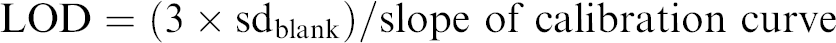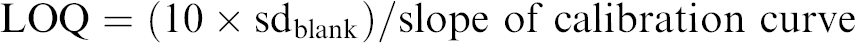Abstract
With a focus on low-cost and low-power consumption, a miniature laser-induced fluorescence (LIF) detection system was assembled using a 635 nm red diode laser as the excitation source and a photodiode element coupled with an operational amplifier for signal collection. The primary elements of the miniature system, namely the laser and the detection system, cost a combined $70 and required only 270 mW of power for operation. When compared to conventional systems assembled using an argon-ion laser source and a photomultiplier tube, this represents a 98% decrease in the cost, and greater than 5000-fold decrease in power consumption. Limits of detection (LOD) and quantitation (LOQ) of the miniature system, evaluated on a microfluidic device for Nile Blue dye diluted in ethanol, were approximately 15 and 40 nM, respectively. Detection of λ-phage DNA on a microfluidic device using the miniature system was performed after mixing with an intercalating dye, TO-PRO 3. The LOD and LOQ of λ-phage DNA after TO-PRO 3 intercalation were approximately 1 and 4 ng/μL, respectively. Quantitation of DNA on microdevices using the miniature LIF detection system was also performed with an error of less than 15%. This detection system is a step in the direction of commercializing microfluidic instrumentation by reducing the cost and power required for operation. (JALA 2006;11:254–9)
Introduction
Analyte detection using laser-induced fluorescence (LIF) methods offers good sensitivity with low detection limits and, as the radiation from a laser source can be focused, provides for detection in very small volumes. This ability makes LIF detection a method of choice for detecting analytes on microfluidic devices, where the characteristic length scales are of the order of micrometers or smaller. Most often microchip LIF detection is applied for the detection of DNA. Because DNA is not naturally fluorescent, this requires mixing DNA with one of several, commercially available, intercalating dyes to generate DNA–dye complexes that fluoresce when excited by light of the appropriate wavelength. The intensity of the fluorescent signal is then directly proportional to the concentration of DNA. The power of this method is evidenced by the single-molecule detection of DNA on microfluidic devices as demonstrated by Haab and Mathies. 1 For these microchip LIF detection systems, a focused laser beam from an argon-ion laser is normally used for excitation, with a photomultiplier tube (PMT) for the collection of fluorescent signal.2, 3
Although there is no question that extremely sensitive LIF systems can be assembled for microchip-based analysis using powerful lasers and ultrasensitive PMTs, 1 the analytical microchip field has evolved to the point where a movement toward detection systems that are more practical is needed to implement microfluidic devices in laboratories not specialized in their use. These systems must be cost-effective, costing tens of dollars not thousands of dollars, and must have reasonable (μM to sub-μM concentrations), but not necessarily exquisite (nM to fM) sensitivity; this will allow direct detection of PCR-amplified DNA and highly expressed proteins or detection of lower abundant materials after preconcentration. Analogous to the miniaturized microfluidic devices used for carrying out rapid analysis,4, 5 the detection systems themselves must also be reduced in size. Moreover, if systems are to be field-portable, or even handheld in nature, power considerations must be addressed.
The laser source and PMT form the primary components of most LIF detection systems, but they can be bulky, expensive, and consume significant amounts of power. For example, the PMT used in the LIF system described by Munro et al. 2 , costs roughly $900, and requires a high-voltage power supply providing up to 1500 V to operate. The argon-ion laser source in this system costs approximately $7500, requires 1500 W of power, occupies nearly 8000 cm3 of space, and weighs over 10 kg. These components are used in conjunction with a microdevice occupying less than 2 cm3 and weighing only a few grams. The size disparity between the devices and the detection system, along with the cost and power required for the instrumentation is a major obstacle to the goal of commercializing microfluidics-based instruments that could be used for on-site analysis.
