Abstract
In vitro screening for oral absorption has become an essential part of drug discovery and development. Recently, a new phospholipid vesicle-based permeation assay was developed which has shown to satisfyingly predict passive absorption of drugs in humans. The purpose of the current study was to investigate whether the assay may be further developed into a high-throughput tool by automating its most time-consuming steps. The following challenges were addressed: (1) to design, build, and test a heat-sealing machine for mounting of the desired type of filter support onto both single wells and 24-well titer plate inserts and (2) to transfer the permeability assay to a robotic workstation with attached ultraviolet (UV) reader. The workstation is able to pipette and transport both plates and filter inserts and perform on-line photometric quantification of the amount of drug permeated. To enable the robot to move single (Standard Transwell; Corning Inc, Lowell, MA) filter inserts, an extension of the gripping arm was designed, built, and tested. Furthermore, in an alternative approach 24-well filter plates (Millicell; Millipore, Billerica, MA) were used instead of single filter inserts. The latter turned out to be more suitable in terms of error-free high-throughput robotic handling. The permeability values of drugs gained by the two automated procedures were compared with those measured by manual handling of the assay. Only neglectable differences in permeability values were seen.
In conclusion, the most time-consuming steps of the assay were shown to be eligible for automation. This represents an interesting addition to the toolbox of
Introduction
Oral bioavailability of a drug, that is, the fraction of an orally administered dose of a drug that unchanged reaches the systemic circulation, is an important property to see if a drug is suitable for oral administration and, for example, in dose finding. The oral bioavailability of a drug is mainly determined by the solubility of the drug in the gastrointestinal (GI) fluid as well as its permeability through the intestinal barrier. To eliminate drug candidates with poor oral bioavailability, permeability screening is today implemented in early stages of the drug discovery and development process. Efficient strategies for screening of permeability properties of large numbers of new drug candidates are needed to facilitate the selection of the most promising candidates for further development.
The phospholipid vesicle-based permeation assay has recently been introduced as a novel method for screening of passive drug permeability through barriers mimicking biological membranes.
1
2–3
The permeation barrier consists of a tightly packed layer of liposomes on a filter support.
1
2–3
For a diverse set of compounds, the apparent permeability coefficients obtained from the phospholipid vesicle-based permeation assay have been found to correlate well with literature data on human absorption
Furthermore, the phospholipid vesicle-based barriers were demonstrated to withstand a pH range from 2.0 to 8.0 without losing their integrity, and are thus regarded suitable for permeation studies at different pH conditions mimicking the drug transit through the GI tract. 7 The barrier has also shown to be compatible with relevant co-solvents and certain tensides. Permeability testing of drugs in presence of commonly used additives appears thus feasible. 7 Taken together, the phospholipid vesicle-based permeation assay, so far, has shown to be a promising approach for low- to medium-throughput screening of passive drug permeability.
However, to fully characterize the permeability properties of potential drug compounds, for example, at different pH values and in the presence of different additives, a large number of experiments are necessary. This is time consuming and cumbersome with the current set-up due to the lack of commercially available filter supports containing the desired type of filter membrane as well as the time needed for preparing the barriers and for performing the permeation experiments manually.
The purpose of this study was thus to investigate whether the most time-consuming steps of the phospholipid vesicle-based permeability assay can be automated: (I) fusion of the desired filter material onto bare filter inserts, (II) preparation of the liposome-based permeation barriers on the filter support, and (III) performing the permeability assay to compare with the manual procedure.
Part I, the fusion of the desired filter material onto bare inserts, has so far been made by hand, insert by insert, and has been the most labor-intensive step. This step was automated by designing, constructing, and testing two prototypes of a heat-sealing machine for automatically fusing the filters onto the filter inserts.
