Abstract
The merger between Astra AB (Sweden) and Zeneca Group PLC (U.K.) to form AstraZeneca in April 1999 placed considerable demands on the compound management capabilities within the merged company. A strategy was developed and implemented to ensure that the compound management infrastructure fully underpinned current and future drug discovery processes.
This overview of compound management within AstraZeneca includes the premerger capabilities, the postmerger activities, and key elements of the AstraZeneca strategy that led to the new fully operational and automated compound management facility at Alderley Park.
The tools in place will allow the technologies to change in the various downstream activities with sufficient flexibility to adapt to new demands. New screening strategies are now enabled, whether materials are chosen as selected sets, by rational theories, better use of the big screening capability, or to support difficult targets and assays; the choice is there for the discovery scientists to now exploit.
Introduction
The compound management capability based at Alderley Park, Cheshire, U.K. has undergone considerable change over the last twenty years. Initial changes were incremental to support technological advances in the bioscience and chemistry laboratories. A significant step change occurred in the late nineties with the introduction of automation within the compound management group, followed by the merger between Astra AB (Sweden) and Zeneca Group PLC (U.K.) to form AstraZeneca in April 1999. The pressures placed on the team to support the new global company and the need to work alongside equivalent teams based at the other research sites necessitated a radical rethink of our working practices. An overview of these events is outlined below.
Premerger Capability at Alderley Park
In the late 1980s, the compound management facility within ICI Pharmaceuticals (Alderley Park, U.K.) was a low-key capability that had a varying degree of usage by the chemistry community. There was a lack of trust in the service, and chemists were not willing to allow their materials to be stored and looked after by a detached service team. This was well founded. Materials were stored in bottles or original reaction flasks in a basement room with steam lines running through the area. Humidity and temperatures were often high. Bottles were stored on shelves or in index drawers raised off the floor (Fig. 1) to give some protection to the occasional flood that happened in the area. The materials suffered as a result. The dry weighing service, as it was then called, however, had very dedicated staff that offered the best type of service they could with the tools they had available.

The basement store, Alderley Park.
With the start-up of high-throughput screening (HTS) in the early 1990s, new liquid handling and bar-code tracking capabilities were introduced into the facility. The need to weigh hundreds and then thousands of samples per week forced a well-needed and radical rethink of both software and hardware requirements for compound management. With this came the recognition by the senior research management that compound management desperately needed investment to preserve and enhance the compound collection and support the technological changes occurring in both the chemistry and bioscience laboratories. The facility was placed under new management with the intention of overhauling the compound management activities and gaining greater acceptance by the chemistry community.
By 1997, the Compound Management Group within Zeneca Group PLC had a new automated facility provided by The Automation Partnership (Cambridge, U.K.), with capability to weigh over 13,000 samples per week, solubilize new stocks to 10 mM concentration in DMSO (dimethyl sulphoxide), and store in both 96-well plate and tube format. The plates and tubes were stored for use almost exclusively by HTS, enabling follow-up cherry-picks of over 15,000 per week. All vessels and containers within the system were bar-coded and fully tracked with the exception of the tubes containing solubilized stock. At the time of system development, the production of unique codes on the base of tubes was not available. The tubes had to be tracked in the system software by positional information only within bar-coded containers. A number of layers of hardware and software backup systems were put in place. However, tubes or racks were inevitably dropped, and without the individual identification, a small proportion of stock was lost over time.
Immediate Postmerger Activities
With the merger between Astra AB and Zeneca Group PLC, an interim measure was implemented to distribute the equal sized solubilized compound collections between four compound management teams supporting the HTS centers. These were based at Alderley Park (U.K.), Charnwood (U.K.), Lund (Sweden), and Wilmington (DE). The materials were to enter into screening as soon as possible.
Materials from Alderley Park were already stored and available in the plate store. They were shipped within a few weeks to the other sites. Likewise, liquid materials were shipped into Alderley Park from Charnwood and Lund, and entered into the tube store. The collection size doubled overnight, and almost reached the capacity of the tube store. Surprisingly, there was only a 10% overlap between the new and original Alderley Park stocks, arising from the fact that both companies had made similar external acquisitions. The tube store enabled the overlap to be removed, and materials to enter HTS quickly. There was, however, little knowledge of the liquid stock quality or global solid stock availability for follow-up activities.
A number of compromises had to be made at this time to support this fast exchange of samples. At Alderley Park, materials were initially registered into the local chemistry system without any structural information, though a unique sample identifier was allocated associated with the unique chemical identifier from the originating system. Structures were later loaded and cross-referenced in a central chemical information area.
