Abstract
The need for a high degree of flexibility within compound screening is a requirement throughout the drug discovery industry. The demands made of automation technology are becoming increasingly sophisticated as assay formats diversify and confidence in screening automation grows. To meet these demands for high-performance and flexibility, MSD with RTS Life Science have developed and installed a dual-robot arm screening system at our Terlings Park site to support early stage drug discovery. Plate transport on the system is via two articulated robotic Staubli arms. The system has two operating modes, combined, with the entire system operating in unison; and independent with each robot arm acting independently to perform two different screens in parallel. Additional flexibility is provided by the SPRINT software ‘dynamic batch scheduling’ feature. This allows for the scheduling of multiple screens to run automatically and for extra plates to be added and new screens to be added once the system is running without the need to stop the system and reschedule the work plan. The utility of the systems is exemplified in a kinase counter screening mode in which four different kinase assays are run against the same set of compounds in one robotic run.
Introduction
When designing a new robotic screening system consideration needs to be given to each component of the system: the robot arm, the liquid handling, plate storage, and plate readers. In addition, powerful and flexible software is critical to maximizing productivity from a robot screening system. We require a high degree of flexibility to meet the demands of the rapidly changing drug discovery process. The expectation from the use of automation in basic research has changed in recent years and the demands made of automation technology are becoming increasingly sophisticated as assay formats diversify and confidence in screening automation grows. 1 –- 3 To meet these demands for high-performance and flexibility, MSD with RTS Life Science have developed and installed a dual-robot screening system at our Terlings Park site to support early stage drug discovery.
Physical System
Two Stäubli robotic arms with associated microtitre plate grippers were selected as the sample transfer mechanism for this system. We also reviewed many liquid handlers and plate readers to compare flexibility, reliability, footprint, cost, and service support. Instruments were chosen to meet the needs of many different biochemical and cellular assay types by purchasing ‘best in class’ items from a variety of vendors. RTS Life Science provides a powerful and flexible integration solution, while minimizing expenditure (Table 1). The overall system was designed to fit an existing rectangular robot room, constraining possible shapes and layouts. The principal design concept was to facilitate multiple simple mix and read assays to be performed simultaneously, while also supporting more complicated assays that would require a greater number of instruments. Each robot arm can access a subset of the system instruments (Table 1, Fig. 1A–D).
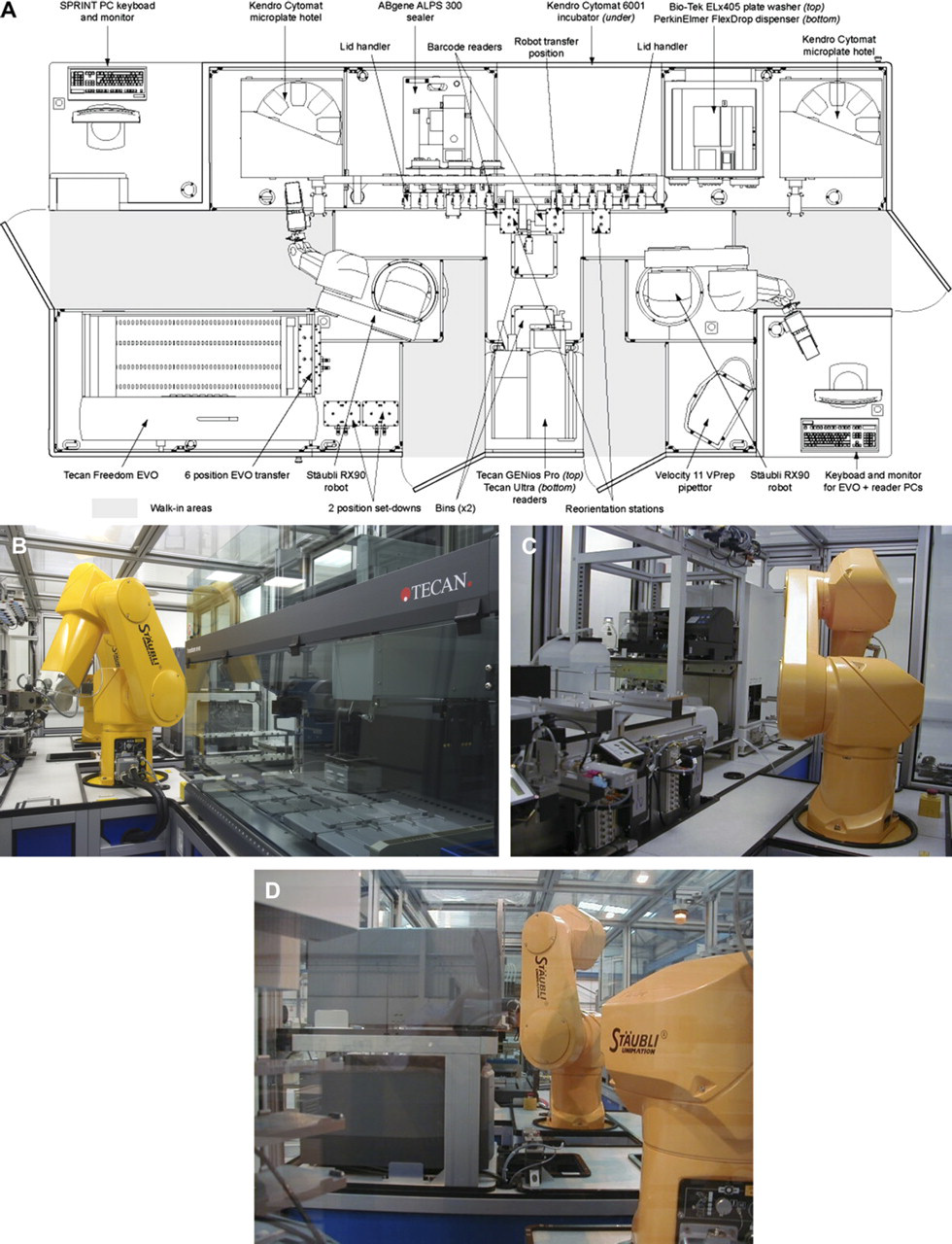
(A) Instrument layout within robot cell, (B) Robot 1, (C) Robot 2, (D) Robots 1 and 2 showing the overlap area.
