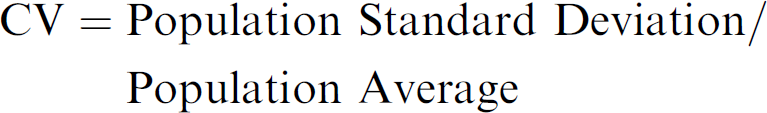Abstract
This review assesses the quality of the data acquired over a 13-week period from a High-Content Analysis screening project that used 297 unique cell lines. This article also evaluates the proficiency of a “tipless” (i.e., does not use disposable tips) full-automation design used for this project that prioritizes intralab system mobility and system configuration mutability. The request to assay a large number of cell lines with poorly characterized growth rates led us to devise an MDS PharmaServices, Inc. proprietary algorithm in an effort to select the proper cell plating density for each cell line. The performance metrics include coefficients of variation (CVs) of Controls for the cell plating data and Data Set Mean CVs for assessing replicate propinquity (i.e., how close the replicates are to each other). The performance of the automation system and our algorithm for this project produced data of superior quality.
Keywords
Introduction
The quest for a single, commercially available, fully automated system capable of adequately handling every single-screening assay known to science remains unfulfilled. As a Contract Research Organization (CRO), we at the Custom Services Group of MDS PharmaServices, Inc. are presented with requests to perform a wide variety of in vitro assays from a global client base of all major and minor pharmaceutical companies and biotech companies. Many of these assay assignments necessitate that the full-automation system physically change to fulfill the unique requests of our clients. This system layout was intended as a purposive response to the ever-changing custom screening projects of our clients. Routinely, the need to rearrange the entire automation laboratory arises when accommodating the arrival of new technologies and subsequential equipment, thus the system has nothing bolted into the laboratory floor. The main body of the system is on wheels thereby conserving the ease of movement of equipment within the automation lab and maintaining flexible use of valuable floor space (intralab system mobility). This particular design also prioritizes system configuration mutability (i.e., the ease or difficulty to make physical changes of equipment to the automation system).
For 13 weeks, we were engaged in a high-content analysis (HCA) screening project that used 297 unique cell lines with varied adherence properties. We lacked vital information on those cell lines and this created many technical difficulties for this cell-based screening project. Among these difficulties was how to automate the predominantly manual and laborious task of cell culture and determining a robotic protocol that was acceptable to a diversity of cell lines. Another large hurdle was developing an assay protocol that would compensate for the lack of information about the cell lines' growth and morphology. All this needed to be accomplished while staying within a narrow budget.
The request to assay a large number of cell lines with poorly characterized growth rates led us to devise an MDS PharmaServices, Inc. proprietary algorithm in an effort to select the proper cell plating density for each cell line. It was based on the morphology, size, and 1 week of growth information. This review assesses the effectiveness of this algorithm on the quality of the data acquired from this HCA screening project.
This article will also evaluate the proficiency of the “tipless” (i.e., does not use disposable tips) full-automation design used. All the operations executed by this system on this particular project exemplify a cross section of the most common operations performed by most fully automated systems. As a CRO, delivering compounds using washed tips is not acceptable. This is because we are unable to unequivocally assure our clients that there is absolutely no compound carryover from one assay plate to the next if we were to use tips that have been washed. The policy is to change disposable tips in the protocol whenever necessary. The cost of these disposable tips, however, can be staggering for some projects, especially when using expensive 384-well disposable tips. This was one of the reasons that motivated us to look at the tipless acoustic droplet ejection (ADE) technology used in the ECHO-550 from Labcyte for the addition of compounds.
Methods and Materials
Tipless Automation System
This system is presently capable of handling 96- or 384-well formats and has the capacity to adopt 1536-well formats. Movable overhead service delivery systems for air, gas, vacuum, electricity, and other utilities are used to accommodate this automation system's requirements. The design is centered on the Velocity 11 BioCel 1200 system which possesses a rapid robotic arm and has event-driven scheduling software that controls all the integrated equipment. The software also allows for offline stand-alone control of any instrument not used in a method/protocol. Two incubators are also incorporated, one for room temperature (Velocity 11 PlateHub) and one with a 4–37 °C (Celsius) temperature range plus CO2 (carbon dioxide) and humidity control (Liconic STR240).
Additionally, the system is equipped with other salient services such as lid removal and replacement (Velocity 11 Lid Station), centrifugation (Velocity 11 V-Spin), and plate washing (Biotek EL-405). Microplate readers are intentionally excluded from the core arrangement for throughput purposes. Empty space on the deck is provided to handle “guest equipment” (i.e., specialty pieces of equipment only needed for certain assays but not for most). Examples of guest equipment might be sealers that are only used for radioligand-binding assays or readers for assays with a short half-life signal. Noncontact liquid dispensers are accommodated to service both compound additions (Labcyte ECHO-550) and reagent additions (Thermo Combidrops or Multidrops) for volumes ranging from 2.5 nL to 300 μL.
