Abstract
An integrated high-throughput tool for forced degradation studies during pharmaceutical development is described. A series of experiments are conducted with the Symyx Technologies (Santa Clara, CA) Automated Forced Degradation System. The hardware and software of the workflow are described. Degradation libraries of stressed samples of liquid formulations of drug A were created, analyzed for room temperature solubility, and then replicates of each library were heated and sampled over time at 55, 70, and 85 °C to monitor for parent product degradation. Library arrays of first-order kinetic plots were generated, and predictions of room temperature stability were made. Several viable solution formulations were identified. (JALA 2005;10:374–80)
Keywords
Introduction
Recently, pharmaceutical development laboratories have been under pressure to stretch their resources to meet a number of new challenges. Advancements in technologies supporting pharmaceutical discovery research methods have led to an increased number of active molecules emerging from drug discovery labs with a growing percentage of these molecules being poorly soluble.1–3 Pharmaceutical companies are trying to identify and solve issues with compounds at an earlier stage to minimize development costs and time. 4 In an effort to fully examine the increasing number of challenging compounds identified by pharmaceutical discovery organizations, pharmaceutical development scientists are looking for more automation and the ability to apply high-throughput methodologies to the realm of pharmaceutical development. Automation is particularly desirable when considering forced degradation studies in which all drug compounds proceed through essentially the same workflow to identify conditions that lead to degradation and to identify degradation products. Whereas a collection of stand-alone analytical instruments offer autosamplers and the ability to analyze a list of individual samples, these instruments do not by themselves enable a high-throughput approach. Fermier et al 5 have described an instrument designed to expose arrays of materials to a wide array of stress conditions in parallel, but it does not integrate the collection and processing of analytical data and so it automates only a portion of the overall workflow. Sims et al 6 have described an approach using online high-performance liquid chromatography (HPLC) analysis to automate and analyze multiple degradation experiments, however, they do not integrate the design or sample generation into their approach. Symyx Technologies’ (Santa Clara, CA) Renaissance software is able to facilitate an entire high-throughput workflow from experimental design, to sample preparation, to screening and data acquisition, to data analysis and querying.7, 8 This paper describes the Symyx Technologies Automated Forced Degradation System, a system comprising solid and liquid handling robotics, environmental stations, and HPLC units, all of which is integrated with Renaissance software allowing scientists to rapidly design, create, and analyze pharmaceutical development formulations (Fig. 1).
Symyx Renaissance Software Suite is a highly integrated suite of software designed to facilitate the process of designing, executing, and evaluating chemical and material research and development. Renaissance consists of several different software applications, each focused on users and specific to the actual day-to-day tasks of laboratory research, all sharing a common environment for data storage–the Renaissance Application Server.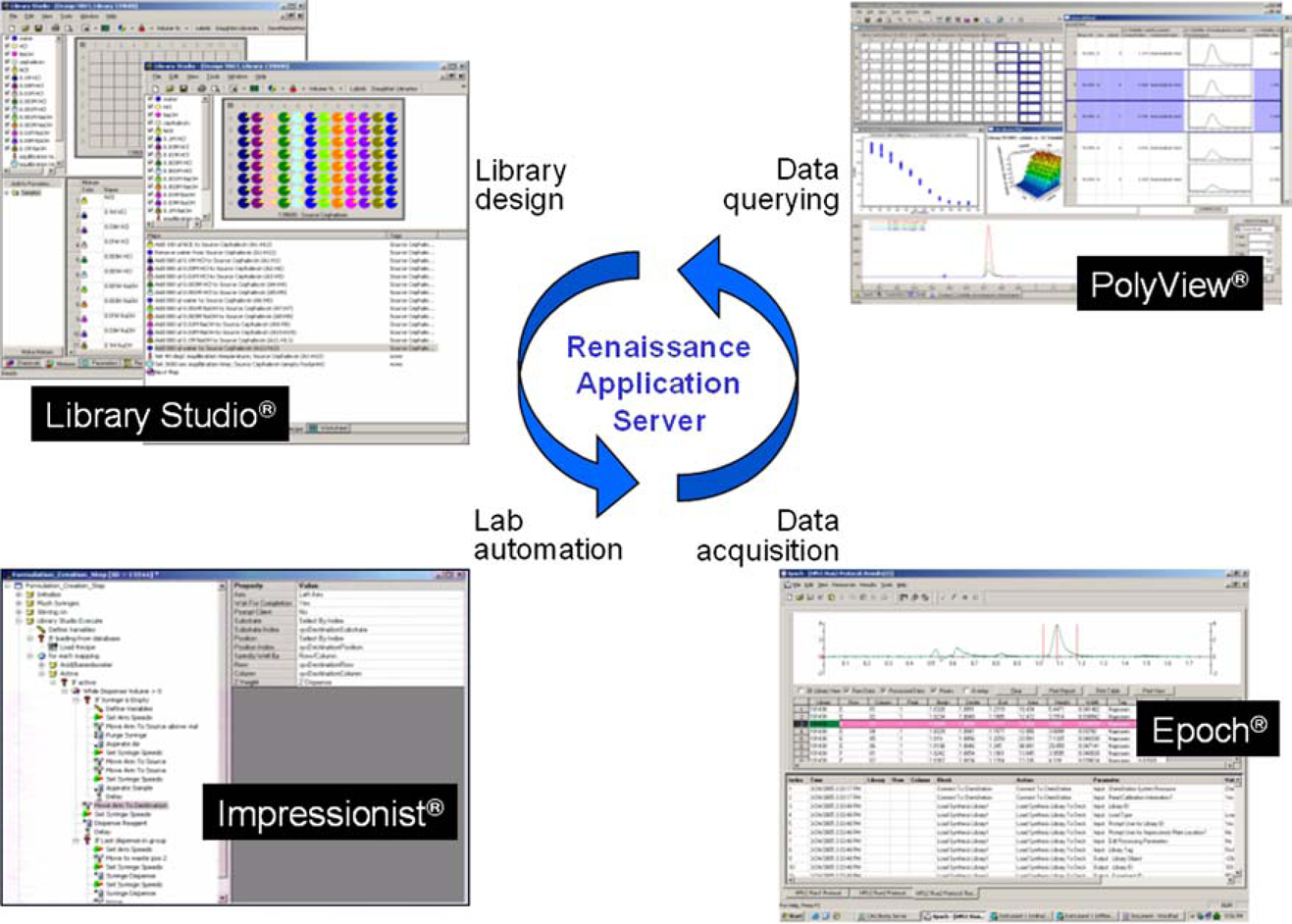
In this paper, the implementation of the Automated Forced Degradation System is exemplified in a case study with the goal of finding a highly stable, highly soluble liquid formulation of a commercial drug compound (compound A). Compound A is currently formulated as an HCl salt in a stable, lyophilized product. A liquid formulation is desired where the parent compound is highly soluble and stable. Whereas the commercial form of compound A is highly soluble under acidic aqueous conditions, it is known to readily degrade to form degradant D. Under the desired neutral aqueous conditions, compound A shows sufficient stability but it has prohibitively low solubility. In the present study, a thorough series of parallel high-throughput experiments allowed for the solubility and stability profile of compound A to be studied as a function of pH, counterion, cosolvent, and complexing agent to find a viable liquid formulation with the requisite solubility and stability requirements.
Experimental
Library Design
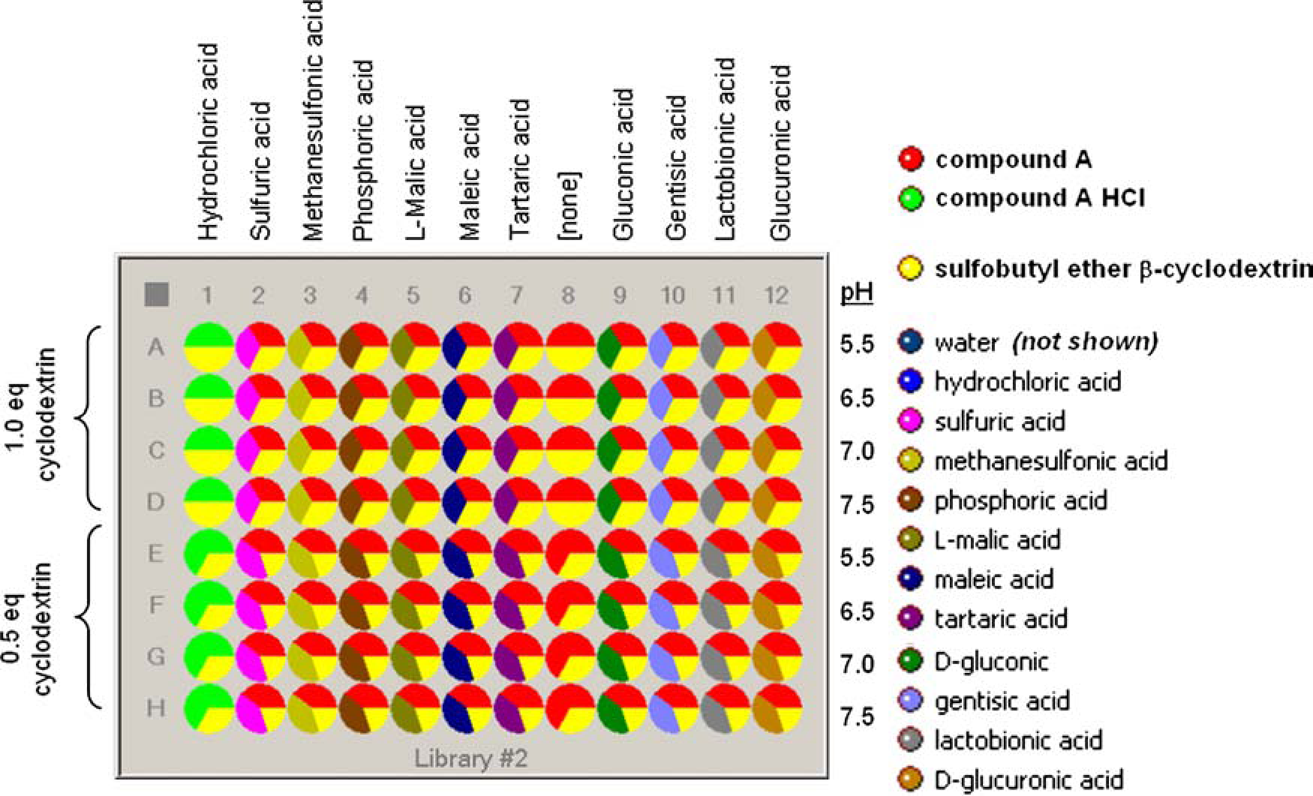
Parallel arrays of experiments were designed using Symyx’ Library Studio, software. Designs for libraries #1 and #2 are shown in Figures 2 and 3, respectively. Each component was entered, and virtual stock solutions of each acid were created within Library Studio. Composition maps were created using equations to specify that each well contained a specific mass of drug compound, specific liquid volume, specific molar equivalence of the counterion, and specific ratio of cosolvent to water.