System instruments available to each robot

Robot A can access a room temperature storage facility, an eight tip liquid-handling workstation with room for shaking and other plate manipulation, and a plate sealer. Robot B can access a different room temperature storage facility, a 96/384 liquid-handling workstation, a 96–1536 bulk liquid dispenser, and a 96/384 plate washer. An area of overlap allows both robots to reach ‘communal’ instruments: a temperature and CO2-controlled incubator, plate waste bins, and two plate readers. Also, there are three plate put-down positions, which both robots can reach, to allow for plates to be transferred from one arm to the other. For future proofing, substantial space for additional instrumentation is also available within the overall initial design. Underlying programming places constraints on each robot arm that preclude potentially expensive collisions in the mutually accessible areas of the system.
The dual-arm system can operate in two modes:
As a combined system, both robots act in unison to ensure plates are transferred around the entire system in accordance with one screening run. This approach can be used to run multiple assays sequentially (batch scheduling). Batch schedules may be created in advance or once the run has started.
As two independent automated screening systems, the two robots act independently to perform two screening runs in parallel. Each robot transfers plates between the instruments in its own subset. Each independent system may run its own batch schedule.
Flexible Screening Management
Flexibility and ease of use were important aspects for designing our new automated screening system. Operators of the system fall into two categories, power users and users; the user log on interface supports this by allowing only the power user to build or modify screening schedules, and the user to only be able to run them.
SPRINT is the RTS software running our dual-arm system; it allows screening schedules to be created, stored, edited, and run in various combinations. The software is dynamic, automatically optimizing throughput and timings for each schedule as it runs. The robot processes to run an assay are constructed using the sample schedule editor (Fig. 2A). Multiple plates may be coordinated according to individual assay requirements. The application allows for plate processing during an assay to be defined. For example, using the transfer step, a plate can be moved from one part of the system to another, during which time a lid can be removed, replaced or discarded or a bar-code can be read. Once placed on an instrument, the run protocol step can be used to link with the instrument third party software and run an assay specific program. Complex schedules can build up from a single primary schedule usually based on the work flow of a compound plate and a series of secondary schedules to load tip boxes to liquid handlers, cell plates, etc. The running of the secondary schedules is triggered from the primary schedule by the use of the launch secondary plate command.
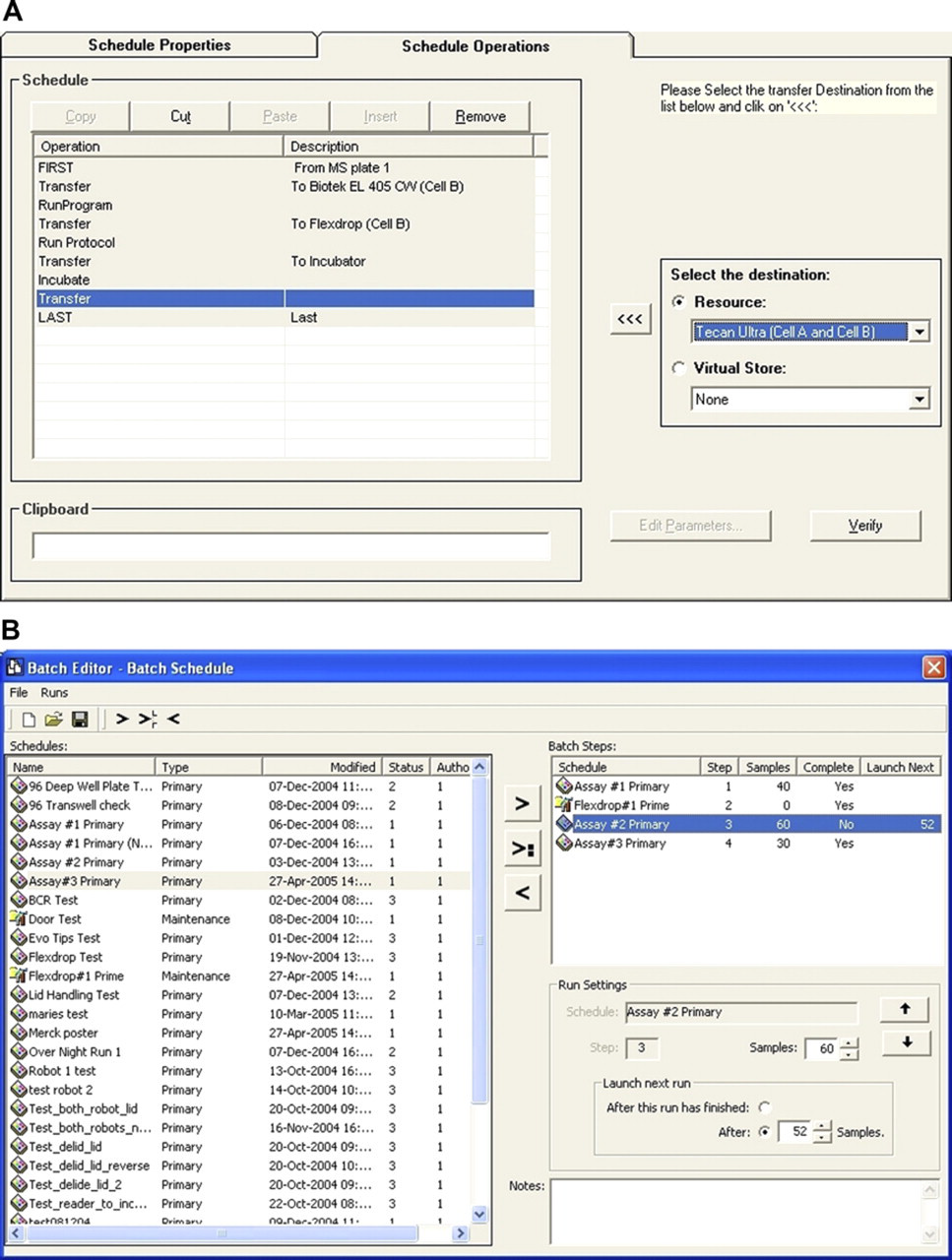
(A) Sample Schedule Editor, (B) Batch Editor.
