Abstract
Most of the medicinal chemistry compounds received from other Merck Research Laboratories (MRL) for evaluation in our in vitro counterscreenings are in pure DMSO solution. This solvent has shown to be a key interference factor in the radiometric evaluation of cytochrome P-450 (CYP) inhibition. To avoid this problem, we have been using a ternary solvent solution that keeps DMSO concentration to a minimum during the titration of the compounds.
The equipment used at our laboratory to perform the dose—response titration of the compounds in the CYP assays is PerkinElmer Evolution Precision Pipetting Platform (EP3) robots with limited microliter capabilities using disposable tips. The need for a nanoliter dispensing technique that keeps the percentage of DMSO at levels that do not affect the assay, and the need to use pure DMSO solutions at high concentrations to avoid solubility issues of the compounds, moved us to design a new automated sample management process for all compounds that were requested to be tested in these assays.
The PocketTips system (nAscent BioSciences Inc., Wellesley Hills, MA) seemed to be a good option for pipetting very low volumes without acquiring new expensive nanoliter liquid handling equipment. Apparent difficulties with implementation were the unavailability of PocketTips for the 96-tips head of the EP3 (PerkinElmer, Waltham, MA) and the need for testing several CYP isoenzymes (up to four) by using the same rack of tips to save run time and reduce costs.
In this article, we discuss the entire process to introduce and validate a new dispensing process for sampling of compounds with multiple nanoliters dispensing at the assay level, indicating in detail how we implemented the use of recycled PocketTips in our PerkinElmer EP3 workstations.
Keywords
Introduction
Cytochrome P-450 (CYP) enzymes catalyze the oxidative metabolism of drugs and xenobiotics. These enzymes belong to a hemoprotein superfamily that has been identified from a wide number of species (ranging from bacteria to higher mammals); CYPs are present in almost every mammalian cell, except in blood and skeletal muscle. Around 2000 different isoforms have been identified and classified by DNA sequencing. 1
During drug discovery, the in vitro CYP inhibition potential of drug candidates is routinely assessed to exclude potent inhibitors from advancing forward in the development process. 2,3 Most of the medicinal chemistry compounds received from other Merck Research Laboratories (MRL) for evaluation in our laboratory for in vitro counterscreenings arrive pure in DMSO solution; however, this solvent is a key interference factor in the radiometric evaluation of CYP inhibition. To avoid interference, we have been using a binary, highly volatile, solvent solution (MeOH/DMSO) (65/35), where the concentration of DMSO is minimized during compound sampling. But with this approach, handling time is a critical factor due to the potential of solvent evaporation and subsequent changes in the titration concentrations.
The use of pure DMSO solutions at all compound management concentrations to avoid solubility issues, and the need for a nanoliter dispensing technique where the percentage of DMSO is maintained at levels that do not affect the assay, motivated us to design a new automated sample management process for all compounds that were requested to be tested in these assays. 4 The equipment available to perform the sampling of the compounds in the CYP assays were two PerkinElmer Evolution P3 (EP3) robots with limited microliter capabilities using disposable tips. 5,6
The PocketTips Technology (nAscent BioSciences Inc., Wellesley Hills, MA) proved to be a good option for pipetting very low volumes without acquiring new expensive nanoliter liquid handling equipment. 7 It is based on typical disposable pipette tips, with the addition of a ‘pocket,’ an open capillary molded onto the inside surface of the tip that loads and transfers precise nanoliter amounts of solutions (Fig. 1). The ‘pocket’ permits transfers of 50, 100, 250, and 500 nL of compounds dissolved in DMSO and delivers these low volumes into the assay plates. The tips are made using natural virgin polypropylene, highly polished to provide crisp edges, and ultra-smooth finishing that eliminates adherence and maximizes the recovery of all fluids; moreover, nAscent BioSciences Inc. can design custom-made pocket sizes for specific customer needs.