The Library Studio design for library #1 indicating relative molar amounts (1 molar equivalent of complexing agent to compound A). The total amount of solvent in each well was 800 μL. The Library Studio design for library #2 indicating relative molar amounts (1 molar equivalent of acid to compound A).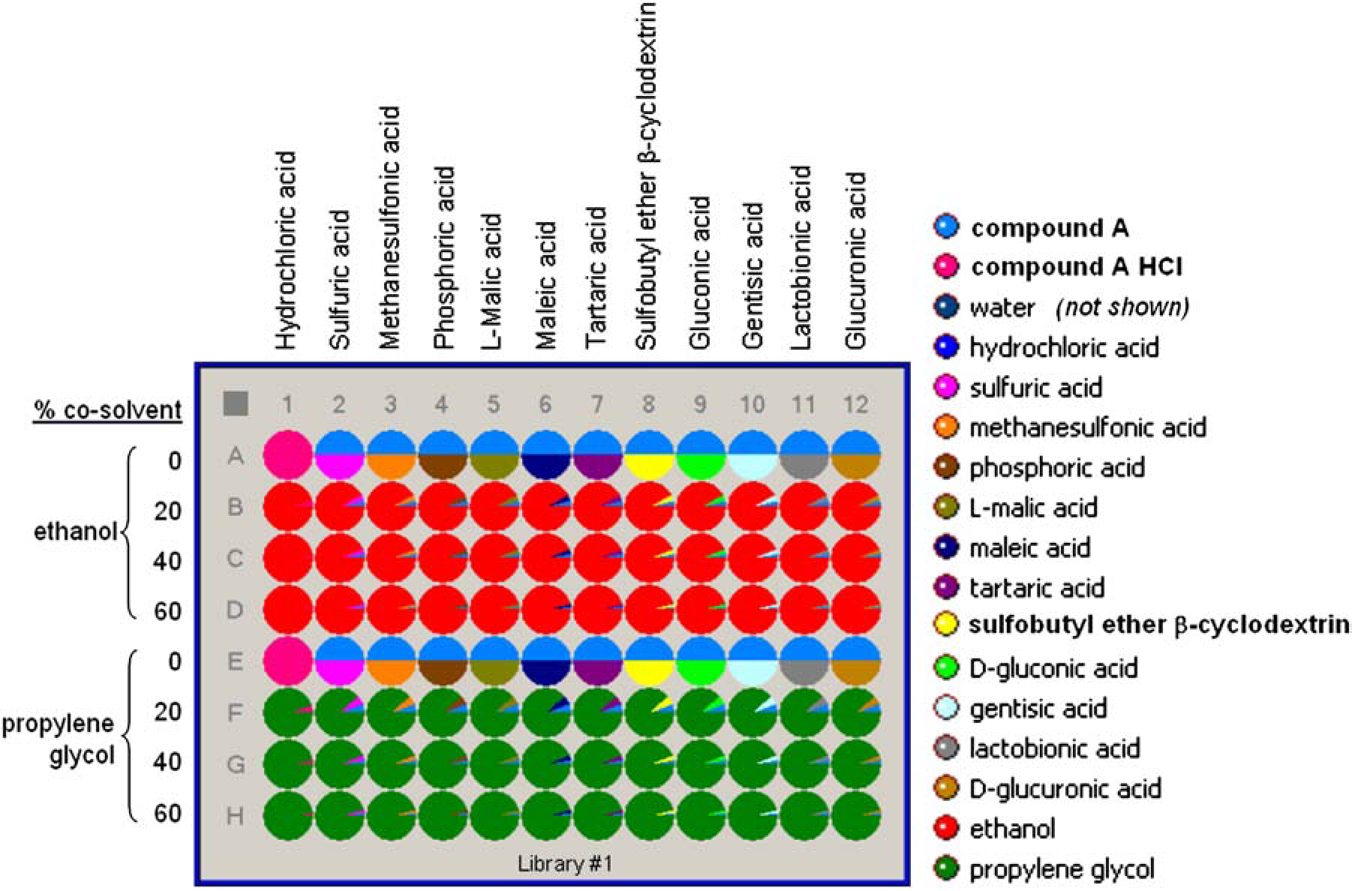
Sample Preparation
To prepare the free-base of compound A, compound A hydrochloride (5.0 g) and potassium carbonate (4.0 g or 1.5 molar equivalents) were added to a 1.0-L round bottom flask. Dichloromethane (350 mL) and ethanol (300 mL) were added, and the contents of the flask were stirred vigorously at room temperature overnight. The milky white solution was filtered with a fritted funnel to a clean bottle. Solvent was removed by evaporation, and the solids were placed under high vacuum for 8 h at 30 °C. Free-base verification was done by 1 H NMR (yield = 86 wt%).