Miniaturization of the LIF detection system instrumentation has been demonstrated, with replacement of the argon-ion laser with a miniature laser diode, and replacement of the PMT with photodiode elements. The combination of a diode laser with a photodiode-based collection device has been previously used for LIF detection in capillary electrophoresis, using an avalanche photodiode, 6 as well as for static measurements on a microchip specific for detection of bacterial spores, using an integrated circuit photosensor array. 7 A diode laser has also been used in the detection system for a commercially available microchip electrophoresis system, but this instrument provides more complex functionality (electrophoretic separations) than required for many DNA detection applications, thus is still costly (~$18,000), heavy (10 kg), and requires significant power (60 W).
This report describes a miniaturized LIF system applied simply for detection and quantitation of DNA in solutions, flowed through a microfluidic device. The system uses a red diode laser module for excitation and a photodiode coupled with an operational amplifier for signal collection. To demonstrate the performance of the miniaturized system, the limits of detection and quantitation for fluorescence in a glass microdevice channel filled with Nile Blue dye were evaluated and compared with those of the traditional LIF system. In addition, the system was used to detect λ-phage DNA concentrations in the glass microfluidic device after mixing with the intercalating dye TO-PRO 3.
Experimental
The LIF system was assembled using cage cubes, and mounting plates and rods purchased from Thor Labs (Newton, NJ), which resulted in a footprint of 45 cm2 with a height of 20 cm. These elements provided rigidity to the system, and ensured that the components of the LIF system, once mounted, remained aligned and stable. The 635 nm wavelength red diode laser module (Meredith Instruments, Glendale, AZ) and the photodiode element (076-12-22, Advanced Photonics, Camarillo, CA) were fixed within plastic rods that served as an interface between the smaller OD of these components and the larger ID of the mounting plates. The dichroic mirror (cutoff wavelength: 650 nm, Omega Optical, Brattleboro, VT) and a front-surface mirror were fixed on two 45° mirror adaptors (Thor Labs, Newton, NJ). These adaptors allowed adjusting the mirror position to manipulate the laser beam and emitted fluorescence. A collimating and focusing lens for a diode laser (Melles Griot, Rochester, NY) was positioned above the dichroic mirror to focus the laser radiation on the microchannel. A bandpass filter (670 ± 20 nm, Omega Optical, Brattleboro, VT) was mounted between the front-surface mirror and the photodiode element to allow through the emitted fluorescence and filter out reflected light from the surface of the microfluidic device. Two cage cubes (Thor Labs, Newton, NJ) were placed one above the other, and the components of the LIF system were mounted on the faces of the cubes using the mounting plates and rods as shown in Figure 1.