Once the individual assay schedules have been constructed using the sample schedule editor, they can be run individually or as part of a batch schedule where a list of several screens is run automatically. Batch schedules contain multiple individual schedules. They are created from individual schedules using the batch editor (Fig. 2B), saved and then called up as required for screening runs. Maintenance schedules can also be built which allow the user to run various system maintenance operations, for example, liquid handlers may be set to be automatically primed or a cell washer could be set to run a clean program to remove any salts that could precipitate after the assay has completed.
Running Screens
There are three ways in which a schedule can be run to perform an assay.
Single run: This mode is used to run a single screen with a known number of plates. Once the run has started, the user can increase the plates within the run.
Continuous run: This mode is used when a single screen is being run, but the operator does not have to define the number of plates to be screened. This can be a very useful option if, for example, the number of available cell plates is unknown at the start of the run.
Batch run: A batch (Fig. 3) allows several screens and maintenance operations to run automatically during the screening period. A batch is built up from a series of predefined schedules and maintenance schedules. Screens are set to run sequentially or may be interleaved within the batch.
Batch run.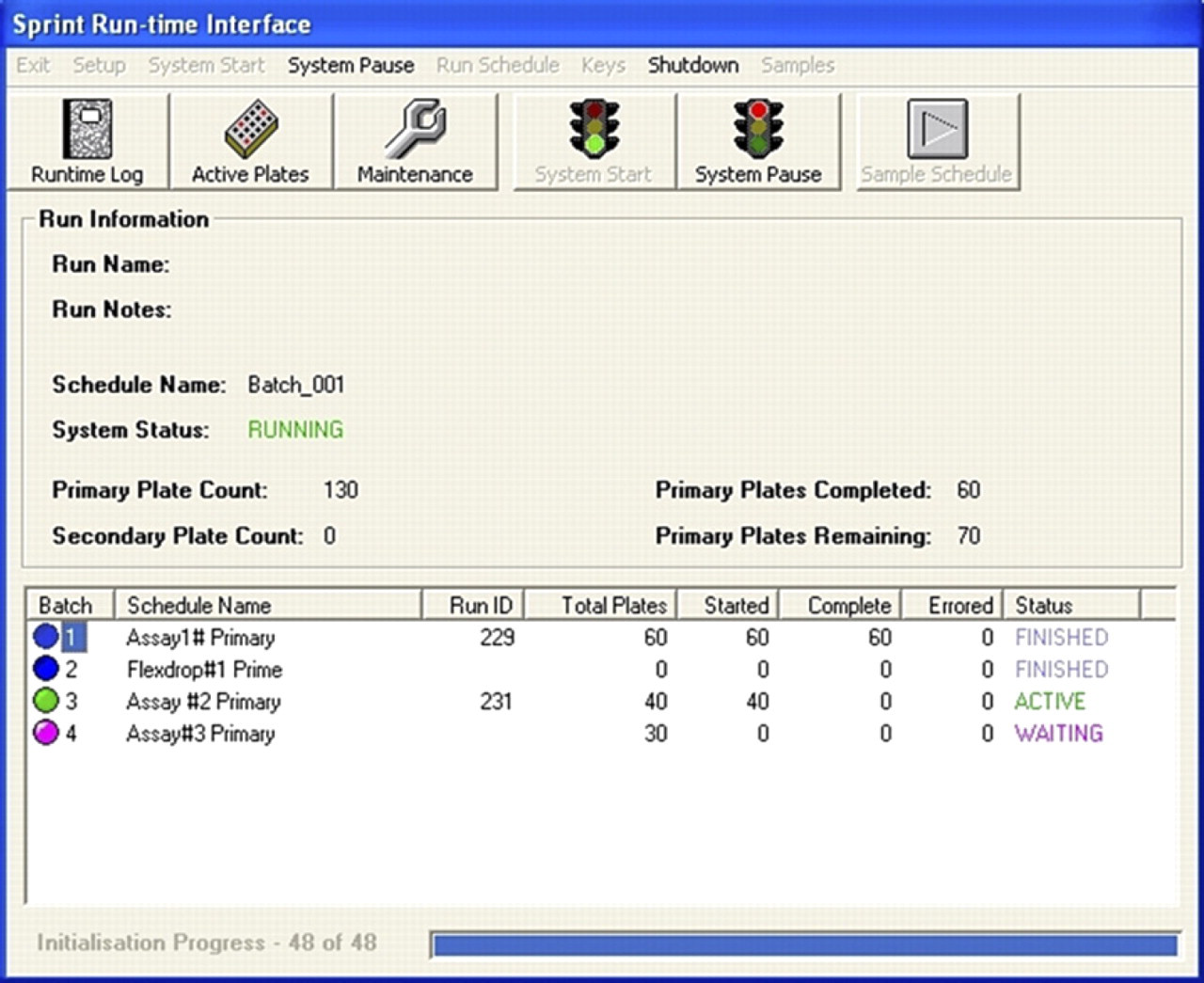
Dynamic Scheduling Option
The expected workload may change during the course of a screening run, for example, cell plates for a particular screen may not be available; the screen must be cancelled or reduced in size; or an extra screen may unexpectedly be required to be run on the same day, but the system has already been started. To meet this need, our dual-arm system runs can be dynamically edited while they are running; this allows the operator to carry out the following options:
Change the number of plates in an individual screen
