Abstract
Nanoscale copolymer membranes that mimic the innate structure and properties of biological lipid membranes possessing hydrophilic and hydrophobic elements to support protein folding were used for a fundamental examination of protein—polymer integration. This study has integrated the neural synaptotagmin II (Syt II) protein, a documented target of the hemagglutinin-33 (Hn-33) protein associated with botulinum neurotoxin type A during the infection process, into polymethyloxazoline—polydimethylsiloxane—polymethyloxazoline nanomembranes. By integrating Syt II into block copolymer membranes, we have developed a neural mimetic membrane toward Hn-33 targeting the applications in nanomaterial-mediated detection. This technology can serve as a robust stand-alone platform for toxin diagnostic studies, or as a coating for integration with micro-/nanofabricated devices and electrodes for protein—protein interaction-based detection. To assess enhanced membrane complexity and toxin specificity, studies assessing the co-insertion of trisialoganglioside-GT1b (GT1b) and Syt II into the nanomembranes were used as a subsequent platform for botulinum neurotoxin type B detection. Protein—membrane integration was confirmed with atomic force microscopy imaging, sodium dodecyl sulfate-polyacrylamide gel electrophoresis, and Langmuir isotherm analysis.
Introduction
Botulinum neurotoxin (BoNT) is an extremely toxic and potent inducer of paralysis due to its evolved mechanisms that preclude neuromuscular communication. 1 Clostridium botulinum in particular is known to cause the food poisoning disease botulism, 2 and a small dosage of 1 ng/kg is lethal to the human body. Due to its extreme toxicity, BoNT acts as a biological hazard to humans and could be a potential biological warfare weapon. By developing a neural membrane that mimics the natural behavior of BoNT, a platform for nanomembrane-mediated diagnostics and related assays can be achieved. Key advantages over conventional lipid systems include increased robustness, 3 as well as tailorable membrane components and lengths. 4 Promise for these technologies has been indicated by various research groups for applications including biosensors, 5 devices for rapid drug screening, 5 devices for drug delivery, 6 and power sources for bioimplants. 7,8 Due to the well-established method and ease of fabrication of the copolymer membrane, its compatibility with standardized biological assays, and capability for optimizing specific processes and applications, this technology sets a basis for the future development of automated laboratory diagnostic-device technologies in studying BoNT and its properties.
Block copolymer membranes can be regarded as biomimetic membranes due to their innate hydrophilic–hydrophobic alternating structure and enhanced robustness; these traits are ideal for potential device-based applications, such as protein biosensors. 4,5,7,9 The enhanced stability and variability of block copolymer aggregates in polymer–protein hybrid biomimetic membranes show promise in the development of protein biosensors in biomedical diagnostics. 10 In addition, the hydrophilic–hydrophobic properties of block copolymer biomimetic membranes allow the potential ability to detect changes in a particular biological environment.
C. botulinum produces BoNT cluster genes that consist of genes for BoNT, neurotoxin-associated proteins (NAPs), and a regulatory gene botR. It has been demonstrated that BoNT attaches to nerve membranes that bind to trisialoganglioside-GT1b and presynaptic membrane protein receptors. synaptotagmin II (Syt II), a neural protein, has been shown to be a protein receptor for botulinum neurotoxin type A (BoNT/A). 11 In particular, Hn-33, a 33-kDa component of BoNT/A NAPs, has been demonstrated to bind with Syt II. 11 Therefore, the protein receptor Syt II can serve as a membrane-embedded element for Hn-33 detection, and therefore subsequently the identification of BoNT. The binding of Hn-33 and Syt II sets the framework for the design and development of hybrid protein–polymer membranes and devices, which specifically and effectively target BoNT.
By taking advantage of the binding characteristics between Hn-33 and Syt II, functionalized neural mimetic membranes consisting of block copolymer polymethyloxazo line–polydimethylsiloxane–polymethyloxazoline (PMOXA–PDMS–PMOXA) for potential applications in nano-membrane-mediated diagnostics were fabricated using Langmuir–Blodgett (LB) film deposition techniques. 12 To increase toxin specificity, additional studies were conducted with the co-insertion of GT1b and Syt II into the copolymer membranes. GT1b- and Syt II-specific binding to botulinum neurotoxin type B (BoNT/B) has been previously demonstrated, 13,14 which provides an additional targeting element for using protein–polymer hybrid film for detection applications. These membranes were characterized chemically and physically with sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) analysis, atomic force microscopy (AFM), and isotherm measurement graphs.