The tube store contained multiple copies of sample stock and was designed for one-hit output to HTS. Requested tubes were not to be returned. In order to effectively use the new stock and make the most of the now-limited space in the tube store, multisipping (repeat sampling of the tube stock) was introduced. To achieve this quickly, an additional compromise had to be made whereby the availability of the stock was not seen while out for processing. This processing took place offline from the main software tracking capability. New in-house tracking routines were therefore written and introduced. When the stock was returned, it was reregistered back into the store and the availability updated to the chemistry system. Each of the compound management areas on the four sites was now handling a collection twice the size compared to a few weeks before and quickly adapting site-specific processes to be able to operate within the new global discovery organization across nine major discovery sites.
Key Elements of the AstraZeneca Strategy
By the end of 1999, a new strategy had been developed and agreed for the compound management capabilities across the new company, covered hardware, software, and key process steps. The strategy included:
Establishment of a compound management network with representatives from all AstraZeneca discovery sites;
Strong interactions with other new global networks, including HTS;
New information systems covering global discovery functions;
A centralized solids store based at Alderley Park, with the most recent project samples available at the local site but also submitted to Alderley Park for solubilization;
Distributed solutions collection based in four centers (Alderley Park, Charnwood, Mölndal, Sweden, Wilmington) fed mainly from Alderley Park. The centers were each to have plate production capabilities supporting downstream activities, and a direct feed into HTS;
Ensuring the high quality of the compound collection.
Process Mapping Activities
According to the strategy, the Compound Management Network (CMN) was formed and held its first meeting early in 2000. Links were quickly established with other global networks, in particular the HTS network. The initial emphasis of the CMN focused on how to support the four HTS centers based at Alderley Park, Charnwood, Lund (soon to be moving to Mölndal), and Wilmington. The next step was for the newly established Compound Management Network to agree how liquid and solid stocks should effectively move around the business according to the agreed strategy. A top level global sample flow was agreed (Fig. 2). A chemistry business reference group for the new global chemistry registration and querying system covered the first area, “New Compound.” The CMN concentrated on the other areas of the flow: “Compound In,” “Storage,” and “Compound Out.” Each area was then expanded with individual activities and decision branches to create a flow of samples and data from initial request to output by the compound management dispatch point. The steps for request initiation included items such as create request, amend request, and check availability.
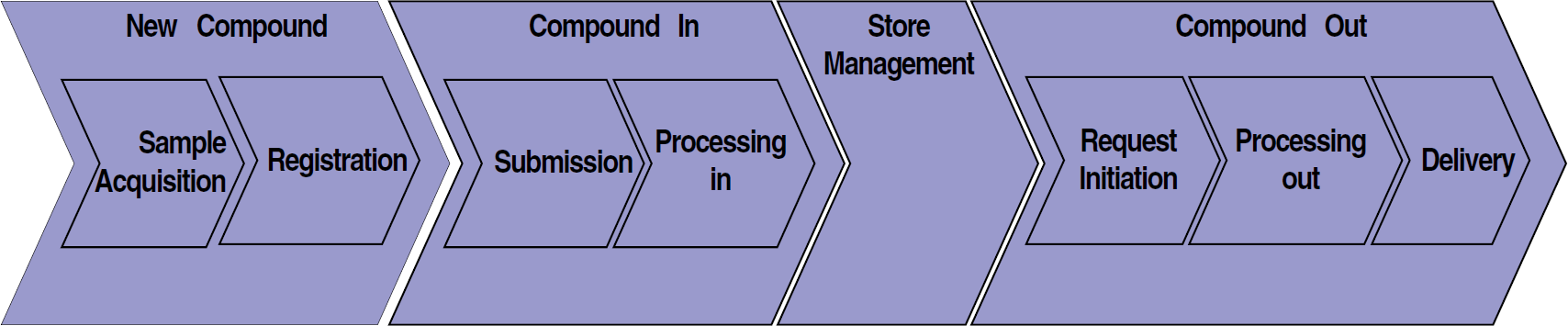
Compound management global process overview.
In order to support the flow, the CMN also agreed the definitions of each step and a core set of global business rules. The business rules included agreeing a common set of bar-code number conventions and global allocation, specific vial types for storage activities, handling, and various storage conditions. This full mapping of the compound management process helped all the teams understand how operations worked on each other's sites, where similarities existed, and what work was required to achieve the strategy. This mapping was fundamental in planning for and implementing the new automation and software capabilities. Scenarios of activities were agreed and reviewed during software walk throughs. In some cases, the global sample flow was challenged and changes were made where required. Overall, the first iteration of the global sample flow and main business rules took four months to complete.