Insert an extra screen (Fig. 4A and B)
(A) Insert new screening run window, (B) List of Screens window.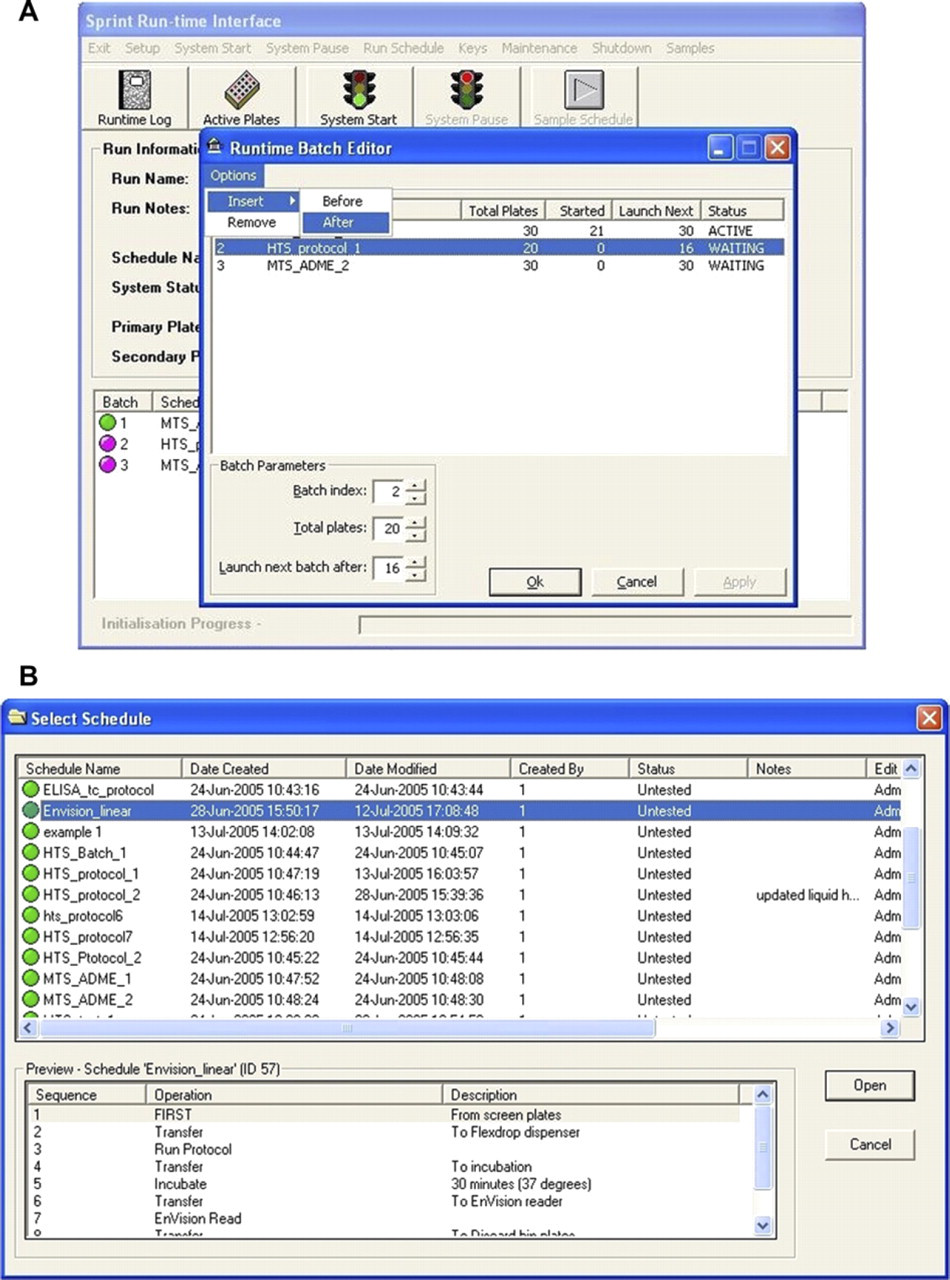
▪ Delete a screen
▪ Alter the rate of plate input (pacing time) for a particular screen (Fig. 5)
Pacing time and delay plate entry window.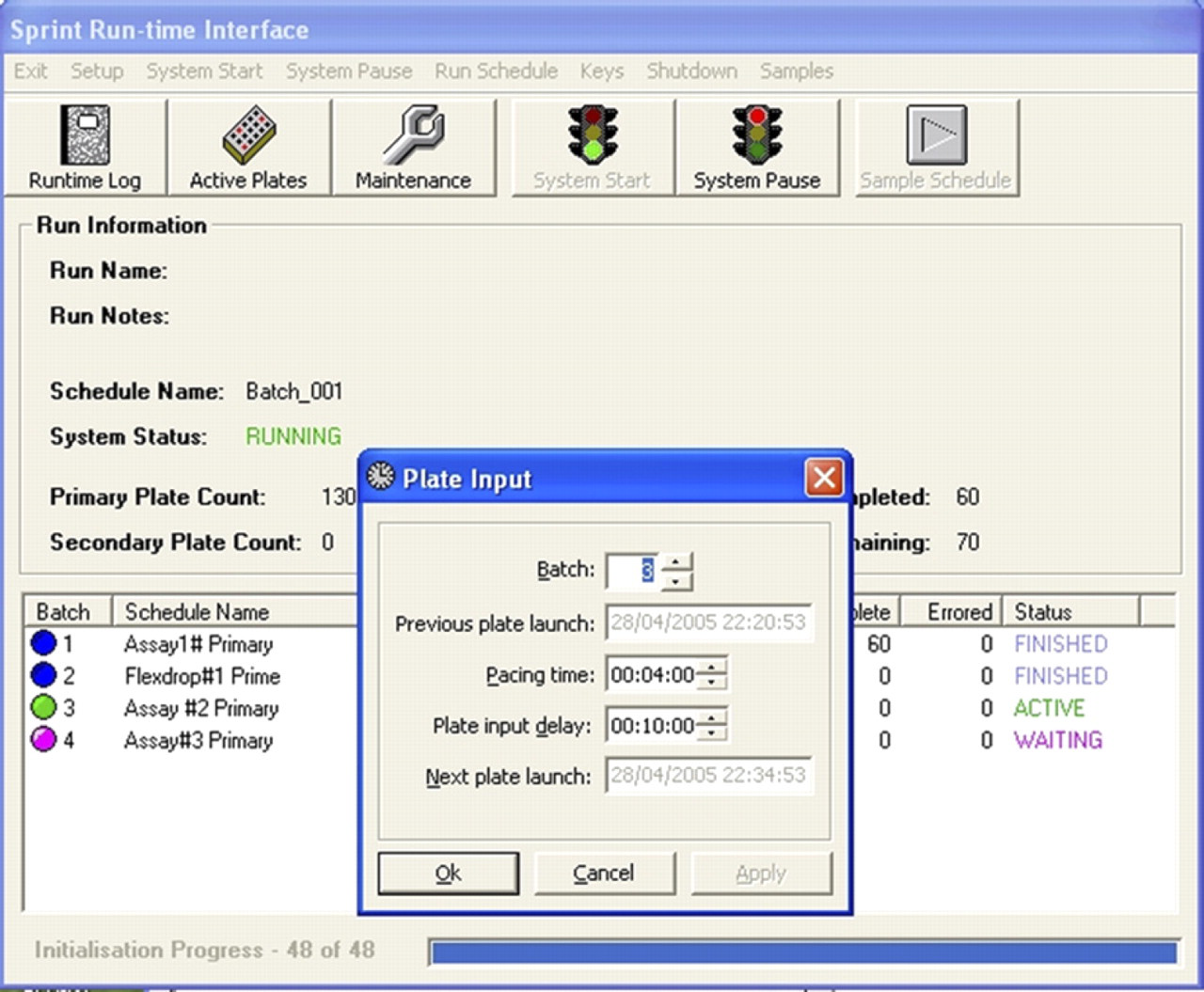
▪ Delay entry of the next plate into the system for a specified time (Fig. 5)
Additional screens are added via the runtime batch editor and by using the “option” tab where schedules may be added to or deleted from the run (Fig. 4A and B).
Manual intervention
During any screen, the operator has the option of safely making physical adjustments to the system. The ‘System Pause’ button brings the system to a controlled stop with all plates in a safe environment, that is, in storage, incubation, or binned. The operator is then able to add new reagents or screening plates to the system, and run instrument checks. Once adjustments are complete, pressing the start button restarts the system (Fig. 6).
