Abstract
AstraZeneca R&D Boston's Compound Management Group (CMG) underwent an extensive workflow redesign project in May 2007. The primary goal of this project was to shift from a solid compound distribution platform to a more robust and efficient liquid compound delivery platform. As a result of the project, there were substantial time savings and efficiency gains in both solid and liquid compound preparation and distribution. Due to the inherent benefits of distributing liquids instead of solid samples, CMG was able to provide assay-ready plates through the use of custom robotics and software tools developed in-house. The implementation of these new platforms drove down upstream processing (solubilization) time by 85%, and allowed internal compound requests to be processed and plated within 24 h. In addition, these compounds are now available for testing at all the AstraZeneca (AZ) sites globally, within one week of compound submission to CMG. Due to the efficiency improvements, CMG was able to not only continue to meet internal customers' demands for solid samples, but also to decrease turnaround time for order delivery, which increases the speed at which a research team can test compounds and subsequently receive project-driving data.
Keywords
Introduction
As one of the world's leading pharmaceutical companies, AstraZeneca's business is focused on turning good ideas into innovative, effective medicines that make a real difference in important areas of healthcare. A major challenge facing pharmaceutical Research and Development (R&D) is the fierce competition, which leads to the urgency of shortening the product development time frame. As a result, any process improvement that can speed up the delivery of a candidate drug is of great benefit to the company. To further this aim, Compound Management Group (CMG) set out to develop an advanced platform for the rapid acquisition of project-driving data that will enable the company to make informed decisions about the future direction of a research project.
Compound Management is a department within R&D Boston whose aim is to curate the potential new medicines (compounds) and to deliver these compounds to research teams on request. Research teams, who are the internal customers of CMG, require compounds in both solid and liquid form. Before the workflow re-design project was started, liquid handling was slow and had an inflexible delivery format. For example, all compounds were delivered in one of only four formats, as it was not possible for CMG to generate assay-ready plates. Also, because compounds were solubilized only once a week, there was often a 7- to 8-day delay between synthesis of a compound and its availability as a liquid. Further, there was no cherry-picking capability; so bioscience departments frequently had no option but to test all the compounds that they received, often wasting time and resources and gathering data on irrelevant compounds. Clearly, a change was needed.
Materials and Methods
The New Solubilization Process
A custom-made high-capacity rack mounted to an integrated shaker mixes the compounds with the appropriate solvent, to the specified concentration. The shaker, shown in Figure 1, replaces the magnetic stir bars previously used for mixing the solutions. The shaker is mounted on a Tecan Evo (Tecan US, Inc., Durham, NC). Using a shaker is a more efficient method of mixing because the vortex generated by the orbit is much stronger and more efficient than stir disks. To ensure that compounds are in solution, the operator removes the rack from the shaker and visually scans the vials through holes in the custom racks. Previous methods required each vial to be removed for inspection, a time-consuming process. This new method significantly decreases processing time, and also increases throughput.

The custom-made high-capacity rack sits on top of the shaker. The shaker, which replaces the magnetic stir discs, creates a stronger and more efficient vortex.
On solubilization, the robot transfers each solubilized sample to four two-dimensional barcoded tubes. Custom software applications determine the amount of compound to be placed in each tube, helping to facilitate downstream processing. The tool also maximizes the amount of compound generated by decreasing the dead volume in the source vial. A conceptual diagram is shown in Figure 2. After the aliquots are prepared, the tubes are uploaded to the database, after which they become available for ordering.