In this example, the system is fully integrated through the software, but each hardware station stands independent. Sample plates are manually transferred between the various hardware stations so that in this implementation a hybrid manual/automated approach is achieved. Renaissance automation software was used to read the Library Studio recipe files and drive an Autodose Powdernium robot; both compound A and the sulfobutyl ether β-cyclodextrin were dispensed to each library plate as solid powders. Plates were manually carried to the Symyx Core Module where Impressionist software was then used to read the Library Studio recipe and to dispense aqueous solutions of the acidic counterions to the appropriate wells. a The Symyx Core Module is a dual-armed robotic unit able to dispense liquids to and from sealed vials, control temperature in three heated zones and one chilled zone, filter samples at elevated temperature, stir samples with magnetic stirring, measure and record the pH of various sealed samples, and sample and dispense viscous liquids using disposable tips. b The plates were dried and then water and cosolvent were dispensed with the Symyx Core Module to the plates according the library designs. Each formulation in library #2 was robotically adjusted to a target pH based on the library design with the addition of HCl and NaOH solutions. For each forced degradation library, a master array of formulations was created and used to daughter out about 800 μL of each sample into three replicate plates to stress the solutions at 55, 70, and 85 °C. Magnetic stir bars were dispensed to each well, and the wells in each library were sealed with a capmat seal. The plates for library #1 were agitated for 42 h at room temperature to allow the samples to reach solution equilibrium for solubility measurements. The plates for library #2 were allowed to equilibrate for at least 2 days after the wells had been adjusted to their final pH.
Gentisic acid has a low water solubility. Consequently, this acid was dispensed as a well-mixed aqueous slurry with the Core Module.
Further information on the Symyx Core Module Automation Station can be found on www.symyx.com under Discovery Tools/Pharmaceutical Workflows/Solubility/Hardware.
Library Analysis
Using the integrated electrode pH meter on the Symyx Core Module, the pH values of each formulation were measured with Epoch software and recorded to the database. Subsequently, the Symyx Core Module was used to filter the saturated solutions of each library of formulations into a sealed microtiter assembly of vials using the Symyx filtration assembly. 7 Aliquots were taken from the sealed plates at room temperature using the Symyx Core Module piercing needle. Each of the replicate plates were heated, and at various times aliquots were pulled and diluted from each of the plates using the robotic piercing tip. Diluted aliquots were stored in a refrigerator at approximately 5 °C until analyzed by HPLC.
All aliquots were analyzed with an Agilent 1100 HPLC by running a rapid isocratic method.