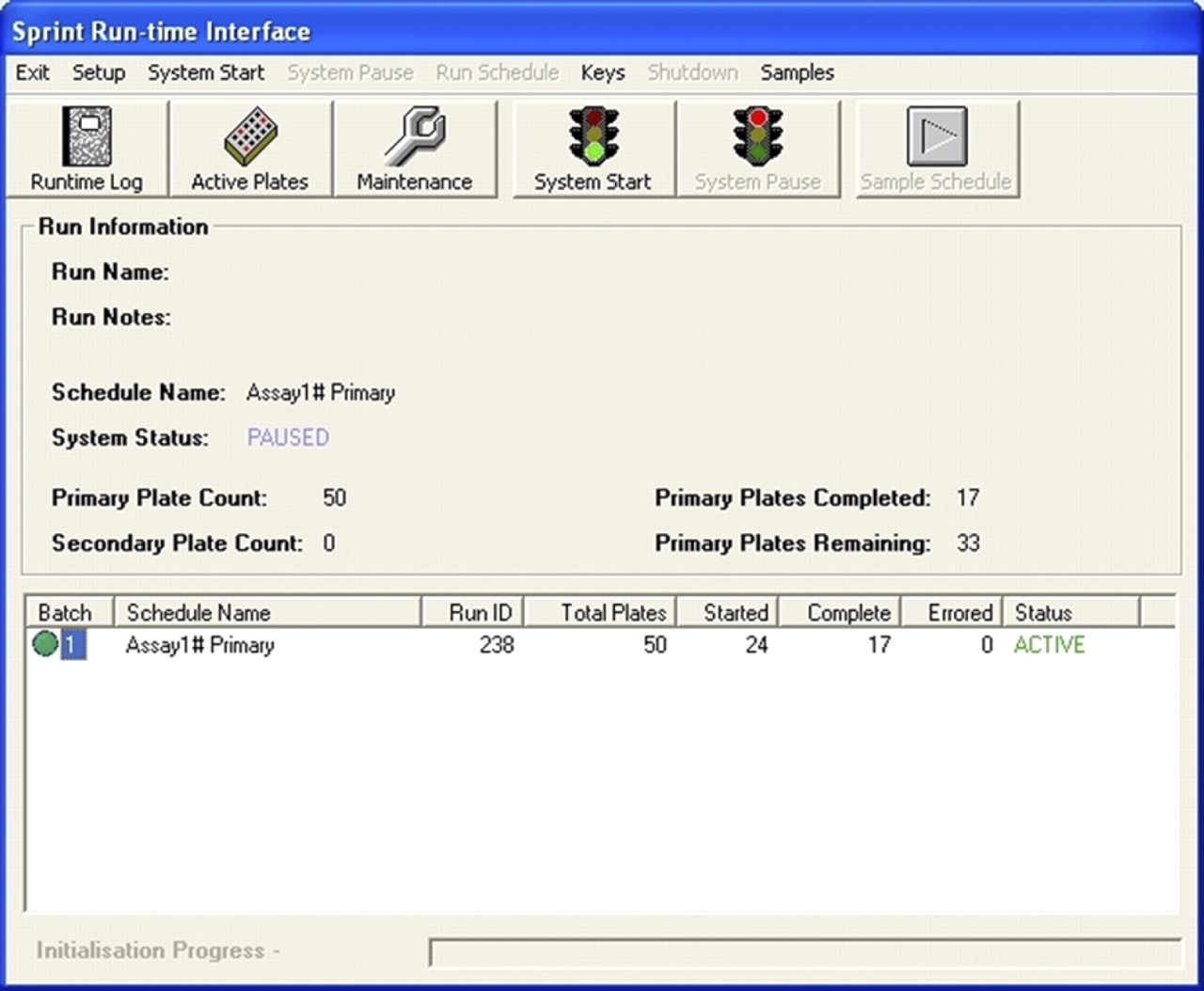
Runtime window showing system passed.
4-Kinase Automated Assay
One of the assays that has been developed and run on the system since its installation at Terlings Park is a kinase IMAP assay in which four kinases (identified as kinases A–D) are screened in parallel against the same set of compounds within a run. The IMAP assay is carried out according to the manufacturer's protocol 4 (Fig. 7). The automation of the assay is divided into three primary components:
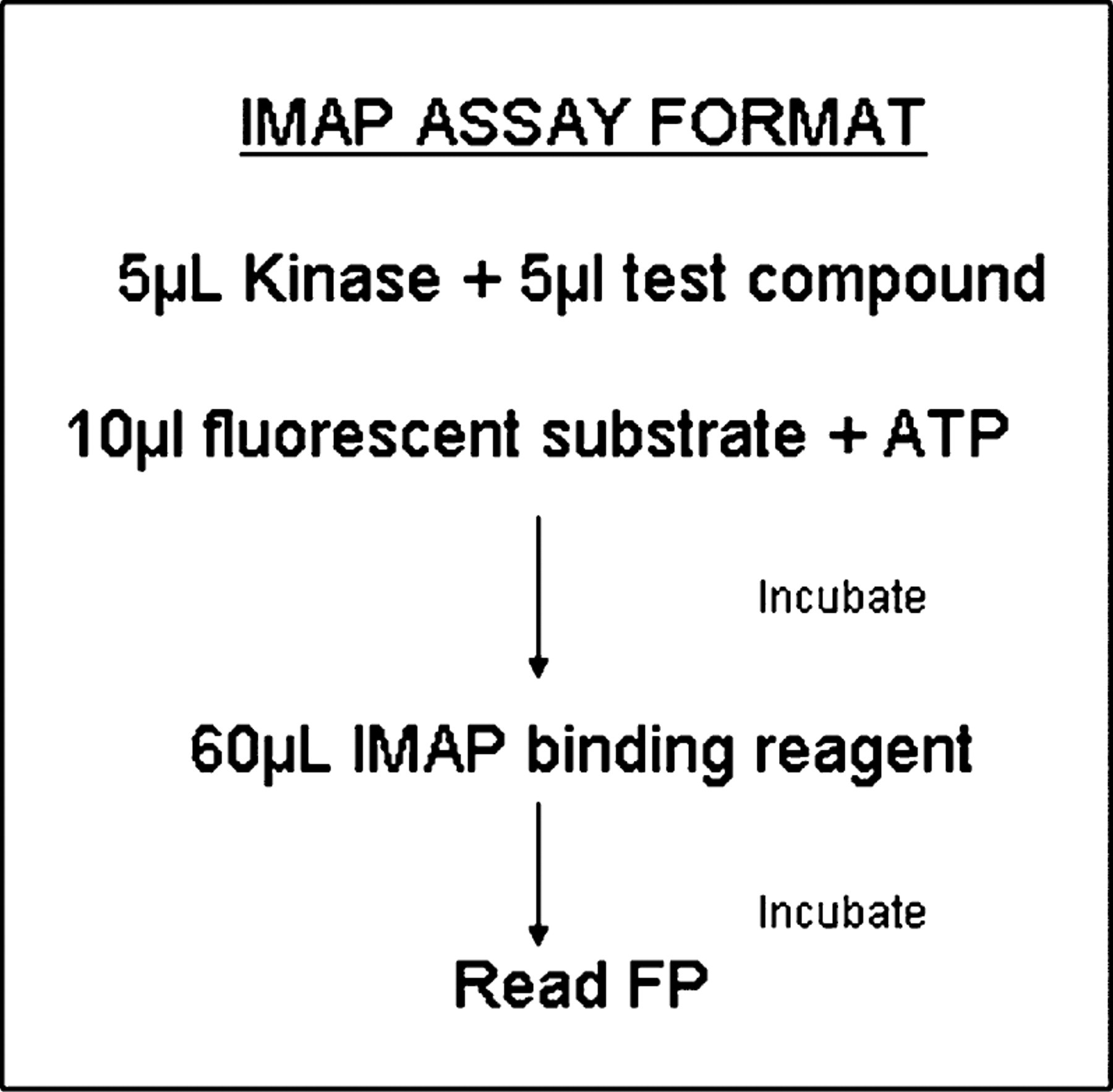
IMAP assay protocol for small molecule kinase inhibitor screening.
