Abstract
We describe a novel nanostructured target plate for laser desorption/ionization (LDI) mass spectrometry, NALDI (Bruker Daltonics, Billerica, MA) target plates. The active surface comprises several layers of inorganic materials that are structured at the nanoscale and then further coated with a hydrophobic organic layer that facilitates sample deposition and LDI performance. These targets have been designed to analyze low mass (below 1500 Da), relatively polar, organic molecules and they have been shown to be up to 10 times more sensitive at detecting analytes in this range than conventional Matrix-assisted laser desorption/ionization—Time of Flight (MALDI)-TOF analysis. The targets can be used on standard LDI-TOF mass spectrometers and have been designed to fit the Bruker Flex series of mass spectrometers. This study demonstrates the utility of these targets for analyzing pharmaceutical compounds and demonstrates their superiority over conventional MALDI both in terms of performance and ease of use. Finally, we also demonstrate that these targets can be used in a unique sample preparation and analysis mode by allowing the capture and analysis of analytes from complex biological solutions.
Introduction
Matrix-assisted laser desorption/ionization (MALDI) mass spectrometry is a well-established technology for the analysis of macromolecules such as peptides, proteins, and oligonucleotides. 1,2 It is less commonly used for the analysis of small compounds such as pharmaceuticals due to the matrix background below 700 Da, which causes a loss of sensitivity. However, the potential for high throughput analyses and the sensitivity and ease of use of this technique have generated interest in its applicability for pharmaceutical analysis. 3 Various approaches have been developed to allow direct desorption/ionization from surfaces, without the addition of chemical matrices, with varying degrees of success. 4 -9 Most previous attempts to create surfaces that allow active ionization/desorption without the requirement of an exogenously added chemical matrix have proved problematic due to poor sensitivity and stability issues. Here, we describe a novel nanostructured surface that provides a highly sensitive, stable, and robust substrate for matrix-free laser desorption/ionization (LDI) mass spectrometry—NALDI targets. These substrates are easier to use because there is no requirement to select an appropriate organic matrix and premix samples prior to analysis. In addition, NALDI target plates (Bruker Daltonics, Billerica, MA) have improved sensitivity over conventional matrix-based analyses on stainless steel plates and they provide a unique functionality; the ability to detect and quantify analytes in complex biological solutions without any upstream sample preparation.
Daniels, Ph.D.

In this study, we compare the detection limits of NALDI targets compared to conventional matrix-based analysis on stainless steel targets for the detection of pharmaceutical compounds. Additionally, we demonstrate that these target surfaces can be used in a unique way that dramatically simplifies sample preparation by facilitating the direct extraction and subsequent analysis of small hydrophobic molecules from complex biological fluids such as urine.
Materials and Methods
NALDI target plates (Bruker Daltonics, Billerica, MA) are available in a ¼-microtiter plate format designed to fit in Bruker Daltonics Flex series of mass spectrometers. The targets comprise a hydrophobic, nanostructured surface coating and they are laser marked to provide identifiable locations for sample deposition (Fig. 1). As the figure shows the surface is comprised of densely packed inorganic nanostructures that are approximately 20 nm in diameter and from 100 to 500 nm in length. We have optimized the morphology and aspect ratio of the structures to provide optimal LDI performance. 9,10 During our development of this surface, it was determined that nanostructures with a diameter of 10–20 nm appeared to be the most effective structure for LDI performance. The other important parameter governing LDI performance is the density of the structures. The current NALDI surface is much more dense than earlier versions with a nanostructure density of > 100/square micron. The figure also demonstrates the hydrophobic nature of this surface. The final layer of the structure comprises a hydrophobic organic layer that allows dense application of droplets, optimizes LDI performance, and provides the novel capability of direct surface adsorption described in more detail below. We have also evaluated the effect of the thickness of this organic layer on LDI performance and we have deposited layers varying in depth from 5 to 40 nm with no significant difference in the final performance of the surface (unpublished data).

NALDI target plates. (A) Increasing magnification of the NALDI target surface. As the scanning electron micrographs show the surface is comprised of densely packed inorganic nanostructures. (B) The application of a final hydrophobic organic layer results in a surface optimized for both performance and for density of droplet application.
To use these targets, analytes in solution can be directly pipetted onto the plate surface without mixing with an exogenous chemical matrix. Spotted or extracted samples do not leave characteristic inhomogeneous dried crystal spots but instead appear clear under normal observation.
To evaluate and compare performance of the NALDI target plates, small molecules (clonidine, propranolol, quinidine, papaverine, verapamil, ketoconazole etc.) were obtained from Sigma (St. Louis, MD). Serial dilutions were made in water/methanol or water/acetonitrile from 100 down to 0.1 pg/μL or lower. Typically, 1.0–1.5-μL spots of diluted analytes in various media were applied to the NALDI plate surface without any organic matrix and either allowed to dry or placed in a humidified chamber to allow passive adsorption of analytes from the spotted solution onto the plate surface. Control samples were prepared with α-cyano-4-hydroxycinnamic acid using polished steel targets. MS and MS/MS spectra were acquired on a Bruker Daltonics Ultraflex II or Ultraflex III TOF/TOF mass spectrometer.
