Abstract
The advantages of using a new automated system, the Hydra-Plus-One System equipped with 96 or 384 precision glass syringes and a non-contact microsolenoid dispenser, in setting up high-throughput low-volume sequencing reactions and PCR are described. Using the syringe-based dispenser, which is the Hydra-PP part of this system, wet dispenses of as small as 100 nL with CVs of less than 10% can be accomplished. The single-channel, non-contact microsolenoid dispenser part of the system can dispense samples as low as 100 nL (with CVs of less than 10%) at a speed of 58s per 96 dispenses into any plate format (SBS footprint). The advantages associated with the use of the Hydra-Plus-One System for setting up PCR and sequencing reactions are high precision at nanoliter-dispense range; speed; and minimal waste of precious and expensive samples. The single-channel dispenser eliminates the dead volume associated with aspirating from reservoirs or troughs and thereby reduces sample waste. In addition, virtually all material can be recovered from the dispenser. Finally, non-contact dispensing enables distribution of sample into wells without any in-between-dispenses washing requirements.
Keywords
Introduction
One of the major economic disadvantages of setting up nucleic acid amplification and sequencing reactions with the existing automated high-throughput dispensing technologies is the waste of precious and expensive reagents. The commonly used 96- or 384-channel dispensers generally aspirate samples from a reservoir with a minimal working volume (the minimum volume needed to cover the floor of the reservoir to allow the filling of each channel) ranging from 1 to 15 mL to dispense microliters to nanoliters of reagents. Therefore, even though the dispensers can be used to scale down the volumes of costly biological reactions (by providing nanoliter-dispensing capabilities), there is still a waste of expensive material. The new Hydra-Plus-One System was designed to eliminate waste issues.
The Hydra-Plus-One System is composed of a single-channel non-contact microsolenoid dispenser (called the NanoFill® dispenser) 1 and a 96- or 384-channel, syringe based Hydra-PP system. 2 The single-channel dispenser transfers from 100 nL to 50 μL of solutions (such as BSA solutions of up to 100 mg/mL) with a dispense-precision variation of less than 10%. 1 The Hydra component of the system can successfully dry-dispense viscous samples (as small as 200 nL) containing up to 30% PEG 8000 and 50% glycerol, with CVs of less than 10% (wet aqueous dispensing capability of as low as 100 nL, with CVs of less than 10%). 3 – 5
The NanoFill system uses a microsolenoid and positive pressure, created by helium gas, to dispense liquid. It has a dispensing nozzle (internal diameter of 125 μm) connected to a 25-inch-long tube. The sample is aspirated from a 0.6 mL micro-tube into the nozzle and is separated from the system fluid (water) by an air gap of 0.75–1 μL. There is no waste of precious and expensive samples (such as proteins) due to virtually full material recovery from the non-contact dispenser. Furthermore, because sample can be aspirated directly from the source tube (0.6 mL microtube), the single-channel dispenser eliminates the dead volume associated with aspirating from reservoirs and thereby reduces sample waste.
Another advantage of this system is its speed of dispense. The speed of the non-contact dispenser is 0.6 second per well. As a result, sample evaporation (a major issue when nanoliter dispenses are performed) is prevented.
While the non-contact dispenser part of the Hydra-Plus-One System can be used for the precise low-volume dispensing of very valuable reagents, the Hydra part of the system can dispense 96 or 384 different (inexpensive) samples simultaneously. Here we demonstrate automated procedures for the setup of PCR and DNA sequencing reactions using both components of the Hydra-Plus-One System.