Implementation of Global Software Systems
AstraZeneca has made a considerable investment in establishing core compound management software platforms. All chemical compounds now registered in the global organization have a unique identifier issued during the registration process. Any scientist can search for information associated with the compound collection and exploit the compound management network linkages by reviewing results, for example, from HTS, seeing what stock is available from any of the compound management centers, and ordering the stocks required for screening campaigns. The process mapping and business rule agreements were essential in agreeing the specifications for the new automated compound management facility and software, and the associated in-house ordering system, closely linked to the other global discovery systems. The relevant compound management business rules were also taken to the chemistry business reference group for review and agreement to ensure the close relationship between the new registration and chemical query tools and the compound management capability.
New Automated Compound Management Facilities
AstraZeneca has worked with RTS Life Science (Manchester, U.K.) in establishing a new automated storage capability across the business, installed in new purpose built buildings. Samples are stored under controlled humidity and temperature. The facility at Alderley Park can store over two million vials and over three million tubes (Fig. 3). Each vessel and container is individually coded to enable the full inventory tracking activities required to support large compound management capabilities. As part of the strategy, Alderley Park now houses the centralized solids store. The latest project stocks are retained at the local sites for a short period of time, and then the stock bottle is transferred to Alderley Park for long-term storage. In order to quickly enter the solubilization processes, however, a proportion of the new local material is sent over to Alderley Park ready-weighed. At Alderley Park, we are able to weigh over 20,000 samples per week using up to 16 weigh booths to feed the solubilization and distribution capabilities required to support the four primary liquid stores and global project, physical chemical, and Drug Metabolism and Pharmacokinetics screening activities (Fig. 4).

The primary liquid store, Alderley Park.

The dispensary weighing area, Alderley Park.
For the solubilization activity, the required amount of sample is weighed into tubes housed in a 96-position rack and a split septum bung is placed onto each one. The rack is then placed onto an input shelf for collection by a tracked robot. The materials now enter processing under controlled environmental conditions. The sample is solubilized in DMSO to the required concentration, purged under nitrogen, and handled in relative humidity between 5-10%. The required amount of material is transferred into multiple tube copies according to the ordering requirements. The tubes for Alderley Park are stored directly in the primary liquid store (PLS), and materials for the other sites are output and dispatched according to requirements.
The PLS is a narrow aisle store serviced by two store robots. These robots transfer trays of tubes between the store and one of four picking stations. Each picking station houses an ABB Flexpicker robot that quickly picks the required tube from the tray to the transport rack, and also returns the processed or new stock to the storage trays. The transport racks deliver stock to the reformatting station. The fast cherry-picking robotics applied to the tubes are able to subset the collection ready for screening campaigns, unlocking HTS and other project teams from rigid plate sets (Fig. 5).

The automated picking area, Alderley Park.
Where working plates are required, these can now have a shorter lifetime, for example, six months, and be supplemented by other selected materials. Small volumes, for example 0.5 μL, are provided in output 384-well plates for thousands of targeted materials and can be turned around to screens within twenty-four hours of request. For support of the variety of downstream processes, the compound management capabilities downstream of the primary liquid stores are site-specific to enable fast introduction of new technologies where required, especially for low-volume transfers. The knowledge each site gains in experimenting with and effectively using these new technologies is shared throughout the compound management network. The liquid material is also monitored by LC-MS (liquid chromatography-mass spectrometry) to confirm the required material is in the tube and details recorded. Over time, the behavior of each material is established to assist in the restocking requirements of the facility.
Compound Collection Consolidation
Chemists are notorious hoarders of stock! The first sweep of materials at Alderley Park occurred during the mid-1990s. An estimated 25,000 samples turned into over 50,000 samples stored in various cupboards and drawers, many of which had never been registered into the registration system. All chemists were involved in this exercise, and where necessary, information was retrieved directly from notebooks.
The decision was taken during the strategy discussions within the new company to complete a similar exercise and bring older stocks into Alderley Park from all sites. In total, 304,000 samples were collected from all the discovery sites. The materials sent were in a variety of containers and data formats, and a number were found to be new to the chemistry database. Six staff worked in a dedicated facility to rebottle and record all the information associated with these materials, including any relevant details written on the label. Details were uploaded into the central chemistry database. This project took 18 months to complete, but now all materials are visible and available for ordering by all sites.