Results
Superior Performance
Figure 2 shows a dilution series of quinidine (m/z 325.191) analyzed either by NALDI or by using a conventional matrix α-cyano-4-hydroxycinnamic acid (CHCA) on a standard stainless steel target. The data are characteristic of the results obtained with a number of other small molecules analyzed in this way ranging from m/z ∼ 180 to m/z ∼ 530. As the figure shows the detection limit on NALDI (approx. 150 amol) is approximately 10-fold lower than using conventional MALDI (approx. 1.5 fmol). In addition to the improved performance, the preparation and deposition of samples for NALDI targets is much more straightforward. Samples in solvent (e.g., acetonitrile/water) can be directly deposited on the plate surface, without the need to mix with an excess of the chemical matrix before deposition and without concern over the crystallization of matrix upon solvent evaporation. After solvent evaporation on NALDI targets, a transparent, homogenous layer of dried sample remains and any part of the region of deposition can be interrogated unlike MALDI samples where “hunting and pecking” for effective crystals is required for optimal results (see below and Fig. 5). In addition to improved detection limits and ease of use, NALDI targets tolerate higher concentration of salt in sample solutions than standard MALDI preparations. Several compounds including clonidine, propranolol, and ketoconazole were detected in 150 mM sodium chloride at the 10 pg/μL level (data not shown).
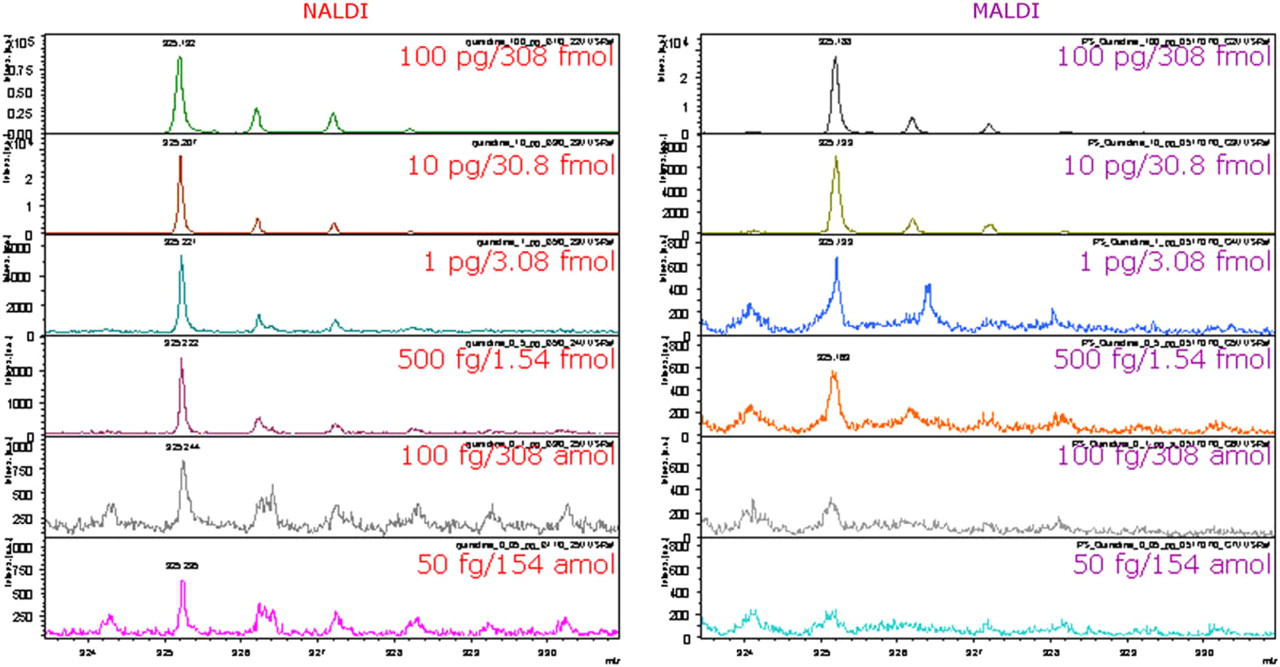
Dilution series of quinidine (m/z 325.191) is characteristic of results obtained with number of small molecules. Detection limit of quinidine on NALDI target (left spectra) is 50 fg (154 amol) compared to 500 fg (1.54 fmol) on polished steel target using conventional matrix-assisted laser desorption/ionization preparation (right spectra).