Materials and Methods
Protein Deposition and Sample Preparation Procedure
Studies have demonstrated the successful fabrication of protein–polymer membranes using LB deposition methods. 15 The KSV 2000 LB system (KSV 2000 Series, KSV Instruments, Standard LB Trough, System 2, Helsinki, Finland) was used for block copolymer and protein deposition as well as for surface pressure and isotherm measurements. Block copolymer with various combinations of Syt II (Syt II, BBTech Inc., BB310, Dartmouth, MA) and trisialoganglioside-GT1b (Ganglioside, Sigma–Aldrich, G3767) were deposited onto glass slides using LB procedures. The deposition process was analyzed with continuous surface pressure and isotherm measurements.
Standard 75 mm × 25 mm × 1 mm glass slides (VMR, West Chester, PA) were sterilized using a solution of 50/50 chloroform and 100% ethyl alcohol. Two sterilized glass slides were clipped to the LB dipping device, and position parameters were chosen based on the number of layers desired. Because three layers are required in this study, the slides are to commence coating beneath the surface of the water at approximately 65 mm below the user-set zero point. To avoid complete immersion in the liquid phase, the zero point was set at approximately 4 mm above the surface. The surface pressure was measured using a platinum Wilhelmy plate sensor (KSV 2000 Series, KSV Instruments, Standard LB Trough accessory, Helsinki, Finland) in direct contact with the surface of the liquid. Approximately 15 μL of triblock copolymer at a concentration of 10 mg/mL was added to the trough containing nanopure water with a glass syringe (Hamilton Syringe, Reno, NV) above the surface of the water. The amount of copolymer may vary depending on the amount required to reach a trough surface pressure of 30 mN/m. Triblock copolymer was prepared by mixing 20 mg of triblock stock (Polymer Source, Quebec, Canada) in 2 mL of chloroform to create a solution with a concentration of 10 mg/mL. To ensure complete evaporation of chloroform from the triblock, the deposited solution was left to settle in the trough for 30 min.
If proteins were incorporated into the triblock membrane, an additional 30 min was also given to allow for proper binding and protein configuration to stabilize. It is important to note that the proteins were added underneath the surface of the water subphase, by directly injecting the syringe with protein within the water, to allow for controlled protein–triblock integration, as opposed to deposition at the surface where proteins can be affected by environmental contamination. Syt II (80 μL) and GT1b (500 μL) proteins were added by direct syringe injection underneath the subphase. Three layers were deposited steadily at a target surface pressure of 30 mN/m.
Eight samples were prepared for each SDS-PAGE electrophoresis analysis with the following combinations: (1) triblock-only negative controls, (2) triblock with Hn-33 (40 μL at 0.0001 mg/mL), (3) triblock with Syt II (40 μL at 0.1 mg/mL), (4) triblock with Syt II (40 μL at 0.1 mg/mL) and Hn-33 (40 μL at 0.0001 mg/mL), (5) triblock only, (6) triblock with Hn-33 (40 μL at 0.1 mg/mL), (7) triblock with Syt II (40 μL at 0.1 mg/mL), and (8) triblock with Syt II (40 μL at 0.1 mg/mL) and Hn-33 (40 μL at 0.1 mg/mL). Hn-33 of 80 μL and 120 μL were incubated atop triblock and/or triblock–Syt II bases for 1 h at 4 °C to reach concentrations of 0.1 mg/mL and 0.0001 mg/mL, respectively. Increased volumes of Hn-33 were deposited to reach a concentration of 0.0001 mg/mL due to the decreased detection level at this concentration.
Imaging Procedure
AFM (Asylum MFP3D, Santa Barbara, CA) images were obtained to identify the structure and interaction between various proteins and triblock using the following combinations: (1) triblock only, (2) triblock with 0.1 mg/mL of trisialoganglioside-GT1b, (3) triblock with 0.1 mg/mL of Syt II, (4) triblock with 0.1 mg/mL of Hn-33, (5) triblock with 0.1 mg/mL of Syt II and 0.0001 mg/mL of Hn-33, (6) triblock with 0.1 mg/mL of Syt II and 0.1 mg/mL of trisialoganglioside-GT1b. Image dimensions were taken at scan areas of 10 μm × 10 μm. Contact mode imaging at line scan rates of 1.0–0.5 Hz were performed at room temperature with 200-μm length silicon nitride cantilevers (TR800PSA, Olympus, Tokyo, Japan). During analysis, images were subsequently flattened to remove the inherent curvature of glass substrates.