A schedule to carry out the serial dilution of the test compound. Using an off-line process prior to the robot run, 10 mM test compounds prepared in DMSO are added to rows A to P of columns 3 and 13, together with standard compounds in columns 1, 2, 23, and 24. Compounds in 384-well plates are then serially diluted using the Tecan Evo on the system, with disposable tips. The serial dilution produces a 10-point half log unit dose response curve and is performed in DMSO to reduce the risk of sample precipitation.
A schedule to predilute the compounds into complete reaction buffer. The VPrep on the dual-arm system is used to transfer an aliquot from the serially diluted compound plate to an intermediate plate containing a uniform volume of complete reaction buffer.
A schedule to build the assay plate and read. Assay plates combining compounds, kinase, enzyme substrate, and ATP are built using the VPrep and Flexdrop. Each individual intermediate plate from part (2) is used to prepare four assay plates, each with a different kinase and its matched substrate. Assay plates are then incubated for 1 h at room temperature (RT) protected from the light, upon which the IMAP binding reagents are added using the PE Flexdrop. Plates are incubated for another hour at RT protected from the light and then read using the Tecan Ultra in FP mode.
The kinases and their substrates were tested across a time course to determine stability at RT over the course of a full run. All kinases and substrates were stable for up to 5 h at RT, and a robust window was achieved in this automated assay yielding a Z factor of ≥0.8 for all conditions (Fig. 8). The IMAP binding reagents were optimized so that all kinases used the same reagents with the optimum composition consisting of progressive binding reagent diluted 1/500 in 85% buffer A, 15% buffer B (data not shown). By running the robot schedules outlined above in a batch mode, with the optimized assay conditions, it has been possible to produce robust selectivity data for small molecule kinase inhibitors (Fig. 9).
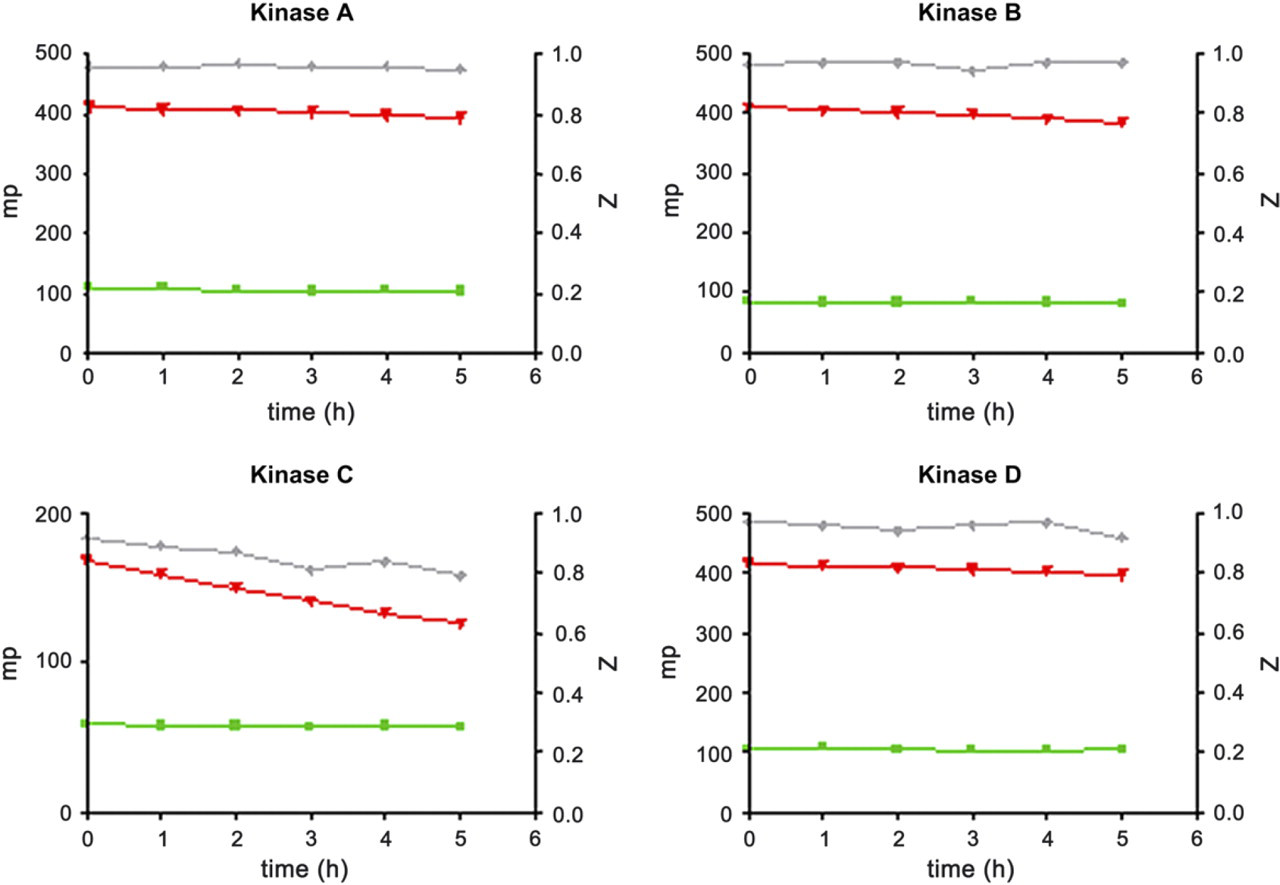
Kinase and substrate stability time course. Kinases were diluted and used in the assay from 0 h to 5 h after being prepared. Kinases A, B, C and D were incubated in the presence of their respective substrates and 10 μM ATP for 1 h at RT protected from the light. 60 μl IMAP binding reagent consisting of 1/500 progressive binding reagent diluted in 85% buffer A and 15% buffer B was added and incubated for 1 h at RT protected from the light. Wells were then read for FP using the Tecan Ultra. ▪ no kinase ▴ max kinase ♦ Z factor (data is the mean and sem of 48 wells).