Epoch software was used to drive ChemStation to collect the data and then to record all the method information, chromatograms, identified peaks, peak areas, and measured concentrations to the database.
Results
Figure 4 shows the pH and solubility of library #1 measured after equilibration for 42 h at room temperature. The tables show that the parent compound was highly water soluble in most of the formulations (rows A and E). The formulations with d-gluconic acid and gentisic acid evidenced lower aqueous solubility relative to the other samples. The addition of cosolvents ethanol or propylene glycol increased the solubility of these systems. Cosolvents had a small effect on the solubility of the other systems. The pH of most formulations in library #1 ranged from ca. 2 to 4.5. Within this pH range, compound A is expected to be very soluble and to undergo fairly rapid degradation. Formulations in column 8 that contain cyclodextrin have an alkaline pH, and yet a very high solubility.
The measured solubility and pH of saturated solutions of library #1 at room temperature.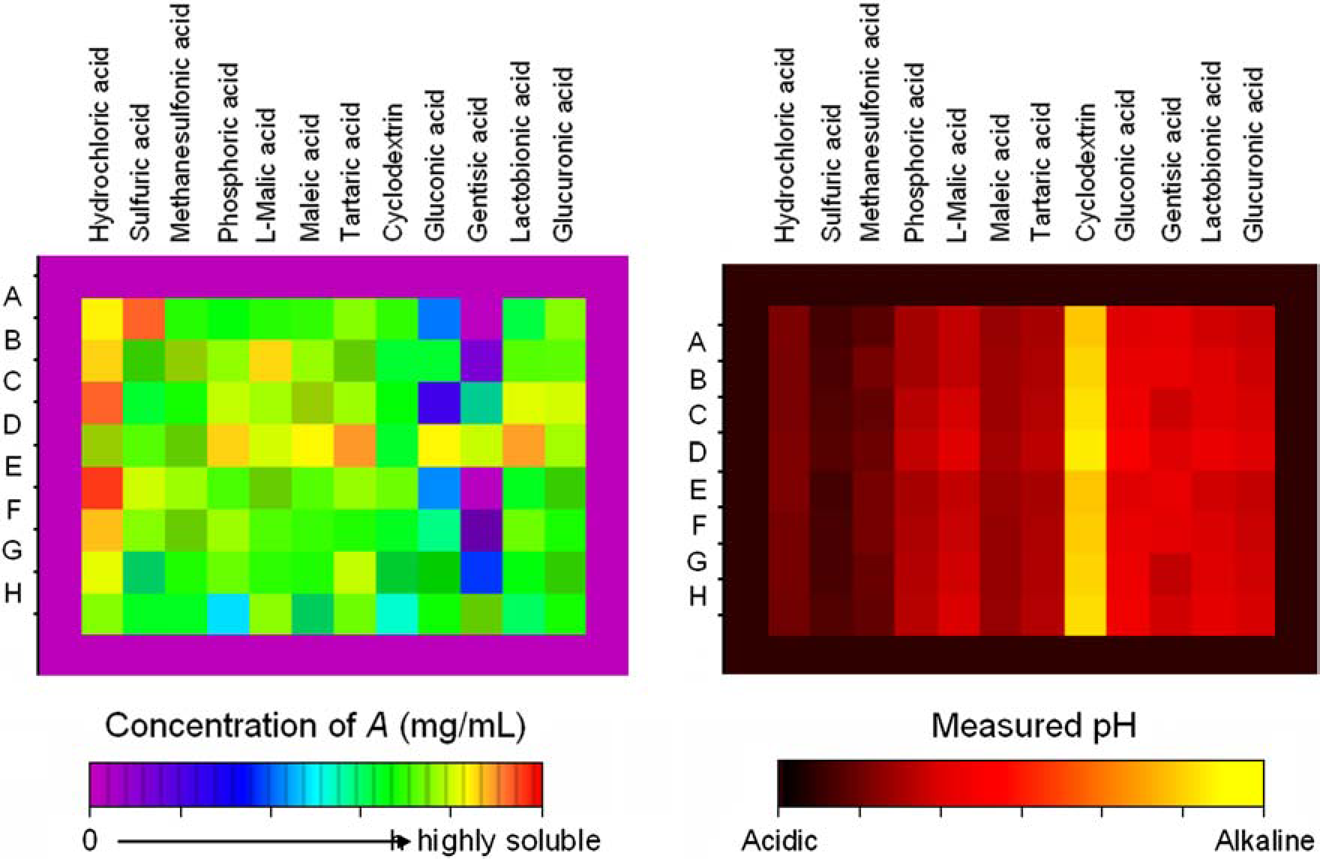
To perform a forced degradation study of these formulations the three replicate plates of library #1 were heated to 55, 70, and 85 °C, and aliquots were drawn and analyzed by HPLC over time for the amount of compound A and the formation of the degradant D. Assuming first-order kinetics and that the rate of disappearance of compound A equals the rate of appearance of degradant D, pseudo-first-order rate constants (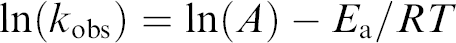
The glass vial for well C3 of the 55 °C plate of library #1 was defective and it did not contain any of its formulation, thus the data indicate that no degradant D was formed.
Pseudo-first-order rate plots for library #1 at 55 °C. Each graph shows the ln{1–[D]/[A]initial} versus time. The degradation data were collected from 2 h to 18 days. The slope of the regression line is kobs at 55 °C. See footnote b.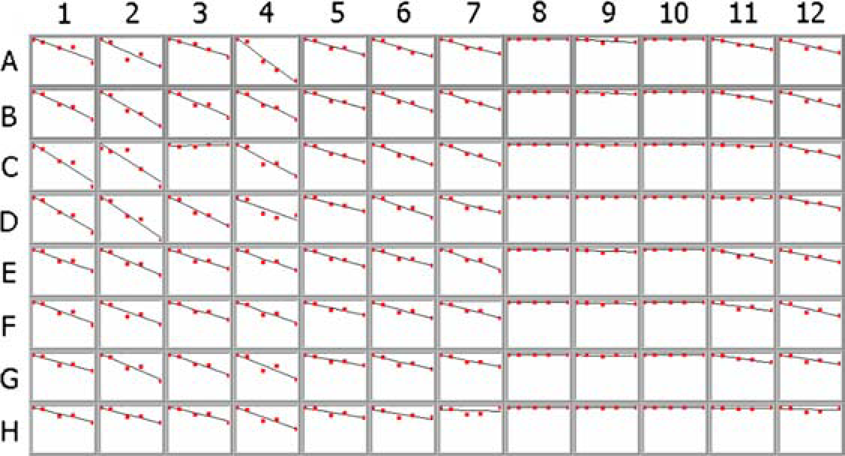
The graphs shown in Figure 5 were generated by using Renaissance software to pull the HPLC data out of the database and analyzing and plotting them using prototype Symyx software that will be included in the next release of Renaissance.
In Figure 6, the ln(
Arrhenius plot of ln(kobs) versus 1/T for library #1. The data were collected at 55, 70, and 85 °C and extrapolated to 25 °C.
Room temperature stability for library #1 expressed as the time in days to degrade 5% of compound A based on Arrhenius calculations (t5% = ln(0.95)/kobs)
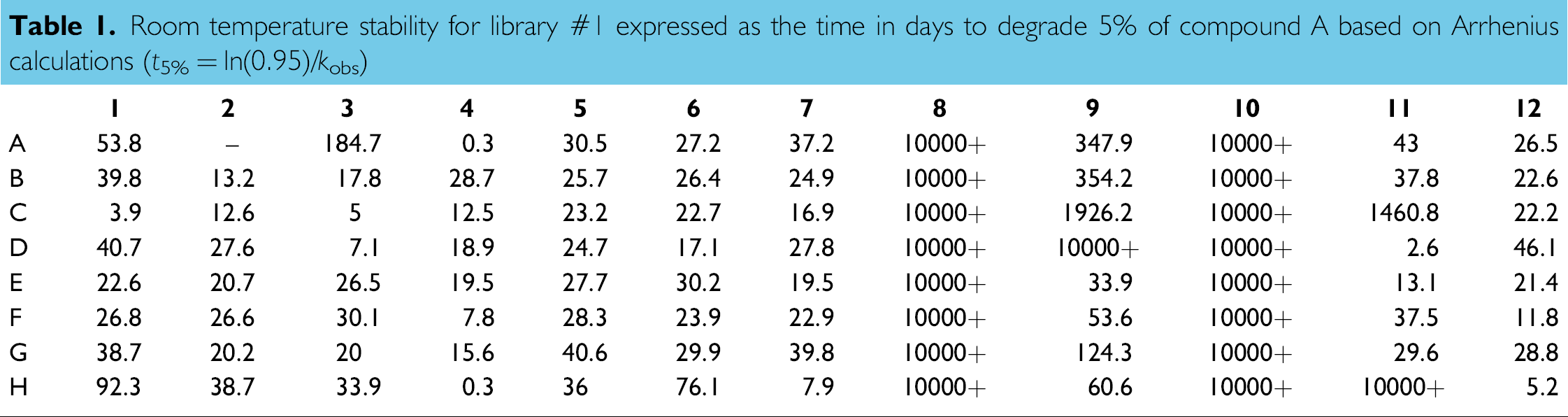
Based on the results from library #1, library #2 was designed, and a subsequent forced degradation study was performed at a range of pH values desired for a parenteral formulation (pH = 5.5–7.5). In addition, library #2 was used to consider various relative amounts of cyclodextrin and whether the combination of the cyclodextrin with various counterions would be advantageous.