Photograph of the miniature LIF system; the 635 nm line from a red diode laser mounted in a plastic rod and the dichroic mirror are mounted on opposite faces of the cage cube on top. The collimating and focusing lens is mounted above the top cage cube. Emitted fluorescence is collected by the lens, transmitted through the dichroic mirror, and reflected from a front-surface mirror through a bandpass filter and onto a photodiode element. The last three components are mounted on the bottom cage cube. The XYZ translational positioning system is located such that the channel of the microfluidic device is above the collimating and focusing lens.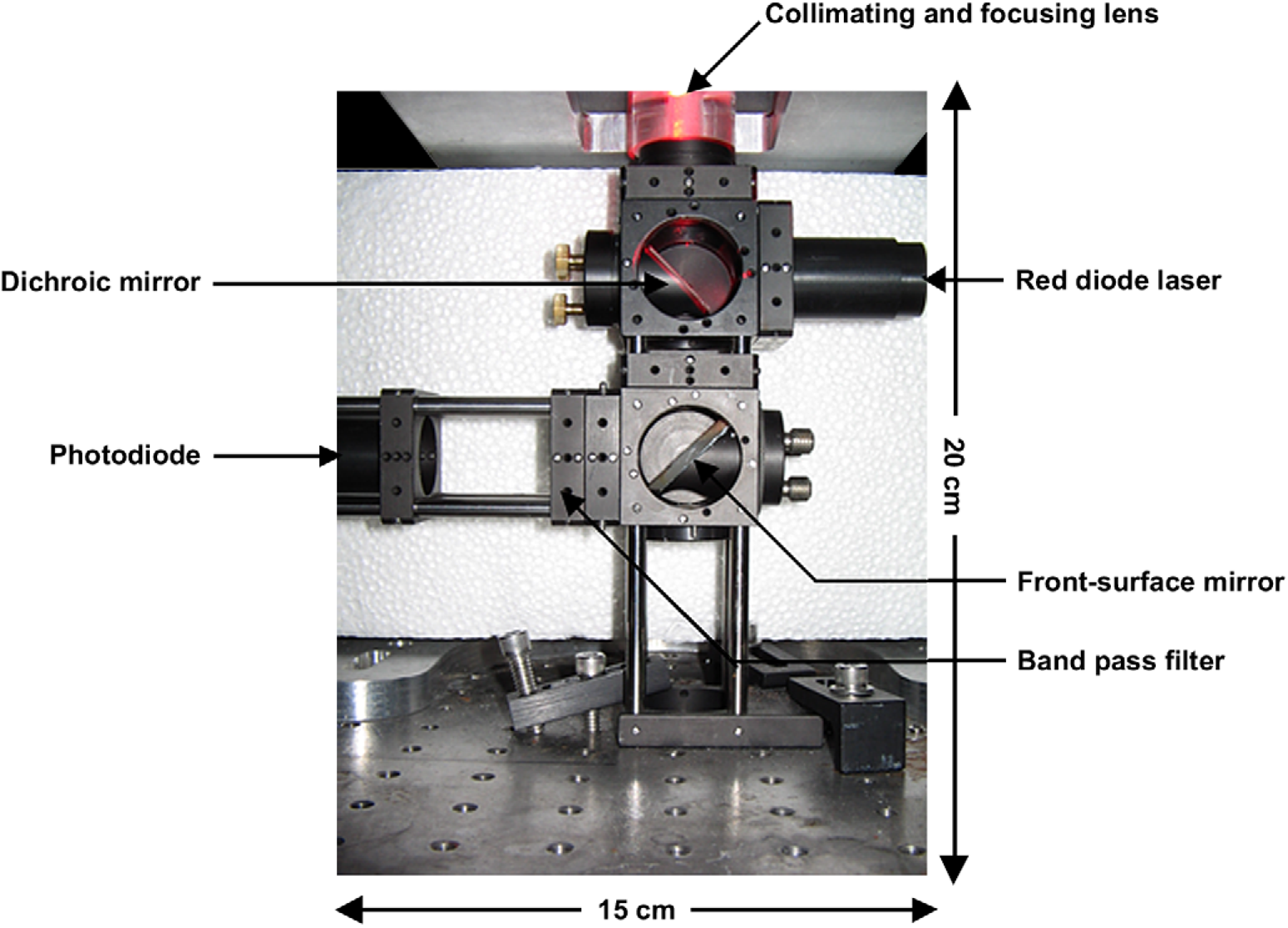
The glass microfluidic device was fabricated from borofloat glass with channels prepared using standard photolithography techniques. 8 The starting width of the channels before etching was 40 μm. The channel depth after etching, determined using a profilometer (Ambios Technology, Inc., Santa Cruz, CA), was found to be 197.6 ± 0.5 μm. The calculated top width of the channel was 435.2 ± 1.0 μm. Access holes were drilled at the inlet and exit using 1.1 mm diamond-tipped drill bits (Crystalite Corp, Lewis Center, OH). The plate was then thermally bonded to another borofloat glass plate at 650 °C. Nanoport reservoirs (Upchurch Scientific, Oak Harbor, WA) were attached at the entrance of the channels to connect the microfluidic device at syringes using PEEK tubing (Upchurch Scientific, Oak Harbor, WA). A syringe pump (KD Scientific, Holliston, MA) was used to control fluid flow.
The microfluidic device was placed on a manually controlled XYZ translational positioning system above the lens to facilitate alignment of the focal point of the laser beam at any location along the length of the channel, as well as at any desired height within the channel cross section. The filtered fluorescence was transmitted through the dichroic mirror and the bandpass filter to the photodiode element. The photodiode transduced the incident light into a current, which was converted to a voltage and amplified using an inverting differential amplifier circuit assembled in-house using a standard operational amplifier (TL082ACP Texas Instruments, Dallas, TX). The feedback resistance in the circuit, which defined the amplification, was selected to minimize the background signal (reflection of the laser light from the glass microchip) while still providing sufficient amplification of the fluorescent signal. The amplified voltage was collected using a USB data acquisition device (National Instruments, Austin, TX) and stored using a LabVIEW program, also written in-house.
