Abstract
Enzyme-linked immunosorbent assay (ELISA) is a standard immunoassay to estimate protein concentrations in a variety of samples. An automated ELISA procedure was established that allows the measurement of the concentration of numerous proteins in a small sample volume. The automated ELISA procedure was tested by measuring the concentration of brain-derived neurotrophic factor in serum samples from humans.
It was shown that the reproducibility of protein concentrations in three assay plates within one automated run was greater than 92%, within independent automated experiments greater than 96%. Automated processing with a maximum capacity of six plates and 240 serum samples per run showed an average interassay precision of 91 %. Serial dilutions of randomly chosen sera displayed significant linearity of recovered concentration.
In conclusion it can be stated that the automated assay provides high flexibility with highly reproducible results and has several advantages compared to the manual method, including less operator dependence and faster sample throughput. The investigated ELISA test system seems to exhibit analytical characteristics indicating further clinical application. (JALA 2007;12:219–29)
Keywords
Introduction
Enzyme-linked immunosorbent assay (ELISA) represents one of the most commonly applied methods providing a simple, rapid, safe, and cost-effective way of protein determination.1, 2 Nevertheless, the process suffers from being highly time consuming and requires a series of liquid supplementations, washes, and incubations before detection leading to both, manpower requirements and an increased risk of failures due to the human factor. This is of special concern when laboratories have to process regularly a high number of samples. Automation of the ELISA technique and application of such methods for clinical and pharmaceutical laboratory efforts constitute, therefore, a significant part of research.3–6 Thus, a fully automated sandwich ELISA in a 96-well format was established using a Beckman Coulter core system and peripheral devices (Fig. 1) supervised by SAMI3.5 software (Sagian Automated Method Interface, Version 3.5, Beckman Coulter) for optimal scheduling of sample processing. As a test setup, the concentration of brain-derived neurotrophic factor (BDNF) in serum samples was determined. BDNF, a member of the neurotrophin family, is a widely distributed growth and differentiation factor for neural cells that can be found in high concentrations in the human brain but also in platelets of the peripheral blood.7–9 Altered BDNF production and secretion have been proven in a number of diseases. For instance, neurodegenerative disorders are known to be associated with decreased BDNF levels in the brain, for example, Alzheimer's and Parkinson's diseases.10–12 There is also evidence that enhanced local BDNF production in the lung contributes to neuronal hyperreactivity and pathologic bronchoconstriction in asthma. 13 Even in more emerging diseases such as diabetes or obesity, there seems to be a correlation with distinct BDNF serum concentrations. 14 An automated BDNF–ELISA is therefore useful for clinical diagnostics and research. To establish the automated protocol, a commonly available BDNF-ELISA test kit (R&D Systems) was used.
Robotic instrument setup.
System Asset
The automation core system that was used here (Fig. 1) consists of a robotic arm (optimized robot for chemical analysis, Beckman Coulter) that is able to deliver required labware to defined positions. As a central device, there is a liquid handling station, providing several pipetting tools (Biomek2000, Beckman Coulter). Moreover, there are several peripheral devices such as an automated washer (ELx405 select, Biotek), a barcode reading and labeling device (Microplate Print &Apply, Beckman Coulter), six shaking units (Variomag Teleshake, H + P, Oberschleißheim, Germany), a hotel with four stacks configured for tip boxes and deepwell plates, one stack for microtiter plates and a dark hotel (also usable as Kryohotel –80 °C) including two stacks for plate incubation (AIGmbH), and two reading device (Fluostar Galaxy, NovoStar, BMG Labtech) integrated. The device integration of reader, shaker, and dark hotel was performed in-house (AIGmbH, Rostock, Germany). The core system is driven by the SAMI3.5 superordinated software (Sagian Automated Method Interface, Version 3.5, Beckman Coulter) for optimal scheduling of sample processing in a medium throughput way.
Materials and Methods
Sample Preparation
Blood samples from cannulation of a cubital vein of 18 voluntary healthy probands were collected into additive-free containers to obtain serum samples (samples were taken several times for each volunteer during a period of 1 year). Blood was placed at rest on ice for 60 min to allow clotting and degranulation of the thrombocytes leading to the release of BDNF from platelets to serum. Subsequently, sample containers were centrifuged at 2000 × g and 10 °C for 15 min, supernatant was aliquoted and stored at –80 °C until measurement.
Elisa
For the quantitative determination of BDNF concentrations, a commercially available ELISA kit was used explicitly following the manufacturer's instructions (R&D Systems, DuoSet ELISA Development Kit). Antibodies, streptavidin-labeled Peroxidase (POX), and recombinant BDNF were supplied with the kit. Chromogen solution (3,3′,5,5′-tetrame-thylbenzidine [TMB] substrate buffer [H2O2], R&D Systems) and stop solution (2 N H2SO4) did not need further dilutions. Phosphate-buffered saline (PBS)/1% bovine serum albumin was used as reagent diluents (RD) with the exception of the capture antibody which was exclusively diluted in PBS. Serum samples were diluted 1:50 for standard procedure and 1:40–1:160 for the serial dilution. The standard maximum concentration was set to 2000 pg/mL and the standard minimum concentration was 62.5 pg/mL. All standard concentrations were measured in duplicates; samples and blanks were measured in two to eight replicates, depending on the assay layout. The calculation of protein concentrations from measured extinctions was performed automatically by a Fluorescence Reader (Fluostar Galaxy, BMG Labtech) facilitating a serial dilution standard curve.