Protein Binding Analysis using SDS-PAGE
Nanomembranes were fabricated within two days before SDS-PAGE analysis. Triblock copolymer with and without Syt II slides were incubated with varying concentrations (0.1, 0.0001 mg/mL) of Type A hemagglutinin-33 (Hn-33, BBTech Inc., BB107) BoNT for 3 h in 4 °C. After incubation, the binding site on the slides were thoroughly washed three times using phosphate-buffered saline (PBS) to remove nonbound proteins within the sample. SDS 2×-loading buffer was then added to the binding area for 10 min to allow for protein incorporation within the loading buffer. After 10 min, the sample was transferred to a 0.5-mL microtube and placed in 37 °C water bath for 15 min.
For SDS-PAGE analysis preparation, a 30% degassed polyacrylamide–bis (BioRad, Hercules, CA) gel was polymerized according to the manufacturer's protocol (BioRad Mini-PROTEAN 3 Cell Instruction Manual). The prepared samples and Precision Plus unstained protein marker (BioRad 161–0363) were injected into the wells of the gel and the cassette was inserted into the assembly system (BioRad, Mini-PROTEAN 3 Cell, 165–3301, 165–3302). This assembly system was connected to a power source (BioRad, PowerPac Universal) of 120 V. The gel was then stained overnight using GelCode Blue Stain Reagent (Pierce 24590, Rockford, IL), destained, and analyzed.
Results and Discussion
SDS-PAGE Analysis
The SDS-PAGE assay is a technique used to separate proteins according to their molecular weight using electrophoresis. With this assay, potential protein–membrane integration between Syt II with triblock and Hn-33 with developed Syt II-functionalized neural mimetic nanomembranes was assessed (Fig. 1).

SDS-PAGE gel examining integration of triblock with Syt II and Hn-33 at various concentrations. Lanes 1–8 correlate with sterilized glass slides and deposited copolymer with proteins. Lanes 1–8 contain the following: (1) triblock, (2) triblock with Hn-33 (40 μL at 0.0001 mg/mL), (3) triblock with Syt II (40 μL at 0.1 mg/mL), (4) triblock with Syt II (40 μL at 0.1 mg/mL) and Hn-33 (40 μL at 0.0001 mg/mL), (5) triblock, (6) triblock with Hn-33 (40 μL at 0.1 mg/mL), (7) triblock with Syt II (40 μL at 0.1 mg/mL), and (8) triblock with Syt II (40 μL at 0.1 mg/mL) and Hn-33 (40 μL at 0.1 mg/mL), respectively. Lanes 6 and 8 produced clear banding close to 33 kDa, the molecular weight of Hn-33. These bands are indicative of Hn-33 presence and provide an indicator for potential Syt II–Hn-33–triblock integration.
SDS-PAGE negative controls (lanes 1 and 5) contained no bands due to the lack of bound or receptor proteins. Triblock with Hn-33 at 0.0001 mg/mL concentration (lane 2) showed no bands due to the lack of experimental sensitivity.
Correspondingly, triblock with Hn-33 at a higher concentration of 0.1 mg/mL (lane 6) showed three clear bands between 25 and 37 kDa, possibly indicative of Hn-33, a protein of molecular weight approximately 33 kDa. The multiple bands can be attributed to protein denaturation occurring between 4 and 37 °C Hn-33-transfer incubation process. Triblock with Syt II at 0.1 mg/mL concentration (lanes 3 and 7) showed no evidence of bands, because Hn-33 had not yet been introduced, explaining the absence of protein interaction. The absence of proteins also signifies the robust integration of Syt II and triblock composites. When Hn-33 was introduced in lane 8, three bands similar in molecular weight to the bands in lane 6 appeared, strongly implying a potential triblock–Syt II–Hn-33 interaction. Interestingly, lane 8 showed three less-dense bands at the same molecular weights than the bands observed in lane 6 despite the same concentration of Hn-33 deposited. This attenuation may be attributed to the reduction of specific adsorption of Hn-33 due to the addition of Syt II and the inert surface properties of the PMOXA group. Lane 4, containing triblock with Syt II and a lower concentration of Hn-33 than that of lane 8, showed no bands, which may have resulted from the concentration of Hn-33 being too low to appear on the gel, similar to the effect seen in lane 2. This study suggests that the capabilities of the Hn-33 protein–polymer hybrids show promise as robust platforms for detection at certain concentrations.