To characterize the miniature LIF system, the limits of detection and quantitation were determined using linearly increasing concentrations of Nile Blue dye (Fluka, St Louis, MO) dissolved in ethanol (blank, 100, 200, 300, 400 nM); ethanol was used as the solvent because the spectrum of Nile Blue in ethanol is available (http://probes.invitrogen.com). These solutions were flowed through the microfluidic device at a flow rate of 100 μL/min using a syringe pump. The emitted fluorescence was collected and recorded using the miniaturized LIF system. A calibration curve was generated by plotting fluorescence response against dye concentration. The limits of detection (LOD) and quantitation (LOQ) were calculated using Eqs. (1) and (2), respectively:
λ-phage DNA (Sigma, St. Louis, MO) was dissolved in TE buffer (10 mM Tris, 1 mM EDTA, pH = 8.0) to generate solutions of linearly increasing concentration (blank, 10, 20, and 30 ng/μL). An intercalating dye solution was prepared by diluting 1 μL of TO-PRO 3 (excit/emiss: 642/661 nm, Invitrogen, Carlsbad, CA) in 1000 μL of TE buffer. Equal volumes of λ-phage DNA and TO-PRO 3 solutions were mixed off-chip and flowed through the microfluidic channel at a flow rate of 100 μL/min using a syringe pump. The fluorescence emitted from each solution was collected and recorded using the miniature system. The fluorescence response was plotted against DNA concentration to generate a calibration curve. The LOD and LOQ of λ-phage DNA were also calculated using Eqs. (1) and (2), respectively.
Results and Discussion
Microfluidic devices have emerged as useful tools for the analysis of bioanalytes owing to their small size, which allows for reduced sample consumption, faster analysis time, and minimal sample handling. Miniaturization of the platform used for analysis represents significant progress in the technology, ultimately allowing microfluidic devices to find application in clinical and forensics arenas. In this work, a miniaturized LIF system is assembled for DNA detection and quantitation in a microfluidic device using inexpensive, low-power consuming, off-the-shelf devices. Miniaturizing the components required for microfluidics-based instruments, and making them cost and power-efficient, represents a small, but necessary and important step toward the ultimate goal of commercializing such instruments.
Comparison with the Traditional LIF System
The miniaturized detection system described in this report uses a miniature (9 mm diameter × 18 mm long; 1.1 cm3) diode laser module as the excitation source that costs less than $50. When compared to the argon-ion lasers found in more conventional LIF systems, the size reduction and monetary savings achieved using a diode laser are obvious. In addition, 1500 W is required to power an argon-ion laser, to provide a maximum laser power of 300 mW; this is significantly more power than required for most LIF detection applications. For the diode laser module, a 5 mW beam can be obtained from an input power of only 270 mW; reducing the power required by an argon-ion laser by almost four orders of magnitude, and easily supplied by battery sources. On the collection side, the photodiode (5 mm diameter × 3.5 mm long) is significantly smaller than a PMT, and does not require a high-voltage source. In fact, the photodiode itself does not require any power, but the ±12 V source, required to operate the signal amplification circuit, can also be powered using batteries. Whereas PMTs offer excellent detection limits with LIF systems, with detection of attomole levels of amino acids having been demonstrated with a diode laser and PMT, 9 replacing the PMT with a photodiode is a viable method for signal collection and has been used in microfluidic devices to detect electrophoretically resolved proteins. 10
The overall comparison between a traditional detection system in our laboratory and the miniature system described here, in terms of cost and power, is given in Table 1. The limits of detection determined for both systems are also reported in Table 1. These were determined using standard fluorescent dyes; fluorescein for the traditional system and Nile Blue for the portable system. For the portable system, the calibration curve generated by detecting linearly increasing concentrations of Nile Blue diluted in ethanol is shown in Figure 2. The curve was linear (R2 = 0.99), and the limits of detection and quantitation for the portable LIF system, calculated using the formula provided in the experimental section, were determined to be 11.6 and 38.8 nM of Nile Blue dye, respectively. This is only one order of magnitude less sensitive than what was achieved in the traditional system for a cost reduction of 98%, and a four order of magnitude saving in power consumed for operation. Additional improvements in the sensitivity of this system are expected with further optimization, including incorporation of more sensitive photodiodes and better signal amplifiers. Additional cost and power requirements incurred through the use of these components will still not be significant when compared to what is required to operate the conventional LIF system. Overall, the system described is significantly smaller than a traditional LIF system and the power required is greatly reduced, providing the opportunity for development of a handheld, battery-operated analytical microchip instrument, without a significant loss in sensitivity.
