by technology change and the “flexible enterprise” model favors renewal of workforce. 15 The author also points out that old equipment is replaced by new equipment that often contains more information technology than the old and the staff therefore need new skills and competencies to meet demands of the new situation. Such a scenario is characterized in clinical chemistry by the skills acquired to manage, for example, automated line, interface to the host computer, and the work patterns that change due to the introduction of autovalidation. The static view of technical determinism 15 –18 proposes that “automation reduces the amount of direct work and increases the number of indirect tasks such as supervision of machinery, quality control, or maintenance.” New skill competencies are required and these also entail modifications of the division of labor. The graduate scientists in the core laboratory fit the criteria for integration as proposed, 15 and in this scenario the scientist performs productive tasks, that is, validation of abnormal results, problem solving of analyses, and quality control and maintenance of the equipment. Integration is evident in the core laboratory as the assays formerly conducted in other disciplines, for example, serology assays are now the responsibility of the core laboratory.
Organizational and technological change is highly correlated with skill changes and in the core laboratory; organizational change that comes with introduction of automated serology assays increases the skills and knowledge, whereas the technological change associated with introduction of the automated line elevates technical skills.
Reasons for Automation in Clinical Chemistry
Various reasons have been given to automate and these include staff shortage and the increase in workload from consolidation and requesting patterns 19 and economics, reduction in errors, and improvement in quality. 4 Others have reported that in their experience the most important impact of combined laboratory reorganization was reduction in full-time employees and this was coupled to decreased error rate in clinical chemistry and hematology that could be attributed to the implementation of automation and the uniformity that is part of standardization of the laboratory operation, 2 while the need to decrease laboratory costs and to improve turnaround time have also been cited for introducing total laboratory automation. 5 It is reported that clinical laboratories need to boost productivity and in many laboratories it is impossible to increase productivity using the equipment they have. 10 This is coupled to low staff numbers these laboratories have and the Association of Clinical Biochemists and The Royal College of Pathologists in UK in a 2002 report pointed out that between 1995 and 2001 the workload increased while the number of staff in clinical chemistry decreased. 20 An automated laboratory system was developed rather than accept an intolerable shortage of technicians at Kochi Medical school in Japan 21 and a decline in the rate of increase of the number of full-time employees in laboratory sections that automate has also been noted. 22 Automation also helps relieve stress on technologists brought about by increased workload 6 and the manual handling of samples that may be infectious constitutes a hazard to laboratory staff, 1,8 hence robotic system improves safety for laboratory staff by eliminating the exposure to pathogens. 23 Automation therefore provides value through improved efficiency coupled with reduction in processing errors, improved turnaround times, automated repeat and reflex testing, enhanced safety, and improved specimen tracking. 10
Development of Automation in Clinical Chemistry
The key to improvement of laboratory services is the implementation of correct automation technology. Clinical pathology automation has evolved from an idea rooted in the mechanical aspects of specimen manipulations in the early 1970s to a more complex information systems-driven technology in the late 1990s. 2 Laboratory automation has a history of over 50 years 24 and the first mechanized clinical laboratory instrument was introduced in 1956. 24,25 In 1957, the first continuous flow analyzer was described and this was a major leap forward in technology. 25 The instruments at that time included Autoanalyzer 1 (Technicon Instruments Corporation, NY, USA), which could assay blood urea nitrogen at the rate of 20 samples per hour. The Autoanalyser 1 was run alone or the sample could be split and the most complex format was to link the Autoanalyzer to a flame photometer resulting in simultaneous analyses of several tests, for example, sodium, potassium, creatinine, and urea. The 1960s onwards saw the release of comparatively fast analyzers 26 capable of performing over 20 analyses at a time; the second generation systems. From 1980s, analyzers were released that were test selective and had throughput of over 2000 tests per hour and therefore required additional technical skills such as instrumentation expertise and for the Technicon instruments, an appreciation of flow dynamics was useful.
Although each generation of automated analyzers improved throughput and turnaround time, they lacked the ability to make quantum improvements that are prerequisites to significant improvement in productivity and this has lead to “total laboratory automation,” a concept which goes beyond automation of analyses by including automation of important labor-intensive manual preanalytical phases. This concept, conceived in Japan, includes robotized specimen processing and delivery and the instruments use embedded computers to govern mechanical movement and data acquisition. 10 Until the late 1970s, most laboratories manually recorded results and the introduction of computers as information systems and as the interface for instruments was significant as this allowed uploading of results (unidirectional interface) into the laboratory information system making the manual transcription of results redundant. A further refinement is bidirectional interface that allows requests to be registered into computer and downloaded onto the appropriate instruments. This is followed by analysis, quality control, verification, and validation of results and, then uploading of results into the laboratory information system. Software was further developed to provide functions that assist scientists in performing analytical work and an important facility in laboratory information systems now is the ability to review results of internal quality control.