Part II, the preparation of the liposome-based barriers was found difficult to automate because the preparation would require automation of centrifugation, heating (50 and 65 °C), and freezing (−80 °C) steps, that are not easily implemented within the robotic platform used. However, the phospholipid vesicle-based barriers have earlier been shown to be stable during storage at −80 °C for up to 2 weeks, which gives the opportunity to produce and store larger batches of the barriers beforehand. 1
Part III of the process, the performance of the permeability assay itself, is not very labor intensive but is the most time-consuming step as it requires manual handling for at least 7 h to move the filter inserts at certain time intervals from one well to the next. At the end of the experiment, the sampling from the compartments and also the UV analysis have to be done. If the assay and analysis steps could be automated, manpower will only be necessary to prepare the barriers and start the experiments. Automation should thus allow a significant increase in throughput. Routines were established and tested to carry out all steps on a common robotic workstation (Tecan Genesis). These comprise pipetting of both donor and acceptor solutions to the filter inserts and wells, respectively, storage of the plates, moving of the inserts from one well to the next at given time points, sampling, and quantification of drug content by UV-absorbance measurements.
Materials and Methods
Materials
Egg phosphatidylcholine, Lipoid E-80, was obtained from Lipoid, Germany. Caffeine, metoprolol tartrate, sulpiride, testosterone, acebutolol hydrochloride, terbutaline hemisulfate, and calcein were purchased from Sigma-Aldrich Co, St. Louis, MO. Bare single filter inserts (Standard Transwell,
Methods

The robotic system, Tecan Genesis RSP 150. A—Robot arm containing four pipettes (needles); B—Hotel for multititer plates (MTPs); C—Gripper for moving single filter units (plus support to park this tool); D—Robot arm (RoMa) for moving plates and filter inserts; E—Working table, nine positions for MTP plates; F—The UV-absorbance reader, Tecan Infinite M200.
Robot software: Gemini v4.2.16.303 (Tecan Deutschland GmbH, Crailsheim, Germany). Reader software: Magellan v6.1 (Tecan Austria GmbH, Salzburg, Austria). As the reader used here does not have a fluorescence option, the samples from the experiment with calcein were measured manually on a Luminescence Spectrometer LS 50B from Perkin Elmer, Beaconsfield, Great Britain. However, a variety of readers could in principle be connected to the robot, depending on what analytical method is needed.
The resistance was measured manually as described earlier at the end of the permeability testing to control the integrity of the single barriers. 2
Results and Discussions
Automation of the Filter Insert Manufacture
Filter inserts with the desired filter type (mixed cellulose ester filters with a pore size of 0.65 μm), used as a support for the liposome-based barriers are currently not commercially available. So far they have thus been prepared in-house by hand, which is labor intensive and error prone. A sealing machine (IBR Heat-Press HP80–3500) to make 24 (single) filter inserts in one run, including software that allows fully automated runs, was therefore designed and built in collaboration with IBR-Ingenieurbüro, Waldkirch, Germany.
The process consists of pressing the filter insert and the cellulose filter onto a hot surface and cutting off the excess filter material. Thus, the instrument included exact temperature control of the areas for sealing (heat piston), a pneumatic unit to press the inserts with the filter toward the heat piston with predefined force and duration, and a cutting device fitting exactly the insert diameter.
A first prototype of the new heat-sealing instrument, as shown in Figure 2, was designed to simultaneously fuse 24 filters onto inserts. To do this, the filter inserts are mounted upside down on an insert holder (Fig. 2A) and a filter disc is positioned on top (Fig. 2B). The machine then pneumatically moves the sandwich consisting of filter holder with the inserts and filter disc until they have contact with the sealing piston, which is electrically heated to a predefined temperature (Fig. 2C). Although the filter holder and piston are in contact and the sealing process is running, round knifes are pneumatically moving down cutting off the excess filter material (see Fig. 2E for picture of the heat and cut piston). The sealing process is continued for a predefined time until the filter material is properly sealed to the insert base. After the sealing process is completed, the insert holder and the sealing piston move back to their initial positions and the excess of filter material is removed manually (Fig. 2D).

The IBR Heat-Press HP80–3500 to automatically make 24 single filter inserts at once. A—Standard Transwell Permeable supports without filters on the support plate; B—Standard Transwell Permeable supports with the filter on top; C—Fusion of the filter to the insert bases; D—Filter-fused inserts; E—The heat and cut pistons (round knifes with heat piston inside) for fusing the filter to Standard Transwell Permeable supports.
Various temperature settings of the heat piston and pneumatic pressure were tested to identify optimal sealing conditions. A piston temperature of 150 °C and a pneumatic pressure of 2 bars applied for 30 s were found ideal in terms of minimal numbers of leaky inserts. The fraction of leakage incidents was reduced from 12% for the inserts handmade by an experienced person to below 2% for the inserts made by the sealing machine.