Figure 7 shows the pH and solubility of library #2 measured at room temperature. Each formulation of library #2 was allowed to equilibrate for at least 2 days at the final pH. The data show that the final pH of the formulations in library #2 was close to the target values. Most of the formulations showed a very weak dependence of solubility on the amount of cyclodextrin in the formulations. The exception was the gentisic acid formulations that show solubilities of 32 and 22 mg/mL for formulations with 1.0 and 0.5 equivalents of cyclodextrin, respectively. Formulations with lactobionic acid showed a solubility of about 50 mg/mL. Formulations with other counterions showed solubilities that ranged from about 37 to 45 mg/mL.
The measured solubility and pH of saturated solutions of library #2 at room temperature.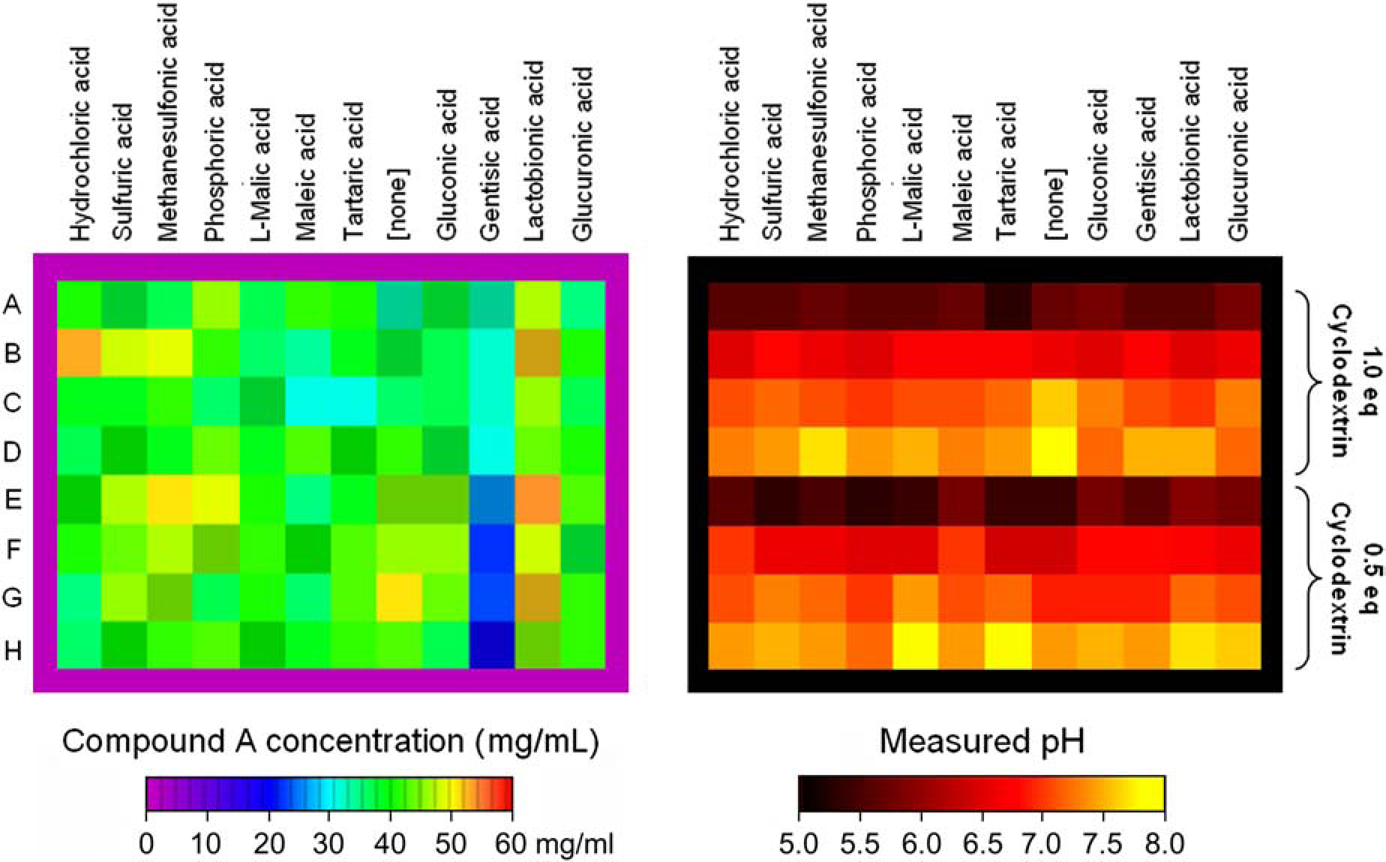
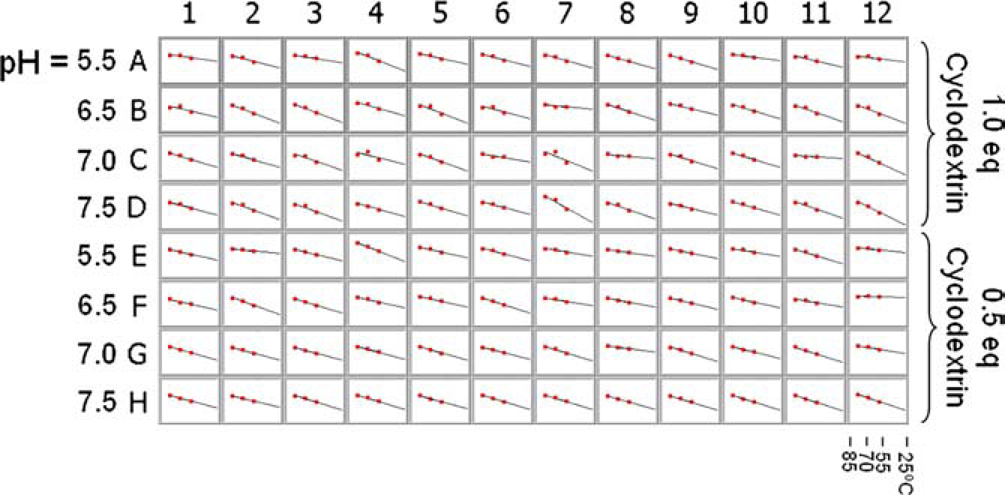
To perform a forced degradation study of library #2, its three replicate plates were heated to 55, 70, and 85 °C, and aliquots were drawn and analyzed as was done with library #1. The pseudo-first-order data for library #2 at 55 °C are plotted in Figure 8. The data show that as the cyclodextrin concentration increases from 0.5 to 1.0 equivalents, the degradation rate drops dramatically to the point where formation of degradant D is almost negligible. Additionally, an increase in pH from 5.5 to 7.5 leads to a decrease in the degradation rate. Similar trends were seen with data collected at 70 and 85 °C. In Figure 9, the ln(
Pseudo-first-order rate plots for library #2 at 55 °C. Each graph shows the ln{1–[D]/[A]initial} versus time. The degradation data were collected from 2 h to 22 days. The slope of the regression line is kobs at 55 °C. Arrhenius plot of ln(kobs) versus 1/T for library #2. The data were collected at 55, 70, and 85 °C and extrapolated to 25 °C.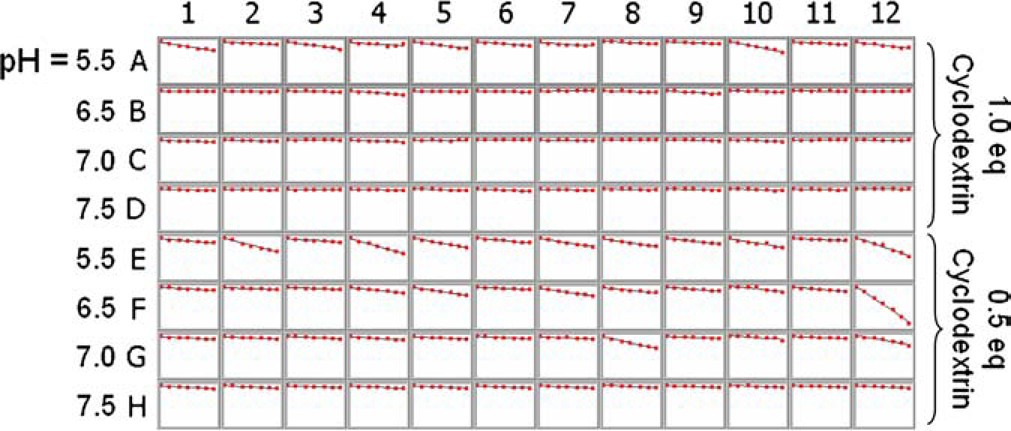
Conclusions