Technical/Organizational Phases in Clinical Chemistry
An overview of the technical phases in clinical chemistry is important in understanding the impact of automation; the pathology cycle involves identification of an appropriate request, preparation of patient, collection of specimen, preparation of specimen, analysis, validation, and reporting of results and these components are important in production of true results. It has been noted that fundamental issues such as test and method selection, patient preparation, quality and handling of specimens, as well as test interpretation and utilization have not, in the past, been given sufficient attention making preanalytical and postanalytical phases more error prone and a reappraisal of the role and mission of clinical pathology laboratories has been argued, with focus on issues such as test appropriateness and test interpretation. 3
Preanalytical Phase
The preanalytical phase constitutes about 60% of the time in a diagnostic process 27 and an error rate in laboratory results of 1–2%, which are mainly preanalytical, that is, sample processing has been reported. 4 It has been argued that the preanalytical phase of laboratory expertise has not been given proper consideration, thereby leading to more errors in this phase. 3 Comparison of the preanalytical mistakes to other types of mistakes found that 84.5% of the mistakes were preanalytical, 28 whereas some studies noted that the errors due to preanalytical factors were 46–68% of total errors 1 and other studies report that preanalytical variables account for perhaps 30% of variables that give rise to results that are discrepant from the clinicians' expectations. 29 Automation, which includes an aliquotter that prepares secondary tubes in clinical chemistry would therefore minimize preanalytical errors. It has also been shown that preanalytical automation enables absorption of increased workload and simultaneously reducing the number of employees. 24
In recognition of the importance of preanalytical phases, the supervisor in the central reception/registration at Monash Medical Centre is often a graduate scientist and this area is also automated. It is usually technical or clerical staff that carries out the tasks of the preanalytical system 4 and in their total automated laboratory, the preanalytical Inlet module is usually operated by laboratory assistants, whereas operations and maintenance of the major components and analyzers are reserved for technical staff. 5 Information technology has improved efficiency in medicine; secretarial time in filing and retrieving paper medical records is reduced and electronic requesting of investigation allows tracking of outstanding results. Voice recognition software also reduces secretarial time and such benefits lead to reduced costs and improve patient care. 30 Medical staff can request tests and access results away from the health facility thus also improving patient care.
Analytical Phase
In the absence of reagent kits (first-generation systems), the scientist prepares the reagents, standards, and quality-control samples. Open-channel instruments allow scientists to use any diagnostic company's reagents and to transfer method(s) to an “open instrument” and these methods are often referred to as “user-defined chemistries.” Most clinical chemistry analyzers have open channels, hence the scientist is able to develop and modify methods for the instrument. Such a facility is essential as it allows new methods to be developed by the laboratory and this is important in situations where the instrument manufacturer no longer provides reagents for a particular method. User-defined chemistries require scientists with technical and theoretical knowledge and in the author's laboratory, examples of user-defined chemistries include copper, fructosamine, and angiotensin-converting enzyme, which are assayed on the Beckman Coulter Unicle DxI 800 Synchron (Beckman Coulter, CA, USA). These chemistries use reagents that are sourced from companies, which may have different formulations, hence require prior evaluation.
The years 1950–1980 witnessed a thrust in method development and considerable expertise of staff in clinical chemistry was used to develop, modify, and validate assays. Japanese reagent manufacturers responded to low-staffing levels in medical laboratories by producing “instant” reagents 21 but the increasing availability of “instant reagents” meant that method development shifted to diagnostic companies from clinical chemistry laboratories. As methods became more complex, the ability to formulate reagents by the scientists in clinical chemistry for in-house methods declined 31 and laboratories began to rely to a large extent on first-generation system reagents from diagnostic manufacturers.