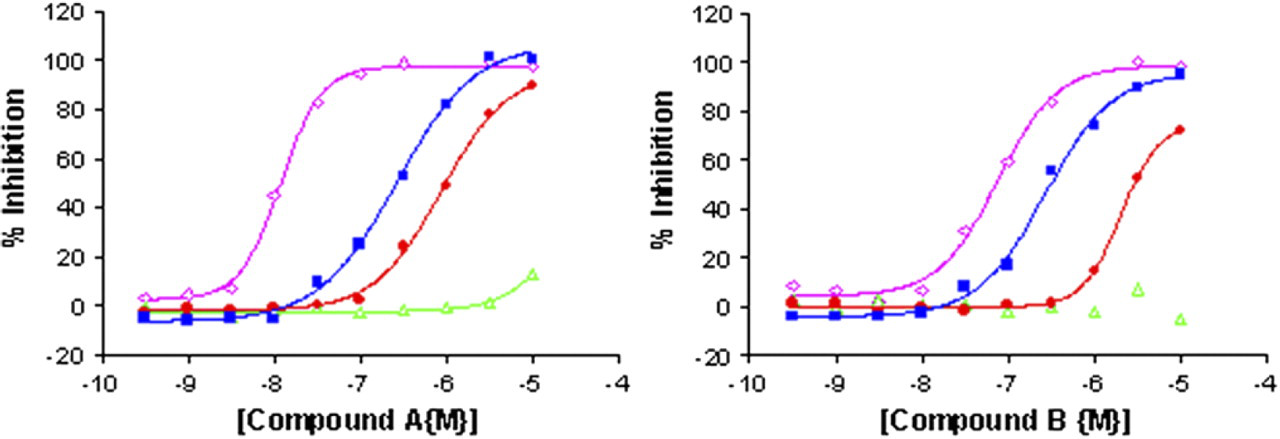
Kinases A, B, C and D were incubated in the presence of increasing concentrations of test compounds (0.03–10,000 nM), their respective substrates and 10 μM ATP for 1 h at RT protected from the light. 60 μl IMAP binding reagent consisting of 1/500 progressive binding reagent diluted in 85% buffer A and 15% buffer B was added and incubated for 1 h at RT protected from the light. Wells were then read for FP using the Tecan Ultra. ⋄ Kinase A, ▪ Kinase B, • Kinase C, △ Kinase D. (mean and stdev of two wells).
Discussion
“MSD have a back-up philosophy that means all assays for key site projects are able to run on more than one robotic system. 5 The option of being able to add a new screen to a run that is in progress is very important for the workflow within our group. If a mechanical failure occurs on a different system, it is possible to move that day's screening set to this dual-arm system, thereby reducing any delay caused in data delivery.” The high level of flexibility in the SPRINT scheduler ensures that assays with less common plate processing requirements are equally transferable to this dual-arm system.
The IMAP assay described is controlled as several independent subschedules: serial dilution, predilution, plate build, and read. For assays which can be subdivided in this way, an extra level of system flexibility is introduced as assays may be easily transferred to an alternate automated or manual processing system after completion of one or more subassays. This SPRINT feature allows redistribution of assays across available automated and manual processing facilities in the event of either failure of an automated system or an unexpected change in screening demands.
Parallel delivery of results from multiple assays facilitates early go/nogo decisions for individual compounds. The SPRINT scheduler's ability to deliver results from multiple assays simultaneously has aided the rapid progression of promising candidates through the early stage drug discovery process.
Conclusions
The novel dual-robot arm system we have designed and built, in combination with the flexible RTS SPRINT scheduling software, has proved to be an excellent addition to our centralized automation medicinal chemistry support assay facility. It has allowed us to provide a highly flexible screening service for multiple project teams. The system's flexibility has been demonstrated by running the kinase counterscreen described in which the same set of compounds was screened against four different kinases within the same process. The ability to add new screens to the system in a truly dynamic way increased our group's ability to adapt to the changing working environment.