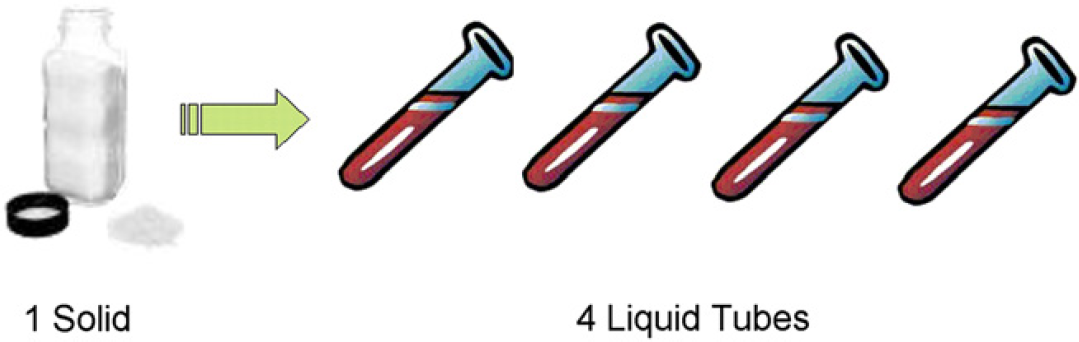
A single solid weighing for each compound is all that is required for the Tecan to make an aliquot of four tubes. Each tube has a unique 2D-barcode, and is uploaded to the database immediately after solubilization. Liquid compound in the tubes is then available for ordering.
Processing Liquid Orders

The major flaws in the previous were identified and corrected in the current system. Flaws and their solutions are marked in yellow boxes, with flaws on the left under ‘previous’, and solutions on the right under ‘current’. One major problem with the previous system was that the huge bolus of weighings for the weekly solubilization run slowed down the whole process. This was resolved in the current system by weighing for solubilization daily.
Through in-house development of software tools for plating compounds, CMG has greatly increased its flexibility with regard to output plate formats. Custom applications allow CMG to readily respond to requests for individualized plate types and formats. New formats and protocols are usually production-ready in less than a week. For the scientist, this upstream processing of compound plates greatly reduces their preparation time and allows for higher capacity runs.
Results
Time to Process Orders Decreases Dramatically
Within one month of the launch of the new workflow, which includes the custom hardware and software applications, the time taken to process both liquid and solid orders decreased dramatically. The average time to complete solid orders, which fell to 20 h in December 2007, is shown in Figure 4. The impact of the new changes is especially notable in the liquid order data, because most of the process improvements were directed toward liquid samples. Liquid order processing time is substantially reduced, even with the additional processing of compounds to assay-specific formats. Solid order processing time also shows improvement; however, not as dramatically as liquid orders. This is primarily due to a drop in the number of solid compounds per order.
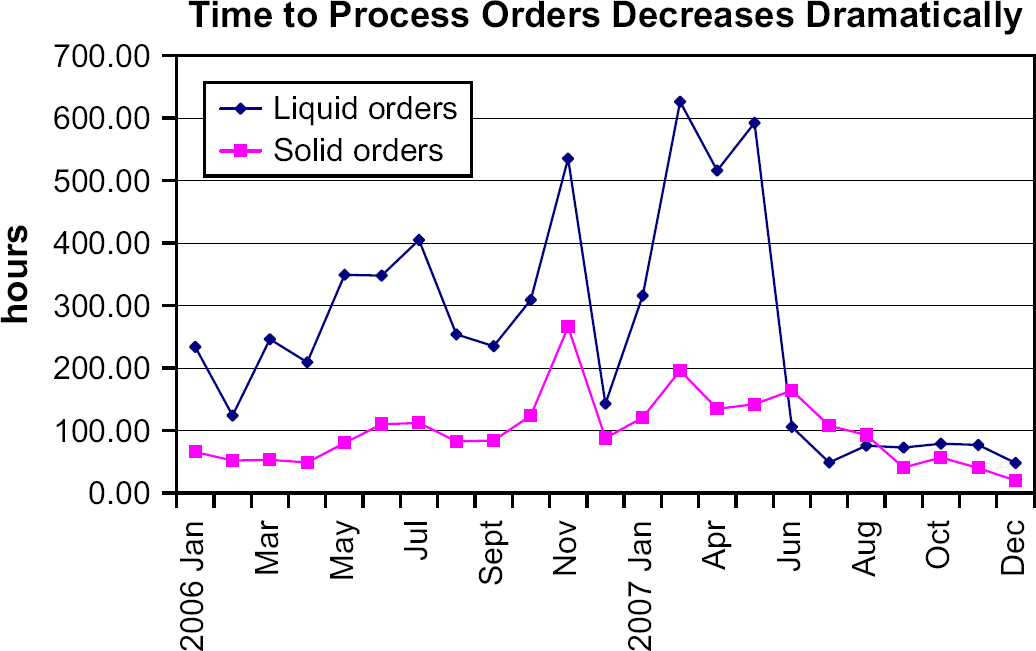
The time to process both liquid and solid orders decreased dramatically after the launch of the new workflow in May 2007. The decrease in liquid order processing time is due to the efficiency improvements resulting from the custom applications developed in-house and the hardware innovations for the Tecan worktable. The increase in demand for liquids consequently decreased the demand for solids, thus bringing down the processing time for solids as well.