Pocket detail of 100 nL PocketTips loaded with tartrazine dye.
The general methodology of PocketTips Technology includes several steps. Firstly, the compound in solution is aspirated from the source plate, passing over the pocket, which is then filled by capillary action. The remaining solution is dispensed back into the source plate. The tips are then cleaned below the pocket by using a common tip washer with deionized (DI) water. The process is concluded by aspirating buffer solution from the assay plate, and mixing 2–;3 cycles within PocketTips to release the compound from the pocket and dispense it into the assay plate. 8
This system neither clogs nor presents carryover, while at the same time it prevents precipitation, eliminates dilution steps, and saves reagents and compounds by attaining the nanoliter range of dispensing. 8,9 Initial implementation difficulties were the unavailability of a PocketTips Technology for the 96-tips head of the EP3 combined by our need to assay several CYP isoenzymes by using the same rack of tips to save on run time and reduce costs.
Our objectives for the project were as follows: (1) to avoid highly volatile solvents for the dosing of the compounds by using pure DMSO and maintaining the quantity of DMSO in the assay within the tolerance limits; (2) to determine the reproducibility, accuracy, and precision of the new EP3 PocketTips Technology; and (3) to design a robust EP3 method using this system in the CYP assays.
Materials and Methods
PerkinElmer Evolution Precision Pipetting Platform (EP3)
Liquid handler workstation with a 96-channel dispense module (Fig. 2). Technical characteristics

Evolution P3 Robotic Liquid Handler with two PlateStak.
Pipetting volume range of 5–;235 μL using P235 tips Accuracy ≤ 1% CV across 96 wells and ≤ 1%, plate to plate, when dispensing 50 μL of water or DMSO into liquid using P235 tips Two PlateStak plate storage modules and a 96/384 Tip Wash Station with automated recirculation system
WinPREP Software Version 2.6.0.21 with Microsoft Visual Basic 6.3
This Visual Basic Graphic User Interface (GUI) allows building complex and versatile methods due to the direct and powerful communication between the software tool and the machine. By using integrated Visual Basic for Applications (VBA), it was possible to create a user-friendly GUI.
Dye Solutions for Volume Measurements
Dilution series of tartrazine in DMSO were prepared from stock solutions at ranges from 8 to 15 mg/mL. A TECAN Ultra Evolution (Tecan Group Ltd., Männedorf, Switzerland) reader was used with a 405-nm filter to determine the optimal concentration of the tartrazine in pure DMSO solution spanning the desired absorbance range. We were able to determine the concentrations of tartrazine in DMSO to be perfectly linear around 10 and 13 mg/mL.
Standard Compounds in DMSO Solutions Plates to be Tested
These compounds were previously prepared in ABGene deep well (ABgene products, US Thermo Fisher Scientific, Rockford, IL) source plates containing two copies of eight different standards, each one titrated at assay concentrations between 100 and 0.1 μM.
nAscent PocketTips [100 nL and 250 nL]
Initially the PocketTips Technology was not available for the 96-tips head of the EP3 platform. This issue required an interactive collaboration between our group and the nAscent design support staff to develop the right pocket for this specific liquid handler system.
Other General Labware
Corning polystyrene 96 flat-bottom microplates (Cat# 3590) (Corning Inco, Corning, NY) were used for the quality control (QC) method. Corning polypropylene 96-well round-bottom microplates (Cat# 3365) were used as the CYP final assay plate, and the ABGene polypropylene 0.8-mL storage–;deep-well plates (Cat# 0765) were used as the compound source plate.
Results
To ensure the optimal performance of the PocketTips Technology using the EP3 instrument, two different pipetting methods were developed in WinPREP EP3 software (PerkinElmer, Waltham, MA) as follows: (1) a ‘general pipetting method’ with 100 and 250 nL PocketTips and (2) a ‘CYP assay method’ with 250 nL PocketTips.
Pipetting Method with 100 nL [and 250 nL] PocketTips
Using the WinPREP software, a unique method for testing PocketTips with two different volumes was implemented (Fig. 3). The accuracy and precision of this method was evaluated using tartrazine dye. This QC method was created with the following pipetting steps
Load Pocket with tartrazine solution at 10 μL/s
Aspirate a 7 μL [10 μL] air gap at a 2 mm [1 mm] height above the bottom of the destination plate well Aspirate 17 μL of tartrazine in DMSO solution (Fig. 4) Dispense an air gap of 2 μL [5 μL] at 3 mm [1.5 mm] height above the well bottom Dispense back 17 μL of tartrazine solution (Fig. 5). The pocket will retain the 100 nL [or 250 nL] desired amount
Rinse Tip below Pocket in Tip Washer (Fig. 6). This protocol is a cycle step to rinse the tips in a Tip Washer unit with DI water below the pocket, to eliminate the residual tartrazine solution
Dispense a 2 μL air gap to empty the tip of any remaining solution Aspirate/dispense 8 μL [7 μL] of DI water in two cycles [three cycles] using the Mix option at 25 μL/s [80 μL/s] Dispense a final 3 μL air gap to empty the tip of residual DI water
Transfer to Plate by using a minimum of 45 μL of DI water based solution or buffer from the destination plate (or plain DI water in the QC procedure). The 100 nL of tartrazine solution loaded into the pocket was transferred into the plate (Fig. 7).
Perform three mix cycles of 15 μL at 25 μL/s [5 μL/s]
Wash Tips cycle to reuse the same Pocket for other possible pipetting processes. This washing step was developed to rinse the tip pocket using DI water in the Tip Washing Station
Aspirate an air gap of 20 μL at a height of 20 mm above the well bottom
Perform five washing cycles with 150 μL of DI water