Over a 6-month period, a high-throughput forced degradation approach was successfully employed to evaluate the feasibility of a liquid formulation for a compound with unsatisfactory solubility and stability characteristics. The Automated Forced Degradation System was used to design, create, and analyze over 5800 samples with varying counterions, additives, solubilizing agents, and cosolvents to identify several promising formulations. The results from this study showed that compound A formulations containing cyclodextrin have several orders of magnitude improvement in stability over the reconstituted current HCl salt product while maintaining a high solubility over a pH range desired for parenteral formulations. Moreover, several other possible formulations were identified as viable, with sufficiently high stabilities and high solubilities. Further optimization studies with these additional formulations are underway with the hope of meeting the needs of the medical community with an elegant, room temperature stable liquid formulation. In addition to the successful formulations that were identified, this study was valuable for widely mapping compound A formulation space.
In the example described in this paper, the Forced Degradation System is fully integrated through the software, but each hardware station stands independent. More generally, the Symyx Technologies Automated Forced Degradation Tool is a modular system that can be set up with a robotic plate handling robot and a choice of stability chambers (Fig. 10). Whereas the material in the example of this paper was highly soluble, this system can be used to spare material for early development studies using only ca. 1 mg of parent compound per sample. This system is designed to allow a scientist to flexibly perform multiple automated stress testing and drug-excipient studies by exploring the effects of environmental exposure (temperature, humidity, light) and chemical exposure (acid, base, peroxide, radical generator, or other oxidizers) on arrays of solid and solution samples. d The system is also being deployed with the new Symyx Extended Core Module (XCM), which incorporates an integrated on-deck balance to enable mass tracking of various dispenses, aliquots, and dilutions to improve the accuracy of these studies. 9 The modularity of this workflow allows one to start with a limited workflow and add modules over time.