Postanalysis
The postanalytical phase is also prone to errors and an error rate of 19–47% of total errors in the postanalytical phase has been reported. 1 Proper reporting of results is crucial and this can be performed electronically and the reporting may be single or cumulative episodes. Pathology reports are normally prepared or reviewed by the chemical pathologist, registrar, or scientific staff who are also available for consultation or interpretation and make decisions on follow-up tests. The introduction of consolidated instruments brought automatic dilution and dilution of specimens which, although a small but significant part of workload, is repetitive and offers little challenge. Another feature of consolidated instruments is reflex testing but this has perhaps removed a challenging task that involves analysis and interpretation of results before a decision is made for further analysis. Autovalidation of analytical data has criteria that govern release of patient results. These criteria are determined by senior scientific and medical staff and this allows the expertise of senior scientists and medical staff to be used uniformly throughout the day. Some have stated that databases that facilitate interpretation and use of laboratory tests will become the norm and these have been shown to improve compliance with established clinical practice guidelines. 32
Staffing
There appears to be an overall shortage of staff in clinical pathology and this been reported in Great Britain and the United States. 33,34 and it has been pointed out that graduates are being employed in alternate facilities such as research and industrial laboratories. 35 This may have resulted in some tertiary institutions opting to teach generic medical science programs to allow versatility in employment. In the United States, the number of programs producing graduates for the clinical pathology laboratory has also declined over the years. 36 Coupled to the fact that in Australia, registration with professional bodies is not a requirement for employment in pathology this perhaps impacts negatively on salaries, professional recognition, and staff numbers.
Automation enables laboratories to increase workload and Uettwiller-Geiger (2001) notes that in the 1990s their laboratory experienced an increase in test volume without commensurate increase in budget or staff and to address the challenges their laboratory moved from a traditional laboratory set-up to an integrated laboratory delivery system or core model. 23 Total laboratory automation allows relatively fewer staff to handle large workloads owing to the efficiency in handling samples, that is, reducing bottlenecks by providing an even delivery of samples to the analyzer. 4 Others have also noted a reduction in the turnaround time and ascribed this to specimens being brought to the automated line immediately after registration whereas previously, samples would remain in reception until collected by scientific staff. 22 Four major time wasting steps are eliminated in the preanalytical phase by total laboratory automation; sorting in reception, waiting until scientists picked up the samples, manual identification of each specimen by reading the barcode, and waiting for analysis to commence. The time wasted in looking for specimens at Monash Medical Centre has been largely eliminated due to the introduction of automated line with a computer program that enables production of appropriate aliquot tubes and sorting of samples into correct locations. The benefit of total laboratory automation is not only the improvement in turnaround time but also the turnaround outliers also decrease. 37 Automation also allows staff to focus on abnormal samples and results 23 and others have concluded that third-generation systems will eliminate “dull, dirty, and dangerous tasks” allowing staff to focus on high value work. 38
Not only should scientists aim to improve the analytical quality of tests but they must also provide state-of-the-art algorithms for interpretation and utilization of tests. Furthermore, clinical pathology laboratories are not only a testing service to provide quality results but are a knowledge and education service that laboratory professionals should maintain as part of health care. 39 Others are of the opinion that decreased requirement for graduates in more routine areas of clinical chemistry will be compensated for by graduates who have a blend of technical, clinical, and managerial skills in more than one area. 40,41 These authors pointed out that a proportionally larger number of graduate scientists will work in nonautomated areas, while non-bachelor degree staff will predominate in the automated laboratory. Robotic automation would allow absorption of increased laboratory testing while withstanding reduction in full-time employment caused by attrition and labor shortage. 23 Besides low staff levels, the other major reason for automation could be cost containment, and cost containment with respect to a reduction in staff with a substantial increase in workload for the new core laboratory is a driving force in laboratory consolidation. 42
Research and Training
There is no doubt that clinical biochemistry needs to devote more time to research and this view is shared by a number of authors. For instance, it has been pointed out that research and development should be practiced by health-care professionals as an on-going process of continual improvement but this had decreased since the early 1990s, largely due to reduction in full-time employees in relation to work volume. 11 Since the 1980s, there has been much emphasis on automation and not enough on research, which has strategic importance to clinical biochemistry. 9 Literature has cited “test” and “diagnostic” research; test research quantifies “characteristics” of a test rather than the test's contribution to estimate diagnostic probability of disease's presence or absence, which is “diagnostic research.” 43 There are schools of thought that improvements in analytical specifications automatically translates into better diagnoses and treatments but there is also evidence that clinical benefits can be achieved by focusing on quality of total testing and in particular, on appropriateness of test requesting and interpretation. 39 As automation continues to play an increasing role in laboratory medicine, it is important therefore that scientists also focus more on research to achieve quality of total testing.