Materials and Methods
The Hydra-Plus-One System (composed of a Hydra-PP equipped with 96, 100 μL syringes with Teflon® coated stainless steel needles and a single-channel, non-contact microsolenoid dispenser) and CyclePlate microplates (cat. # 1047–00) were provided by Apogent Discoveries, Sunnyvale, CA. The PCR core kit (cat. # 1 578 553) and Human Genomic DNA (cat. # 1 691 112) were purchased from Roche, Mannheim, Germany. Human β-actin primers were obtained from Clontech, Palo Alto, CA. Sequencing template M13mp18 single-stranded DNA (cat. # N4040S) and primer M13 Sequencing Primer (cat. #S1224S) were obtained from New England BioLabs, Beverly, MA. DNA sequencing reactions were performed using ABI PRISM® BigDye™ Terminator Cycle Sequencing Ready Reaction Kits (cat. # 4303150) from Applied Biosystems, Foster City, CA. Agarose-gel electrophoresis was performed as the standard protocol to check the quality of the PCR products. 1 Kb Plus DNA Ladder (cat. # 10787–018) was purchased from Invitrogen, Carlsbad, CA. Electrophoresis images were detected with a NucleoVision™ Imaging Workstation from NucleoTech® Corporation, San Mateo, CA. Micro®-90 soap (cat. # Z28, 156–5) and Coulter® Clenz (cat. # 8546930) were purchased from Sigma-Aldrich, Milwaukee, WI, and Beckman Coulter, Fullerton, CA, respectively.
DETERMINING THE DISPENSING PRECISION OF THE HYDRA-PLUS-ONE SYSTEM
Prior to the use of the Hydra-Plus-One System, the uniformity and consistency of the volumes dispensed across the array of syringes for the Hydra-PP component were determined by the coefficient of variance (CV) (Apogent Discoveries Application Note 6; http://www.apogentdiscoveries.com). In short, different volumes of a 10 μg/mL fluorescein solution were dispensed into each well of a 96-well plate containing 0.1 M Tris buffer. The final volume in each well after dispensing was 100 μL. Each plate (after an incubation period of an hour) was then read in a TECAN SpectraFluor™ fluorescence plate reader and the CVs were determined across each plate for each of the dispensed volumes. A high uniformity for dispensing volumes equal to and greater than 100 nL was evident, with CVs of less than 10% (Figure 1). The uniformity and consistency of the volumes dispensed by the single-channel microsolenoid dispenser was also determined by the coefficient of variance, with CVs of less than 10% for dispensing volumes of 0.1–2 μL. 6
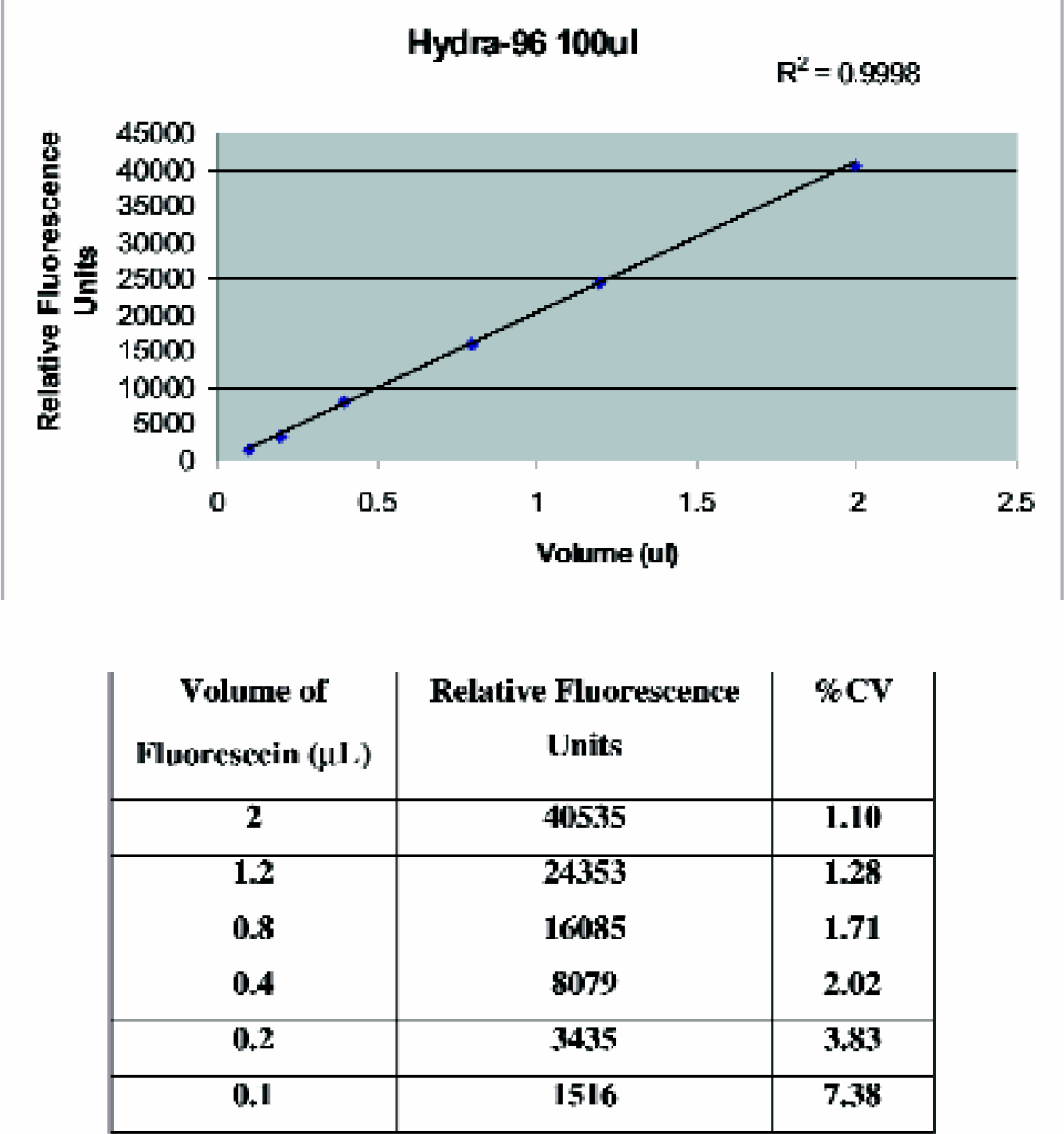
Dispensing precision of a Hydra-PP system equipped with 96, 100 μL stainless steel syringes.