AFM Analysis
The protein–membrane interactions between Syt II, Hn-33, GT1b, and block copolymer were analyzed through AFM analysis. AFM analysis enables the determination of feature sizes of each component, which may be indicative of any combinatory effects between Syt II with triblock copolymer and Hn-33 and GT1b with our Syt II-functionalized nanomembrane. The observed features serve as potential indicators for protein insertion compared with control sample images. Each AFM image was flattened before analysis due to the inherent curvature of glass to standardize the comparison between the height differences of each component.
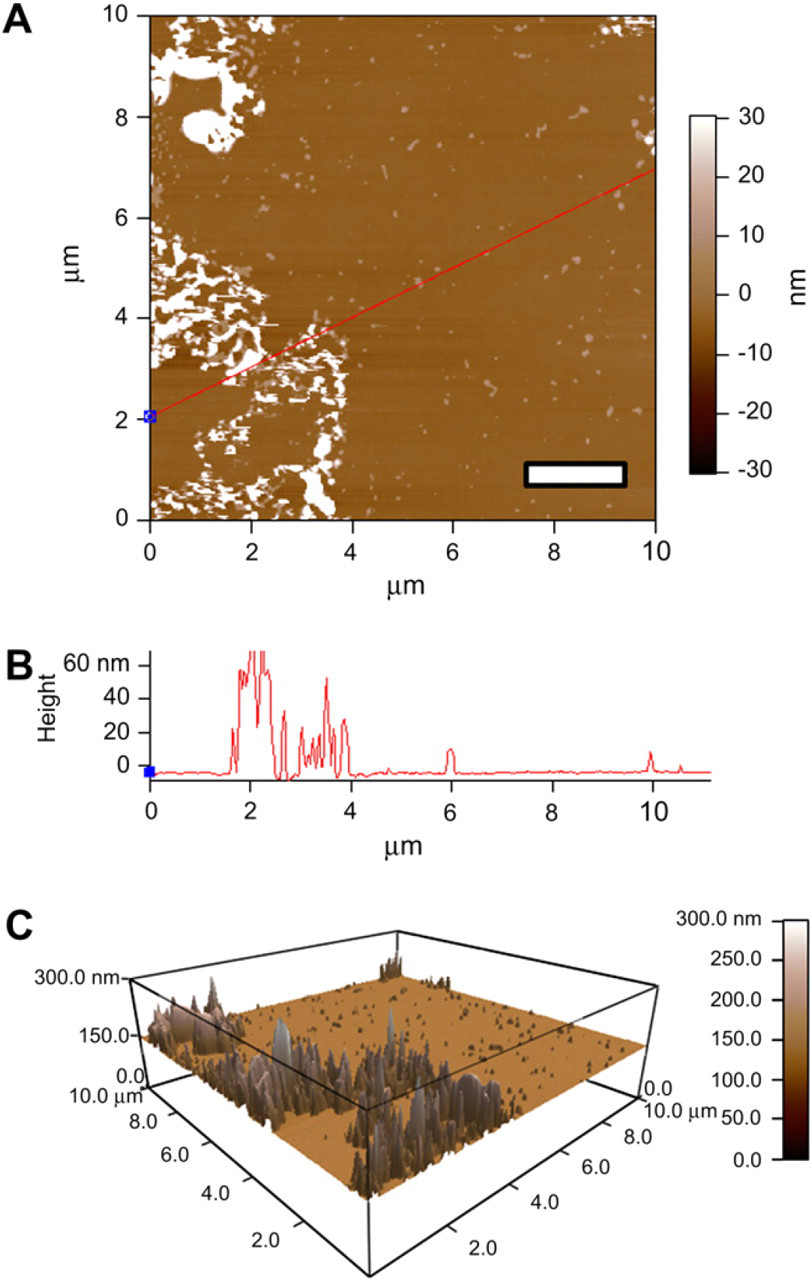
AFM of triblock only. (A) AFM image of a glass slide containing triblock copolymer in varying sizes and states of aggregation: scale bar = 2 μm. (B) The height profile displays the range of heights associated with triblock aggregates (∼60–100 nm). (C) Three-dimensional (3D) representation of the triblock copolymer deposition, correlating aggregation with feature sizes.