Since the 1980s, clinical chemists have allocated a large proportion of resources to support automation as opposed to maintaining and promoting research. 9,44 It is our view that the preanalytical, analytical, and postanalytical phases are interdependent and research should equally be directed to all the phases. The main issues associated with laboratory re-engineering include; multidisciplinary training, greater need for skills in systems control and support compared to analytical skills, and the need for training in information technology and management techniques. The American Association of Clinical Chemistry Task Force (1996) on the Changing Practice Environment listed core competences expected of clinical biochemists and they include: assessment of new technologies, application of informatics and appropriate research, management skills, professional development, and continuing education. 9 Some authors have also expressed concern at the excessive enthusiasm for purely technological aspects at the expense of issues such as test and method selection, patient preparation, quality and handling of specimens, as well as test interpretation and utilization and argue for a focus on issues such as test appropriateness and test interpretation. New tests, especially molecular techniques, rely more on laboratory advice and interpretation of results, hence by default laboratory staff are increasingly sharing the responsibility of patients' treatment and management. 3 Such requirements therefore call for changes to the curricula of the laboratory specialist and also for programs dealing with continuous education to emphasize test appropriateness and interpretation of pathology data.
Scientists in the future will be aided by intelligent software to bring together results from physical experiments and analysis to formulate hypotheses and plan future experiments. The quality of software will be important to the successful laboratory and will expand the number of decisions and inferences from data; the final state will be the automation of the whole experimental cycle with only the highest strategic decisions made by the scientist. 45 If this is the case then scientific duties with regard to method development will need a high component of computer expert knowledge and this has implications for the staff required and knowledge base in clinical chemistry. Curriculum development is changing to incorporate computer-aided experimental design and the use of computers for data mining as well as modernizing traditional scientific material. 33 Postgraduate degrees should offer a curriculum that encompasses systems engineering, systems management, contract management, finance, and human resource management with a strong emphasis on team building and marketing. 41
Staff Structure and Skills
Efficiency declines when there is an inappropriate level of staff and this leads to stress from overwork, delays in processing, and increase in errors ultimately decreasing productivity. A blend of staff with technical, clinical, information technology, and management skills is therefore required in clinical chemistry. The establishment of a core laboratory should preserve scientific experience and expertise and needs to be accompanied by a reduction in base grade scientists who would be replaced by technicians and laboratory assistants. 46 The submission from Eastern Health to the Review of the Victorian Pathology Services Accreditation Act 1984 states that the majority of tests, particularly in clinical chemistry and hematology, are performed by automated analyzers with on-board computers linked to laboratory information systems capable of data validation and ensuring quality assurance. Other submissions suggest that a considerable number of tests in the laboratory are now automated and this has reduced the skill requirements for those conducting the tests. 47 The Audit Commission in the United Kingdom noted that pathology staff was slow to change working practices and staff skill mix in response to changes in technology. The commission recognized that clinical pathology was complex and more than just analysis and that there are other factors that impact on staff in clinical chemistry, which are advice on appropriate investigations, selection of suppliers, interpretation of results, quality of results, training and education of laboratory staff and users of pathology service, participation in clinical audit and creating effective management structures (Price and Barnes, 1999). 11
Employment in clinical laboratories increased between 1960 and 1980 due to a growth in demand that was generated by increased laboratory capabilities and availability of tests. The decade from 1980 however saw a growth in capacity of automation without an increase in number of staff and from the 1990s there was introduction of automation for labor savings and at the same time consolidation of laboratories that lead to a decline in labor requirements but this was projected to plateau in laboratories with advanced automation. 8 It can therefore be seen that there is a requirement to review the number and balance of staff as responsibilities change and a further consideration is whether staff of the core laboratory is deployed to other specialties of clinical chemistry that are less automated and more technical. If rotation of staff occurs into these areas then the requirement for tertiary educated staff remains the same because more complex analytical work is not usually performed by technicians. This view is shared by others who consider that many areas of clinical chemistry will continue to require staff with very high technical skills and knowledge. 11
In the past, manufacturers have developed instruments for each discipline but now there is a trend to develop a single instrument capable of analyzing samples for a range of tests, which cross the traditional departmental boundaries. 4 The central core laboratory usually consists of sections of previous technology-related laboratories 48 and the majority of assays that were carried out in endocrinology, toxicology, serology, and special clinical chemistry are now part of automated central laboratory. Therefore, the staff who will interpret results from samples now processed on the automated line, which were previously in a specialist laboratory requires consideration. One approach is for intellectual ownership of the assay to be retained by the original specialist laboratory, which is possible with a two-stage validation process. This involves the core laboratory performing the analysis and validating results. This is followed by the specialist laboratory reviewing the validated results, making clinical interpretation and then releasing results into the laboratory information system. 49 Alternatively, rules may be written into the laboratory information system that contains the expert knowledge of the specialist laboratory and its comments.