SAMI Methods
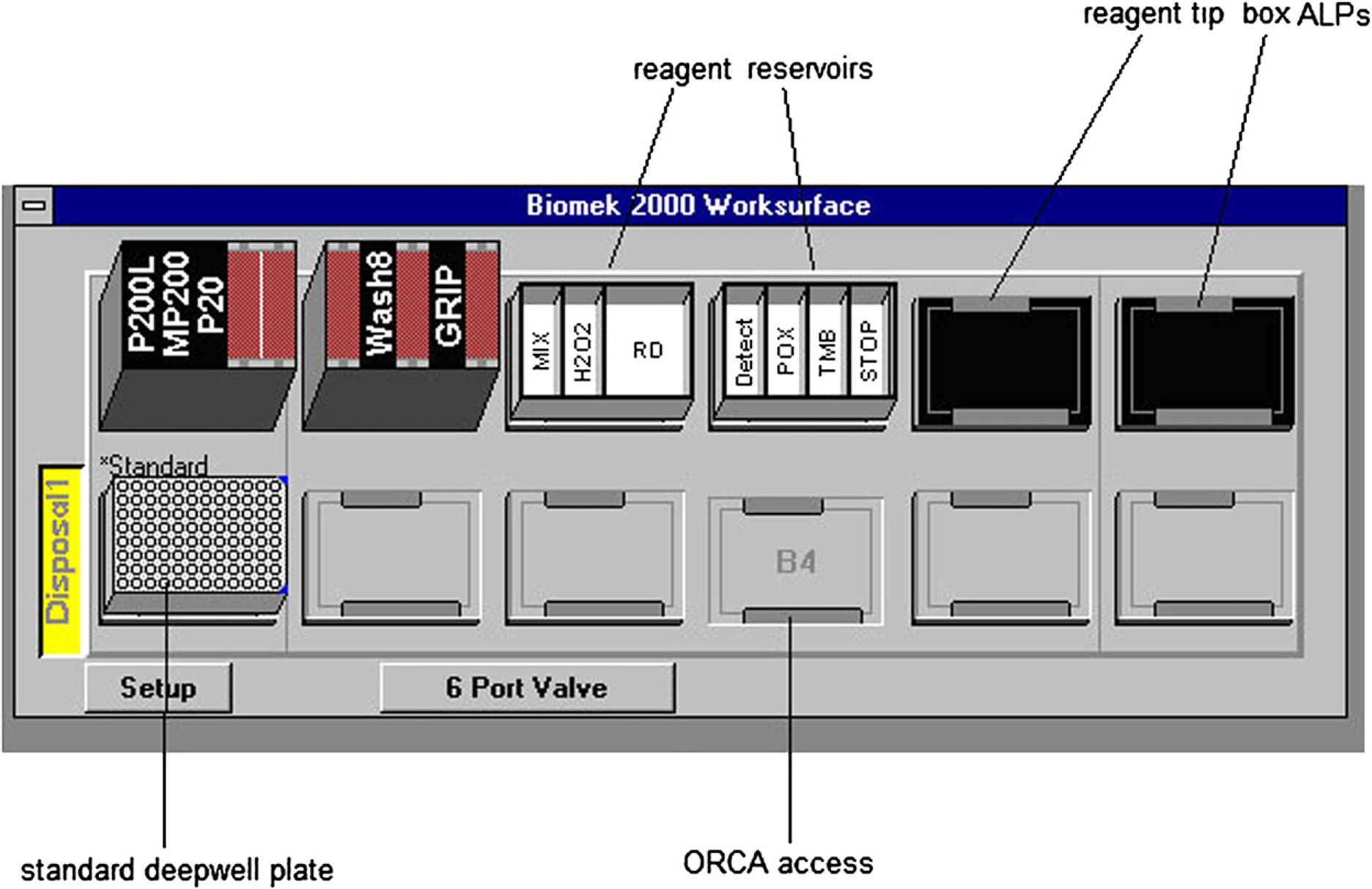
Hardware setup Biomek2000 workstation. ALP, Automated Labware Positioners.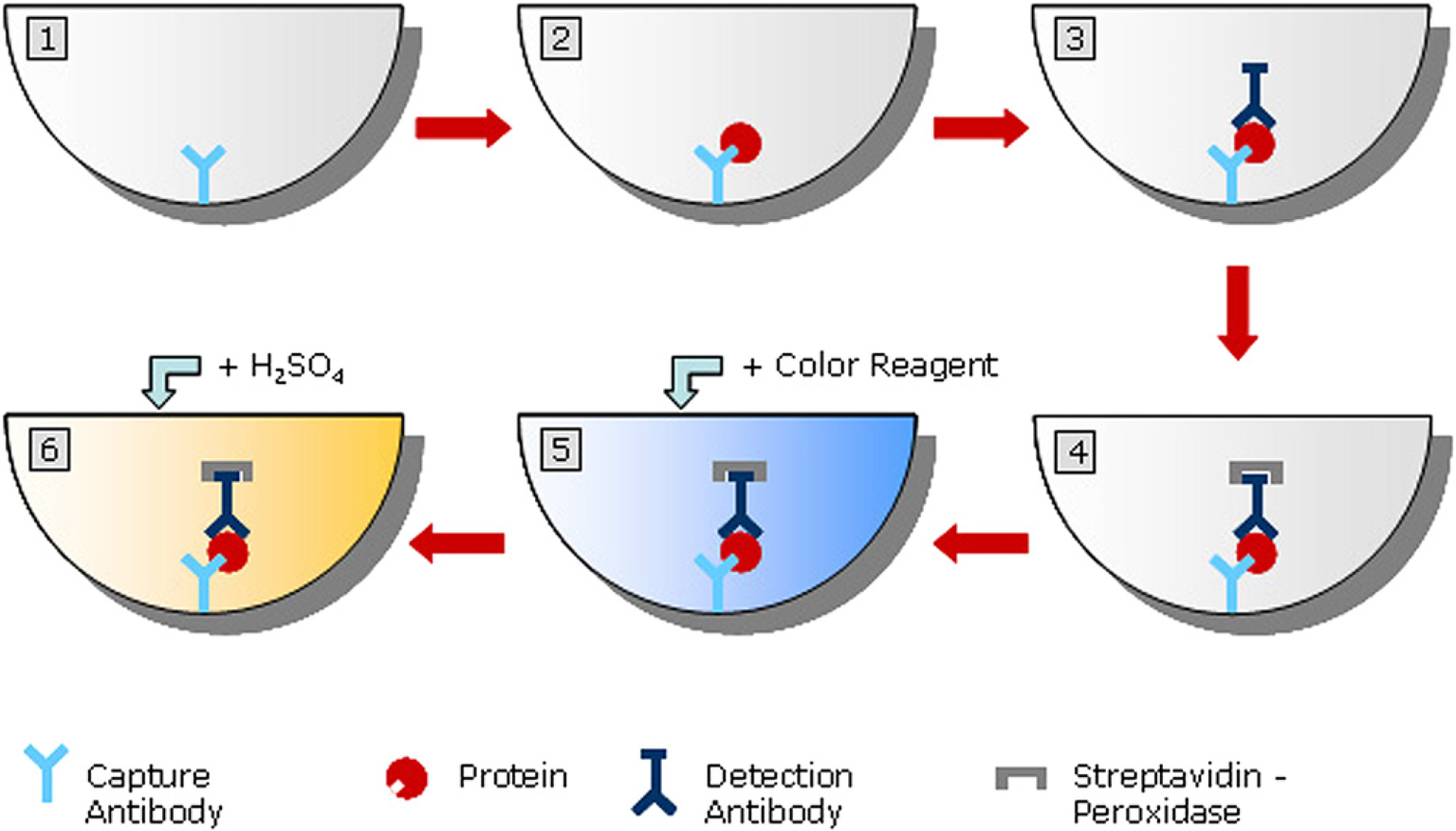
The subsequent ELISA processing comprises six distinct methods on the Biomek2000 workstation combined to one SAMI protocol (see Fig. 3, and for a schematic overview Fig. 4).