Figure 3 reveals the presence of additional nanostructures after the introduction of GT1b to triblock copolymer. GT1b was shown to be in a circular conformation on a scale of –2 to 4 nm in size according to its relative position on the substrate. Interestingly, GT1b introduction appears to indent themselves in the substrate, which explains the “negative” height profile. This engraining characteristic attests to the robustness of integrating GT1b into the nanomembrane.
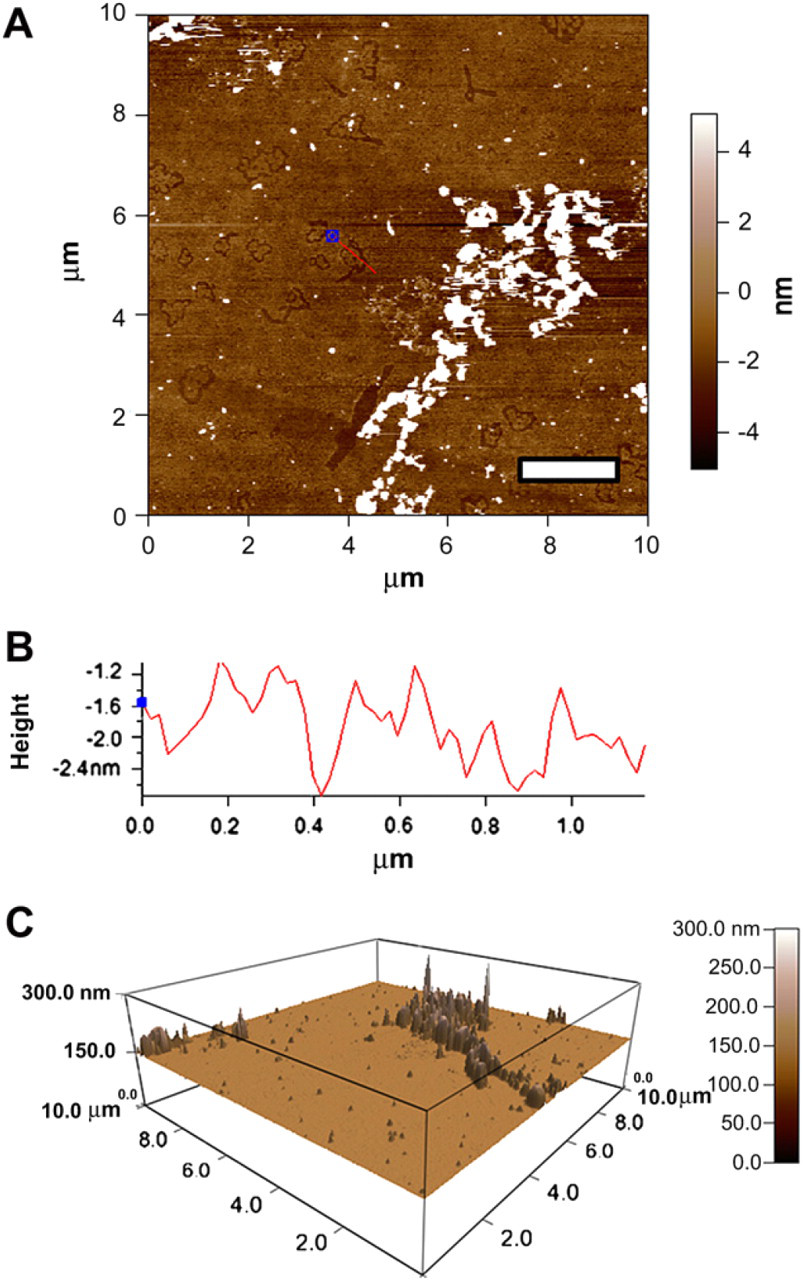
AFM of triblock with trisialoganglioside-GT1b. (A) AFM image of a glass slide containing triblock copolymer and GT1b at 0.1 mg/mL: scale bar = 2 μm. (B) The height profile of the GT1b features indicates an embedded nature, possibly due to a property of GT1b, or physical reaction between triblock and GT1b. GT1b features were measured to be roughly –2 nm in depth. (C). In comparing feature heights, a 3D representation shows the dramatic scale differences between triblock copolymer features and the nonvisible GT1b, at 60–100 nm and –2 to 4 nm, respectively.