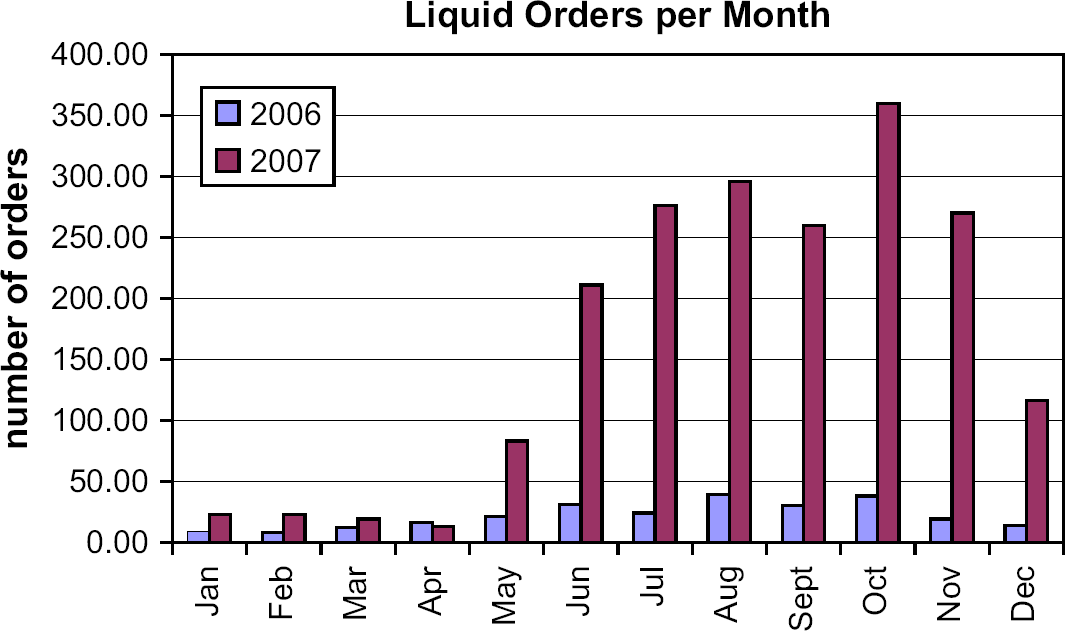
There was a fivefold increase in the average number of liquid orders handled pre-launch (20.8 liquid orders/month) as compared to post-launch (196.5 liquid orders/month). From May to December, the blue bars, (representing 2006), are much shorter than the red bars, (representing 2007), which graphically contrasts the higher number of liquid orders processed post-launch, as compared to the prior year.