Workflow of an automated ELISA. Schematic overview of a sandwich ELISA. 1: coating with capture antibody; 2: capture of antigen; 3: Secondary antibody (e.g., biotinylated) binds to the complex; 4: streptavidin-coupled peroxidase binds to biotinylated-Fc of secondary antibody; 5: addition of enzyme substrate and resulting enzymatic reaction; 6: reaction stop with 2 N sulfuric acid and measurement (photometer).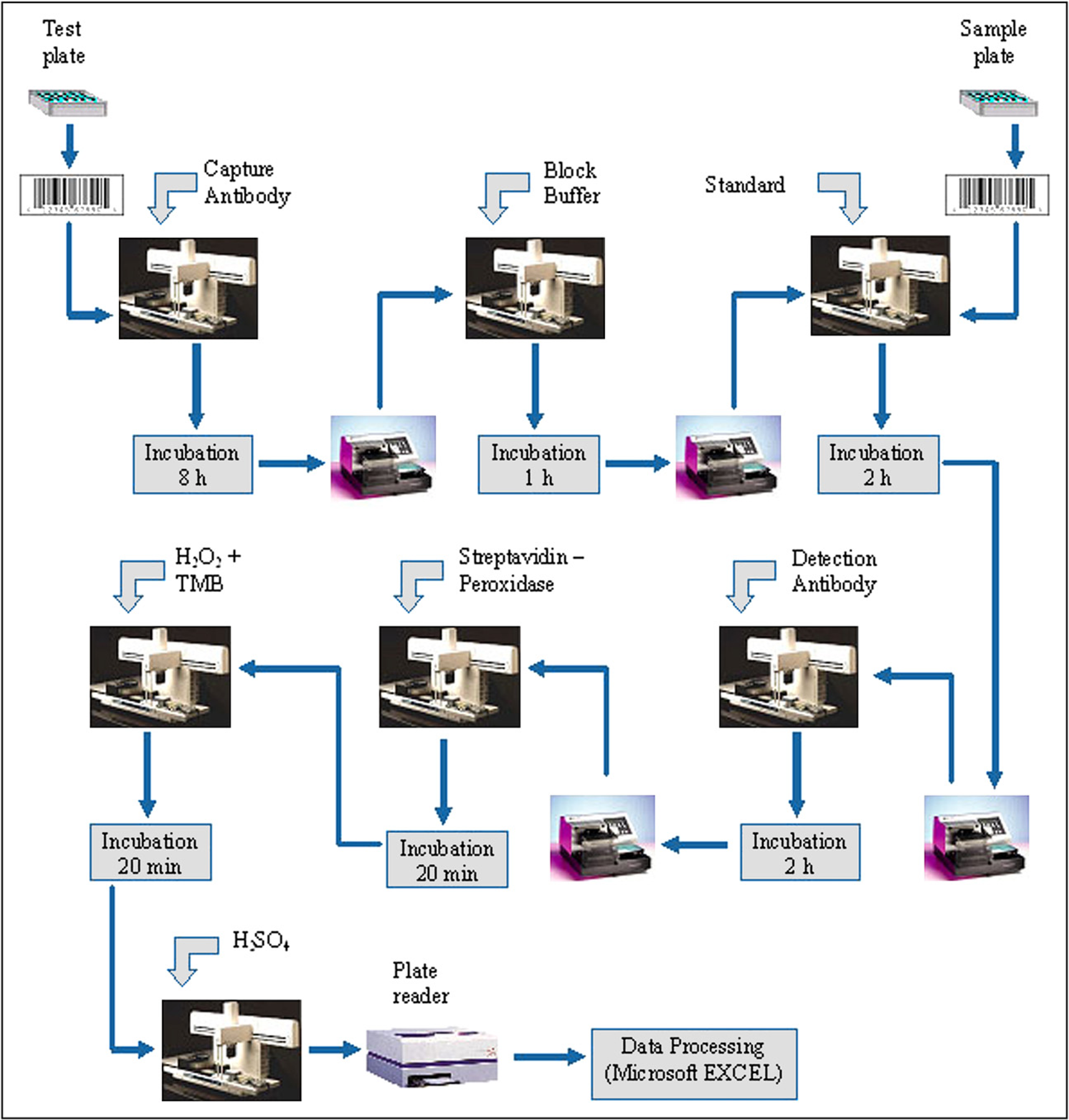
Transfer of blocking solution (RD)
Standard and sample transfer to ELISA plate (standards for all six plates were diluted manually and provided in a deepwell plate)
Addition of biotinylated anti-BDNF antibody
Addition of streptavidin–HRP conjugates
Mixing of TMB and H2O2
Transfer of mixed solution to test plate
Addition of stop solution.
After each transfer step, plates were placed on one shaking unit for 30 s for optimal reagent distribution. The ELx405 was used to wash all plates in-between transfer steps with provided wash buffer. Absorption readings were taken with a Fluorescence Reader (Fluostar Galaxy) at 450 nm and 590 nm. The values obtained at 590 nm served as correction factor that was used to subtract plate imperfections.
Each experiment was accompanied by an online visualization software (in house development, AIGmbH), which generated layout-dependent “heat maps” and calculations such as average absorption (AV), standard deviation (StDev), and
These three independent methods were attached to run in one time line.
Results
Running Time and Scheduling
The running time of one assay plate was determined to 6 and a half hours. Due to optimal duling of families by SAMI3.5, the whole process for six assay plates is completed within 8 hours and 45 minutes (see Fig. 5). The running time of the coating method varied between 5 min and 45 min, depending on the amount of assay plates. The effort for automated sample preparation was dependent on the assay layout (two or four replicates, serial dilution of samples) and number of sample plates, but did not exceed 1 and a half hours.