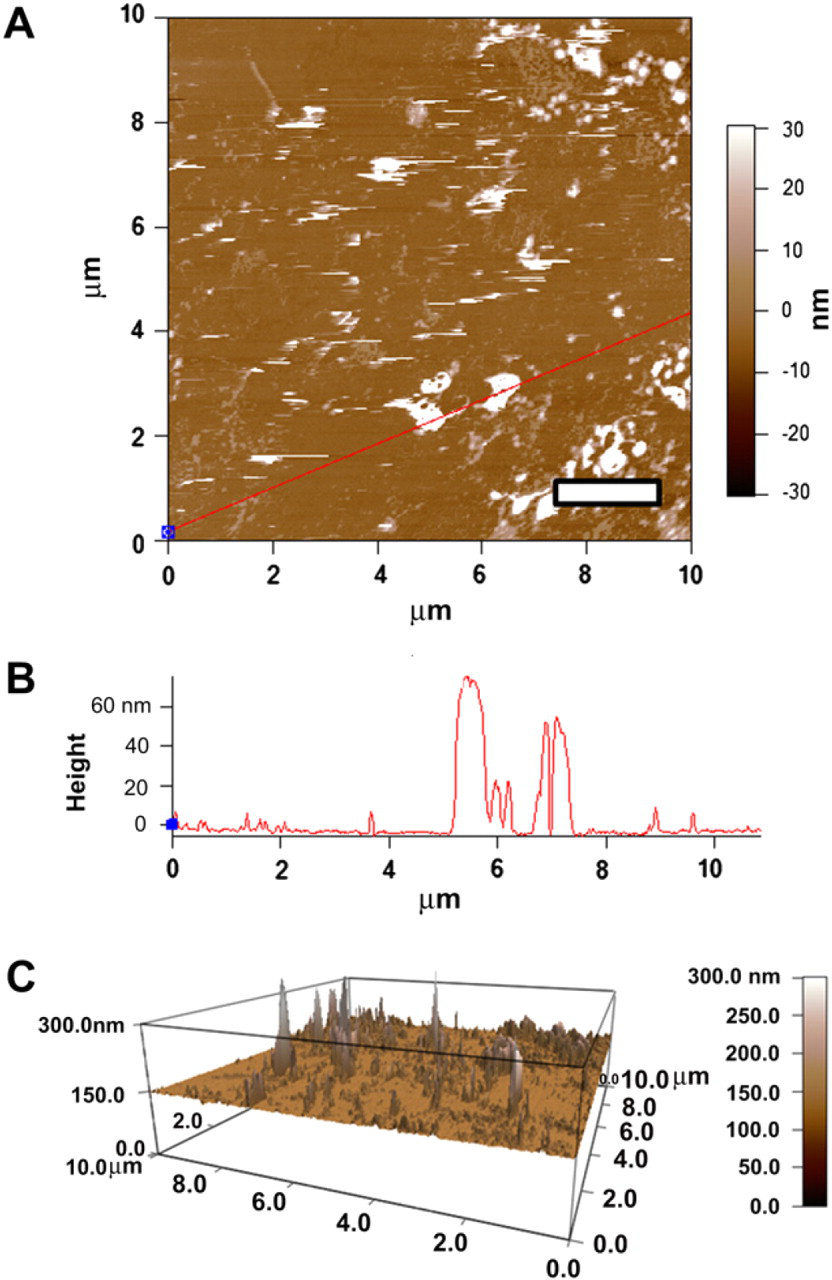
AFM of triblock with Syt II. (A) AFM image of a glass slide containing triblock copolymer and Syt II at 0.1 mg/mL reveals the smattered nature of Syt II against a background of triblock: scale bar = 2 μm. (B) The height profile indicates the triblock with Syt II combination to be on the same order of size as triblock alone (∼60–100 nm). (C) Because triblock features have been shown to be approximately 60–100 nm in feature size, 3D images indicate potential Syt II protein integration at approximately 20–40 nm.