Taken together, the process of sealing filters onto the insert bases went from being the rate-limiting step of the permeability assay to the most efficient step and this consequently improves the throughput of the assay significantly. Within the same time span we earlier used for producing one insert (5 min), we can now make 48 inserts by using the sealing machine (two runs). The efficiency of the process is thus increased by a factor of almost 50 and the time used for 18 inserts, which is needed for one assay run, reduced by about 1.5 h. Furthermore, the quality of the machine-made inserts is better than those handmade in terms of tightness of the joints between the filter and the insert base.
In parallel, a second heat-sealing machine was designed in collaboration with IBR-Ingenieurbüro, Waldkirch, Germany, to handle 24-well filter plates. This second prototype (IBR Heat-Press HP60–250-ESM) works after the same principle as that described above, but here the filters are sealed one by one to the bottomless wells. All together it takes approximately 15 min to seal all the 24 wells with filters. The same sealing parameters as described above (150 °C, 2 bars, 30 s) were also used here. It was even more important that the sealing in the 24-well filter plate is performed properly because leakage in only one filter would lead to discarding of the whole plate.
Automation of the Permeability Assay by Using Filter Inserts
The requirements for the robot to be able to run the permeability experiments are that it can perform pipetting, move the plates, and take off and put back the lids as well as move the inserts. To run the minimal experiment with six inserts, two 24-well plates with lids and one 96-well UV plate are needed. However, the goal of developing the automated procedure is to investigate more drugs/compounds at the same time.
The manual procedure for the permeability assay involves moving of the filter inserts to fresh acceptor wells at specific time intervals. For the automated assay, we decided to stick to this procedure rather that replacing the acceptor medium in the well by pipetting. Although the single filter inserts (Standard Transwell) used here have holes in their upper edges (see Fig. 3), which principally would allow the pipettes to stick down into the acceptor solution it has to be taken into account that each filter insert is free to rotate in the plate wells, which could potentially lead to collisions between the pipettes and the inserts. Pipetting for replacement of acceptor solution was thus regarded too risky.

Photograph of the Standard Transwell inserts. Holes that could be used for emptying the respective multititer plate wells can be seen in the upper parts of the inserts.
The robot was therefore programmed to do the following: The robot first moves the 24-well plates from the storage rack to the working table to give the pipetting arm access to the wells. The lid is removed from each plate and placed on the working table next to the plate. The pipettes now fill equal amounts of acceptor medium into all wells. Single filter inserts with the beforehand-prepared permeation barriers are then picked up from a 24-well plate on the working table by a especially designed finger tool (see Fig. 4) and placed in row 1 (assigned

Photographs of the specially designed finger tool enabling gripping of filter inserts. A—from the front; B—from the side; C—from beneath; D—from above.
Within 30 min, which is the minimum time span for moving the inserts to the next row of wells, the robot is in principle able to handle six compounds with six replicates each in parallel. With the robot equipment used here only three drugs (six replicates each) could be handled in parallel due to limited storage capacity of the hotel (see Fig. 1B). However, an increase of the hotel space would allow handling of the maximum number of compounds as well as sequential starting of more than one set of permeability assays. For a maximum of six compounds with six replicates each, the automated procedure is assumed to take about 12 h.
The finger tool is placed on the two gripping arms in a 90° angle position (see Fig. 5A). The depth of the tool is 80 mm, which gives a stable contact between the finger tool and the gripper arms. When the gripper moves the arms toward each other, the finger tool and the fingers do the same. The width of the whole tool can thus be varied from 87 to 114 mm. The fingers are curved at the tip and have a soft rubber material on the inside of the fingertips to improve the gripping performance. An important aspect was that the gripper should be able to collect the finger tool when the inserts have to be moved and park the tool when plates have to be moved. The finger tool is therefore placed on a station when not in use, where it is freely reachable for the gripper, as shown in Figure 5B. By this design the gripping arm can serve two functions, both moving plates and lids as well as moving smaller items by fetching the finger tool first. This tool is also very useful for handling all kinds of round vials up to 30 mm. Due to the shape of the fingertips, the gripper is optimal for vials from 10 to 20 mm diameter.