Evaluation of limits of detection and quantitation for the miniature LIF system. Nile Blue solutions (blank, 100, 200, 300, and 400 nM) diluted in ethanol were flowed through the microfluidic device at a flow rate of 100 μL/min and detected using the miniature LIF system. The signal measured from the increasing concentrations was linear (R2 = 0.993). Each point is an average of seven measurements. The standard deviation of the blank (sdblank = 0.07) and the slope of the calibration curve (0.0018) were used to calculate the limit of detection and the limit of quantitation.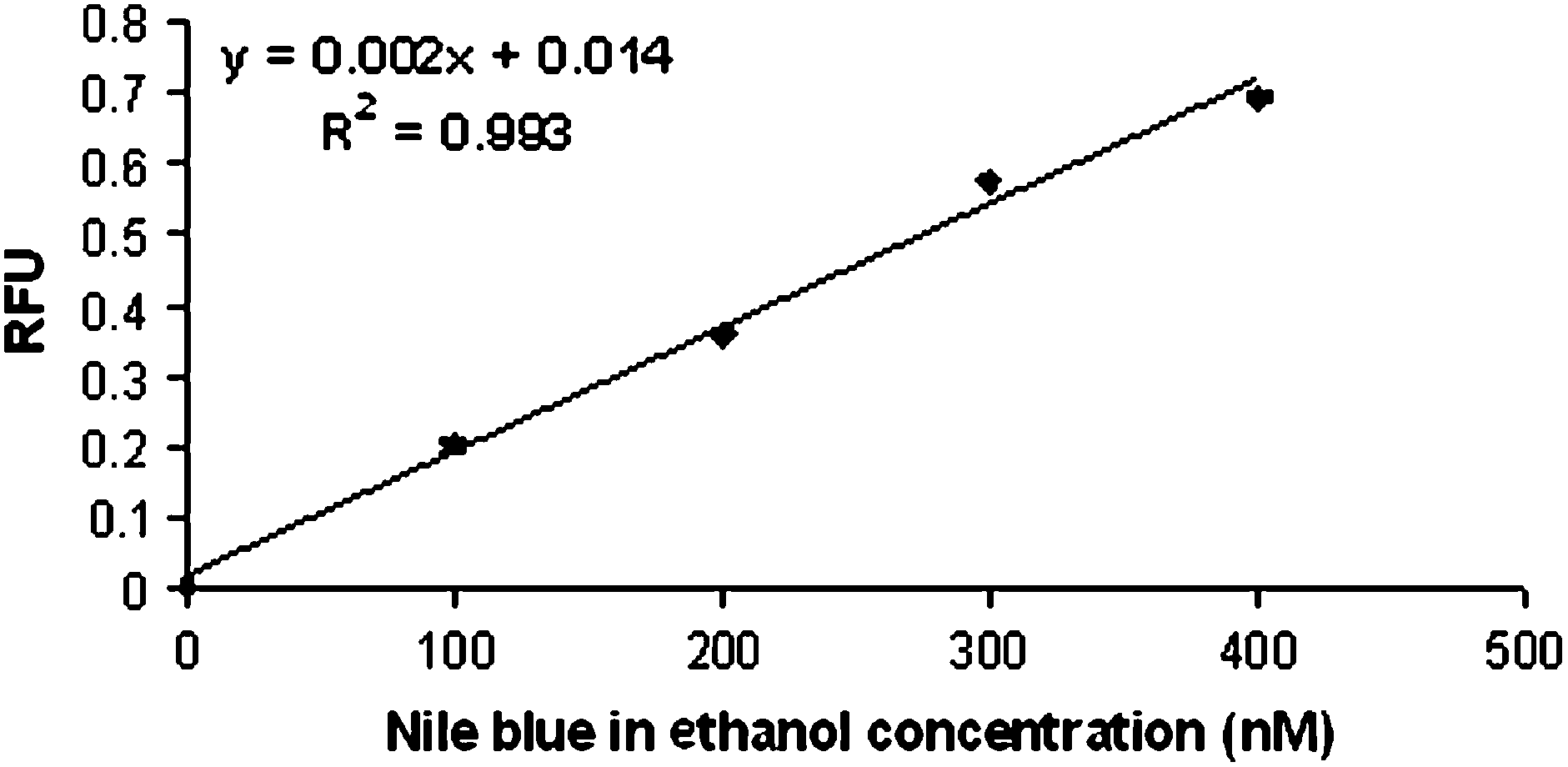
Comparison of conventional and portable LIF systems

One of the other issues to be addressed in the development of this miniaturized LIF system is the issue of applicability. The traditional argon-ion systems have wide utility because of the large variety of dyes available, which can be excited with the radiation supplied by this laser. Because of the growing popularity and decreased cost of the red diode lasers, a number of flourescent dyes and tags that use the red laser excitation have been developed and are commercially available. These tags have been implemented with both protein and DNA detection providing broad applicability to the miniaturized LIF system.