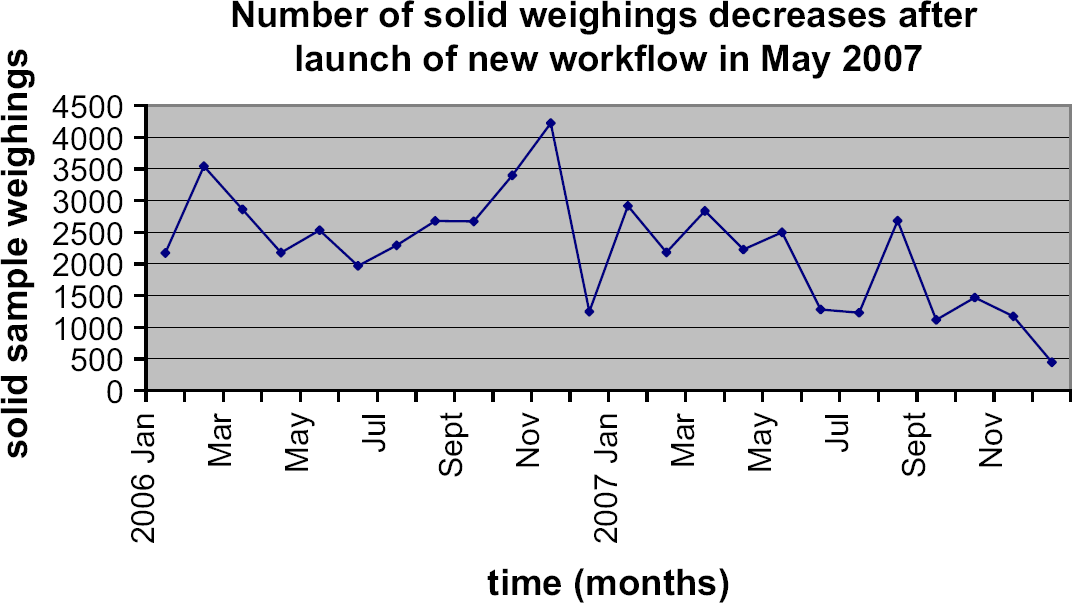
After the launch in May 2007, the number of solid weighings decreased due to the substitution of liquid orders for solid orders. The average number of solid weighings processed per month fell from 2614 to a mere 1342.
Discussion
As pharmaceutical companies continue to grow into increasingly large global entities, the ability to curate compounds for global testing is a growing challenge. Companies with sites around the globe must invest heavily in infrastructure to facilitate efficient distribution of compounds to all R&D sites. This undertaking involves major investment in IT-related applications for the tracking, archiving, and locating of compounds that are dispersed around the globe. In addition, pharmaceutical companies need the appropriate hardware to support the physical distribution and reformatting of the compounds into assay-ready plates. Of course, there are risks associated with the investments put forth into these endeavors. With the rapid discovery of new technology in scientific fields in general, plus the high obsolescence rate of equipment, major overhauls of these systems are required on average every 5–10 years. Continued improvements and enhancements are needed to keep up with the pace of changes in technologies and methodologies.
Conclusions
Shifting the focus from solid to liquid distribution has dramatically improved the efficiency of AstraZeneca Boston's CMG. Customization of liquid-handling robots, and the development of in-house software were major factors in this improvement. Specifically, whereas before it would take 7–8 days to prepare an in-house sample for testing, in the new process it takes merely 24 h (an 85% reduction in lag time). The custom hardware and robotic deck configurations have made same-day solubilization a reality, decreasing the runtime from 3.5 h/100 compounds down to 45 min/100 compounds. The in-house software applications have allowed CMG to generate assay-ready plates, giving greater flexibility to and saving time for bioscience departments. Despite processing quintuple the number of liquid orders compared with prelaunch, both liquid and solid orders are processed more quickly; and by December 2007, the average was 20 h from solid order placement to delivery. These technological innovations and efficiency improvements enabled CMG to deliver assay-ready plates, and to make compounds available globally. Ultimately, because CMG delivers compounds to the project teams sooner, the time it takes to generate data is markedly reduced.
Acknowledgements
The authors would like to thank John Rosamond, Timothy Ikeda, Nigel Tester, Irene Yershov, Jeff Andrews and the entire Discovery Informatics team for implementing the changes and for their valuable feedback. All are at AstraZeneca R&D Boston.