The AZFILTERS are comprised of chemical and property filters. The chemical filters include those that identify reactive or unwanted chemical functional groups in the structure, for example bland structures, toxiphores, dye-like and ugly structures, and frequent hitters. The property filters use calculations to predict certain physical properties of the structure and compare them to a desired range, for example, ClogP, of molecular weight and polar surface area. Core compounds are those that pass the entire chemical and property filters in AZFILTERS. The back-up compounds fail one of the property filters, and ugly compounds fail either two or more property filters or any of the chemical filters. The AZFILTERS were designed, and are periodically refined, to provide some common ground amongst the chemistry community on types of compound that would generally not be rejected for structural reasons if they appeared in a list of hits from primary HTS or other screening activities. The filters do not, by themselves, delimit a subset considered to be representative of the compound collection. Different filter criteria were set up for different purposes, for example for screening, compound acquisition, and library synthesis. These are regularly refined depending on the feedback from various teams.
The physical quality of the solids collection was also reviewed. A new team was established in the dispensary to prepare materials for analysis and weigh materials for the new solubilized collection. Shift working was introduced into the facility to support this activity and maintain operations to the global screening campaigns. Over a two-year period, the dispensary weighed over a million samples for this activity alone. Where designated minimum amounts of core solid samples were available, these were weighed out by the dispensary staff and analyzed by two dedicated staff over an 18-month period. Analysis was by LC-MS. A proportion was also reanalyzed, for example, where mass was unconfirmed. Flow NMR (nuclear magnetic resonance) was also used as quality control to evaluate false positive and false negative rates. A purity cutoff was established at 85%. Of those reviewed, two-thirds were greater than 85% pure and of the correct mass. These were selected for onward processing into the new solubilized collection. The remainder is undergoing a variety of different process steps, for example, those less than 85% pure and available in sufficient amounts are selected for purification and reregistration into the collection. A small percentage of the collection is being disposed of.
With this information on the collection highlighting, only 66% of the selected core and back-up materials were suitable for immediate solubilization, equating to only 50% of the combined collection; a new strategy was established for compound collection enhancement (CCE) through synthesis and acquisition. The aim of the acquisitions area is to give the collection an overall coverage of leadlike chemistry space, to minimize the number of singletons, and to identify and fill holes. Biological areas are also targeted for in-house synthesis. Dr. John Steele of the Charnwood R&D leads the CCE initiative. Four synthesis teams based on different sites are addressing selected therapeutic target classes involving over fifty chemists with a strong link to the medicinal chemistry community for ideas exchange and prioritization. The materials are produced to specific quality criteria, and are supported by bioscience and compound management teams, new databases for recording and exploiting the required information, and the outsourcing of chemical building block production.
AstraZeneca has focused hard on physical, data, and chemical quality. Key principles in our activities in the compound management capability are fast sample accessibility through high-speed picking; maintenance of sample integrity; quality and error-free equipment handling; having available and reliable inventory data; and material and process efficiency and effectiveness. The tools in place will allow the technologies to change in the various downstream activities, with sufficient flexibility to adapt to new demands. New screening strategies are now enabled, whether materials are chosen as selected sets, by rational theories, better use of the big screening capability, or to support difficult targets and assays; the choice is there for the discovery scientists to exploit.
Summary
With the significant investment in the global compound management capabilities and software systems, the Alderley Park facility is now fully operational supporting the AstraZeneca compound management strategy. The compound collection quality is quickly being enhanced and the scientists are learning to exploit their resource, which is more akin to a production facility, and to gain the maximum benefit from this investment.
Acknowledgments
The author would like to thank the following colleagues for their help and support: Mike Cox, AZ Global Compound Management; Paul Leeson, AZ Global Chemistry Forum; John Steele, CCE Initiative; Jin Li and Sorel Muresan, Computational Support; Tony Wilkinson, Lee Griffiths, and Ian Sinclair, Collection Analysis; Mark Divers, AZ HTS Network; Ian Yates, Philippa Skinner, Philip Spencer, Dalin Nie, Swier Copinga, Lars Ogren, Denise Penkalski, Chris Porter, Michael LaPlante, Srinivasa Bachally, and David Camp, AZ Compound Management Network; Gill Gowler, Paul Owen, and Isabel Charles, Compound Management Team Leaders; and Andy Morton, AZ Engineering.