DNA Detection and Quantitation
Microfluidic devices are also advantageous with respect to the small volume of reagents used, and could be used in conjunction with the miniaturized detection system for small volume analyses. Cuvette-containing fluorometers are routinely used to determine the concentration of dye-intercalated DNA in a sample, with the intensity of the fluorescence proportional to the DNA concentration. The volume of reagents required for fluorometer-based assays can range from hundreds of microliters to milliliters. When DNA detection was performed using the microfluidic device described in this report, the DNA and intercalating dye solutions were mixed off-chip and loaded into the device where flow through the microchannel allowed exposure to the focused laser beam. Reproducible fluorescent signals (Figure 3) were obtained from only 10 μL volumes of linearly increasing concentrations of λ-phage DNA mixed with equal volumes of the fluorescent intercalating dye, TO-PRO 3 (RSD < 10%). The limits of detection and quantitation of λ-phage DNA, calculated using the formulas given in the experimental section, were 1.05 and 3.5 ng/μL, respectively.
On-chip detection of λ-phage DNA mixed with TO-PRO 3 intercalating dye. λ-Phage DNA solutions diluted in TE were mixed with equal volumes of TO-PRO 3 (1 μL in 1000 μL of TE) off-chip. The DNA–dye solutions (10 μL) were flowed through the microfluidic device at 100 μL/min and detected using the miniature LIF system. The signal measured from the increasing concentrations was linear (R2 = 0.981). Each point is an average of seven measurements. The standard deviation of the blank (sdblank = 0.07) and the slope of the calibration curve (0.016) were used to calculate the limit of detection and the limit of quantitation.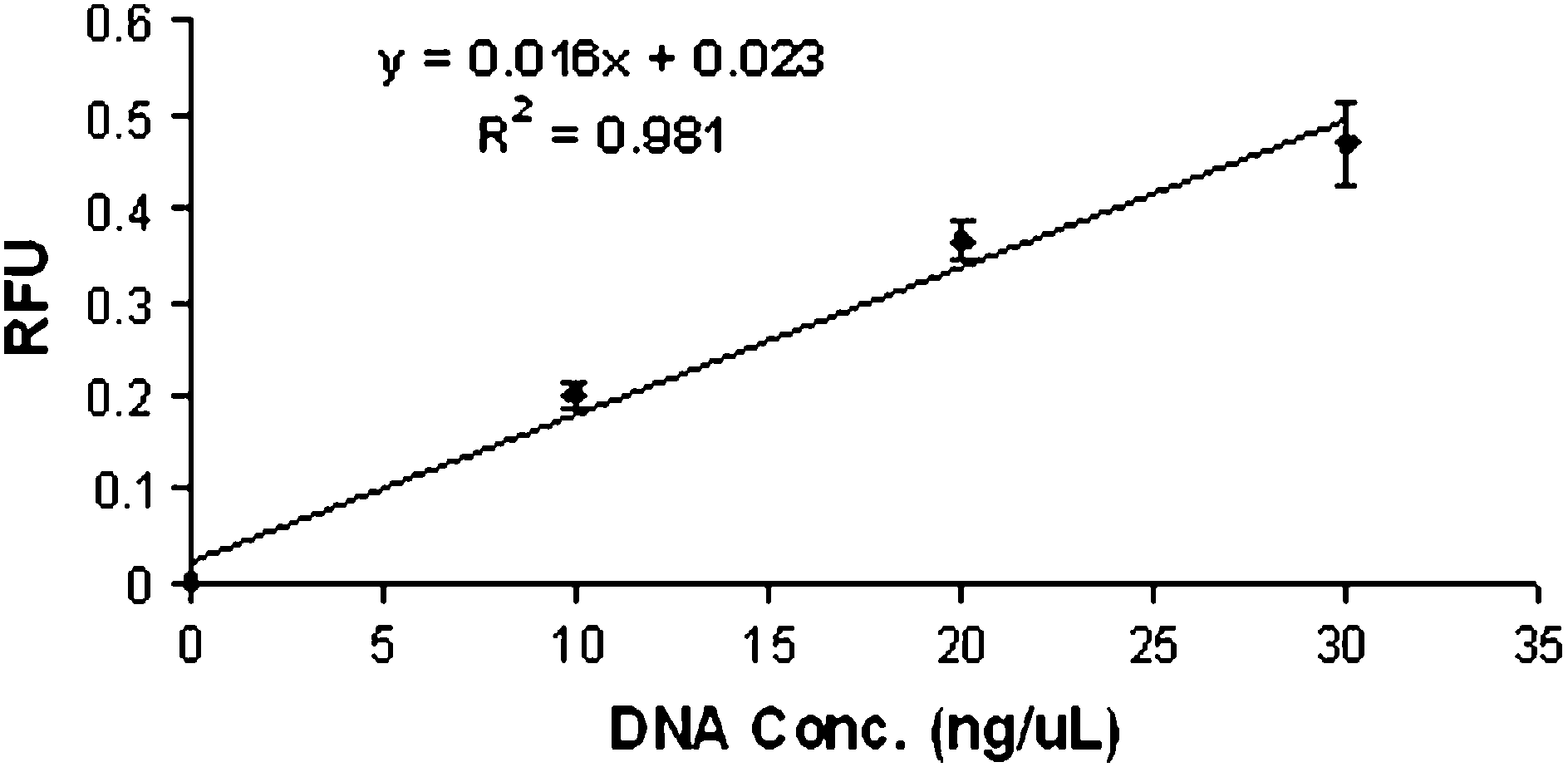