Designed WinPREP EP3 workstation and method for QC method.

Aspirating tartrazine solution to load pocket.

Dispense back the rest of dye solution.

Rinse tips below pocket.

Transfer 100 nL of dye to assay plate.
Validation Absorbance Method for 100 nL [and 250 nL] PocketTips
To validate this method for accuracy, precision, and re-producibility, the following steps were performed
A reference six-point, standard two-fold serial dilution plate with tartrazine was prepared manually (Figs. 8 and 9). This plate was used to determinate a calibration curve to assess the volume accuracy of the PocketTips EP3 method. Plate preparation included the following: (1) use of a Corning Polystyrene 96 flat-bottom microtiter plate (Cat# 3590) as calibration plate, (2) dispensing 75 μL of DI water into every plate well using a RAININ eight multichannel pipette (20–;200 μL) (Rainin Instrument, LLC, Oakland, CA), (3) removing 8 μL from the first column of wells, (4) mix 8 μL of a tartrazine solution with the remaining solution in the first column using a eight multichannel pipette (5–;50 μL), (5) serial manual titration and mixing of 75 μL from the first to the last column of wells by using a eight multichannel pipette (20–;200 μL), and (6) discarding half of the volume of the 12th column. Absorbance of the calibration plate at 405 nm was read in a TECAN Ultra Evolution spectrometer. Tables 1 and 2 summarize the results for the pipetting 100 nL and 250 nL of this calibration curve with PocketTips, respectively.

Tartrazine serial dilution standard curve for 100 nL PocketTips.