Optimal SAMI3.5 scheduling process of six ELISA assay plates. Explanations of the scheme are given in white blocks, representative for the first assay plate.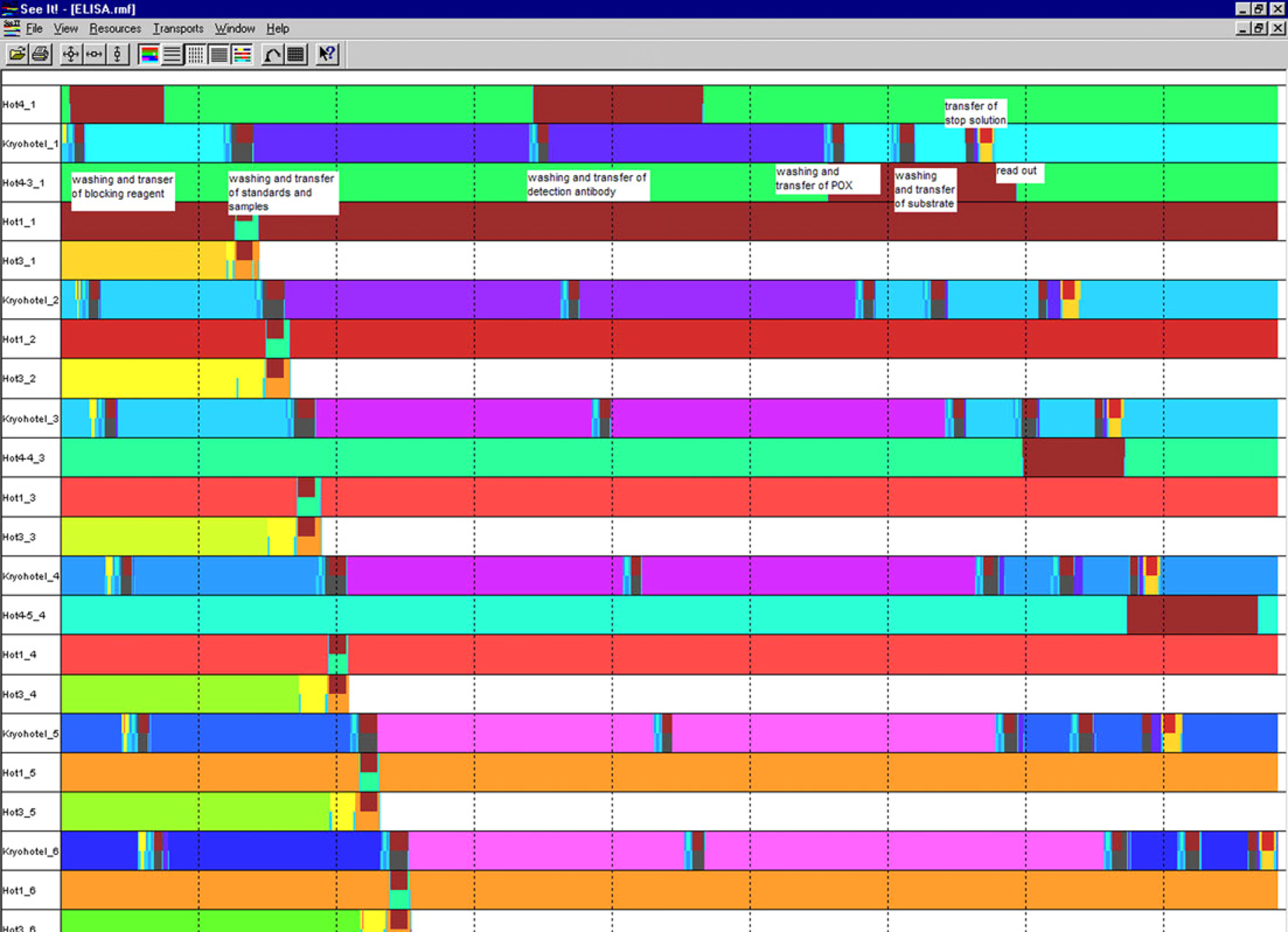
Consistency of Results
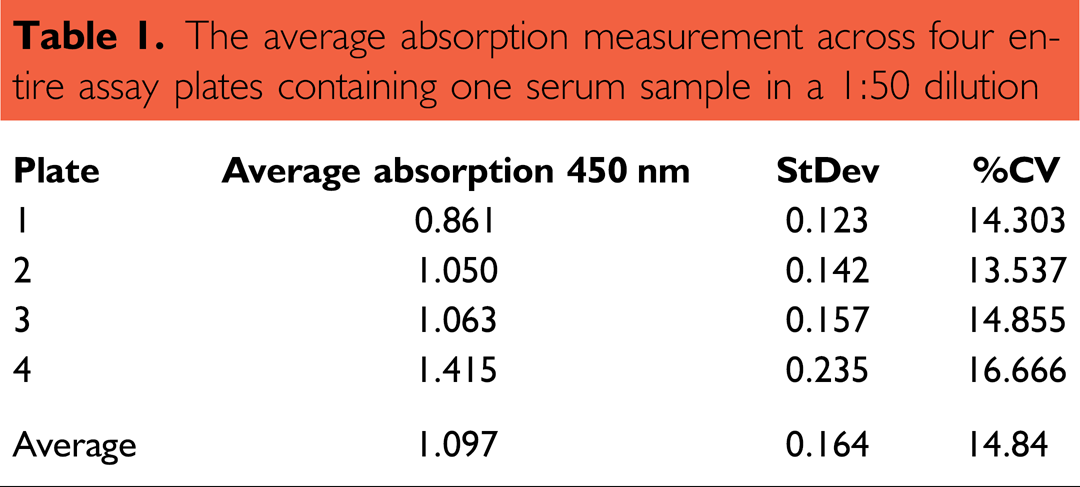
A major concern while setting up ELISA assays is about the result consistency. To elucidate the test system with regard to this problem, the assay was first run with one sample with a constant concentration (1:50), including four blank controls across the entire plate (intra-assay variation). The average intra-assay variation was determined to be between 13.5% and 16.7% (four plate replicates, see Table 1). The signal to noise ratio
16
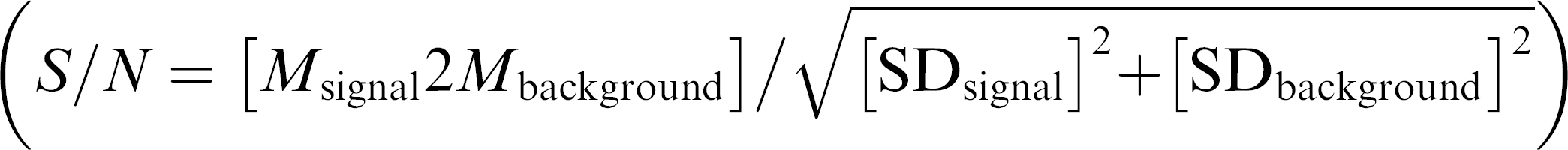
The average absorption measurement across four entire assay plates containing one serum sample in a 1:50 dilution
was approximately 3:1 (2.8:1) and thus within the expected range.
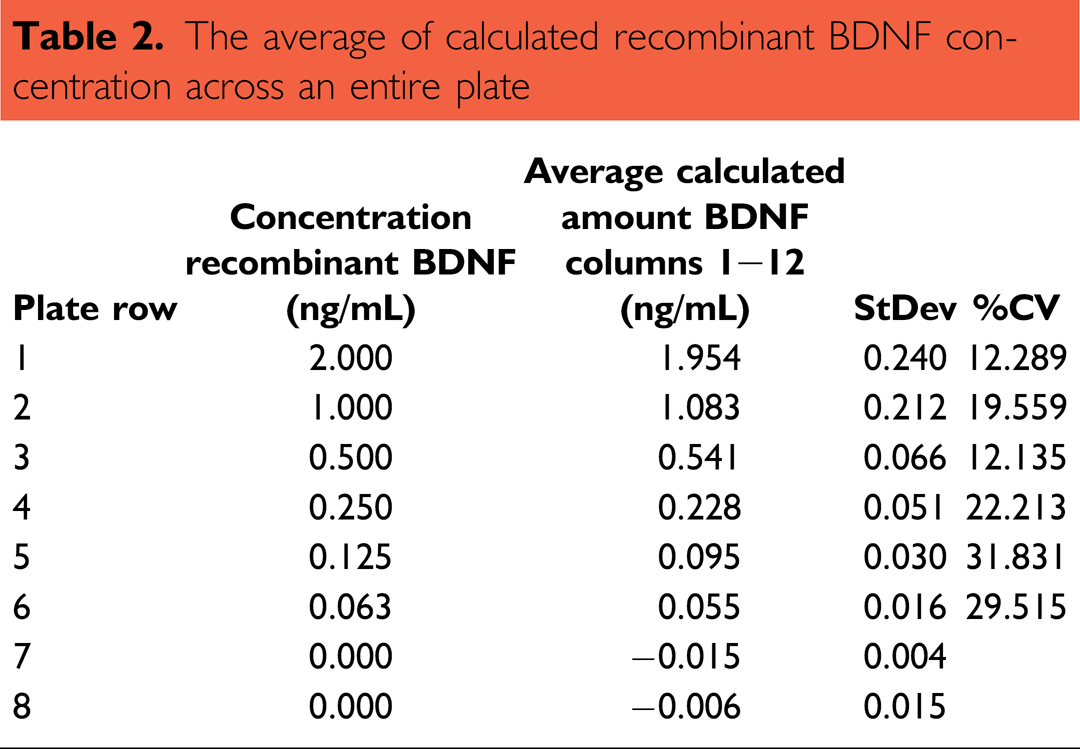
The consistency of the exact BDNF amount was analyzed by running a standard dilution across an entire plate (Fig. 6, Table 2). The standard was provided in one row of a deepwell plate, and then transferred onto the assay plate. Here, the absorption measurements and calculated concentrations of BDNF were in excellent correlation (
The average of calculated recombinant BDNF concentration across an entire plate
Comparison between defined BDNF concentrations (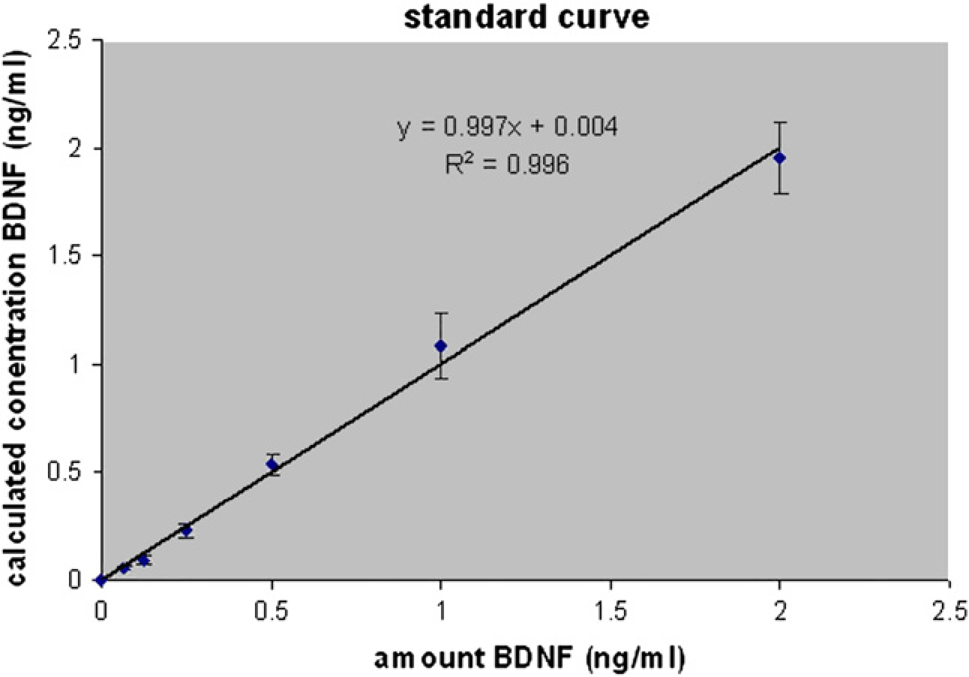
Comparison Between Manual and Automatically Generated Results
To estimate whether the data from an automated procedure are comparable with a manually performed ELISA, the assay was done manually step by step. Here five serum samples with a constant concentration (1:50) in four replicates were compared. Subsequently, the results were aligned with data achieved by the automated workflow.