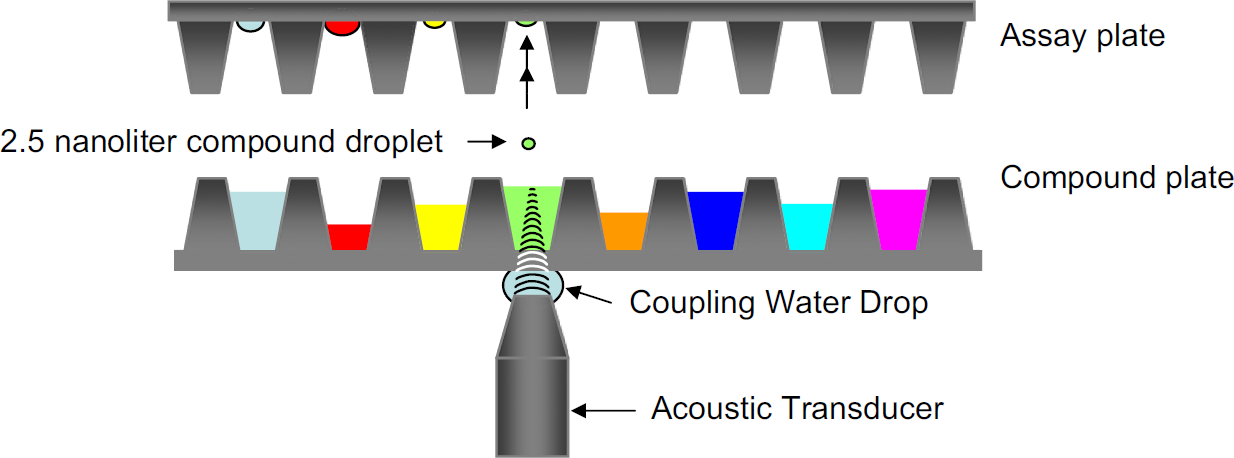
During the compound addition step, the noncontact acoustic-based liquid dispenser ECHO-550 inverts the assay plate upside-down before entering the ECHO-550 chamber. 1 This is done because the compound droplets are expelled upward from the compound plate into the assay plate (Fig. 1). As both the assay plate and compound source plate enter the ECHO-550 chamber, the plates are deionized to prevent any deleterious effects from static charges. These charges have the potential to swerve the miniscule 2.5-nL droplet away from its upward path upon ejection.
Acoustic Droplet Ejection (ADE) Technology.

Velocity 11 BioCel® 1200 system

The two main activities performed in ADE technology are audit and ejection. The purpose of the audit stage is to identify the location of the meniscus of each well of the 384-well compound source plate. The ejection stage is responsible for the actual dispense of compounds.
During the audit stage, each of the 384 wells of a prequalified compound source plate is examined. The acoustic transducer of the ECHO-550 focuses on each well of the source plate and emits a pulse of sound through a layer of water the size of a large drop. This same pulse continues onward to pass through the bottom of the polypropylene plate and finally through the DMSO which fills the wells of the compound source plate.
The purpose of the drop of water is to couple the acoustic transducer with the bottom of the source plate, thus providing a consistent acoustic medium between them. Via a pump system, this drop of water is constantly supplied, thereby maintaining contact between the transducer and the bottom of the source plate at all times.
The quantity of energy used in the audit pulse is purposely set low to avoid any ejection of a droplet during this stage. Known are the acoustic energy absorbing properties of the coupling water, the polypropylene bottom of the compound source plate and the DMSO in which the compounds are dissolved. Thus, the low setting of the auditing sound pulse required to penetrate these materials is a known quantity.
As the audit pulse travels from the transducer through these known entities, any energy that is not absorbed and reflected by these materials is echoed back at the surface (the meniscus) of the DMSO liquid. The reflected energy is measured by the acoustic transducer and the data are analyzed to calculate the location of the meniscus of each individual well. The data are stored for use in the next stage.
During the ejection stage, the acoustic transducer returns to each individual well of the source plate from which a transfer must take place. Again, it focuses on a well and then quantifies the amount of acoustic energy needed to eject droplets based on the data that were previously obtained during the audit stage. The increased level of acoustic energy that is emitted by the transducer results in the ejection of the required volume of compound. This ejection is done in increments of 2.5 nL/droplet.
Nuclei-Staining Cell-Based Assay
After a lengthy and thorough evaluation process involving plates made by various manufacturers, a polystyrene 384-well clear, flat-bottom plate was chosen for use in this MDS PharmaServices, Inc. proprietary method. During the week before being plated into 384-well imaging plates, each of the 297 cell lines was observed in their flasks for growth doubling information along with its respective cell morphology and size. This newly acquired information was used to assess a plating density for that cell line. Each cell line was plated using two densities. Smaller cells and those with slower growth rates were plated at higher densities. Larger cells and those with faster growth rates were plated at lower densities. For each cell line, the better of the two plating densities (judged to be those with the lowest coefficients of variation [CVs]) was chosen for optimal microscopy analysis and growth performance as measured by doubling efficiency.
Cells were automatically enumerated using the New Brunswick NucleoCount. Using Thermo Combidrops, a non-contact peristaltic-based liquid dispenser, a volume of 60 μL of cells/well in antibiotic-free proprietary media was plated by technical staff under the protection of a biosafety level 2 (BSL-2) hood. The plates were then incubated at 37 °C, 5% CO2, and 95% humidity for 24 h.
The compound source plates used in the ECHO-550 were Labcyte ECHO-qualified, 384-well, clear, flat bottom, polypropylene microplates (Catalog #P-05525). The reason for prequalification is due to the requirement of ADE technology to focus acoustic energy at the meniscus to properly eject each 2.5-nL droplet. Critical to the success of this focus is consistency in the acoustic-absorbing properties of the plate (i.e., no warping of plate bottom, material quality, equal thickness of well floors, and others). A volume of 60 nL of compound in DMSO was then added to each well of the assay plate using the ECHO-550. After compound addition, the assay plates were again incubated at 37 °C, 5% CO2, and 95% humidity for another 72 h.
There was no postaddition mixing or vortexing of the assay wells in an effort to avoid disruption of the monolayer and maintain cellular integrity. The compound addition was performed in a clean but uncontained environment.
At the end of the 72-h incubation, cells were fixed using a proprietary method and blocked with 0.5% bovine serum albumin and 0.005% Triton X-100. The contents of the wells were aspirated with a Biotek EL-405 and the cells were then incubated after the addition of primary antibody. All reagents were added using Thermo Combidrops or Multi-drops. After 30 min, the cells were washed with a Biotek EL-405 and secondary antibodies were added. (The results of the antibody staining are not covered in this article.)
After incubation, the cells were washed again and a deoxyribonucleic acid-binding (DNA) dye, 4,6-diamidino-2-phenylindole (DAPI), was added in phosphate-buffered saline to quantify nuclei. All centrifugation steps in this protocol used the Velocity 11 V-Spin.
One plate of Hela cells with reference compounds was assayed at the beginning and one plate at the end of each assay run as a quality control measure.
The percentage of control (POC), as a ratio of the treatment/control fluorescent intensity, was plotted versus the concentration of inhibitor and analyzed using nonlinear regression analysis (IDBS XLfit4) for the determination of an effective concentration value at 50% POC (EC50).