Tartrazine serial dilution standard curve for 250 nL PocketTips.
Results of the 100 nL PocketTips
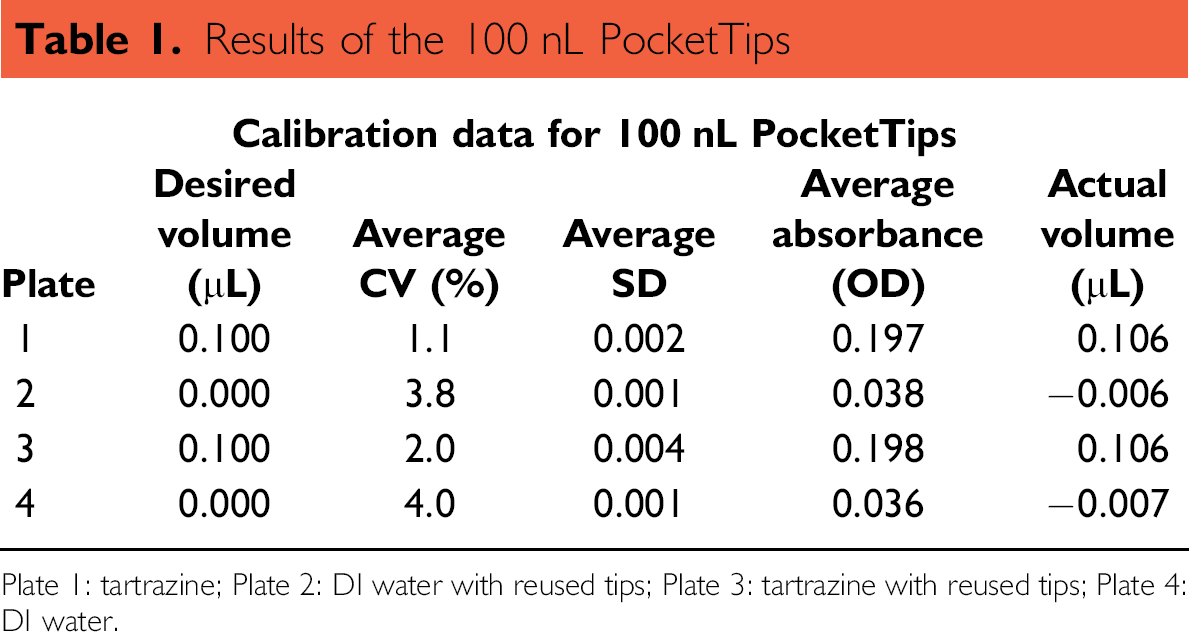
Plate 1: tartrazine; Plate 2: DI water with reused tips; Plate 3: tartrazine with reused tips; Plate 4: DI water.
Results of the 250 nL PocketTips
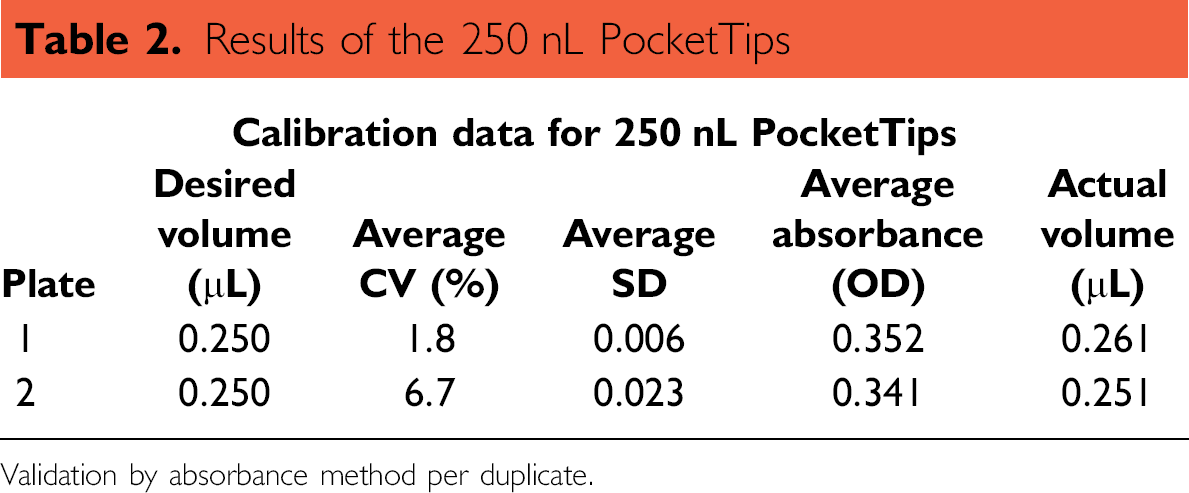
Validation by absorbance method per duplicate.
The multichannel pipettes (20–;200 μL and 5–;50 μL) used to prepare the calibration plate were new pipettes. These pipettes passed RAININ tests performed by qualified technicians in accordance with the ISO 8655–;6 standard with the following results: (1) gravimetric data for multichannel pipettes (20–;200 μL): 20 μL systematic error (accuracy) = 20.10 μL; 20 μL random error (precision) = 0.19 μL; 200 μL systematic error (accuracy) = 200.38 μL; 200 μL random error (precision) = 0.22 μL and (2) Gravimetric data for multichannel pipettes (5–;50 μL): 5 μL systematic error (accuracy) = 5.042 μL; 5 μL random error (precision) = 0.042 μL; 50 μL systematic error (accuracy) = 50.303 μL; 50 μL random error (precision) = 0.069 μL
Transfer 100 nL [250 nL] of 13 mg/mL tartrazine solution in DMSO to the destination plate using the EP3 described method (Tables 1 and 2/Plate 1)
Re-run the EP3 method after the wash tips cycle using DI water instead of tartrazine as the source plate (Tables 1 and 2/Plate 2)
Manually dispense only DI water into a plate (Table 1/Plate 3)
Re-run the EP3 method with tartrazine as the source plate (Table 1/Plate 4)
Read the absorbance of these four plates using a TE-CAN Ultra Evolution
Readings from the 100 nL Plates 1 and 3 were used to calculate the coefficient of variation (CV) between the 96 tips, allowing us to assess the precision of the method. Using the tartrazine calibration curve, the volume accuracy of the 96 tips could also be calculated. The reproducibility of the process was confirmed by comparing the calculated volumes of Plates 1 and 3(0.106 μL both). By comparing the CVs of Plates 2 and 4 (3.8 and 4.0%, respectively), the efficacy of the washing step was demonstrated. Also, the calculated volumes of Plates 2 and 4 (–;0.006 μL and –;0.007 μL, respectively) indicated that potential carryover effects due to tip reuse did not occur.
CYP Assay Dispensing Method with 250 nL PocketTips