Figure 3 shows that improved performance relative to MALDI is also achieved when more complex mixtures of analytes are used. Comparison of dilution series of mixture of four compounds (propranolol, quinidine, papaverine, and verapamil) on NALDI target plates and conventional stainless steel plates (with added matrix) shows similar detection limits of 50 fg for verapamil (110 amol). However, signal abundance of verapamil is three times higher on NALDI targets. Detection limit of papaverine is two times lower on NALDI targets than on polished steel targets (Fig. 3). Two additional components of the mixture (propranolol and quinidine) were detected at 500 fg on the nanostructured targets and with the conventional preparation on polished steel targets but signal abundance of quinidine was four times higher and signal abundance of propranolol two times higher on the NALDI targets. In addition, MS spectra acquired from NALDI targets have significantly less background signals and are easier to interpret.
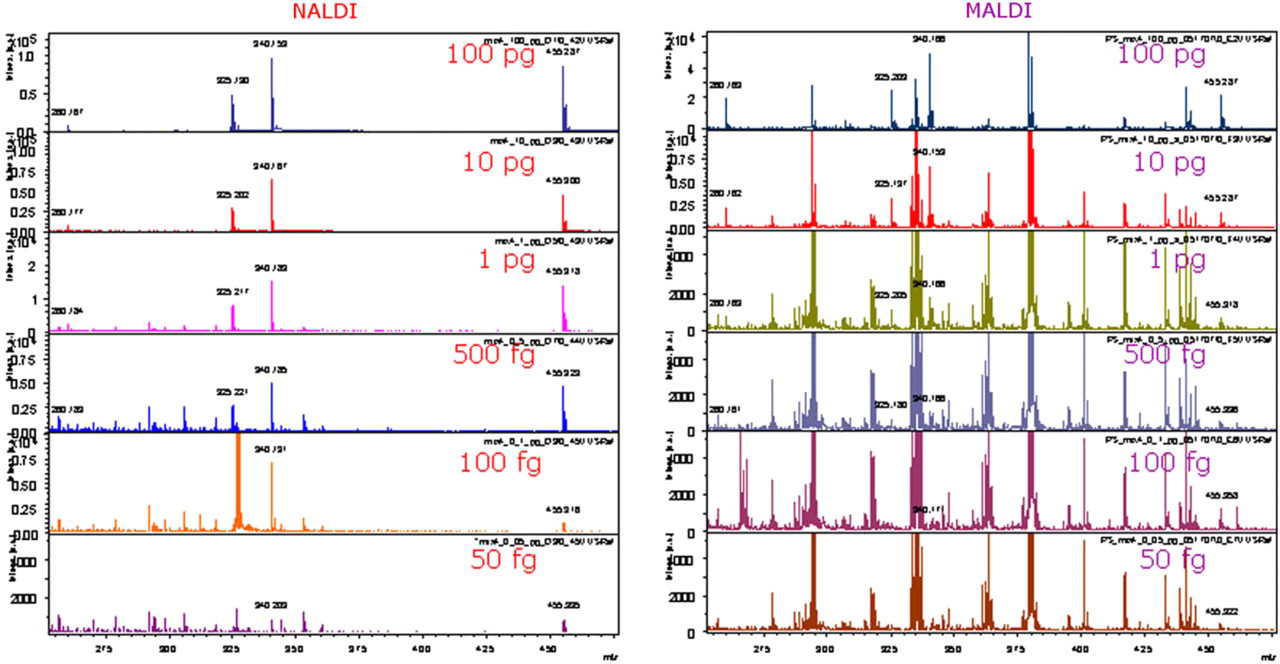
Laser desorption/ionization spectra of mixture of four pharmaceuticals obtained from NALDI target (left) are significantly different from matrix-assisted laser desorption/ionization (MALDI) spectra (right). The relevant molecules are notated by mass values shown next to each peak (propranolol, quinidine, papaverine, and verapamil). Note the ease of distinguishing the labeled peaks in the NALDI spectrum from the same set of molecules analyzed using MALDI. The reason for this difference is that the level of chemical noise is substantially lower when NALDI is used (because no chemical matrix is required) and spectra are easier to interpret because there are no matrix adduct signals.