Another consideration is whether scientists work only in one discipline or are multiskilled, for example, capable of working in clinical chemistry and hematology. Co-location of instrumentation is a solution often implemented. 46,50 In this model, work is performed by staff for each discipline, for example, clinical chemistry and hematology on analyzers with a shared central specimen reception and the other model is of core laboratory. Co-location obviates the need for retraining a large number of staff and the constant updating of knowledge for the several disciplines. The close proximity of instrumentation and staff retains the benefits of central reception; ancillary facilities; and cooperation in day, evening, night, and weekend shifts. There is a need to assess the skills and interests of staff before implementing automation and this is important if the laboratory has separate specialist laboratories because there may be a need for cross-training. 10 An important point mentioned was customized training for individual's existing skills but this may have negative financial implications.
As throughput increases so will the implementation of core laboratories into clinical pathology, and a complete core laboratory includes all automated analysis for clinical chemistry, hematology, coagulation, serology, bacteriology, and blood bank and in this model staff is cross-trained to operate most of the instrumentation. Experience elsewhere has shown that implementation of a core laboratory necessitated the transformation of a laboratory's operations from separate professional disciplines to an integrated core model and this included merging of clinical chemistry and hematology, removal of physical barriers to accommodate a core model, integration and cross-training of staff, work station consolidation and consolidation of proficiency testing, quality control, and educational programs, among others. 23
At Southern Cross Pathology Australia, Southern Health, the introduction of the automated power processor and attached analyzers lead to a decrease in Grade 1 level staff in several outlying laboratories. Outpatient and nonurgent inpatient work was sent from other Southern Health laboratories to the central laboratory at Monash Medical Centre. Productivity also improved in the core laboratory and this outcome has also been the experience of others 24 who reported a reduction in technical staff while increasing productivity. The reduced staff requirement at Dandenong and Moorabbin laboratories was used to open a new laboratory at Casey Hospital. The redeployed staff constituted 4.4 full-time equivalents. In addition four staff took voluntary redundancy packages. Minor staff changes were instituted in the main clinical chemistry laboratory and the reduction of staff by one was due to consolidation of instrumentation. At the North Shore-Long Island Jewish Health System, where a core laboratory was conceived and subsequently implemented, no employees had lost positions as a result of the exercise. 5
The changing role of clinical chemistry was succinctly put 3 in the following categories:
Real-time results
More effective tests
Involvement in therapeutic decisions
Increased clinical consultation
Emphasis on prevention as well as cure of disease
Use of evidence-based medicine and outcome assessment of laboratory tests.
This role has also demanded a change in the knowledge base and technical skills of the scientist. 3 At the North Shore-Long Island Jewish Health System, which uses an automated system they removed technical staff from routine tasks and placed them where they handle complex tasks relating to analytical processes. The technical staff handles “backups,” for example, failed quality control that requires user intervention or calibration, and the overall maintenance for clinical laboratory automated system is under the automated laboratory manager. 5
It was forecast that in the 2000s, the skills for the medical scientist will include cellular and molecular diagnostics 38 and it was noted 3 it was that laboratorians (scientists) must fulfill the following roles in the future:
Guarantee the quality of analytical results
Ensure quality of pre- and postanalytical phases
Continuously improve use and interpretation of laboratory data in patient management
Participate in laboratory and clinical research to ensure the best use of laboratory data.