The manually performed ELISA displayed a mean variation factor of 17.8% whereas the automated procedure showed a variation of only 14.6%. Both assays displayed a correlation coefficient greater than 0.93, and the calculated serum concentrations were comparable to each other (see Figs. 7 and 8).
Comparison of estimated BDNF serum concentration resulting from manual and automated ELISA procedure. P1–5: randomly chosen serum samples. Comparison of standard curve resulting from manual and automated experiments.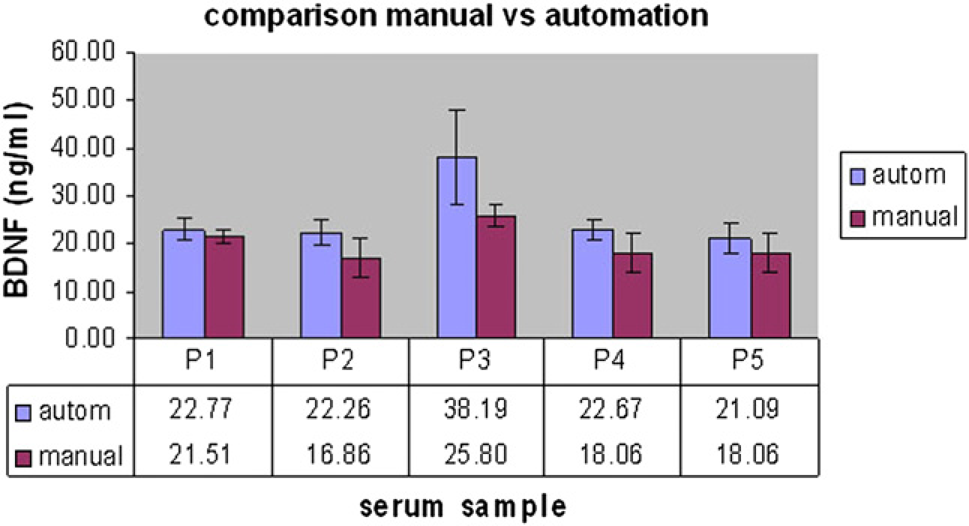
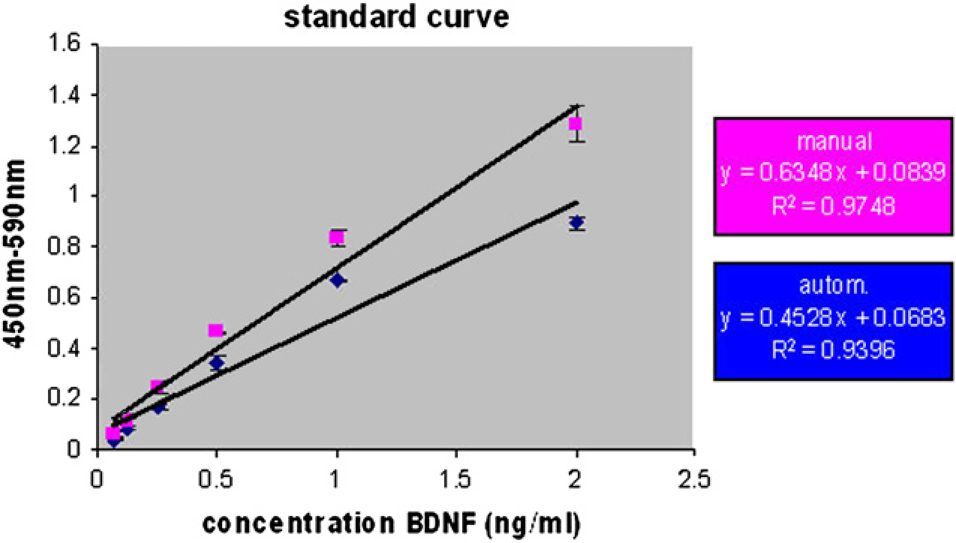
Reproducibility of Data
To investigate the reproducibility of the data that have been received from an automated run, one run with three plates and three different 1:50 in RD-diluted serum samples/plate was performed and the same experiment was repeated on another day (Table 3A). Here, high reproducibility within one automated run was obtained (intra-assay variance). Although the precision varied between serum samples, it did not exceed 11% and the resulting average variability was lower than 8% (see Fig. 9 and Table 3B). The estimated BDNF serum concentration of the same samples between two independent automated runs was even better (ranging between 92% and 98.9%).
Reproducibility of concentration measurements over three plates within one automated run, and comparison of two independent automated runs 1, 2, 3: randomly chosen serum samples; 1_1: assay plate_experiment
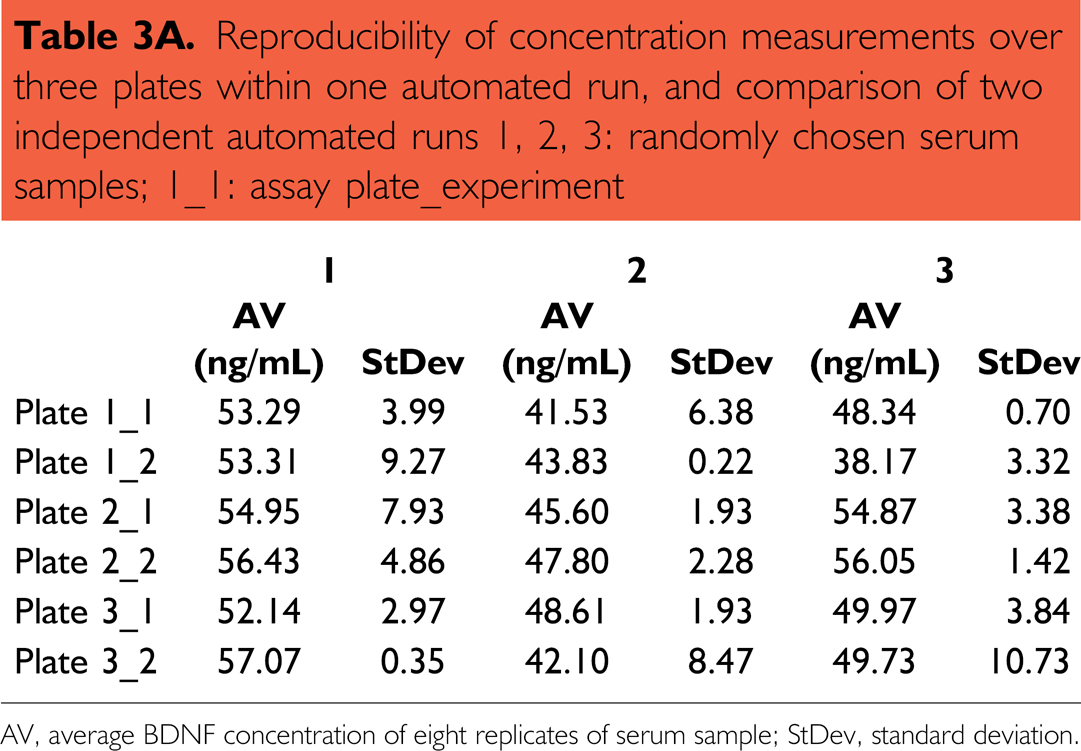
AV, average BDNF concentration of eight replicates of serum sample; StDev, standard deviation.