MS/MS Mode
The improved detection limits seen in reflectron mode when comparing NALDI to MALDI are also improved in MS/MS mode. As Figure 4 shows the detection limit for verapamil in MS/MS mode is 110 amol on a NALDI target compared with 1.1 fmol using conventional stainless steel targets and CHCA matrix. At the 1.1 fmol level, it can also be seen that parent ion signal on the NALDI target is approximately twice as abundant as the signal on the steel target. Characteristic fragment ions at m/z 165 and 303 dominate the MS/MS spectra acquired on the NALDI target down to the 110-fmol level. These fragments are less abundant in spectra acquired using conventional MALDI at the 2.2-fmol level and lower. In MS/MS spectra, it is very important to have a fragmentation pattern that can be easily interpreted and structure specific fragment ions that give intense signal. Thus, in Figure 4 we have demonstrated that MS/MS spectra of verapamil obtained from NALDI targets have an easily recognizable pattern of structure specific fragment ions at m/z 165 and 303 that dominate all six spectra from 220- to the 110-amol level. In contrast, fragment ions at m/z 165 and 303 are significantly less abundant in spectra obtained from steel target using conventional MALDI preparation at 2.2- and 1.1-fmol level. A likely explanation for this effect is that there is more chemical noise in the parent ion selection window in conventional MALDI spectra at low levels which results in some abundant, but unassigned, fragment ions suppressing the signal of the fragment ions at m/z 165 and 303. This explanation is supported by a detailed analysis of the parent ion selection window for verapamil (455.290 ± 3 Da) on both NALDI and MALDI targets (data not shown). In the case of the NALDI spectrum, the chemical noise level is low and thus the detection limit is improved by an order of magnitude (to 50 fg). In the case of the MALDI spectrum, from a polished steel target, chemical noise level is substantially higher with unassigned signals at m/z 454, 457, and 458 and thus 500 fg represents the detection limit using a conventional MALDI preparation for this analyte. We are currently evaluating this benefit of NALDI targets for analyzing small molecules by MS/MS in much more detail and will present the findings in subsequent publications.
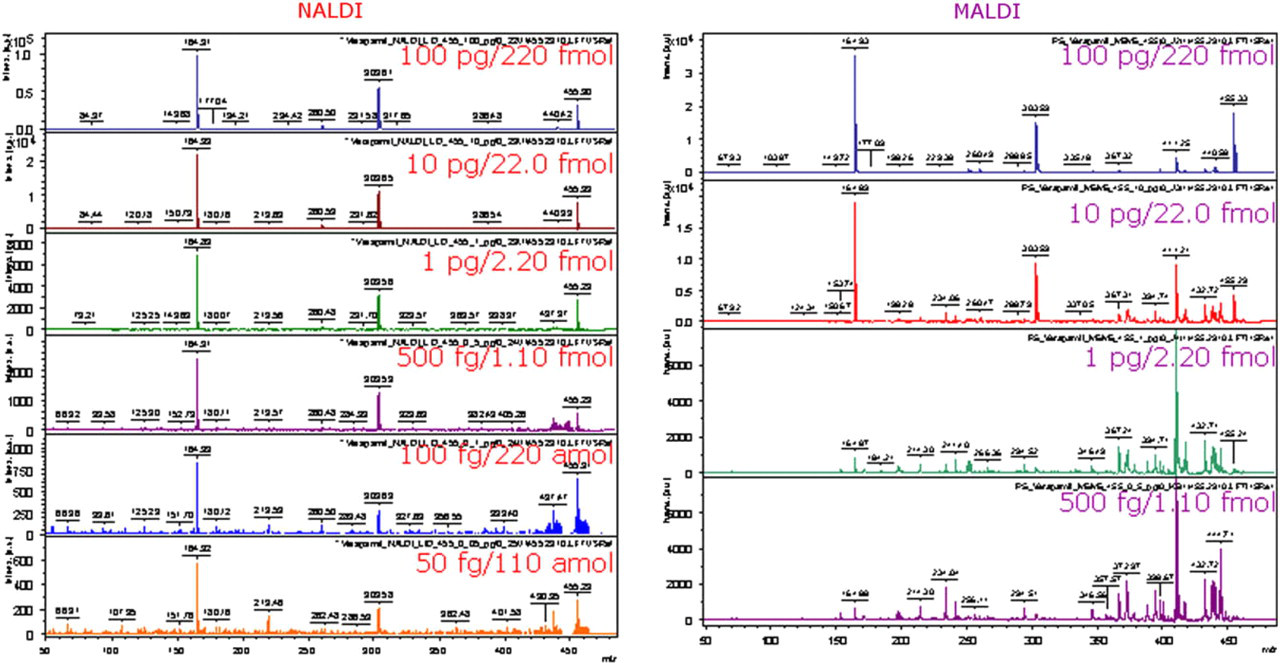
Use of NALDI targets in MS/MS mode. Detection limit of verapamil in MS/MS mode is 50 fg (110 amol) on NALDI target (left) versus 500 fg (1.10 fmol) on steel target using conventional CHCA matrix (right). Note that at the 1 pg level, parent ion signal obtained from NALDI target is significantly more abundant than the signal from steel target.