Key Performance Indicators
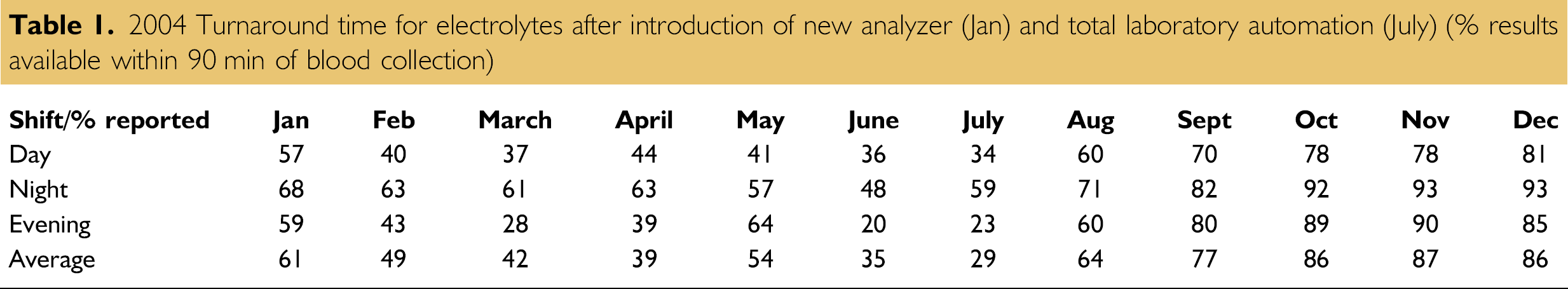
A consideration in evaluating automation after its implementation is comparison of key performance indicators and in our experience the change in performance is usually reflected when work processes have stabilized. A decrease in performance may be seen in the short term but this often reflects the inexperience of staff with new instrumentation and the bedding-in of new work processes. The authors' experience is that the laboratory achieves maturity 24–30 months after introduction of a major stand-alone analyzer and the changes in work patterns, which ensue. The experience in the Biochemistry Unit at Monash Medical Centre was that the Key Performance Indicators (KPI defined as % results available within 90 min of blood collection) for potassium, sodium, chloride, and bicarbonate increased significantly after 12 months compared to preintroduction of the automated line. This was after an initial decline in the first 6 months, which was followed by an almost arithmetic increase in improvement and this performance has been maintained. (Tables 1 and 2).
2004 Turnaround time for electrolytes after introduction of new analyzer (Jan) and total laboratory automation (July) (% results available within 90 min of blood collection)
Turnaround time for electrolytes for the emergency department at Monash Medical Centre (% results available within 90 min of blood collection)
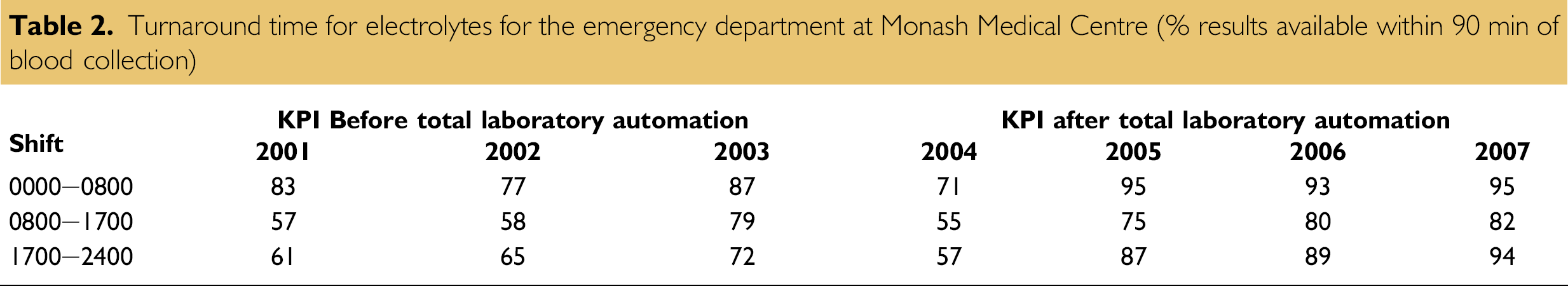
The volume of work passing through the analyzers has increased at the Monash Medical Centre laboratory (7% per year since 2004) and similar experiences have been reported at North Shore-Long Island Health System with the laboratory performing outreach testing. 5 The outpatient and nonurgent specimens from three other hospital-based laboratories now pass through the main analyzers in the clinical chemistry unit at Monash Medical Centre as well as urgent samples from Emergency, Intensive Care, Coronary Care, and Hemodialysis Units, which are placed on automated line. Urgent specimens are placed on the Inlet module to the automated line because this ensures timely analysis and reporting of results and special handling procedures are not required and this demise of the urgent laboratory was postulated. 4
Instrument Function Associated with Automation
A description of instrument function at Monash Medical Centre is necessary to consider staff requirements in relation to automation. The instrumentation consists of the Beckman Coulter Power Processor Automated Samples Processing System, Intelligent Aliquotter, Specimen Stockyard, Outlet rack, the UniCel DxC 800 Synchron Clinical System (Beckman Coulter, CA, USA), and Unicel DxI 800 Immuno-assay System (Beckman Coulter, CA, USA).