The CVs of Controls for each cell line was calculated from the Control well population on all plates of each of the two densities.
For each cell line, one density set showed a lower CV of Controls value than the other density set. For this evaluation, all the data from each cell line were chosen from the density set with the lower CV of Controls value.
Each cell line was exposed to several different test compounds. Each compound was run at 10 concentrations in triplicate. The CVs of the triplicates for the compound-treated wells were calculated from the standard deviation divided by the mean of the fluorescence of the control cells for each individual cell line. As compound concentration and inhibition of cellular proliferation increases, our experience has shown that for the most part, the CVs of the replicates decrease.
A Data Set was defined as 30 data points (three data points for each of 10 concentrations per compound per cell line) that comprise a curve demonstrating the EC50 values (see Fig. 2). The CV of each of the 10 triplicates was averaged and will be referred to as the Data Set Mean CV. In this study, a Data Set Mean CV value was used to assess one value per curve rather than 10 separate triplicate CV values per curve. This mean value was used as a general indicator to indirectly measure the overall accuracy of the compound addition and gauging overall replicate propinquity (i.e., how close the replicates are to each other).
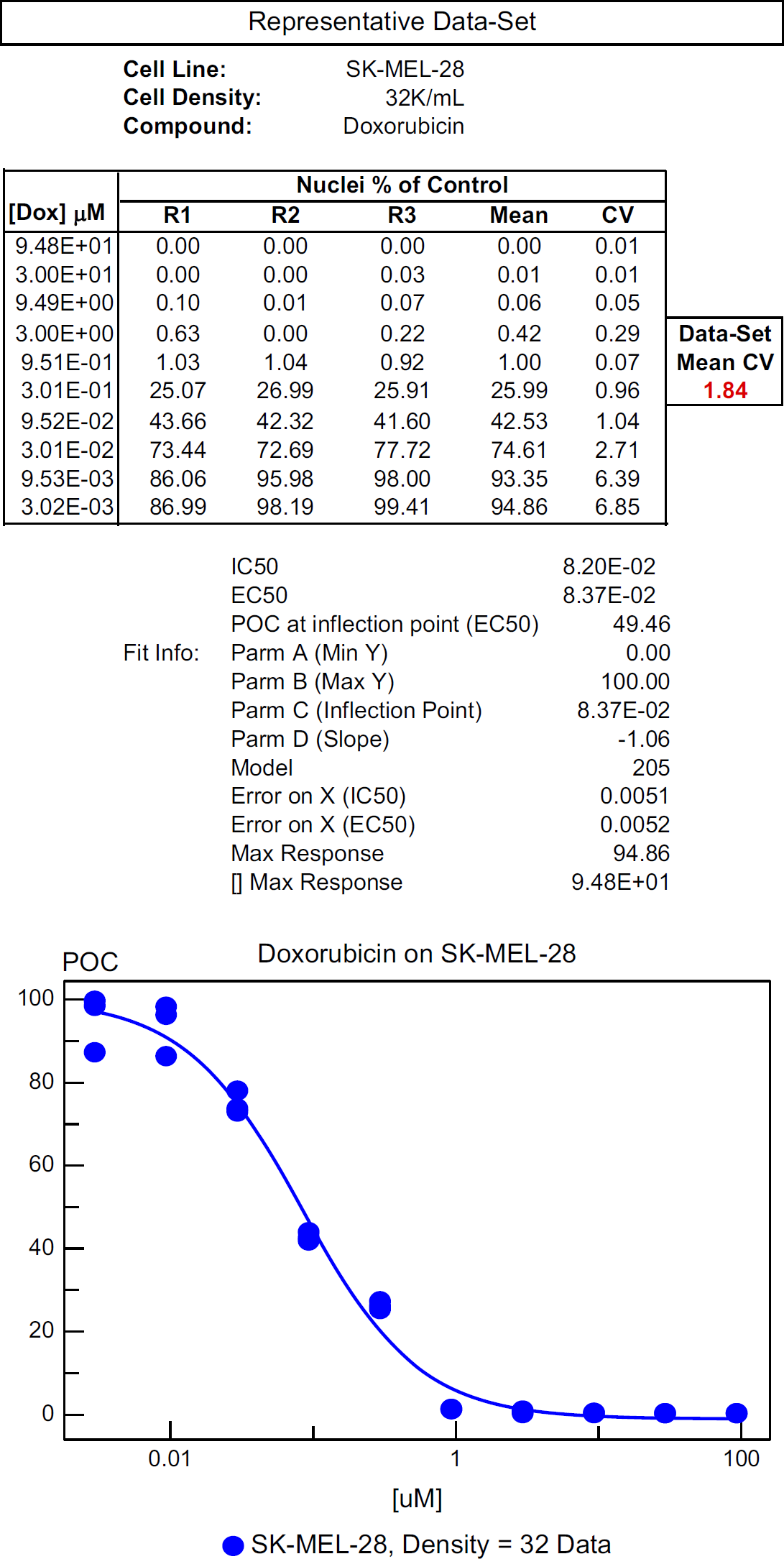
Example of a Data-Set.
The general criterion we use for deeming cell-based assay data as acceptable is having a less than 300% (3-fold) variability of the EC50 values of the reference compound acting upon the control cell line. With respect to the algorithm, growth performance was measured by doubling efficiency.
The performance metrics for the automation system include CVs of less than 20% for the control wells from the cell plating data. With regard to replicate propinquity, the Data Set Mean CV should be less than 20% to be considered acceptable. The CVs were divided into nonsuspension and suspension cell lines given the problematic nature of fixing, staining, and washing nonadherent cells.
Results and Discussion
An analysis was performed on the Hela cells which were assayed on the same compounds along with every batch of 15–17 cell lines. One plate of Hela cells was assayed at the beginning and one plate at the end of each assay run. The interassay variability (expressed as an EC50) of this assay for the same compound and cell line was 13% (0.13-fold) where n = 18. The intra-assay variability of this assay was 6% (0.06-fold) where n = 3.