Photographs of the specially designed finger tool in the robotic system. A—The finger tool mounted on the gripping arm; B—the finger tool parked at the station from where it can be picked up by the gripper.
Gripping of the single inserts, which have only a very small and flat rim, which can be used for gripping, turned out to be a critical step in the automation of the assay because the contact area between the fingers and the inserts is quite small (see Fig. 3 and Fig. 7). Due to the geometry of the 24-well plate, the filter inserts are also placed quite close to each other as shown in Figure 6. This requires a high precision in positioning the fingers in between the inserts. To meet this demand, the fingers are programmed to grip the inserts not parallel to the plate edge but at a 45° angle as shown in Figure 7. Using the finger tool, the robot was now mechanically able to move inserts from one well to the next during the permeability assay. However, the inserts are placed in wells on a 24-well plate and the single inserts can rotate 360° in the well and have some space to move out of the central axis. Thus, it turned out to be challenging to grip the inserts perfectly, which in worst case led to a premature stop of the program, which means loss of all results from this run. We decided to try with another filter plate format, which circumvents the need for the gripper.

Photograph of a 24-well plate with single filter inserts, shown from the top.

Photograph of the finger-tool gripping one single filter insert from a 24-well plate.
Automation of the Permeability Assay by Using 24-Well Filter Plates
So, as an alternative for using the single filter inserts, a plate with 24 bottomless wells (see Fig. 8) was tested in parallel. The advantage of using this plate is that 24 wells can be moved at once, which circumvents the challenging movement of single inserts and drastically reduces the total number of moving operations within one run. As for the single filter inserts, the 24-well filter plates are at present not commercially available with the desired type of filter (0.65-μm pore-sized mixed cellulose ester filters) and have to be preformed by the heat-sealing instrument (see Automated Preparation of the Filter Insert).

Millicell cell-culture plates: insert tray (on top) and receiver tray (in the middle) (reprinted with the permission from Millipore).
An important difference between the single filter inserts and the 24-well filter plate is the surface area of the filters. The effective filter area for a 24-well filter plate is 0.6 cm2 per well, b whereas the corresponding area for the single filter inserts is 0.33 cm2. c Based on this difference, the preparation procedure for the barriers was adjusted so that the amount of liposome dispersion per area remained the same.
When programming the modified assay procedure to handle 24-well filter plates again a decision had to be made whether to move the insert tray at different time points from one plate to the next or instead to replace the acceptor solutions in the wells by pipetting. However, because the shape of the wells and the design of the plates make it difficult to empty the wells completely (see Fig. 9) we decided to move the 24-well filter plate to fresh acceptor wells. This alternative is also considerable faster than replacing the acceptor medium by pipetting. A four-drug script, with six replicates for each drug, was developed for 24-well filter plates after the same principles as for the single insert system, except that the whole plate with the 24 filter units was moved over to new plates at the desired time points.