PREPARING THE HYDRA-PLUS-ONE SYSTEM
Because the Hydra-PP component of the Hydra-Plus-One System is equipped with fixed needles, the need for an efficient syringe-cleaning procedure for the inhibition of sample to sample carry-over and cross contamination is evident. Following a proper syringe washing procedure also prevents the syringes from clogging. Extensive numbers of wash methods (published elsewhere) have been developed for different applications to address these issues. 1,3,4 In short, prior to the setup of experiments, the Hydra syringes were washed with water for six cycles (“one wash cycle” was defined as a fill and empty of the syringes at full syringe volume). If the syringes were used for dispensing nucleic acids, a manual wash procedure using 2% bleach was performed and was followed by six wash cycles of water in the wash reservoir (see Application Notes 5 and 10 at Apogent Discoveries web site at http://www.apogentdiscoveries.com). After the last use of the system for the day, the Hydra syringes were washed three times with Coulter Clenz (a detergent containing proteases) and then rinsed with water for an additional six wash cycles. 7
Before the microsolenoid dispenser was used to aspirate samples, it was automatically washed with 1500 μL of water through the execution of wash commands. An air gap (0.75–1 μL) was then aspirated into the dispenser, followed by aspiration of the sample. The preceding wash step was repeated between aspirations of different samples. After the last use of the system for the day, the microsolenoid dispenser was washed with Micro-90 soap and then rinsed with water (through the execution of the software command called Daily Wash). The outside of the microsolenoid dispenser was rinsed and wiped clean. 1,7
SETTING UP THE PCR
Both components of the Hydra-Plus-One System were used to set up PCR. The inexpensive buffers and DNA templates, which might vary from one well to another, were dispensed using the Hydra component of the system, while the expensive reagents such as the dNTP, primers, and Taq DNA Polymerase were dispensed using the non-contact dispenser component. Using the Hydra syringes, 0.4 μL of 5 ng/μL human genomic DNA were dispensed into a PCR plate. Next, a master mix of PCR primers, PCR buffer, dNTP and Taq Polymerase was aspirated by the single-channel microsolenoid dispenser and dispensed (3.6 μL per well) into the same PCR plate (total reaction volume of 4 μL per well). The plate was sealed and placed in a thermal cycler.