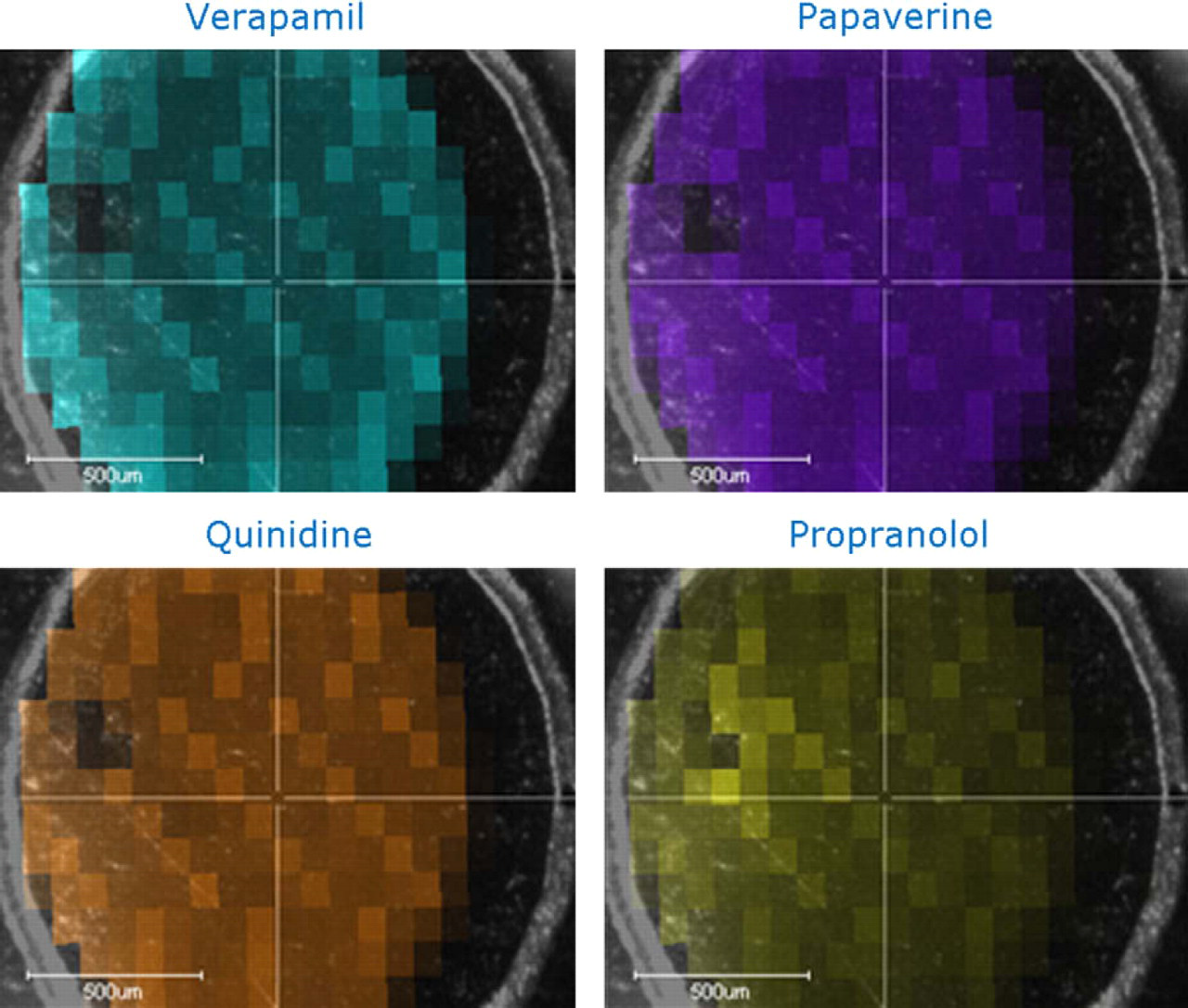
Laser desorption/ionization MS image of a sample spot containing mixture of verapamil, papaverine, quinidine, and propranolol at 10 pg/μL shows even distribution of all four compounds within the area where the 1-μL droplet was deposited. This is represented in this figure by a relative colorimetric intensity scan (relative to the highest intensity signal obtained in the imaging data set) of the zone in which the sample was deposited. The software assigns a different color output for each analyte (specific peak) identified in the deposited mixture. There is no evidence of the analytes spreading outside the deposition region, in fact the limits of spreading of the deposited 1-μL droplet within the marked deposition zone are clearly delineated (i.e., the 1-μL droplet was smaller in size than the diameter of the etched circle on the NALDI target).
Homogenous Distribution
Another clear advantage of NALDI over MALDI is that the samples are homogenously distributed over the region of deposition rather than concentrated in crystalline “hot spots” that need to be manually selected. This will facilitate the automated analysis of samples that have been applied to the target plates and will also improve the speed of analysis. To demonstrate that the applied sample is actually distributed evenly over the deposition zone, we used the FlexImaging 2.0 software package (Bruker Daltonics, Billerica, MA). The image has 80-μm spatial resolution, which required automated acquisition of 225 spectra with 100 laser shots per spectrum from a single spot on a NALDI target. Figure 5 shows the results of this study using a deposited mixture of four compounds. It is clear that the analytes were evenly distributed and there is no evidence that they spread outside the area of deposition. This facilitates automated analysis of samples deposited onto the targets.