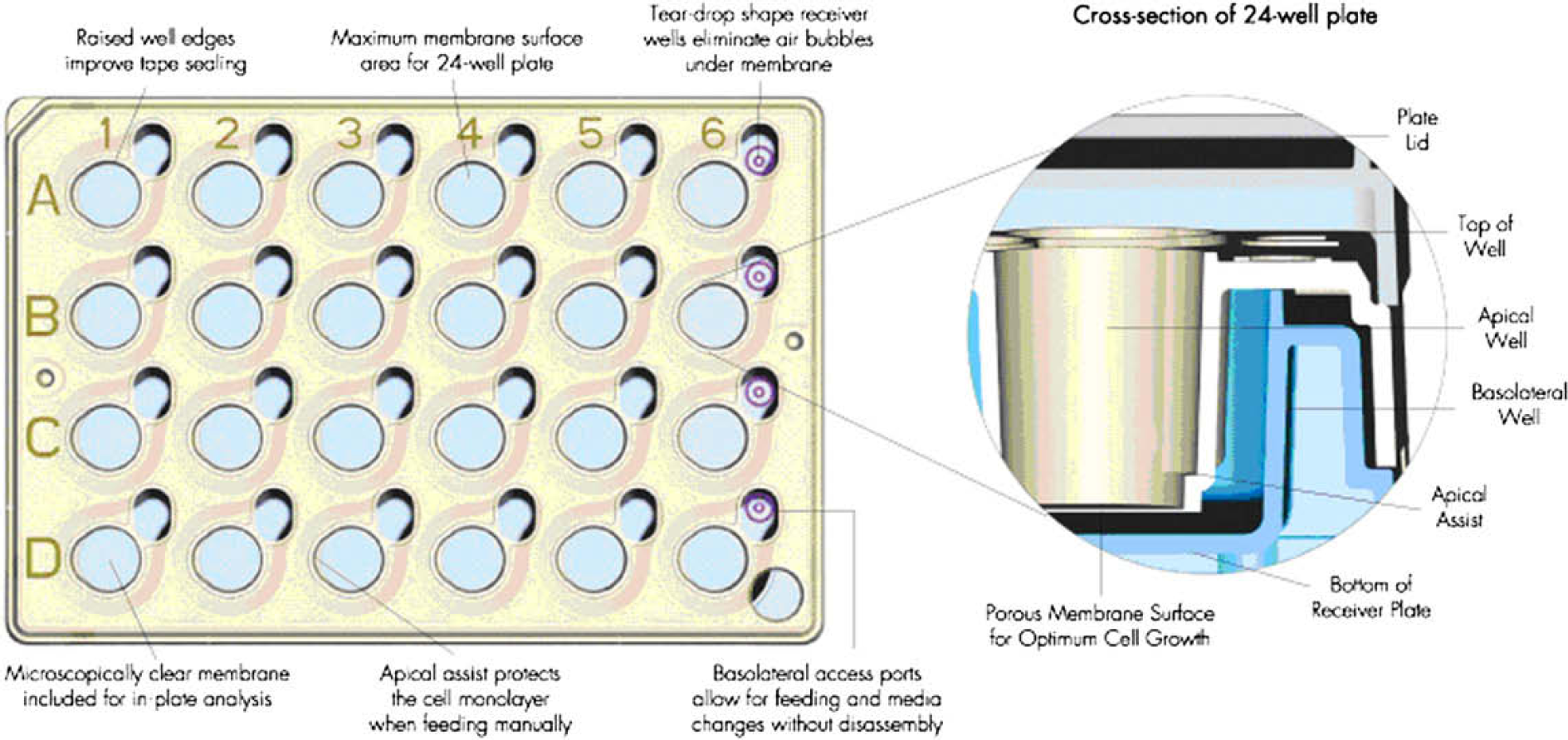
The insert tray at the Millicell cell-culture plates (reprinted with permission from Millipore).
Compared to the script for the single filter inserts, the number of movements is now drastically reduced. For example, a three-drug assay (six replicates) with single filter inserts needs 144 moving steps, whereas for a four-drug assay (six replicates) with 24-well filter plates only a total of 28 moving steps is necessary. Another advantage with using these plates is that due to fewer moving steps and thus a faster process time, it is possible to run about 20 drugs with six replicates each at the same time.
Due to the difference in filter area, a systematic increase of the flux of the drugs was observed when changing from the single filter inserts to the 24-well filter plates. The flux through barriers made on the 24-well filter plates was about twice the flux through single filter inserts, which correlates with the expected value when the filter area is doubled. The increased flux and hence the increased concentration of drug in the acceptor chamber allows a decrease of the assay time by decreasing the time span between the time points for moving of the inserts or alternatively allows for detection of drugs with slow flux. Thus, the 24-well permeability assay allows a considerably higher throughput.
A currently unused possibility with the robot is that the workspace can be connected to an incubator. It is thus also possible to run the experiments at other temperatures than room temperature and by that get information on how the temperature is affecting the permeation across the barriers. Furthermore, connecting the robot with an automated hotel unit, it would be possible to further increase the number of drugs, which could be automatically handled.