DISPENSING THE DNA SEQUENCING REAGENTS
The Hydra syringes were used to dispense 2 μL of M13mp18 DNA (50 ng/uL) into a CyclePlate. Next, a master mix of sequencing primer (M13 forward) and Terminator Ready Reaction Mix (containing the AmpliTaq DNA polymerase, rTth pyrophosphatase, magnesium chloride, dye terminators, and buffer) was prepared. The single-channel microsolenoid dispenser was used to aspirate the mix and then dispense it (2 μL per well) into the CyclePlate (total reaction volume of 4 μL per well). The plate was sealed and placed in the thermal cycler.
Results
REACTION SETUP TIME
The dispensing speed of the non-contact microsolenoid dispenser is approximately 58 seconds per plate of 96 samples. The dispensing speed of the Hydra-PP system for transferring 96 samples simultaneously was approximately 20 seconds. The total setup time for a 96-sample PCR or a 96-sample DNA sequencing reaction plate, using the Hydra-Plus-One System, was less than 2 minutes (washes not included). The rapid speed of the instrument prevents evaporation and thus enzyme deactivation, ensuring high precision and reproducibility for setup of amplification and sequencing reactions.
The washing procedure is a separate process that can be carried out after the PCR or the sequencing reactions are assembled. Washes are performed through the execution of wash commands from the software and take a couple of minutes.
QUALITY OF THE PCR AND DNA SEQUENCING REACTIONS
To determine the quality of the PCR set up by the Hydra-Plus-One System, the amplified samples were analyzed by gel electrophoresis analysis (Figure 2). Uniformity and consistency in PCR yield among the different wells indicated that the Hydra-Plus-One System offers a reproducible method of automated PCR setup.

Electrophoresis analysis of the PCR product. Samples were loaded manually onto a 1.2% agarose gel. Human β-actin primers were designed to amplify 838bp of template human genomic DNA. The total PCR reaction volume was 4 μL. All 4 μL were loaded into the gel.
To determine the quality of the sequencing reactions set up by the Hydra-Plus-One System, random samples were sequenced on an ABI 3700 sequencer. Result showed an average read length PHRED Q>20 of 547.8 (for 30 samples sequenced), with a standard deviation (STDEV) of 35. Figure 3 presents one of the sequence chromatograms.
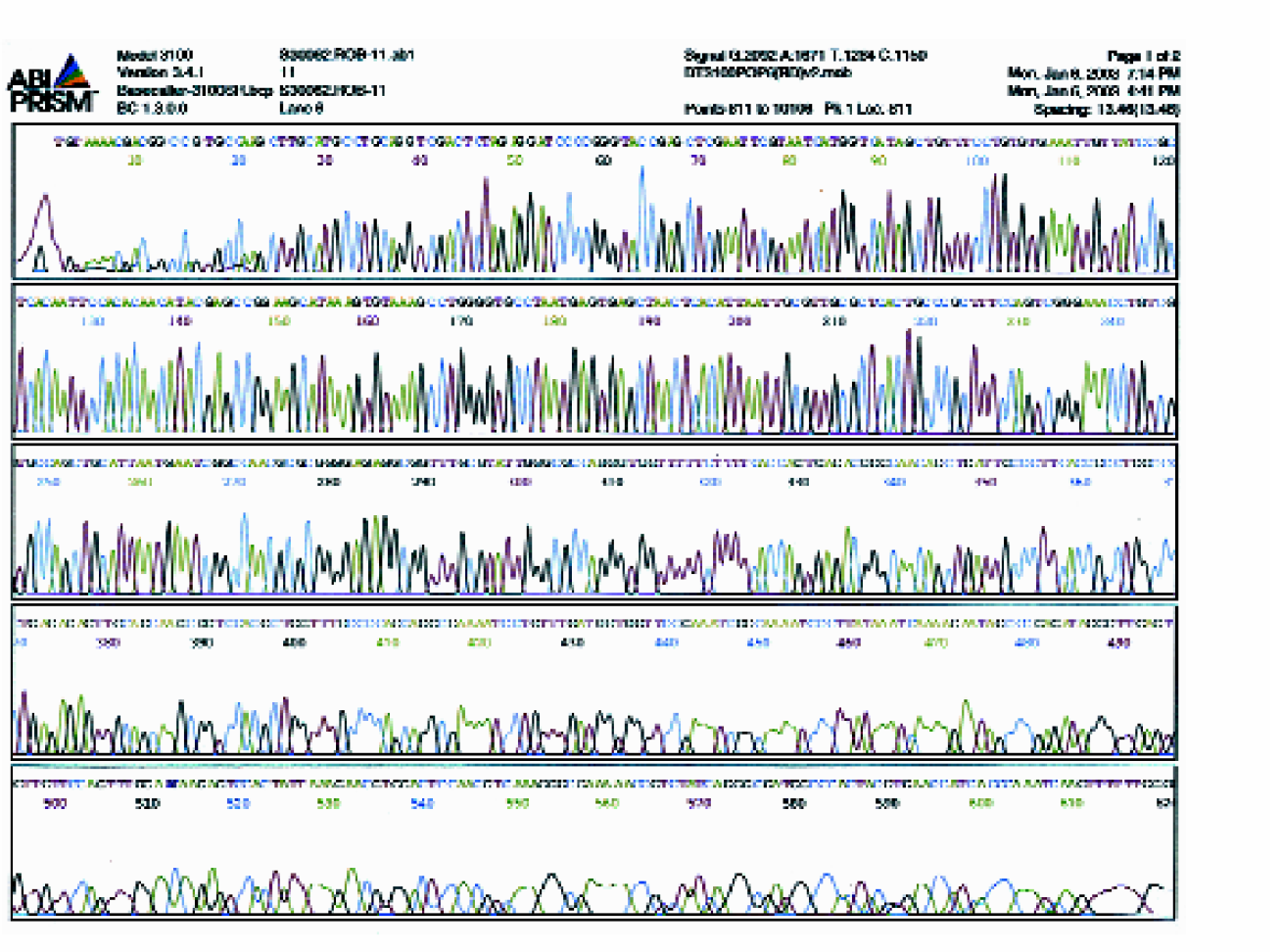