Out of 227 nonsuspension cell lines, 217 had CV of Controls below 20%. Of the remaining 10 nonsuspension cell lines, the CV of Controls was above 20% (Fig. 3). From the resulting 6952 data sets within the nonsuspension cell group, 6776 (97.5%) had acceptable levels whereby the Data Set Mean CVs fell below 20% (Fig. 4). Of the remaining 176 Data Set Mean CVs deemed unacceptable, six nonsuspension cell lines with poor CV of Controls comprised over 70% of this subset of unacceptable values.
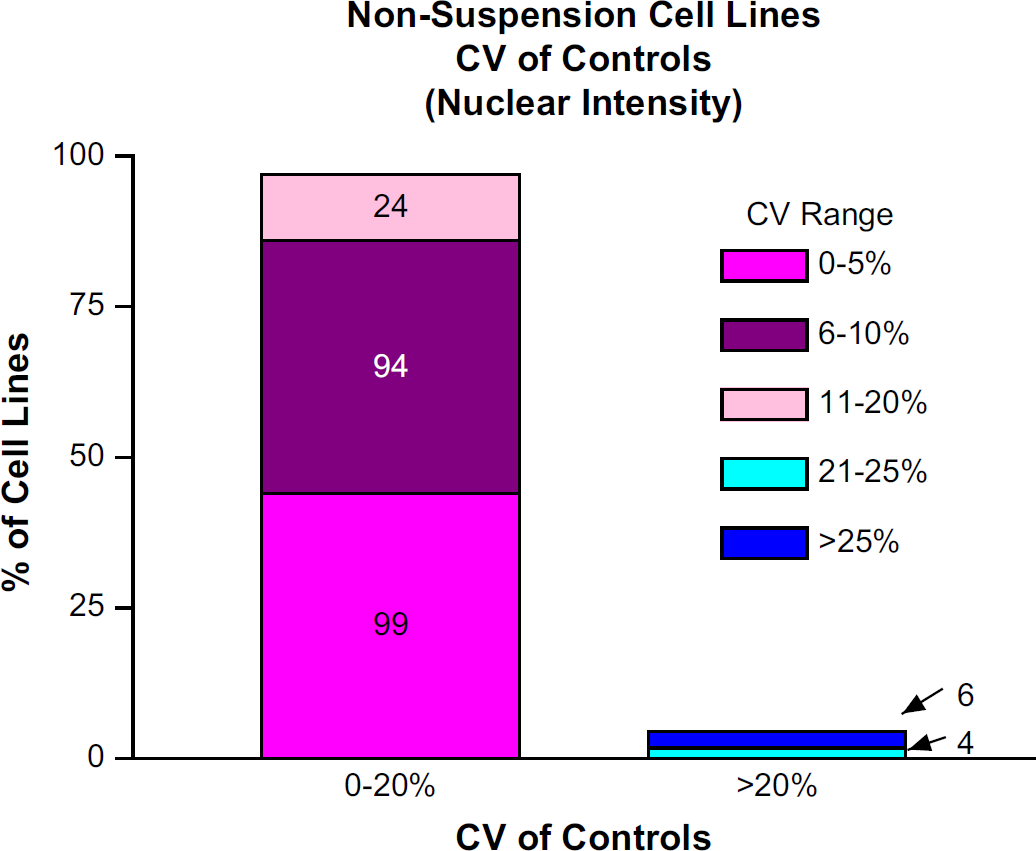
The percentage of cell lines with Mean CV of Controls <20% and >20%. The absolute number of cell lines is written within the bar chart.
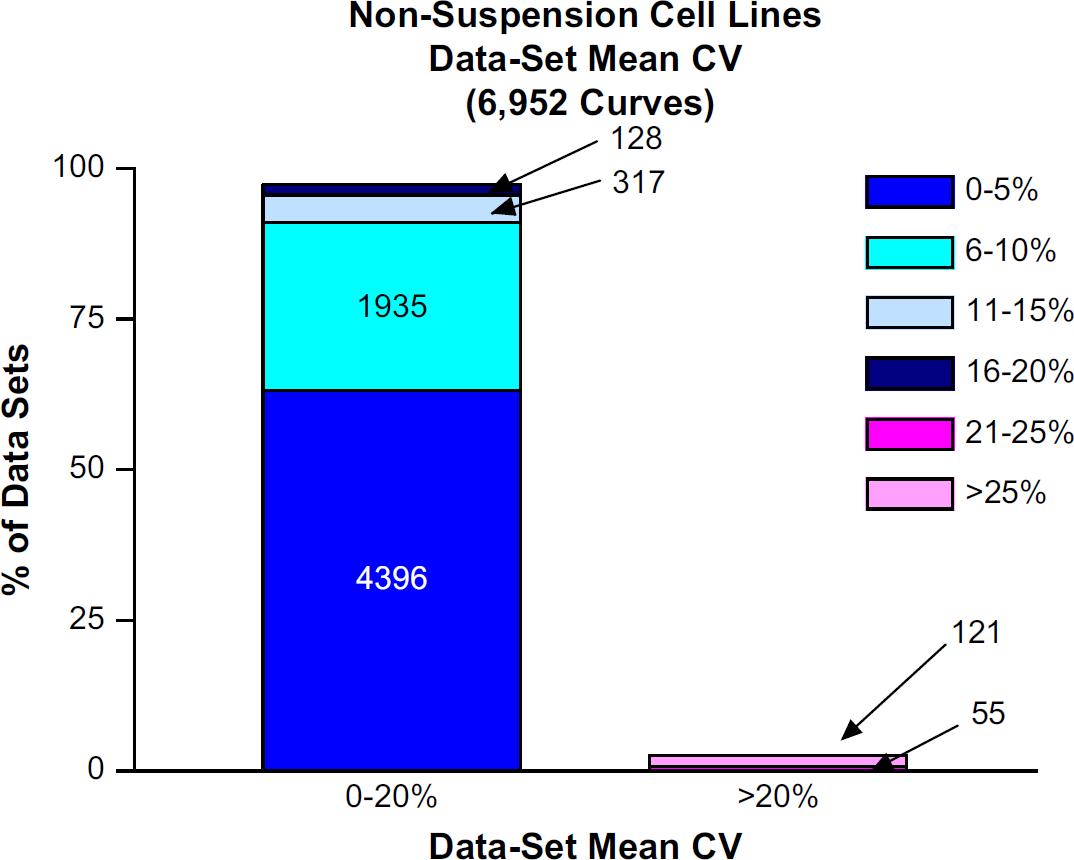
The percentage of data sets with Data Set Mean CVs <20% and >20%. The absolute number of data sets is written within the bar chart.