Inter- and intra-assay variance of three serum samples/three plates and two experimental runs
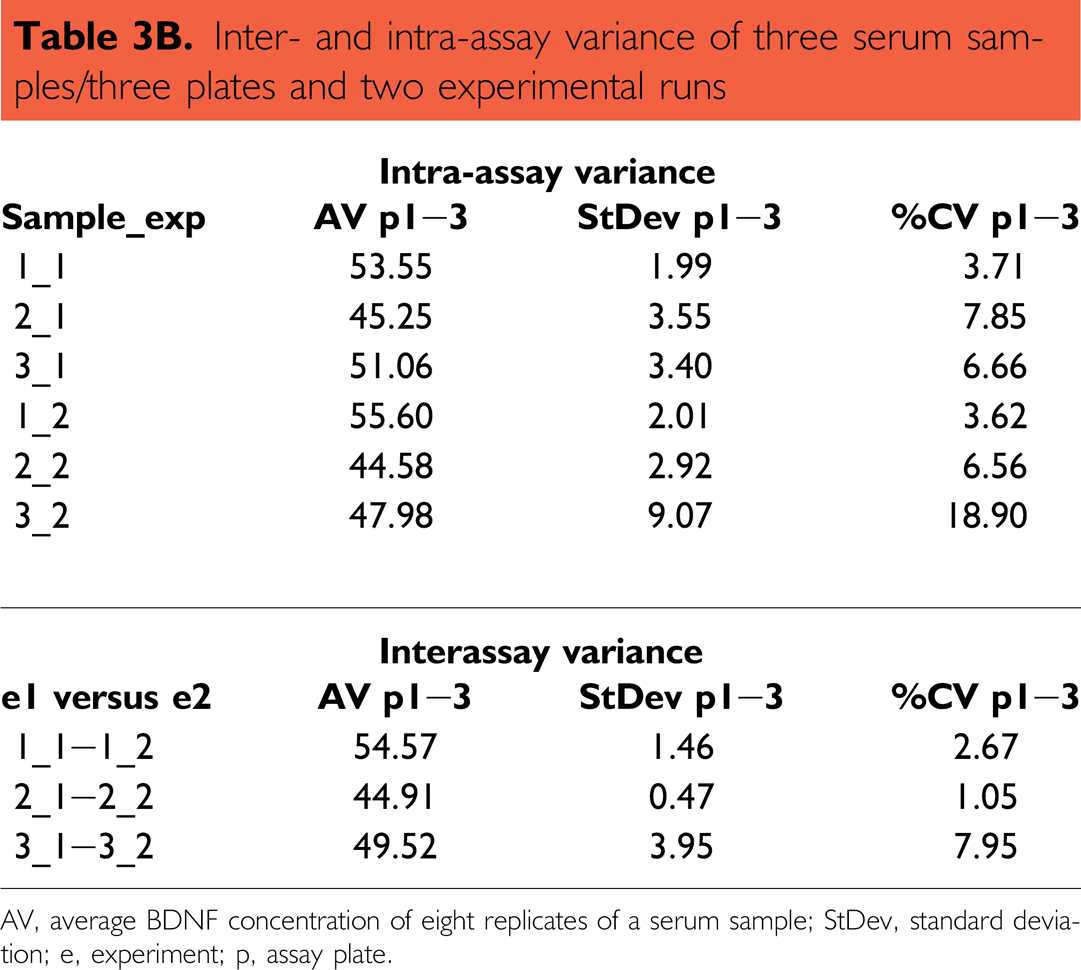
AV, average BDNF concentration of eight replicates of a serum sample; StDev, standard deviation; e, experiment; p, assay plate.
Reproducibility of concentration measurements over three plates within one automated run, and comparison of two independent automated runs.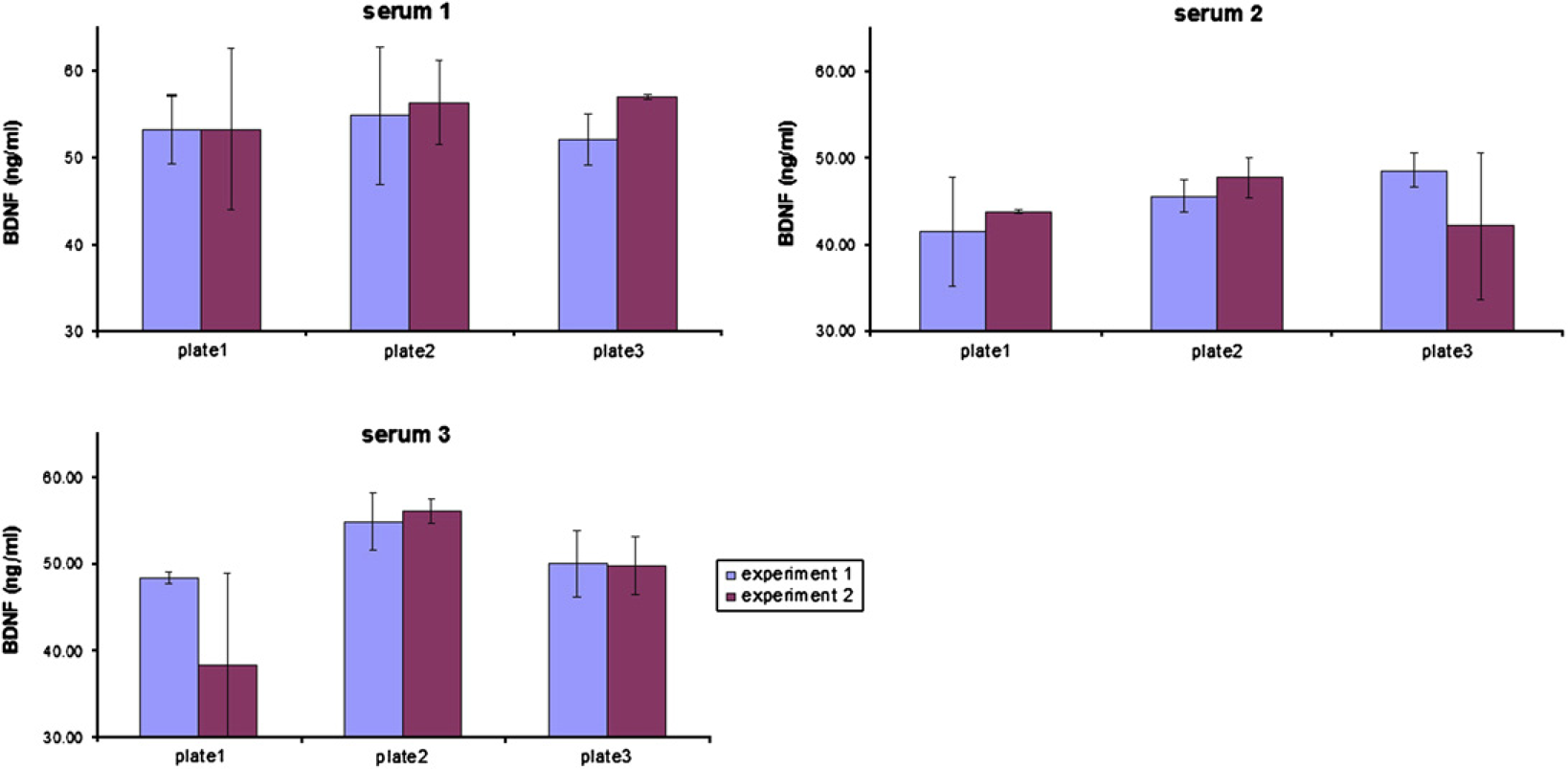
Stability of Results at Maximum Capacity
To estimate the stability of the assay results at maximum possible capacity, the ELISA method was performed with six assay plates. Here two different protocols were used:
two sample replicates and
four sample replicates.