Sequencing chromatogram of a representative sample. Sequencing template was M13mp18 single-stranded DNA and the sequencing primer was M13 forward. Total sequencing reaction volume was 4 μL.
Discussion
Using an automated dispensing system equipped with 96 non-disposable precision glass syringes and a non-contact microsolenoid dispenser, high-throughput PCR sand sequencing reactions were set up. While the Hydra-PP part of this system was used to simultaneously dispense 96 inexpensive DNA and buffer samples, which might vary from one well to another, the expensive reagents such as the dNTP, dye terminators, primers, and Taq DNA Polymerase were dispensed using the non-contact dispenser component of the system.
The advantages associated with the use of the Hydra-Plus-One System for setting up PCR and sequencing reactions were high precision at nanoliter-dispense range (CVs of less than 10% for volumes as low as 100 nL by both the Hydra and the NanoFill components of the system); speed (the total setup time for a 96-sample PCR or a 96-sample DNA sequencing reaction plate was less than 2 minutes); and minimal waste of precious and expensive samples.
An additional advantage of this system is that the single-channel dispenser eliminates the dead volume associated with aspirating from reservoirs or troughs and thereby reduces sample waste. Furthermore, virtually all material can be recovered from the dispenser. For example, to dispense 2 μL of a master mix of sequencing primer and Terminator Ready Reaction Mix into a 96-well plate, a total of 195 μL of this sample was prepared in a microtube and 194 uL of this mix was aspirated into the NanoFill dispenser. Next, 0.5 μL of this mix was prime-dispensed back into the source (the microtube) three times. Prime-dispensing is highly recommended because it increases the precision of dispense. This was followed by the dispensing of 2 μL of the mix into each of the 96 wells. Therefore, the waste volume associated with this dispense was approximately 3 μL (including the 1.5 μL returned to the source, 1 μL dead volume of the microtube, and 0.5 μL dead volume of the nozzle), or less than 2% waste per plate of 96 reactions.
Conclusion
Using the automated Hydra-Plus-One System, a total of 96 PCR or sequencing reactions can be set up in less than two minutes. This system provides maximum precision and speed while minimizing the waste of precious and expensive reagents.