For the most part, replicate propinquity was directly proportional to the cell line CV of Controls. As the CV of Controls for each cell line increased, the likelihood increased that the Data Set Mean CVs would approach over 20%, the level of unacceptability (Fig. 5).
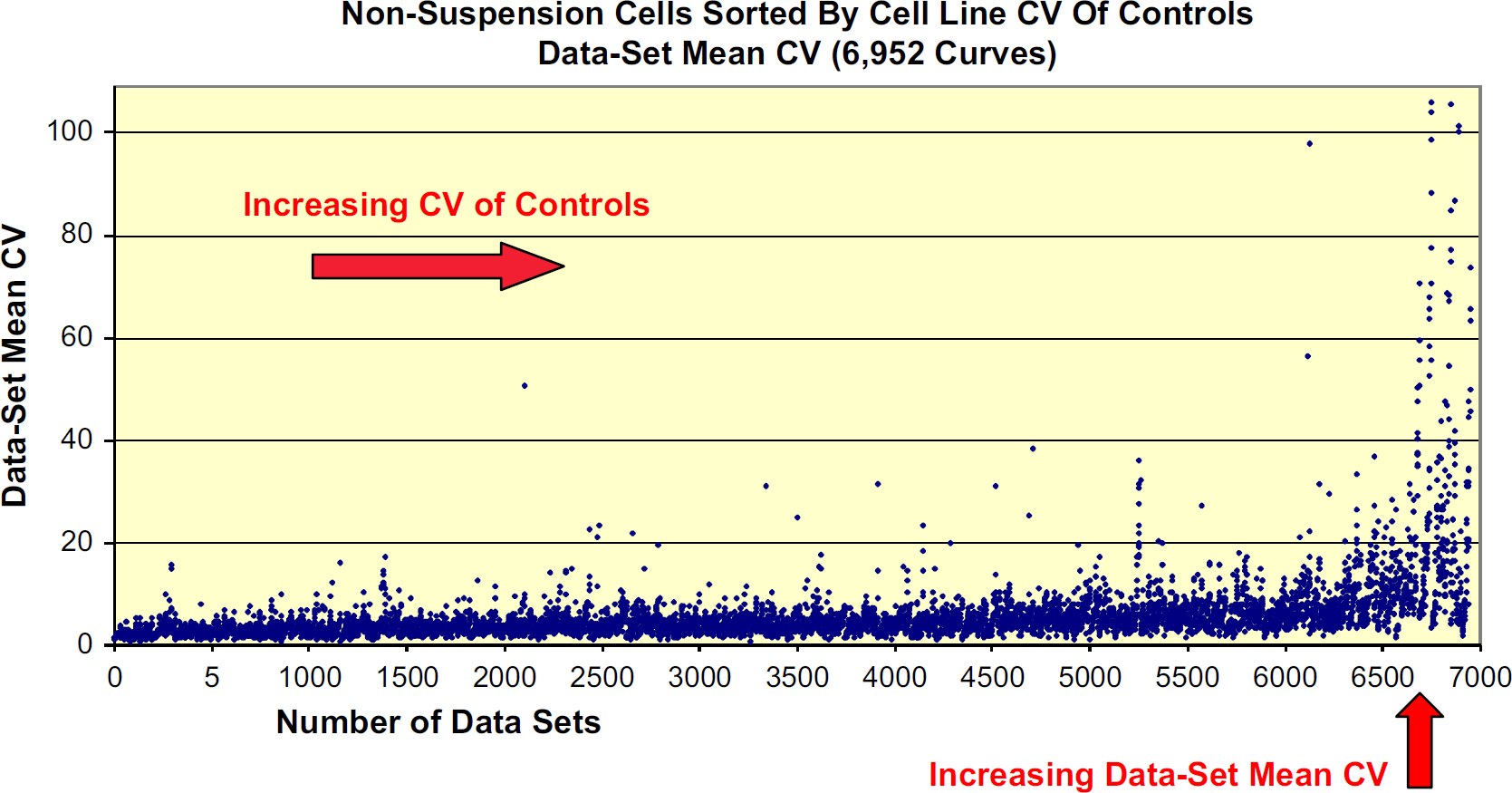
Each individual point represents a Data Set Mean CV value.
Of the 70 suspension cell lines, 47 had CV of Controls below 20%. The remaining 23 suspension cell lines had CV of Controls above 20% (Fig. 6). Here, it is probable that the numerous washes acting on the less adherent cells were responsible for the larger number of cell lines with poor CV of Controls. Out of 1758 data sets within the suspension cell group, 1357 (77.1%) had Data Set Mean CVs below 20%. Of the remaining 401 data sets, 21 suspension cell lines with poor CV of Controls comprised over 85% of this subset (Fig. 7). Again, for the most part, replicate propinquity was dependent on the cell line CV of Controls (Fig. 8).