Novel Capabilities
The multilayered, nanostructured surface allows passive adsorption of small hydrophobic analytes from complex biological solutions onto the surface of the plate without any prior sample clean-up steps. This opens up a whole new range of application possibilities for NALDI targets because it has the potential to allow rapid analysis of small molecules directly from untreated samples.
To achieve extraction of low-mass organic compounds from complex biological solutions, a droplet of solution is placed directly onto the surface of the NALDI target and incubated in a humidified environment to prevent droplet evaporation (Fig. 6). Droplets, as small as 0.5 μL, can be applied to the surface at high densities and without cross contamination (due to its hydrophobic nature—see Fig. 1B). After an appropriate amount of time, the droplet can be washed from the surface removing the contaminating salts and nonadsorbed species and the target plate is dried. The extracted molecules can then be directly analyzed without any further processing of the target plate.
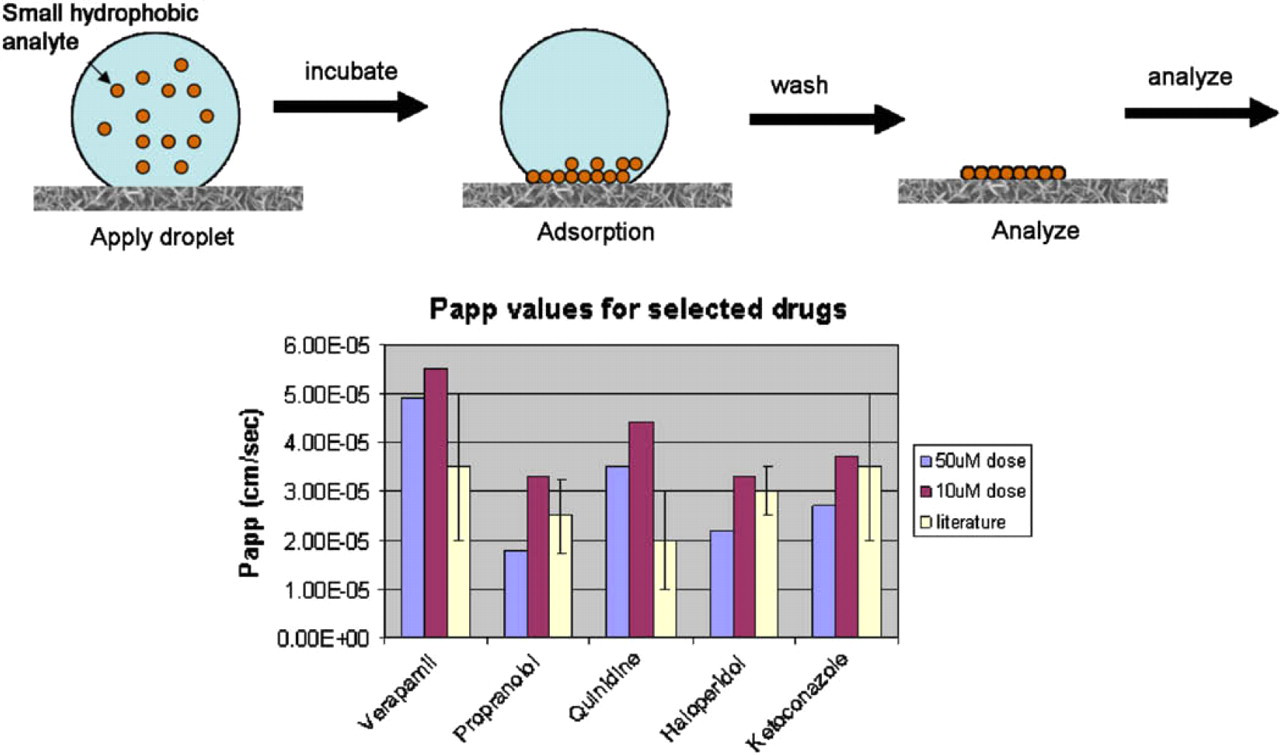
Schematic representation of the passive adsorption of small hydrophobic molecules deposited as a 1-μL droplet and allowed to incubate for 20 min before removal of the unbound material in the droplet. This can be applied to the direct analysis of apical and basolateral solutions of transport buffer directly from a caco-2 transport assay plate. The histogram on the right side shows that caco-2 assays analyzed in this way give data comparable to gold standard literature values. Quantitation was achieved by including an internal standard in all analyses.
We have evaluated a number of different biological fluids of varied complexity including serum, tissue culture media, complex assay buffers, and urine. As Figure 7 shows we can monitor the adsorption of molecules over time by washing the droplets from the target surface at defined time points. By carrying out this analysis we determined that, for the molecules evaluated, extraction onto the surface was detectable after about 1 min and plateaued at approximately 20 min (Fig. 7).