The automated run of six assay plates with 1:50 in RD-diluted serum samples in duplicates displayed an intra-assay variability ranging from 1.8% to 21%. The average variation was 9%. The same experiment performed with four replicates have shown an intra-assay variability ranging from 7.8% to 14.8%. The mean was also nearly 9% (see Table 4). These results are in consistence with previous received data (see point 4). Even the automatically calculated
Intra-assay variability

Linearity of the Serial Dilution
To estimate the linearity of BDNF concentrations in different sera, a protocol for automated serial dilution was set up. Here, six assay plates were run, each consisting of three serum samples in eight replicates (all together 18 serum samples). The serial dilution resulted in a final dilution of serum samples in RD of 1:40, 1:80, and 1:160, respectively. The results are shown in Table 5 and Fig. 10. The analysis of the row data showed that the automated run is in acceptable linearity within one serum sample over three dilution steps. The StDev varied between 7% and 35% (see below). The average deviation across all sample dilutions was 14.6%. The average deviations of the distinct serum dilutions ranged from 19.8% for 1:40 sample dilution to 22.6% for 1:160 sample dilutions. However, there are some exceptions with relatively high deviations (e.g., sample 5 or 13), caused by some outliers (for a summary, see Table 5).
Serial dilution of randomly chosen serum samples
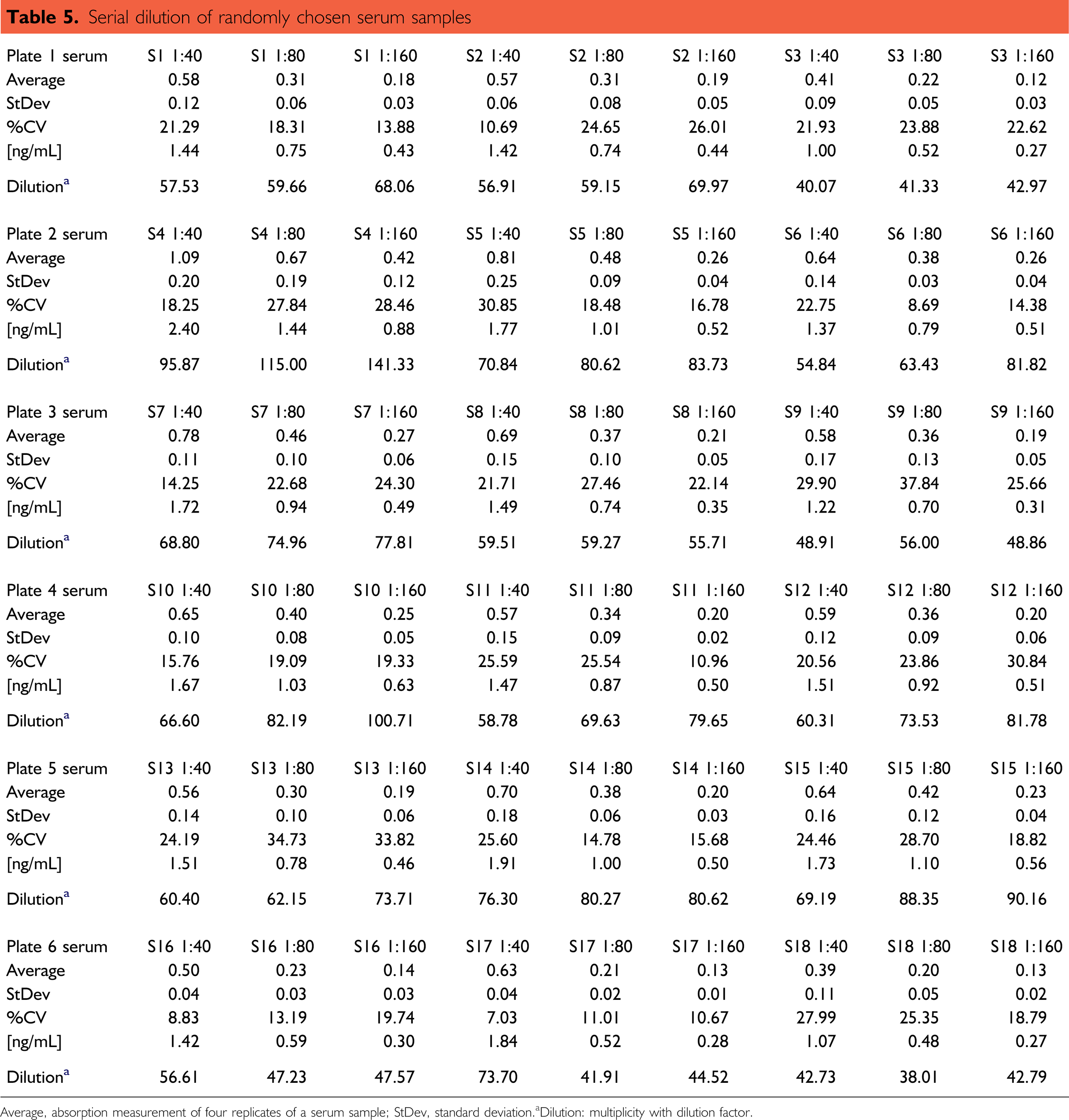
Average, absorption measurement of four replicates of a serum sample; StDev, standard deviation.
Dilution: multiplicity with dilution factor.