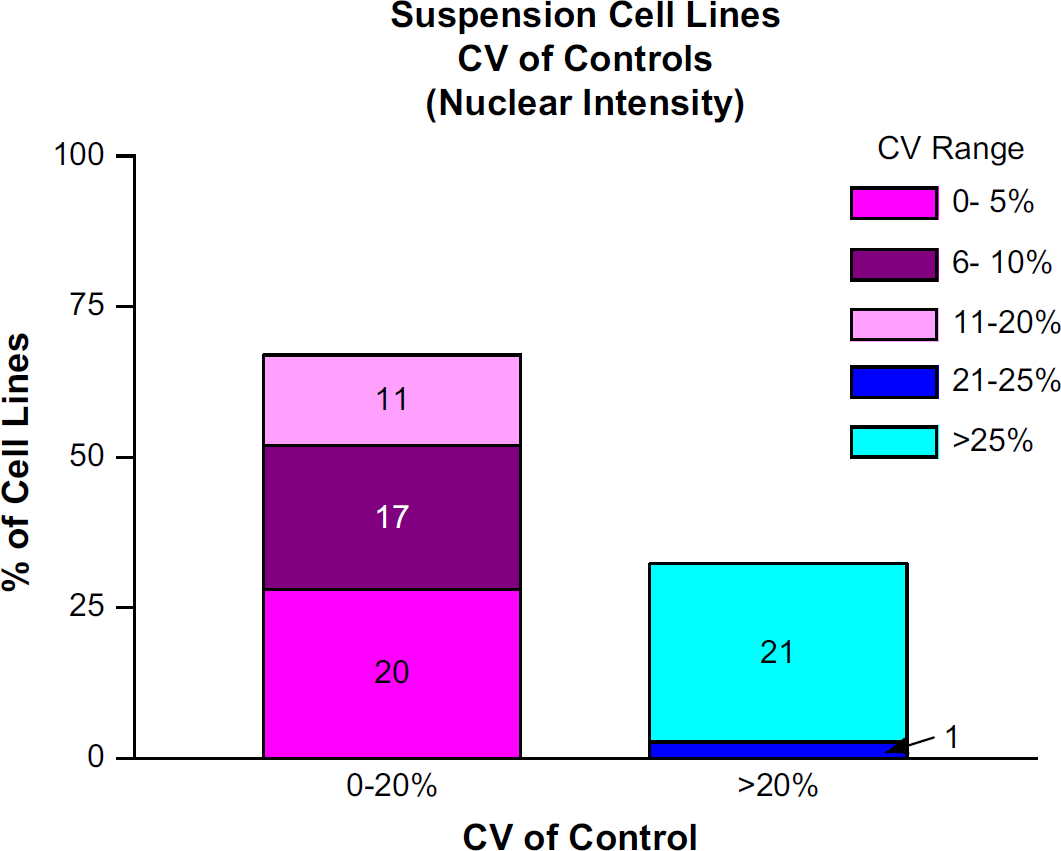
The percentage of cell lines with Mean CV of Controls <20% and >20%. The absolute number of cell lines is written within the bar chart.
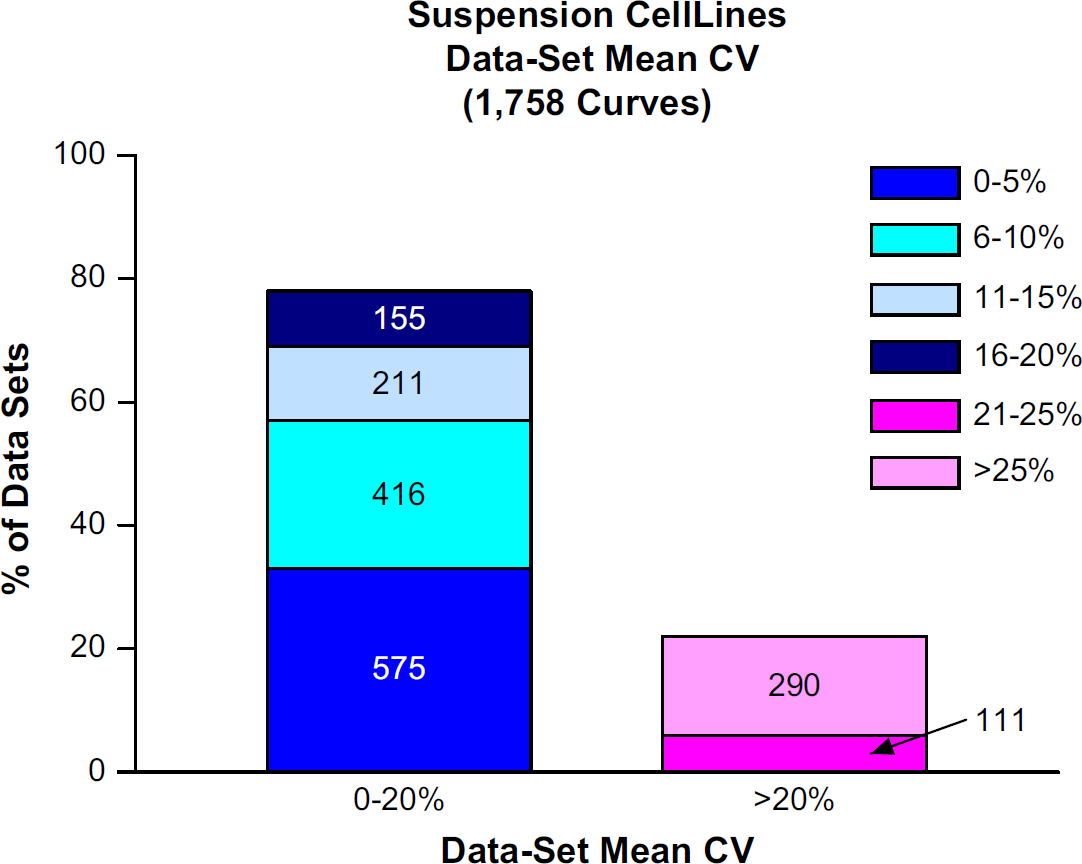
The percentage of data sets with Data Set Mean CVs <20% and >20%. The absolute number of data sets is written within the bar chart.