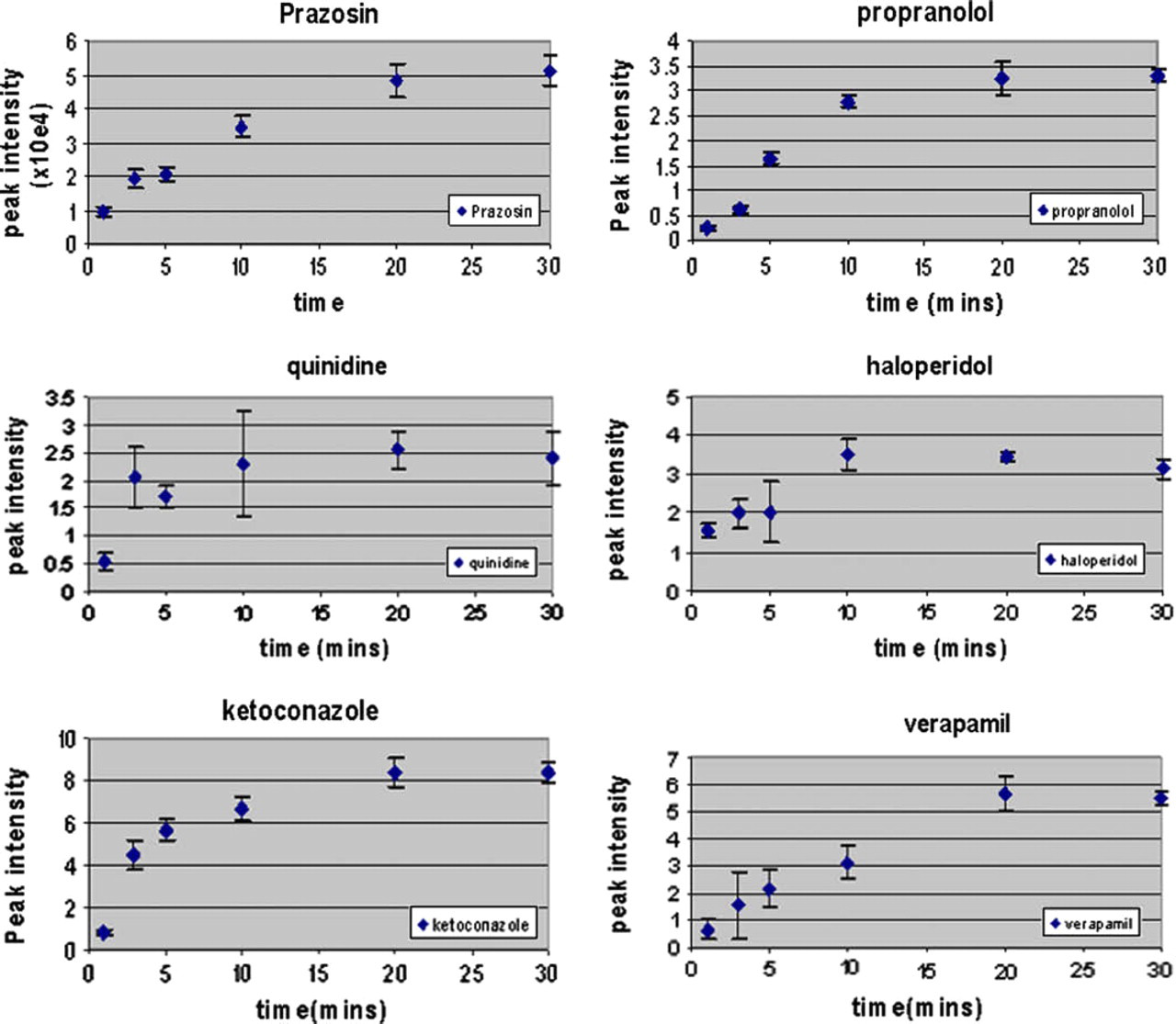
Determining extraction rates. Drugs from a diverse small molecule panel were diluted to 10 μM in medium and then 1 μL was applied directly to the NALDI plate surface for various times before the removal of the droplet and washing in water. The figures below show the time course data for the extracted compounds. The x-axes represent the time of incubation and the y-axes represent the signal intensity obtained at the appropriate m/z ratio for each analyte.
This unique capability of our nanostructured target plates is the ability to reduce upstream sample processing steps and thus reduce the cost and increase the throughput of mass spectrometric analysis of analytes in complex media. To demonstrate this capability, we used a small molecule panel that was representative of structurally diverse pharmaceuticals and diluted these molecules directly into human urine. 1 μL of the “spiked” urine was then deposited directly onto the plate surface and incubated in a humidified chamber for 20 min. After this time, the plate was washed and analyzed as described above. As shown in Figure 8, all molecules were detected at a concentration of 100 ng/mL and four out of five were detected at 10 ng/mL. In addition to human urine, we have analyzed analytes diluted into various biological solutions including tissue culture media, microsomal assay buffer, caco-2 transfer buffer (Fig. 6), and diluted plasma with similar results. These experiments indicate that the use of matrix-free, nanostructured target plates could streamline the sample preparation and analysis of small molecules in early stage in vitro absorption, distribution, metabolism, excretion, and toxicity workflows or high-throughput diagnostic applications.