Linearity of automated sample dilution during one automated run of six assay plates. Bars: serial dilution of randomly chosen serum samples. Corresponding data are provided in Table 5. Graph: linear regression of standard curve. Error bars: standard deviation of two replicates.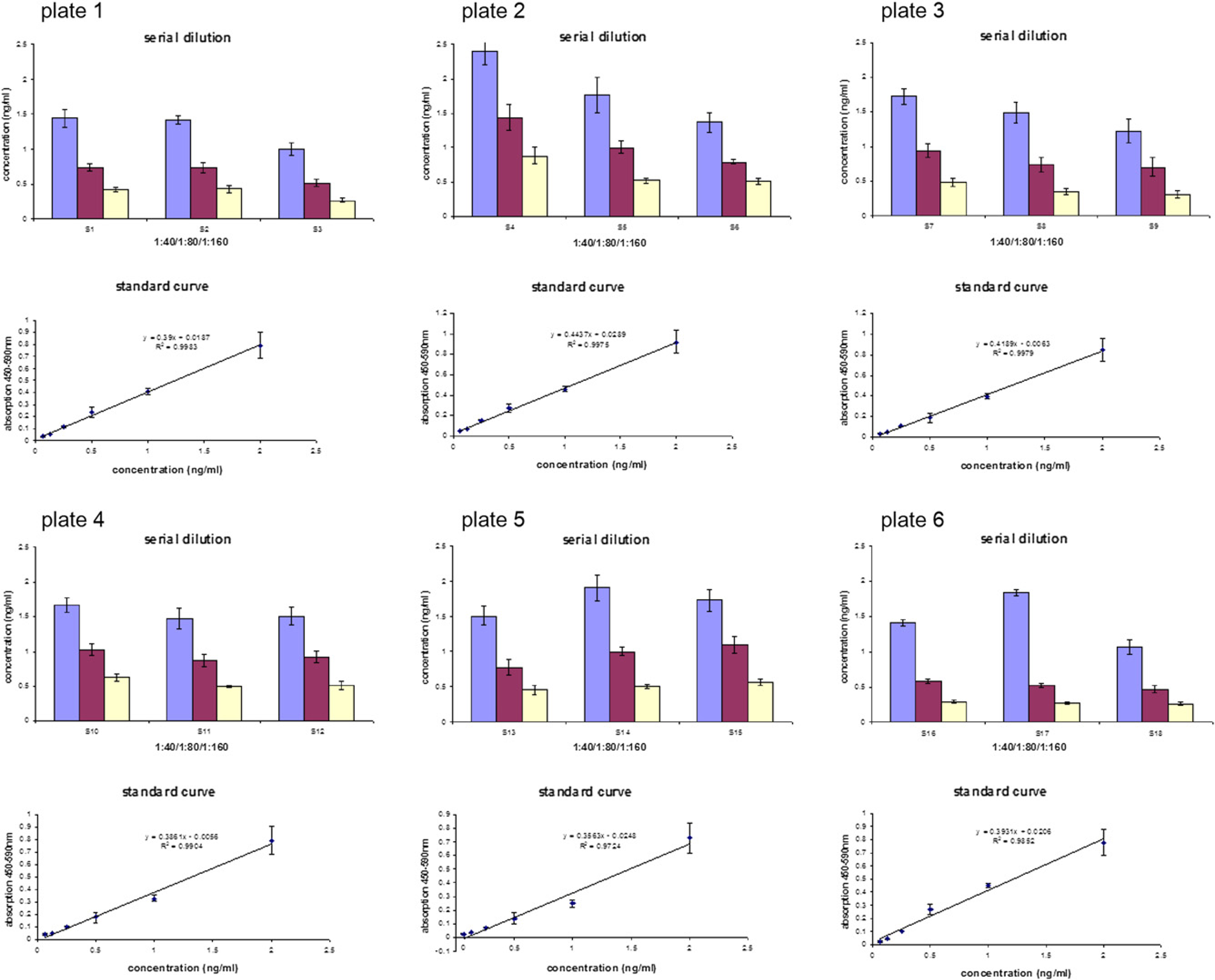
Discussion
Due to the multiple applications of ELISAs in the fields of clinical diagnosis and biological research, a fully automated test procedure on a Beckman Coulter robot facility was developed. Because of its increasing relevance for the clinical diagnosis and research, the BDNF was chosen as a test target for the system.16, 17 The protein was detected via a commercially available ELISA, which was actually specified for manual operation.
Using the robot system, one is able to analyze 1–240 samples in replicates, provided in one to six assay plates per run, with a running time of 6 and a half hours to 8 hours and 45 minutes, respectively (see Fig. 5). After the assay is started, there is no need for manual interference, thus it is a truly walk-away setup. Nevertheless, due to the variety of assay layouts, this system also provides the flexibility which is demanded in the area of scientific and clinical applications.
The automated process was validated and the received data were reproducible. StDev of processed samples ranged from less than 1% to 21%. Additionally, there was no reproducible difference between assay plates in a series. Discrepancies arising from processing on varying daytimes where not detectable, indicating a reliable and stable procedure (see Table 4 and Fig. 9). The average intra-assay variance was 9% (see Table 4). According to recently published data, the intra- and interassay precision of automated immunochemical assays varied dependending on the discrete assay and target, but was ranging from 1.5% to 7% 5 or to 23% coefficient of variation (CV). 18 Putting these results into consideration, it can be deduced that the values represent dynamic biological ranges.
The native and the recombinant BDNF protein was detectable in all automatically handled serum samples. The calculated protein concentrations in test serum samples were comparable to those from manually operated assays (see Fig. 7).
The measured serum concentrations ranged from 3.27 to 95.9 ng/mL, the mean was 47.8 ng/mL. These data were in agreement with recently published data obtained from the largest study conducted on neurotrophin concentrations. 19 According to this study, the average BDNF concentration in serum samples of healthy donors is 22.6 ng/mL, ranging from low levels (1.9 ng/mL) to 51.5 ng/mL. Nevertheless, some of the test sera showed increased levels of BDNF. The maximum determined concentration was 95.87 ng/mL, which means up to about twofold in comparison to literature values. Additional investigation on sample aliquots of the same donor resulted in comparable calculated BDNF concentration. Whether these unexpected values were caused by a failure during manual preparation or if this donor represents a specific phenotype cannot be answered here, because donor samples were given anonymously.
Using a serial dilution method for sample processing, StDev increased faintly with the dilution factor. But deviations of discrete samples were sometimes far exceeding the accepted range of agreement (over 30%). A close look at the row data revealed that StDev resulted from single mistaken data points (outliers), for example, one or two outliers of eight replicates. High deviations are always a problem in automation processes, especially when promoting miniaturization or more throughput. To decrease StDev and to increase throughput, high-throughput screening technologies are working preferably with commercially available one step assays (add reagent and read out). 20 Even on the overwhelming market of commercially available ELISA kits and products, companies try to develop and offer ELISA technologies with a reduced number of process steps, for example, precoated plates, no washing steps, and so on. However, an ELISA remains a multistep assay and therefore maximum throughput is kept lower to achieve reliability and good assay quality.
Another problem in automation of processes is the instability of solutions over longer periods of time. 21 While setting up the assay it was shown that the BDNF standard degraded with increasing detention times leading to decreased extinctions during ongoing experiments. The aberration of standard values of plate one and six achieved 35% (data not shown). Eventually, other standards are more stable and this should be further evaluated using comparable assays. The problem can be avoided by decreasing the room temperature (below 20 °C) and refreshing the standard after half of the assay plates. Generally, to avoid such problems, it will be necessary to integrate a cooling automated labware positioner on the Biomek2000 deck.
The data presented here demonstrate that the automated robot-based workstation can be used for executing an ELISA under stringent conditions as a method to detect and determine immunological relevant parameters such as BDNF serum concentrations. The establishment of an automated ELISA has led to a stable and accurate application providing results that can be considered equivalent to manually operated assays. The results for recombinant BDNF standards and for BDNF concentrations that have been derived from healthy donors were reproducible for the automated run. As intended, the automation of the assay has shown to be a time-effective tool for future medium throughput analysis of protein concentrations.
Since this complex assay protocol was established, it is considerable to perform a broad range of ELISA assays without hardware changes and the technique can be extended to virtually all soluble protein targets and immunological questions, respectively.
Footnotes
Acknowledgments
The project was supported by the Federal Ministry of Education and Research.