Automated Line with Processing, Analytical, and Storage Modules
Automated centrifugation, although expensive, may be considered the first requirement for efficiency in automation. One-third of sample workload was delayed in clinical chemistry for more than 5 min because centrifuges were not emptied immediately and the correct time for centrifuging was not used in 35% of runs. 51 Centrifugation is labor intensive due to batch processing and nonautomated centrifuges must be loaded and unloaded manually. If a centrifuge is not opened when samples are ready there is a delay and similarly if the staff unloads the centrifuge prematurely then there is inefficiency. Preanalytical automation can reduce labor costs by up to 50% 52 and it is the lower cost staff who are replaced by preanalytical automation. 4 An observation in the clinical chemistry unit at Monash Medical Centre into registration and manual handling of specimens, revealed centrifugation to be the major bottleneck and this is in accord with findings elsewhere. 4 This is not however universally accepted and it has been pointed out that people could do this task more efficiently and the process was largely without error, thus negating one of the reasons to automate the process. 19 It is the experience at Monash Medical Centre that automated centrifuging is more efficient and requires less staff to run and studies suggest that automated centrifuge loading is advisable to avoid the discontinuity of batch-wise centrifuge loading 38 (Fig. 1). Centrifuge.
The Intelligent Aliquotter, comprising a serum-level detector and secondary-tube labeler and aliquot unit, has brought about fundamental change in processing of samples in clinical chemistry. Before installation of the automated line, the clinical chemistry unit had a policy of minimal aliquots, that is, aliquots were prepared only when necessary for stability of the analytes due to the risk of incorrectly labeling aliquot tubes. An approximate 1% error rate in labeling aliquot tubes has been reported, 6 whereas a 0.2% error each has been attributed to mislabeling and spilt and dropped tubes. 4 The laboratory at Monash Medical Centre was in essence a serial laboratory with regard to testing, that is, a sample was placed on one analyzer and when analysis was completed the sample was moved to a second analyzer resulting in a longer turnaround time. The aliquotter station removed this barrier with automated labeling of aliquot tubes and pipetting from the primary tube. The clinical chemistry laboratory became a parallel testing laboratory with consequent improvements in turnaround time for the analytical work in the manual and low volume work stations in the special clinical chemistry laboratory and in handling of specimen tubes and such improvement has also been noted by others 24,38,53 (Figs. 2 and 3). Aliquotter. Aliquotter, pipetting station.

The third improvement was linking the automated refrigerated 3060 tube stockyard via the Power Processor to analyzers to allow sample tubes to be retrieved for retrospective and reflex testing and dilutions, hence time is no longer wasted in locating samples. Others consider this important along with automation and laboratory information system interfacing. 2 The tubes are returned to the refrigerated stockyard and filled racks are mapped and have descriptors. Mapped racks, which are removed from the stockyard are stored in a walk-in refrigerator and the retention time for mapped racks is 14 days, which is the stability time for most common analytes. Retention allows retrospective tests to be added and those requested within 3 days, the average time racks are retained in the automated stockyard, are automatically retrieved, de-capped, sent to the analyzer, and after analysis automatically re-capped and returned to the stockyard. Specimens can be retrieved from mapped racks or from the stockyard by interrogating the line computer. Postanalytical automation is not considered to be a major consideration when reviewing the need for automation and sample handling, smooth workflow and handling an increased workload of specimens are considered more important. 4 This view is contrary to observations in clinical chemistry at Monash Medical Centre where retrieval of specimens from the stockyard for retrospective analysis and reflex testing saves time. Manual filing of specimens is subject to error, consequently time for manual retrieval of specimens is increased and the retrieved specimens need to be loaded onto analyzers and after analysis they are retrieved and re-filed with the possibility of mis-filing, hence postanalytical automation therefore removes these activities and saves time.