To adapt the EP3 method to meet the needs of the CYP assays, two additional steps were included in the same Win-PREP process as follows: one to dispense an internal Control Curve and a second to dispense the control blanks.
To avoid any possible cross contamination between the Control Curve transfer step and the titrated compounds transfer step, this curve with eight different concentrations placed in the first column was dispensed by using a different PocketTips rack 9 (a customized tip box with only eight tips placed in the first column was prepared for this purpose). The rest of the assay process steps, including the addition of 10-μL blank points, were performed using an additional 88 PocketTips rack.
The following sequence outlines the pipetting steps for this assay dispensing method (Fig. 10).
Load the Pocket with the Control Curve with only eight PocketTips at the first column of an empty tip rack:
Aspirate an air gap of 10 μL at 1 mm height above the well bottom Aspirate 17 μL of the control curve compound from the source plate to load the pocket at a speed of 10 μL/s Dispense a 5 μL air gap at a 1.5 mm height above the well bottom Dispense the remaining 17 μL at a speed of 10 μL/s by leaving only the volume of the Pocket full with the 250 nL
Rinse Tips below Pocket in Tip Washing Station by a cycle step to remove any residual compound remaining inside and/or outside the tip below pocket
Dispense a 2 μL air gap in the wash station Aspirate/dispense 7 μL of DI water in three cycles using the mix option at 80 μL/s speed Dispense 3 μL to empty the tip of any solution
Transfer Control Curve to assay to transfer control curve compounds from the pocket to a destination plate with 45 μL DI water
Aspirate/dispense 15 μL of DI water at a 5 μL/s speed in three cycles
Change PocketTips: replace the eight tips used to transfer the CYP control curve with another new set of 88 PocketTips (columns 2–;12) from a new tip box also located in the EP3 workstation.
General pipetting method with 250 nL PocketTips including the full four steps described in the previous section, ‘Pipetting method with 100 nL PocketTips [and 250 nL].’ During these steps, tips in column 12 were not used because any compound in solution was placed at those wells.
Transfer blank control points to assay (placed at the last column of the plate):
Aspirate 10 μL of blank solution at a speed of 10 μL/s Dispense 10 μL of blank solution at a speed of 10 μL/s