Comparison of Automated and Manually Obtained Permeability Values
The automated permeability assays using single filter inserts and 24-well filter plates were run with five and four drugs, respectively, already tested in the manual permeability assay, and the
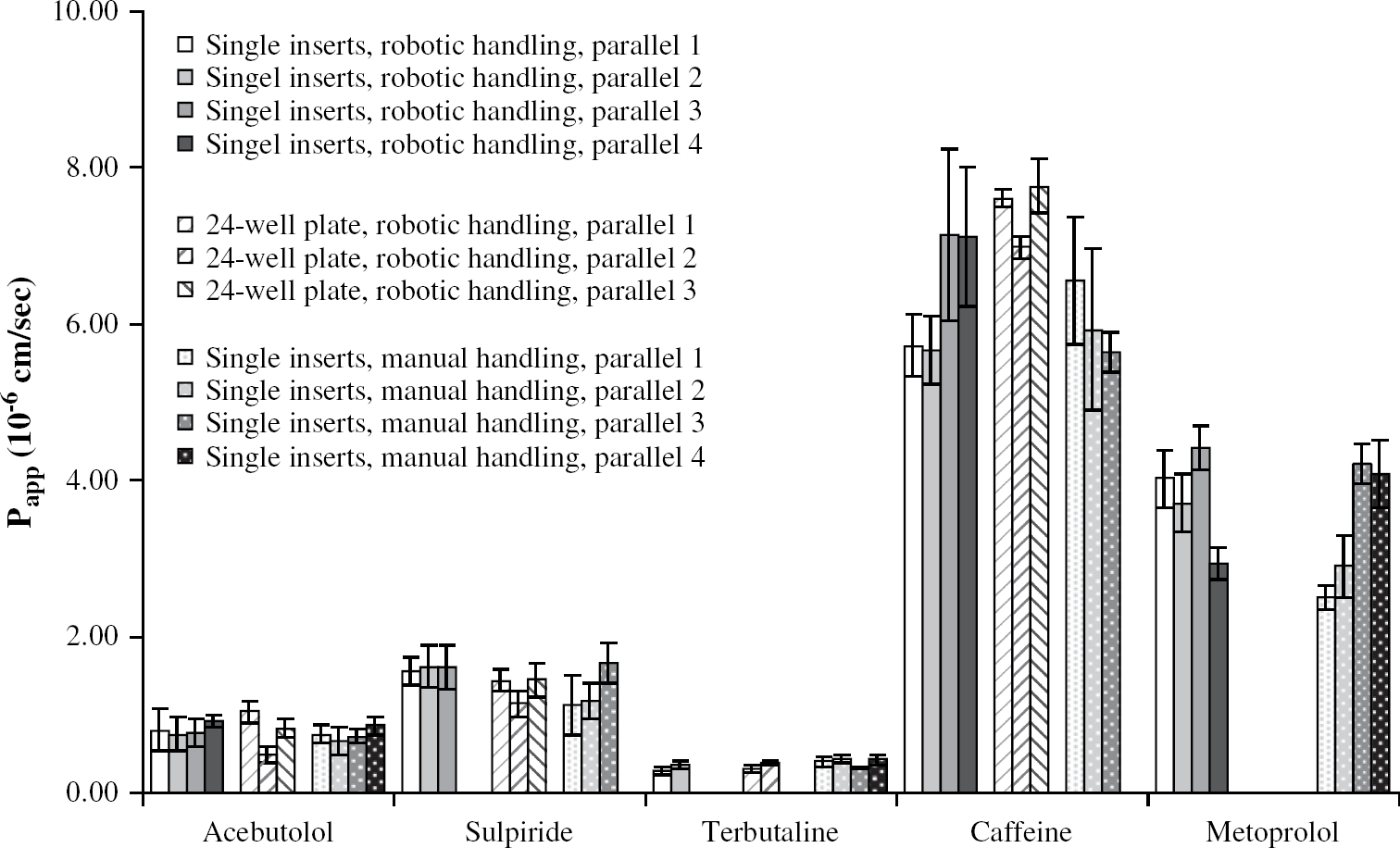
Results from the single parallels with the automated assay using single filter inserts and 24-well filter plates together with permeability data from the single parallels obtained with the manually run assays. Standard deviations of the results from six inserts are reported for each parallel.