This sets the stage for DNA quantitation on a microfluidic device based on fluorescent measurements made using the miniaturized LIF system. Unknown samples could be evaluated using this system, and quantitated based on the calibration curve generated, replacing the traditional large volume fluorometer assays, and significantly reducing the amount of expensive intercalating dye required for an analysis. To evaluate the miniature system for DNA quantitation, two samples (containing 15 and 25 ng/μL of λ-phage DNA mixed with TO-PRO 3) were evaluated on a microfluidic device following generation of a calibration curve on that same device. Based on the equation from the generated calibration curve, the fluorescent signals obtained provided concentrations of 17.1 ± 0.6 and 23.1 ± 1.6 ng/μL, respectively, for the two samples; this represents an error of less than 15% for the 15 ng/μL solution and less than 10% for the 25 ng/μL. This indicates that utilization of this LIF system for DNA quantitation on microfluidic devices is possible.
Conclusions
The potential for microfluidic device analysis at the site of sample collection will require cost-, space-, and power-efficient components to be incorporated into the instrumentation that will be commercialized. Toward this end, a miniaturized LIF system was assembled that used off-the-shelf, inexpensive, low-power consuming, and space-saving components as the excitation and signal collection sources. When applied to DNA detection on a microfluidic device, the performance of the miniaturized LIF system was only slightly reduced from that of a conventional LIF system assembled using bulky, expensive components requiring significant power for operation. Additional optimization of the miniature LIF system components may provide further improvements. This preliminary work suggests that it is not unreasonable to expect that small, cost-effective systems with reasonable sensitivity can be assembled from off-the-shelf components for battery-operated, potentially handheld microfluidic analysis of DNA or other bioanalytes.