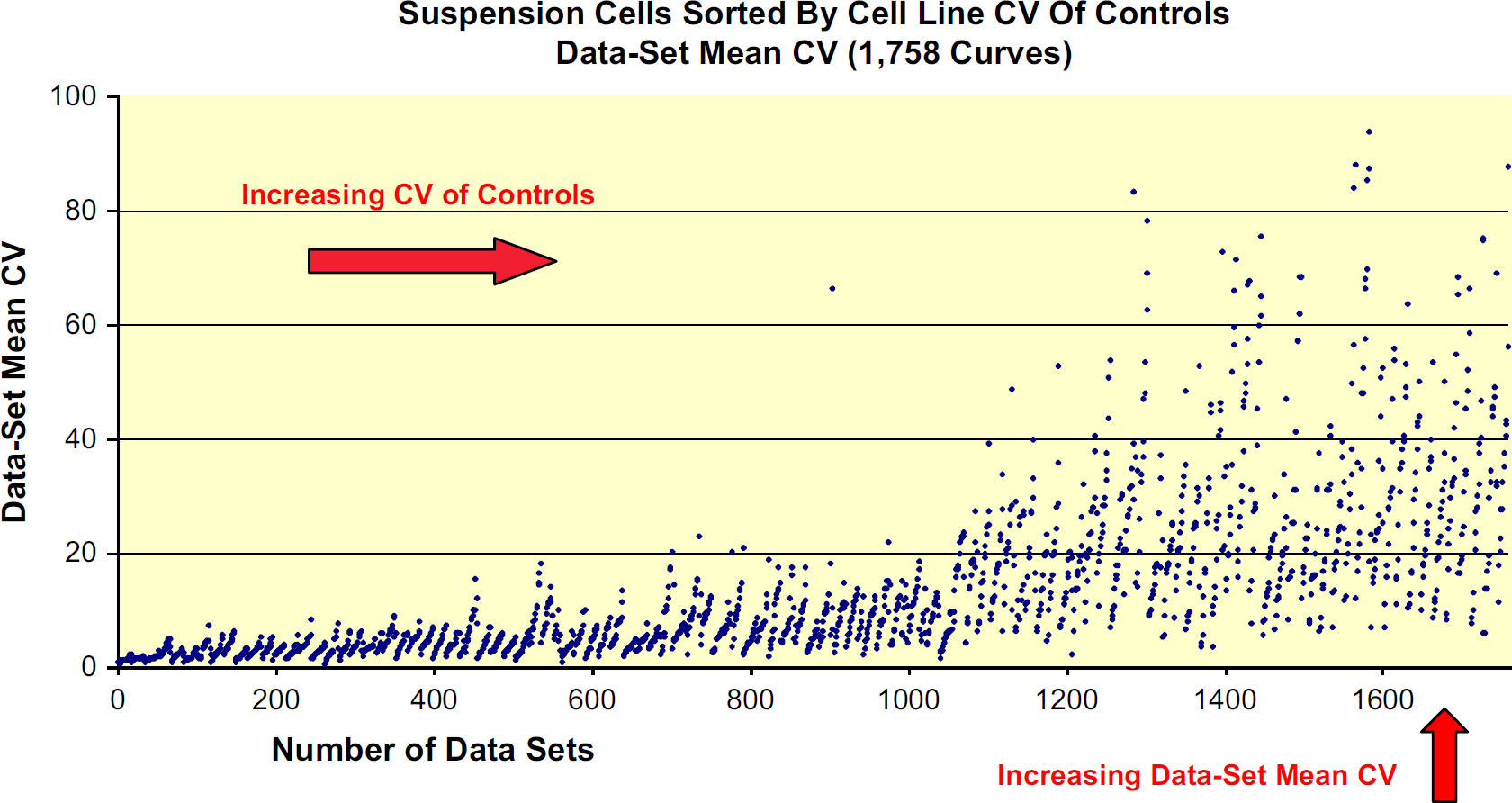
Each individual point represents a Data Set Mean CV value.
With regard to growth performance as measured by doubling efficiency, 90% of the 297 cell lines doubled at least once during the 72-h period. Fifty-three percent (53%) of the cell lines doubled more than twice during the same period, indicating the algorithm met reasonable growth parameters in plating the cells.
Although we lacked the knowledge concerning optimal cell density of each cell line, our algorithm provided a wide enough window to generate quality data. The TØ values allowed us to calculate a growth rate of cells while on the plate and the two densities allowed us some knowledge as to whether the number of doublings increased or decreased with increasing density. The approach of quantifying total fluorescent intensities instead of individual cells was superior for nuclear counts and allowed quantitation of “colony forming” cells along with nonadherent cells. The comparison of nuclei counting to fluorescent-intensity analysis gave similar EC50 values that ranged less than 2-fold.
The time frame in which to complete the cell-based project was 13 weeks. To complete this project within the desired time and cost interval, full validation of 297 cell lines was impossible. However, by developing the algorithm, we achieved a success rate in excess of 89% (< 20% CV of Controls), with only 15% of the cell lines needing to be repeated. Even adding a 15–20% repeat cost, it still results in an overall savings of over 100% relative to the estimated cost of prevalidating each cell line. Another benefit of the algorithm is obtaining the data sooner as a result of the short timeline.
None of the 297 cell lines run in this project became contaminated pre- or postcompound addition. Originally, we had 298 cell lines and had one contamination leaving 297 cell lines whose data are presented here. This contamination was attributable before the cell-plating step. Unbeknown to us at the time, these same infected plates were processed through the system early in the project (Week 2 of 13) and yet, for this project, no other cell line processed afterward was cross-contaminated. This lack of contamination for all 297 cell lines was confirmed by a well-by-well analysis of each 384-well assay plate with the GE Healthcare IN Cell Analyzer 1000 as the nuclei stain in the analysis identifies fungal, bacterial, and mammalian DNA.
Prefixation procedures were handled aseptically with the exception of the compound addition by the ECHO-550. This system exposed over 1200 unlidded, cell-containing assay plates outside the protection of a BSL-2 hood. In comparison to the standard manner for most cell-based assays whereby compound addition is performed inside a BSL-2 hood, this automated design has demonstrated in a very convincing manner that it can equally or better maintain sterility of cell-based assays that use 384-well plates and whose cells are grown for 72 h in antibiotic-free media.
The lack of contamination may be attributed to some notable features of this system. The expeditious plate lidding/delidding abilities of the Velocity 11 Lid Station and the high-speed plate transportation of the Velocity 11 robotic arm minimized the overall environmental exposure of the uncovered plated cells. It took less than 3 s to convey the microplate to the ECHO-550 stage after the lid had been removed. The deionization and inversion of the assay plate by the Labcyte ECHO-550 and its distinct noncontact ADE technology may have also contributed to the favorable outcome.