Summary of permeability data from the automated assay using single filter inserts and 24-well filter plates and the manual run assay
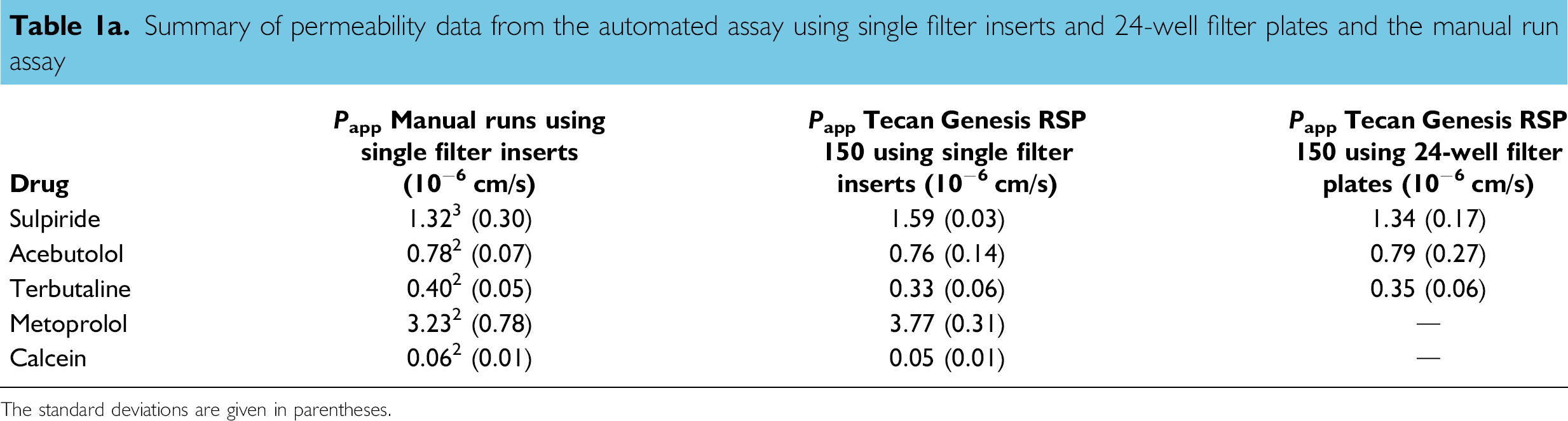
The standard deviations are given in parentheses.
Summary of permeability data for caffeine from the automated assay using single filter inserts and 24-well filter plates and the manual run assay

The standard deviations are given in parentheses.
As can be seen in Tables 1a and 1b, there were only negligible differences between the mean permeability values obtained with either of the automated assays, compared to the values obtained with the manual assay. For the compounds in Table 1a, no significant difference was found using the
The phospholipid vesicle-based assay seems to have a somewhat higher day-to-day variation compared to the PAMPA assays. The reason for this is most probably that the present model is based on a considerably more complicated barrier system. However, the higher day-to-day variation does not decrease the ability of the model to differentiate between drugs with different permeability values or to classify them correctly according to their
Conclusions
This study demonstrates that the two most time-consuming steps in the phospholipid vesicle-based permeation assay can be automated. Firstly, the fusion of the filters to the bare inserts has successfully been automated by using specially designed heat-sealing machines, resulting in a faster and less error-prone filter insert production process. Secondly, the permeability assay was successfully transferred to a common laboratory robot. Both tested filter insert systems, the single filter inserts and the 24-well filter plate, were found suitable. It could be shown that transferring the assay to an automated system does not influence the outcome of the permeability assays. The 24-well filter plates were found to be easier and faster to handle, which render them more suitable for high-throughput screening. The single filter inserts allow, on the other hand, for more flexibility and are thus suitable for medium-throughput screening.
This phospholipid vesicle-based permeation assay thus represents an interesting alternative to the established intestinal permeability screening tools due to its easiness, transferability to other laboratories, and its good correlation to in vivo data on fraction absorbed in humans. 2
As automation increases the throughput considerably the assay is also useful for studies of the influence of a range of factors on drug permeability. For example, the effects of varying the pH in the donor compartment can be efficiently studied, also varying the lipid composition in the liposome barrier can be performed to model various epithelial barriers.
Acknowledgments
We would like to thank to the Norwegian Research Council for financial support.
We are very grateful to Mr Reichenbach of IBR-Ingenieurbüro, Waldkirch, Germany for his genius ideas and the very nice collaboration.