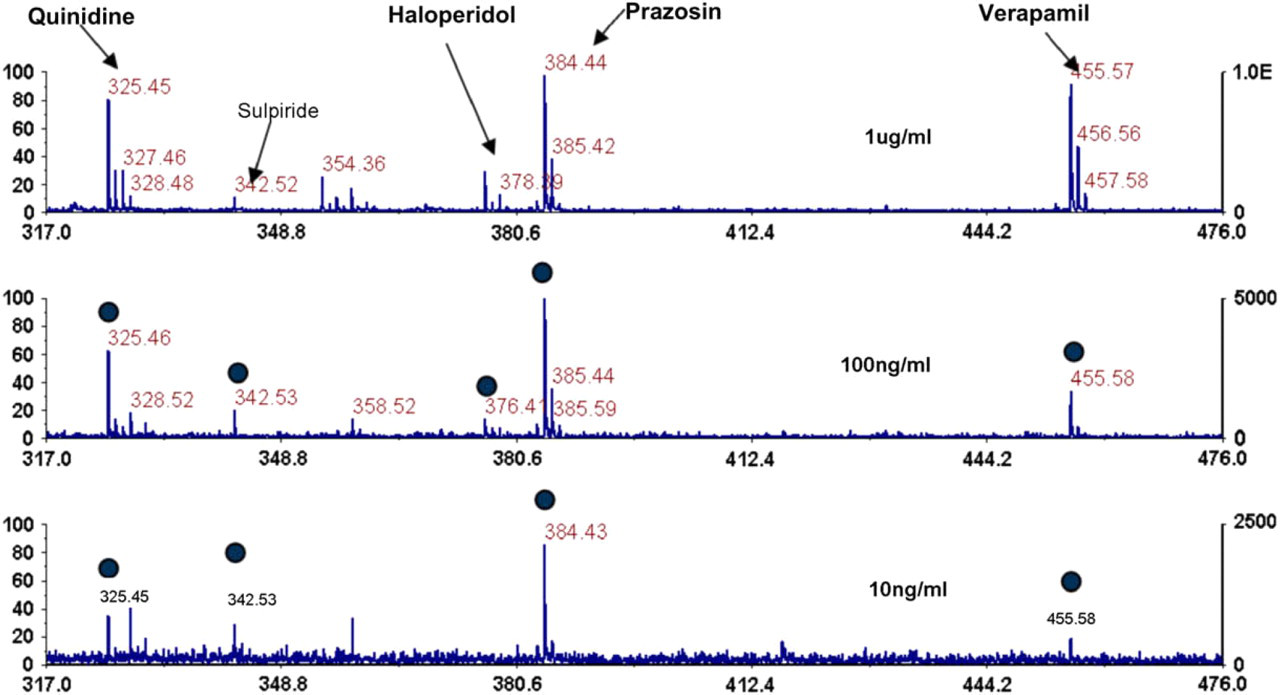
Direct detection of drugs from urine. Five drugs were spiked at various concentrations directly into human urine. One microliter of urine was deposited onto the plate surface, incubated for 20 min, washed clean, and then the plates were directly analyzed. As the figure shows all five drugs are still detected at 100 ng/mL and four out of five at 10 ng/mL.
Conclusions
Analysis of low molecular weight pharmaceuticals with NALDI target plates provided higher quality spectra with improved limits of detection over conventional MALDI analysis. Typically, detection limits were 2–10-fold better with NALDI targets than when using conventional MALDI. Even in cases where signal abundance were equivalent, NALDI targets demonstrate improved performance due to significantly higher signal-to-noise ratios as a result of reduced background interference. At low levels of analyte (1 pg and below), characteristic fragment ions in MS/MS spectra were significantly more abundant when compared with conventional steel targets and chemical matrices.
In addition to the enhanced performance of NALDI targets, the nanostructured surfaces provide a sample preparation and an active LDI–MS surface for one step analyte extraction and analysis. This functionality works best for small hydrophobic organic compounds characteristic of pharmaceutical compounds. Extraction of the analytes of interest occurs in less than 30 min and the process requires no specialized sample preparation or extraction steps before on-plate analysis.
Therefore, the improved analytical performance and novel sample extraction capabilities offer a unique method for streamlining mass spectrometry-based analyses of small molecules in drug discovery and diagnostic applications. As NALDI target are evaluated in various application areas, we would predict that novel applications will emerge as a result of applying this nanostructured surface coating to mass spectrometric analyses.