In Louis Pasteur's classical experiment where he demonstrated that the fermentation of nutrient broths is caused by the growth of microorganisms, he boiled nutrient broths and then exposed these broths to the environment. Air was admitted to the broths through a tube that curved downward and thus prevented dust particles from entering. The ECHO-550 first inverts the microplate causing the wells to face downward (Fig. 1) and then deionizes the microplate whereby dust may no longer be attracted to it. By doing so, the conditions of that classical experiment may be emulated in the sense that microbial laden dust is resisted from entering the microplate wells filled with cells in antibiotic-free media. Using ADE technology, there are no disposable tips involved in liquid transfers, thus that source of contamination is eliminated entirely.
Delivering compounds to assay wells in 100% DMSO limits the detrimental effects of compound precipitation. Use of the Labcyte ECHO-550 circumvents the need for a predilution step in aqueous buffers before its addition into assay wells. The compound addition process is slower (approximately 4min/plate) compared with some other liquid handlers. However, with regard to replicate propinquity, the quality of the data produced using the Labcyte ECHO-550 was outstanding when using our 20% criteria for Data Set Mean CV. Out of the 6952 curves generated in nonsus-pension cell lines, the overall mean of the acceptable Data Set Mean CVs was 5.05%. Out of the 1758 curves generated in suspension cell lines, the overall mean of the acceptable Data Set Mean CVs was 8.52%. Considering the 60 nL volume of the compound additions and that there was no mixing in the assay wells, this level of quality becomes even more remarkable.
With some liquid handlers dispensing in a cell-based assay, a bolus of compound in 20–100% DMSO may be delivered near the monolayer. This bolus causes a condition above the monolayer whereby the concentrations of DMSO and compound are higher than the final assay concentration conditions dictated by the experimental protocol. Until diffusion occurs, this temporary state can be deleterious to the cells in the immediate vicinity of the bolus. It can create a pattern that becomes evident on the microplate floor after plate washing whereby cells are aspirated after being loosened by the effects of the bolus. Such a pattern was not evident in any of the 297 cell lines used in the HCA assay as confirmed by the fluorescent microscope imaging system.
Use of the ECHO-550 prevents monolayer damage due to this bolus effect. The compounds are added while the assay plate is inverted. The 2.5-nL droplets are launched upward at the meniscus. While the assay plate is upside-down, the compound then diffuses and accumulates at the meniscus until completion of compound addition to the entire 384-well plate. When the plate is reverted upright, the compound (which is now accumulated at the top of the well) then begins to descend toward the monolayer of cells. As the compound diffuses downward, it distributes slowly and evenly, reaching the monolayer with a concentration of compound and DMSO at final assay concentration conditions (i.e., one of the 10 serial-dilution concentrations used for that compound's EC50 curve).
Because we eliminated the purchase of liquid handling tips and the associated hazardous waste disposal costs for those consumables, the cost savings on this project were estimated to be about $60,000.
System configuration mutability takes into account the disconnection and physical removal of equipment from the automation system which is then replaced by another instrument. The new instrument then requires reconnection, reframing, and a quality control test run to ensure proper function. Minimal system morphing was built into this design through the choices of equipment on the deck. These choices are responsible for maintaining daily maximum flexibility to execute the wide variety of in vitro assays we run daily in our lab.
To run a cell-based assay, some previously discussed components of this system's design preclude the costly addition of custom biosafety cabinets or other expensive laminar flow specialty structures. By avoiding such extraordinary biosafety constructions, we conserve precious floor space in the automation lab. This maintains efficient intralab mobility thereby preserving the flexibility to handle a wide variety of assays. This is accomplished by providing open space to shuttle guest equipment on and off the system. Intralab mobility is further advanced by the Velocity11 BioCel 1200 design whereby this entire system, which is on heavy-duty lockable casters, can be moved to any part of the laboratory with relative ease. The task of moving the BioCel 1200 is facilitated as movable overhead service delivery systems are in place. None of our equipment is bolted into the laboratory floor as are some commercially available automation docking stations which sacrifice laboratory adaptability.
Whenever the read-window (the range of time within which an assay plate can be read and produce reliable data) is long, then our philosophy is that it is better to remove that reader from a fully integrated system, thus maintaining system economy of space. It also improves assay throughput because there is one less time-consuming station to schedule in the process. This was the case with the HCA assay, whose read-window is indefinite and whose plate read-time is about 45 min/plate. Therefore, the two GE Healthcare IN Cell Analyzers 1000 were not integrated onto the main system. Instead, each was integrated with a Hudson PlateCrane robotic arm (models XL or EX) and the readings took place over a 24-h period of time.
Summary
During this project, we faced many challenges. We had a 13-week time frame to complete the project and dealt with formidable logistics involving management, cell biology, automation, information technology, and supplies. During the course of the assay, the microplates underwent numerous aspirations, washes, and reagent additions, all of which contribute to increased variability in the data. We dealt with unknown cell lines whose growth rates and optimal densities were unknown. We worked in a 384-well plate format that can be difficult for cell-based assays (so much so that some labs continue to use 96-well formats for many of their cell-based assays).
After recognizing the valuable contributions of the scientific team who worked hard on this project, upon review of the data, we concluded that the automation design, the performance of the automation system, and our algorithm were responsible, in large part, for producing data of superior quality for this project.
Acknowledgments
I would truly be remiss in not recognizing the extraordinary accomplishments of the following dedicated personnel without whom this cell-based project absolutely would not have been possible to complete: Kathleen At-well, Jamie Baumgartner, Cheryl Bogucki, Sidney Bounds, Ed Gontermann, Mary Gross, Mike Harges, Jim Hnilo, Rob Keyser, David Kirk, Karen Marcoe, Lisa Martel, Daniel Menard, Phuong Nguyen, Rod Shively, and Karen Yoshino. As a token of recognition and appreciation, this acknowledgment pales in comparison to the remarkable efforts, notable acumen in their fields, and distinguished professionalism manifested in the achievements of this talented and multidisciplinary crew. It was truly an honor for me to have been associated with this project.