Command Central allows a scientist an overview of the power processor and the linked instruments. The operational status of UniCel DxC 800 Synchron Clinical System and Unicel DxI 800 Immunoassay System is displayed and the screens for laboratory information system validation may be displayed for UniCel DxC 800 Synchron Clinical System and Unicel DxI 800 Immunoassay System. This facility is important for the evening and night shifts when there is only one scientist in clinical chemistry. Process control allows for significant improvements in laboratory efficiency 54 and the basic process control recognizes instrument failure and alerts the operator of any repair action. Intermediate process control alerts the scientist to the status of onboard reagents, waste management, and instrument failure. Advanced process control adds the ability to turn the instrument on and off remotely, to schedule samples for dilution and/or repeat analysis, and for the instrument to have ability for self-diagnosis and repair. At its fullest implementation, the main instruments at the other Southern Health laboratories could be reviewed for operational status and also quality control by Command Central at Monash Medical Centre.
The work processes of central reception at Monash Medical Centre have been significantly altered by the power processor. The sample tubes for UniCel DxC 800 Synchron Clinical System and UniCel DxI 800 Immunoassay System are placed directly on the Inlet module after registration and the barcode on each sample tube is read and the tube proceeds to line centrifuges and then to aliquotter and analyzers; this process is supported as desirable. 55 In effect, technical assistants now load the analyzers, a task formerly performed by scientists (Fig. 4).

Inlet.
The number of manually loaded samples has decreased to less than 50% and a significant proportion of these are pediatric samples. Scientists from serology also place sample tubes onto the line to make aliquot tubes for permanent storage and this is a significant saving in time and removes manual labeling of aliquot tubes, a potential source of error. Other studies have noted that their robotic system automated many time-consuming and labor-intensive processes including sample login and sorting, centrifugation, and cap removal and specimen sorting. 23
Laboratory Information System
One of the priorities in clinical chemistry is rapid access to appropriate information and laboratory information systems continue to develop to meet this demand. Laboratory information systems facilitate all aspects of pathology work from registering of samples to interpretation of data. It has been noted that there is abundance of information in biomedical sciences, which is continuously changing. Laboratory specialists and clinicians need access to scientific-based information anytime and anywhere. Various forms of personal digital assistant are under trial or in use and such tools have information on reference and critical ranges, interferences, pathophysiology, sample collection among others. 56
Registration
Registration of patients and pathology tests is carried out for all of clinical pathology by central reception staff and all requests are entered using test codes. The laboratory information system has programs that electronically register samples from outlying laboratories to the Monash Medical Centre and this allows the tests to be downloaded onto the power processor or to other load lists. This activity minimizes errors and saves time.
Autovalidation
Automated validation of results has been in use in core laboratory at Monash Medical Centre since January 2006 for SYNCHRON LX20PRO (Beckman Coulter, CA, USA) now replaced by the UniCel DxC 800 Synchron Clinical System and from March 2006 for UniCel DxI 800 Synchron Clinical System and this autovalidation is approximately 50% for DxC 800 and 75% for UniCel DxI 800 Immunoassay System. The autovalidated samples list on an electronic load list. Manual validation of remaining results on the load list gives rise to an average delay of 9 min between uploading of results from UniCel DxC 800 Synchron Clinical System to the laboratory information system and validation. Autovalidation removes this delay and this finding is also supported by others. 57,58 In some laboratories, 80–85% of results are released by autovalidation results with the remainder manually released by medical scientists, 59 whereas other laboratories report lower levels of results being autovalidated. 60
The responsibility for the autovalidated results also needs to be considered. It has been stated that computer released or interpreted results should be checked by the chemical pathologist or the requesting practitioner 37 and that a computerized interpretation of a result only becomes valid if there is a signature of the interpreting specialist. 38,61,62
Conclusion
There have been significant changes in clinical biochemistry over the last 60 years, particularly the last 10 years with the introduction of total laboratory automation. Automation of the main chemistry analyzers, including immunoassay and linking them together with preanalytical and postanalytical automation to give total laboratory automation has given predictability to result availability. Diagnostic companies that supply reagents have largely obviated the need for the development of methods, which previously was a major responsibility of the laboratory scientist. In addition computers are not only capable of running highly automated instruments but also of implementing expert rules to allow validation of results and their immediate release. The constitution of the staff profile in the laboratory with total laboratory automation and autovalidation requires examination to match the required knowledge and skill base with the new environment. Scientists need to devote more time to research and as automation increases so should the training requirements to reflect the changes in responsibilities. As we look ahead to the continued development and demand of more specialized techniques using, for instance, liquid chromatography mass spectrometry/mass spectrometry and microarrays for nucleic acids and protein analysis in investigations of gene or genome expression in various diseases software that can interpret such data needs to be developed for expert systems in total laboratory automation.