AFM of triblock with Hn-33. (A) AFM image of a glass slide containing triblock copolymer and Hn-33 at 0.1 mg/mL: scale bar = 2 μm. (B) The height profile displays spikes of > 50 nm indicative of triblock, and smaller such features in the range of ∼40 nm, indicative of the Hn-33 features. (C) 3D representation of larger triblock features (∼60–100 nm) and smaller Hn-33 features (∼40 nm).
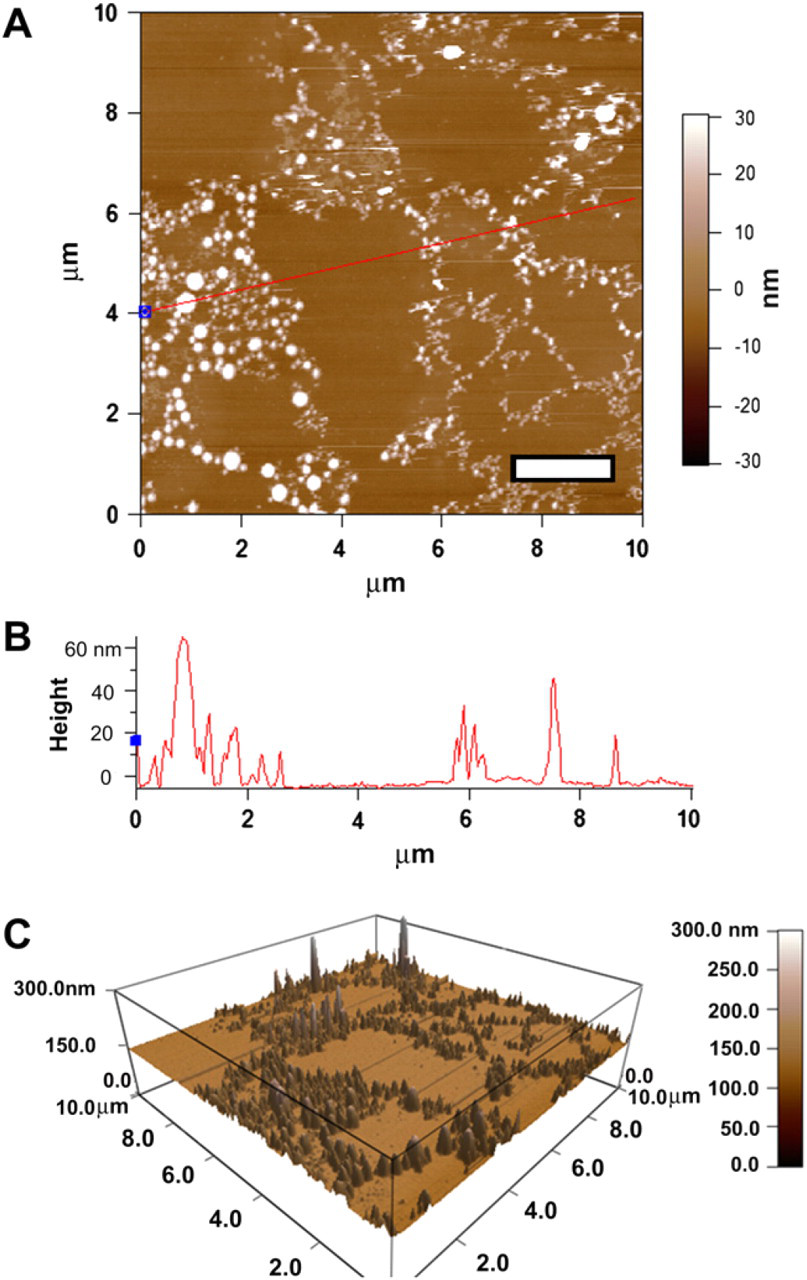
AFM of triblock with Syt II and Hn-33. (A) AFM image of a glass slide containing triblock copolymer and Syt II at 0.1 mg/mL and Hn-33 at 0.0001 mg/mL: scale bar=2 μm. (B) The height profile shows the presence of triblock features (∼60–100 nm), Hn-33 features (∼40nm), and Syt II features (∼20–40 nm). (C) 3D representation of triblock copolymer, Hn-33, and Syt II coexistence on the glass slide, indicative of binding between the three components.