Designed WinPREP EP3 workstation and method for CYP sample preparation assay.
Results from CYP Assays
Once the final assay-ready plate was generated by the EP3 liquid handling using the CYP pipetting method described here, the plate was processed in the CYP2C9 isoform that was selected for the validation process. Gemfibrozil, a known commercial inhibitor of this isoform, was chosen as the test standard because of its good correlation in IC50 results compared with previous standard dispensing techniques involving mixtures of MeOH/DMSO (see Fig. 11).

Dose–;response curves for gemfibrozil in the CYP2C9 inhibition assay and calculated IC50s, using the standard pipetting system or PocketTips Technology.
Discussion
The main difficulties encountered during the development of the dispensing methodologies described above could be summarized in the following sections.
Reducing the Excess of Liquid in the Tips
The dispense step was difficult to adjust because we had residual drops residing at the tip ends after dispensing. If we increased the air gap, the compound could be lost in the pocket. We tested different dispensing speeds and heights, but we could not totally dry the tips. Finally, we were able to completely eliminate the drops by both reducing the dispensing speed to the machine's minimum and adding a tip-touch step at the liquid top.
Avoid Empty the Pockets during the Rinse Step
During the rinse step we tried different aspiration heights. We also needed to renew the distilled water in the tip washer and increased to a maximum aspiration height to avoid the mixing of water with the compound in the pocket. For the case where the CYP control curve was dosed with the first column and there was residual water that could not be removed from the rest of the tips, we offered the user the option of changing rack between the Control Curve and the compounds.
Obtaining a Good Mix to Dispense the Pocket
We had previous problems with the speed and height in the transfer to assay step. This step required a strong reduction in the mixing speed and an additional tip-touch at the liquid top.
Reducing the Risk of Cross Contamination
In the validation method we had no problems with cross-contamination, but in the CYPs method we had several problems because of the ‘sticky’ nature of several compounds. In those cases, we decided to use disposable tips by including two PocketTips boxes in the CYPs method, although this additional step would cause increased turnaround time and costs.
We have also piloted another technology for nanoliter dispensing, the GENESIS NPS (NanoPipetting solution) by TECAN. This machine was equipped with VCC (Volume Control Centers) based on XP3000 Plus dilutors, a nanopipetting device (Active Tip M), nanopipetting-specific tubing, and a 50-mL syringe system (1% n-propanol). The system was controlled by Gemini 3.2 software. The Active Tip M range of aspiration was from 17 nL to 50 μL, and had a dispensing range from 0.5 nL (by single droplets of aqueous solution) to 10 mL. Dispensing volumes more than 700 nL required larger excess volumes. This technology proved more expensive than PocketTips Technology because upgrading both, the software and the hardware, was necessary to setup the robots. An advantage of the Active Tip M was that the equipment could be used for variable volume ranges, whereas PocketTips Technology required a different type of tips for each specific volume. 11
Other technologies for nanoliter dispensing such as ECHO550 Acoustic Liquid Handler and Mosquito nanoliter pipette were demonstrated by LABCYTE (Labcyte Inc., Sunnyvale, CA) and TTP LabTech Ltd (TTP LabTech Ltd., Herts, United Kingdom). The ECHO550 Acoustic Liquid Handler was a good option, but it was more expensive than the Mosquito and PocketTips Technologies together. Volume range available to transfer was from 2.5 to 250 nL and the system could determine the water content of DMSO and the depth of fluid in each well. These data would be important for the users because they would be informed about the quantity and quality of samples in the source plates.
The Mosquito was a low-volume liquid handing instrument that combined a positive displacement pipette to ensure zero cross-contamination with a low cost of a disposable tip system. The system permitted pipetting and mixing volumes ranging from 1.2 μL down to 50 nL and tip washing was not possible. Pipettes could aspirate and dispense a variety of viscosities of liquids such as EtOH, H2O, DMSO, and glycerine. The major drawback of this instrument was the tips. Tip blockage was common because the barrel tips or pipettes are in a tape at a horizontal position and change to a vertical position during use. This system was cheaper than the ECHO550 but more expensive than PocketTips Technology.
Conclusions
PocketTips Technology was useful to maintain the DMSO content at the minimum required levels for testing compounds in CYP inhibition assays. PocketTips Technology reduced the work and materials used in compound management steps. The preparation of intermediate dilutions with MeOH, previously necessary to dispense the minimum quantities of the desired compounds, was avoided. This system also provided accurate, precise, and reproducible nanoliter pipetting in an EvolutionEP3 with a 96-tips head and disposable tips. In conclusion, the PocketTips Technology assay dispensing method has been currently implemented in our PerkinElmer Evolution P3 workstations and has proved to be a powerful tool that will be used in other assays at our laboratories.
Acknowledgments
Authors thank Dr. Gerald F. Bills and Dr. David P. Overy from Merck Research Laboratories–;CIBE for the support and help during the preparation of this manuscript.