There are several methods to do oxidative challenges with this system. The most straightforward is to use the robotic units to add a chemical oxidizer (e.g., HOOH) and pull samples over time. There is also the potential opportunity to study the effects of elemental oxygen by placing the arrays of samples in a Symyx sealed high pressure (HiP) reactor and providing a defined pressure of oxygen or oxygen/air at a defined temperature.
Layout of Automated Forced Degradation System.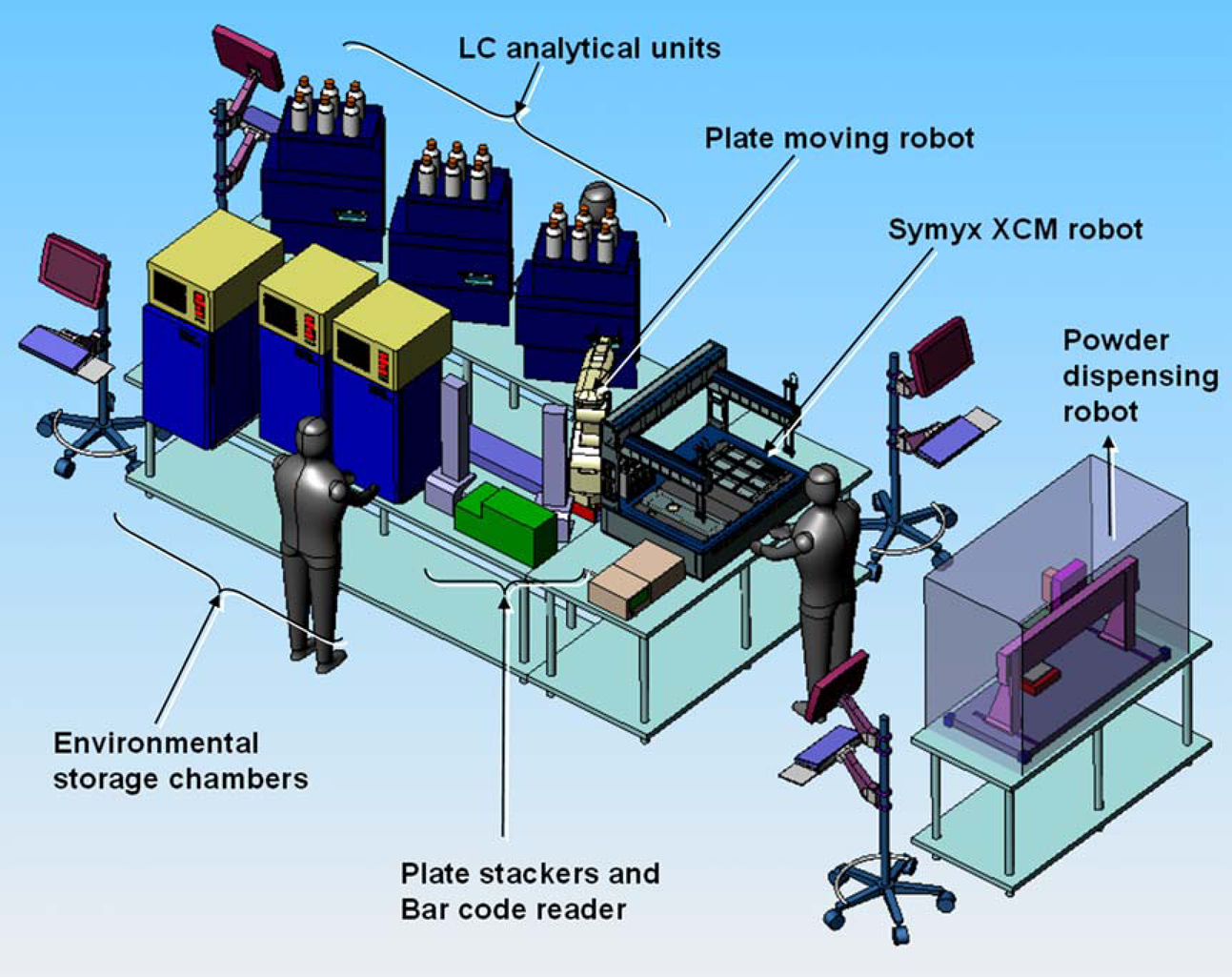
The Symyx Automated Forced Degradation System integrated with Renaissance offers a high degree of value to a pharmaceutical development lab. First, it allows a given staff to perform an increased number of experiments at an earlier stage. Second, this system allows researchers to expand the breadth of experimental space being tested when compared with what a similar team of researchers could study using traditional techniques and a comparable amount of time. This yields a better chance of initially identifying an optimal formulation and the potential of an early discovery of alternatives for patent protection and life cycle management. Finally, the software integration of this tool offers easy and extensive data management and the ability to maximize the advantages of higher throughput and expanded experimental breadth. By capturing all the data to a common database, the Symyx Automated Forced Degradation System has the ability to rapidly analyze and tie together hundreds or thousands of pieces of data from the library design, to the sample creation, to the sample screening. These data can be rapidly collected and compared between users, between different tests, and even between different geographic testing sites to generate comprehensive reports.
Footnotes
Acknowledgments
We would like to thank Dr. Stephen Baertschi of Eli Lilly and Company for helpful scientific discussions and a critical review of the manuscript.