According to AFM images of triblock copolymer with Syt II and GT1b shown in Figure 7, several protein-indicative domains are abundant on the substrate with a background of triblock. GT1b does not appear when Syt II is introduced with triblock and GT1b. Due to the large-scale difference between the smaller GT1b and the larger 20–40-nm Syt II expected feature sizes, it became challenging to resolve both proteins simultaneously. The general arrangement of the proteins is similar to previous scans and further attests to the integration of the aforementioned proteins into the membrane. As previous studies have demonstrated the straightforward approach of subphase-mediated insertion of proteins into copolymeric membranes, the co-insertion of Syt II and GT1b is expected to display highly similar insertion properties. 3,4,20
AFM of triblock with Syt II and trisialoganglioside-GT1b. (A) AFM image of a glass slide containing triblock copolymer and Syt II at 0.1 mg/mL and GT1b at 0.1 mg/mL: scale bar = 2 μm. (B) The height profile shows the presence of triblock (∼60–100 nm), Syt II (∼20–40 nm), and GT1b (∼–2nm). (C) 3D representation of the coexistence of triblock, Syt II, and GT1b, indicative of the binding between these three components.
LB Isotherm Measurements
Isotherm measurements were plotted as surface pressure (mN/m) versus area (cm2) for our four parameters of interest: triblock copolymer only, triblock with GT1b, triblock with Syt II, and triblock with Syt II and GT1b (Fig. 8).
Isotherm measurement graphs plotted as surface pressure (mN/m) versus area (cm2) for triblock only, triblock with trisialoganglioside-GT1b, triblock with Syt II, and triblock with Syt II and trisialoganglioside-GT1b. The increase in substances added to the triblock base shifts curves to the left, indicating an increased duration of time for surface pressure to reach 50 mN/m/solid phase when correlated with a decrease in surface area.
Isotherms were shifted left as more proteins were subsequently added to the triblock copolymer. This leftward shift indicated that it took longer to achieve a surface pressure of approximately 50 mN/m due to the decrease in surface area (cm2). Because GT1b possesses a small surface area (Fig. 3), the isotherm shifted the least amount to the left. The curve representing triblock with Syt II shifted farthest to the left, suggesting a surface area decrease in the trough of approximately 25 cm2. This decreased space caused by Syt II and triblock integration increased the time taken for isotherm completion. As previously explained, the addition of GT1b to triblock did not dramatically alter the surface area, thus minimizing the changes in surface pressure properties during compression. However, once Syt II protein was introduced to the triblock and GT1b, the surface pressure took longer to reach its target. This could be explained by an attenuation effect created by the addition of Syt II to GT1b due to the potential competition between the two elements for interfacial insertion, resulting in a curve which falls between the curve of triblock with Syt II and that of triblock with GT1b. Langmuir film isothermal analysis demonstrated the ability to examine protein–polymer interactions and applying subsequent surface pressure changes as indicators for protein–polymer integration. Variation in surface pressure changes corresponding to decreases in surface area during compression from the addition of GT1b and Syt II demonstrated a delayed progression toward the solid phase serving as evidence for the interaction between GT1b and Syt II with the triblock copolymer nanofilm.
Conclusion
In this study, we developed Syt II-functionalized neural mimetic nanomembranes by integrating PMOXA–PDMS–PMOXA triblock copolymer with Syt II to serve as a protein–polymer membrane platform for nanomaterial-mediated detection applications. We incorporated Hn-33 and GT1b into these nanomembranes to study the interaction fundamental to detecting BoNT. We analyzed protein-membrane integration using multiple analyses methods. AFM and Langmuir isotherm analyses provided imaging and quantitative assessment of protein–polymer interaction and integration coupled with straightforward methodologies for inserting membrane proteins into copolymeric nanofilms. Further analyses using LB isotherm measurements also support the development of protein–polymer membranes for subsequent applications in detection and diagnostics.
Additional studies were conducted to assess the co-insertion of GT1b and Syt II into our nanomembranes as a platform for BoNT/B detection. This concept of GT1b and Syt II binding specifically to BoNT/B has been previously demonstrated and further serves as a foundation for using a protein–polymer hybrid film for detection applications. Specifically, rapid drug screening or general diagnostics could be achieved with an assay of neural mimetic membranes for analysis and detection of drug.
In conclusion, these Syt II-functionalized neural mimetic protein–polymer nanomembranes have the potential to be used as robust stand-alone platforms for toxin diagnostic studies or as a coating for integration with micro-/nanofabricated devices and electrodes for protein–protein interaction-based detection.
